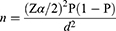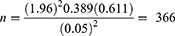Back to Journals » Neuropsychiatric Disease and Treatment » Volume 17
Alcohol Use Disorder and Its Associate Factors Relating to Patients with Severe Mental Disorders Attending Psychiatric Follow-Ups in Northwest Ethiopia
Authors Kassew T , Kiflie M, Minichil W , Dejen Tilahun A, Liyew B
Received 5 March 2021
Accepted for publication 26 May 2021
Published 4 June 2021 Volume 2021:17 Pages 1801—1810
DOI https://doi.org/10.2147/NDT.S309704
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Roger Pinder
Tilahun Kassew,1 Mihretu Kiflie,2 Woredaw Minichil,1 Ambaye Dejen Tilahun,3 Bikis Liyew3
1Department of Psychiatry, College of Medicine and Health Sciences, University of Gondar, Gondar, Ethiopia; 2Department of Psychiatry, University of Gondar Comprehensive Specialized Hospital, Gondar, Ethiopia; 3Department of Emergency Medicine and Critical Care Nursing, College of Medicine and Health Sciences, University of Gondar, Gondar, Ethiopia
Correspondence: Tilahun Kassew
Department of Psychiatry, College of Medicine and Health Sciences, University of Gondar, P.O.Box: 196, Gondar, Ethiopia
Tel +251918410710
Email [email protected]
Background: Patients with mental disorders have high rates of co-existing alcohol use disorder and vice versa. Alcohol use disorder has emerged as a major challenge to intervene patients with severe mental disorders. It is under-recognized and has not been investigated well in low-income countries like Ethiopia. The aim of this study was to assess the prevalence and associate factors of alcohol use disorder among patients with severe mental disorders attending psychiatric follow-ups at the University of Gondar comprehensive specialized hospital, northwest Ethiopia.
Methods: A total of 384 patients with severe mental disorders selected by a systematic random sampling technique took part in the interviews for this cross-sectional study. The alcohol use disorder identification test was used to assess the problem. Univariate and multivariate binary logistic regressions were computed to examine the associated factors. An adjusted odds ratio with a 95% confidence interval (CI) was used for reporting the result.
Results: The prevalence of alcohol use disorder among patients with severe mental disorders was 36.2%. Of the participants, 23.5%, 8.6%, and 4.1% had hazardous, harmful alcohol drinking and alcohol dependency, respectively. In the multivariate analysis, male sex, younger age, psychotic and bipolar disorder diagnoses, having poor social support, moderate and high perceived stress, and current tobacco smoking were factors significantly associated with alcohol use disorder.
Conclusion: The prevalence of alcohol use disorder among patients with severe mental disorders was found to be high. Early intervention and integrating treatment services for co-existing mental disorders and alcohol use disorder is important. The special treatment, where it is offered for young male patients and those with the diagnosis of psychotic and bipolar disorders, is recommended to promote the uptake of alcohol and mental health treatment services.
Keywords: alcohol use disorder, co-morbidity, severe mental disorders, Ethiopia
Background
Alcohol use disorder has been defined as a pattern of alcohol consumption that involves problem of controlling drinking and places the individual at an increased risk for acute or chronic harm.1 Patients with severe mental disorders have high rates of substance use disorders. Among the substance use disorders, alcohol is the most widely consumed by such patients.2,3 Again, people with an alcohol use disorder are more likely to have co-occurring mental disorders for which they frequently receive no treatment that addresses both conditions.4,5
In the United States, an estimated 18.7 million people have been diagnosed with alcohol use disorders.5 The prevalence of alcohol use disorder (AUD) is higher among those with severe mental disorders as compared with the general population.6–8 Different studies conducted in developing countries report that the co-morbidity rate of AUD in affective disorders and schizophrenia ranged from 11.3% to 56%5,6,9,10 and 9.7% to 74%5,6,11 respectively. The large variation in outcomes of such prevalence studies was attributed by the differences in study settings, study design, and AUD screening tool. Although there is a dearth of prevalence studies in sub-Saharan Africa countries; the existing literatures have noted high rates of AUD among individuals with severe mental disorders. For instant, the prevalence of AUD was 13.1% in Nigeria,12 29.6–45% in South Africa,13–15 59.3% in Tanzania16 and 68% in Kenya.17 Moreover, in Ethiopia reports showed that 24.5% to 39.1% of the patients with mental disorders had AUD18–20 which showed there is discrepancy across different geographical settings and at different periods.
Co-morbid AUD adversely affecting both the course and treatment outcomes of mental disorders.21–24 It can increase the incidence of physical conditions25–28 such as hypertension, cardiovascular disease, HIV/AIDS infection, cirrhosis and accidental injuries, and behavioral disorders29–31 like suicidality, violent/aggressiveness and social relationship problems. Again, it can result in non-adherence to treatments, relapse of psychiatric disorders, an increase in the cost of health care, and the reduction of the quality of life among patients with severe mental disorders.21,22,32 Researchers reported that younger age,15,20 male sex,10,18 being unmarried,13 low educational level,10,20 unemployment,10,33 longer duration of illness,30,34 early age of onset of illness,34,35 and having schizophrenia and/bipolar disorders12,15 are the main factors that influence individuals with severe mental disorders to have AUD. Having poor social support,10,11 high perceived stress,11 use of other psychoactive substances like tobacco smoking,6,12,19 and family history of alcohol and other substance use disorders16,36 are also important contributing factors to AUD among patients with severe mental disorders.
Home-made alcoholic beverages like “tella” (local beer with alcohol content 2–4%), “areqe” (strong distilled liquor with alcohol content is up to 45%) and “tej” (honey wine with alcohol content 7–11%) are common in north-west Ethiopia (the study area) and used by anyone without the restriction of gender, age, and mental health status by ignoring its unfavorable consequence.37 AUD has emerged as a major challenge to clinicians who try to intervene patients with severe mental disorders. Apart from the above reasons, some of the previous studies done in Ethiopia did not use a standard AUD measurement scale like alcohol use disorder identification tool (AUDIT)18 and some of these studies were not exclusively focused on patients with severe mental disorders.19,20 Moreover, this study incorporated and examined the relevant clinical and psychosocial factors which were not considered in the previous studies. Thus, this study aimed to determine the prevalence and associate factors of AUD among patients with severe mental disorders attending psychiatric follow-ups in northwest Ethiopia.
Methods
Study Setting and Population
A cross-sectional study was conducted on patients with severe mental disorders attending psychiatric follow-ups at the University of Gondar comprehensive specialized hospital (UoGCSH), northwest Ethiopia, from May 2019 to June 2019. A total of 384 outpatients with severe mental disorders (psychotic, bipolar, and major depressive disorders) diagnosed by the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) criteria were selected for interviews using a systematic random sampling technique. Patients aged 18 years and above who were on psychiatric follow-ups during data collection and taking alcohol at the moment were included. Patients with severe mental disorders (eg, major depressive and bipolar I disorder with psychotic feature and schizophrenia) who were too ill to communicate were excluded.
Sample Size Determination and Sampling Procedure
The minimum required sample size was estimated by using single population proportion formula considering the following assumption38:
where
n= minimum sample size required for the study.
Z= standard normal distribution (Zα/2= 1.96) with confidence interval of 95% and α=0.05.
P= the prevalence of co-morbid alcohol use disorder taken from a previous study that was conducted in southwest Ethiopia: 38.9%.19
d= Absolute precision or tolerable margin of error (5%).
Five percent non-response was added (366x 0.05= 18) and 366+18= 384, and then the final calculated sample size for this study was 384.
On average, there had been an estimated of 1120 patients with severe mental disorders and yet taking alcohol visited the hospital in 2 months. A systematic random sampling technique was employed to select the participants until the required sample size fulfilled. The sampling fraction (k) was calculated to be 1120/384≈3. The first participant was chosen randomly by a lottery method from numbers 1–3. Then, every third patient was interviewed and then the chart was reviewed regularly in each follow-up day. Of those, 370 patients were willing and interviewed the questionnaire.
Data Measurement Instruments
Alcohol use disorder was assessed using the Alcohol Use Disorders Identification Test (AUDIT),39 which had a 10-item self-report alcohol screening instrument that emphasizes the identification of alcohol use disorder in the last 12 months. It is developed by the World Health Organization that has been found most effective in identifying persons with drinking problems such as hazardous drinking, harmful drinking, and alcohol dependence as well as AUD. It provides professionals with the opportunity to take preventive measures and, hence, reduces the problems caused by this consumption. The questionnaire covers the domains of alcohol consumption (items 1–3), drinking behaviors (items 4–6), and alcohol-related problems (items 7–10). Each question was scored from 0 to 4. The score ranged from 0 to 40, and the scores 0–7, 8–15, 16–19, and ≥20 stood for non-hazardous drinking, “hazardous alcohol use”, “harmful drinking”, and “alcohol dependence” respectively.39 For this study, a cut-off point of “8” and above was considered as AUD.19
The Oslo Social Support Scale (OSSS), which had three items, was used to assess the level of the participant’s social support.40 The scale ranged from 3 to 14, and the scores 3–8, 9–11, and 12–14 showing “poor”, “moderate”, and “strong” social support, respectively. Perceived stress was measured by a 10-item perceived stress scale (PSS)41 which evaluated the level of perceived stress in the last month. The tool consisted of a 5-point Likert scale to which responses varied from never to very often. The PSS-10 scores were obtained by reversing the scores on the four positive items (4, 5, 7, and 8) and summing across all 10 items. The total score ranged from 0 to 40 and the scores 0–13, 14–26, and 27–40 indicated “low”, “moderate”, and “high” perceived stress, respectively. Those patients who use other substances such as Khat, tobacco, cannabis and shisha for non-medical purposes in the last 3 months considered positive for current other specified substance use. Items on socio-demographic, illness-related, and family-related conditions were prepared in accordance with previous works of literatures.18,35 The socio-demographic, illness and family-related characteristics of the participants included gender, age, marital status, educational level, religion, occupation, type of diagnosis, duration of illness, age of onset of illness, history of admission, family history of alcohol use problem, family history of other substance use, and family history of mental illness.
Data Collection Method
The data were collected using a structured questionnaire by both interviews and reviews of patient records. The questionnaire was first prepared in English and then translated to Amharic, the working language of Ethiopia, and back to English to maintain consistency. The questionnaire contained socio-demographic characteristics, family-related conditions, other substance use conditions, PSS, OSSS, illness-related, and AUDIT questions. First, every participant was interviewed about their socio-demographic characteristics, family-related conditions, other substance use conditions, PSS, OSSS, and AUDIT, and then reviewed the patients chart records regularly regarding illness-related variables (eg, type of diagnosis, number of hospital admission, duration of illness and treatment). Data were collected by five trained mental health professionals by the Amharic version of the questionnaire for 2 months. Furthermore, the overall data collection process was closely supervised by the principal investigator and one mental health professional.
Statistical Analysis
The data were analysed using the Statistical Package for Social Sciences (SPSS) version-20. Descriptive and binary logistic regression analyses were employed to see the frequency distribution and assess the correlation between independent variables and AUD, respectively. Variables with p-value ≤0.2 in univariate logistic model were selected for inclusion in the multivariate logistic regression. The adjusted odds ratio (AOR) with a 95% confidence interval was employed to report the results of factors associated with AUD. A p-value of <0.05 was considered as statistically significant. The model goodness of fit was assessed by Hosmer and Lemeshow test. The model was well fitted for this study as the test p-value result was 0.15. In addition, multi-collinearity was tested using the variance inflation factor (VIF). In this study, the VIF was less than 5 for each independent variable, indicating there was no significant multi-collinearity between independent variables.
Results
Socio-Demographic Characteristics of the Participants
A total of 370 patients took part in the study with a response rate of 96.3%. More than half of the participants (52.7%) were male; 168 (45.4%) were between 25 and 34 years of age, and 192 (51.9%) were unmarried. Majority (66.5%) were Orthodox Christians; 144 (38.9%) were unemployed and 166 (44.9%) unable to read and write (Table 1).
 |
Table 1 Socio-Demographic Characteristics of Patients with Severe Mental Disorders at UoGCSH (n=370) |
Illness, Psychological, Substance Use, and Family-Related Characteristics
Of the respondents, 135 (36.5%) were living with the illness for 6–10 years and 139 (37.6%) were developed the illness before they were 25 years of age. More than half of the participants (54.1%) had psychotic disorders (schizophrenia, schizoaffective, and brief psychotic disorders), and one-fourth (25.1%) had a history of hospital admission. Nearly half of the participants (46.8%) and (47.8%) had moderate social support and moderate perceived stress, respectively. At the moment, 131 (35.4%) and 80 (21.6%) of the participants were using Khat and tobacco, respectively. More than one-fourth (26.8%) had a family history of heavy alcohol use (Table 2).
 |
Table 2 Illness, Behavioral, Psychological and Family-Related Characteristics of Patients with Severe Mental Disorders at UoGCSH (n=370) |
The Prevalence of Alcohol Use Disorder (AUD)
The prevalence of AUD among patients with severe mental disorders was 36.2% with a 95% CI of 31.6 to 41.3. Of the participants, 87 (23.5%), 32 (8.6%), and 15 (4.1%) had hazardous, harmful alcohol drinking and alcohol dependency, respectively. The prevalence of AUD among patients with psychotic, bipolar, and major depressive disorders was 44.5%, 44.8%, and 16.04%, respectively.
Factors Associated with Alcohol Use Disorder (AUD)
As indicated in Table 3, the univariate binary logistic regression showed that sex, age, religion, social support, perceived stress, type of diagnosis, admission, duration of illness, current tobacco use, family history of heavy alcohol use, and family history of mental disorders were fulfilled the p-value <0.2. In multivariate logistic model: male sex, younger age (18–24 years), bipolar and psychotic disorders (schizophrenia, schizoaffective and brief psychotic disorders), poor social support, high and moderate perceived stress and tobacco smoking at the moment were significantly associated with AUD at p-value <0.05.
 |
Table 3 Univariate and Multivariate Binary Logistic Regression of Factors Associated with AUD Among Patients with Severe Mental Disorders (n=370) |
The odds of having AUD among male patients with severe mental disorders was 3.15 times [AOR = 3.15; 95% CI: 1.68–7.89] higher as compared to females. The odds of AUD was 4.7 [AOR= 4.71; 95% CI: 1.44–9.46] times higher in young adult patients (18–24 years) than older individuals. Individuals who had poor social support had 2.54 times [AOR= 2.25; 95% CI: 1.04–4.82] higher odds of AUD compared with patients who had strong social support. Patients diagnosed with psychotic [AOR= 2.96, 95% CI: 1.56–7.45] and bipolar disorders [AOR= 3.56, 95% CI: 1.59–8.33] were 3 and 3.56 times more likely, respectively, to develop AUD than patients diagnosed with major depressive disorder. The participants who had moderate [AOR=2.47, 95% CI: 1.21–5.04] and high [AOR=4.25, 95% CI: 2.14–9.38] perceived stress were 3 and 4 times more likely, respectively, to develop AUD than those with low perceived stress. Individuals who were smoke tobacco at a moment had 3.41 [AOR= 3.41; 95% CI: 1.34–8.39] times higher odds of AUD as compared to those who had not smoke tobacco.
Discussion
This study attempted to assess the prevalence and associate factors of AUD among patients with severe mental disorders attending psychiatric follow-ups at public hospital in northwest Ethiopia. This study reported that 36.2% of the patients with severe mental disorders had AUD. This finding was in line with the studies done in south-west Ethiopia,19 South Africa14 and Australia.32 This result was lower than the studies conducted in Kenya17 and Tanzania.16 The reason for the discrepancy could be due to the differences in study population and study design. In this study, the participants were patients with severe mental disorders using cross-sectional study, whereas the participants of Kenya’s study were patients with depression before and after the treatment, and the Tanzania’s study was a case study on substance use patients with mental disorders. Furthermore, the degree of openness with which people report their experiences of alcohol consumption might be varied across countries that might account for the variation.
On the other hand, the prevalence of the current study was higher than those of studies done in Addis Ababa, Ethiopia,20 Nigeria,12 Sri Lanka,10 Singapore6 and India.21 The discrepancy might be due to most of the indicated studies were measured by using different tools like ASSIST. And the participants were patients with bipolar disorders, and depression and schizophrenia in the Addis Ababa’s and Singapore’s studies, respectively. While the participants in this study were patients with severe mental disorders, and AUDIT was used to measure AUD. The other important factor for the variation is the difference in availability and patterns of alcohol use in the general population across countries.37,42 Alcoholic beverages like Tella, Areqe, Tej and Beer are common in the study area37 that might be contributed to the high prevalence of AUD. This study had shown that many patients with severe mental disorders have co-morbid AUD and suffering from negative health effects of alcohol drinking in Ethiopia. So, early intervention and integrating treatment services for co-existing mental disorders and AUD is important to detect cases at early stage and prevent further consequences of the problem. Again, appropriate treatment of co-morbidities helps to improve patient and family quality of life.
Some of the factors associated with AUD in this study were different from those of previous studies in Ethiopia. In this study, we observed that age of the patients is the important contributor for AUD in which the odds of developing AUD was higher among young (18–25 years) patients than older ones. This report was consistent with other previous studies.16,20,30 This might be because there are multiple influences on higher rates of alcohol and other drug use among young people, and it is a common finding across countries.43,44 Furthermore, the young part of the patients have higher rate of substance use, with adolescence is the critical period for initiation and the young age is the peak age for substances use. Secondly, patients with the diagnosis of psychotic and bipolar disorders was more likely to develop AUD compared with those with the diagnosis of major depressive disorder. This finding is consistent with other previous studies.5,12,45 This might be because people with such diagnoses have highly impaired judgment and insight as well as aggressiveness and uncontrolled emotions. These have been consistently associated with heavy and problematic alcohol use among psychotic and bipolar disorder patients.46
Poor social support was also significantly associated with AUD among patients with severe mental disorders. The result is consistent with the finding of previous studies10,11 which showed that strong social support decreases the odds of AUD and protects patients against alcohol use–related problems. The possible reason might be that, lack of social support might results in psychological distress, feeling of loneliness, helplessness and perceive themselves as disadvantaged. Then, they drink alcohol to avoid such negative feelings. Fourthly, moderate and high perceived stress was more likely to lead AUD compared to low perceived stress. This finding is consistent with that of another study.11 This could be due to individuals with stress and negative emotions drink alcohol to cope with stress/depression and escape from negative feelings. Moreover, patients who had smoked tobacco currently were more likely to develop AUD compared to those who had not smoked tobacco. This result is in line with those of previous studies.19,47,48 This might be patients with bipolar and psychotic disorders smoke tobacco as self-medication which helps in reducing positive symptoms and improving cognitive functions. Additionally, it might be due to behavioral concordance of using one type of substance with other substances, and, perhaps the illness dictates the use of multiple substances at the same time.
One of the main limitations of this work might be social desirability bias in that individuals might have provided socially palatable to face-to-face interview questions relating to alcohol and other substance use. The AUDIT is a screening instrument, not a diagnostic tool that could potentially influence magnitude estimates. Besides, the research could not show the cause–effect relationships between factors and outcomes owing to its cross-sectional nature.
Conclusion
In conclusion, the prevalence of AUD among patients with severe mental disorders was high. Male sex, younger age, psychotic and bipolar disorders, poor social support, moderate and severe perceived stress, and tobacco smoking at the moment were the factors associated with the problem. Early intervention and integrating treatment services for co-existing mental disorders and alcohol use disorder is important. The special treatment, where it is offered for young male patients and those with the diagnosis of psychotic and bipolar disorders, is recommended to promote the uptake of alcohol and mental health treatment services. Screening for current tobacco smoking is also useful to detect cases at early stage and prevent further consequence of the problem. Prospective studies are also recommended to assess the cause–effect relationships between psychotic and bipolar disorder diagnosis, tobacco smoking, social support and perceived stress level with AUD in local setting.
Data Sharing Statement
All the datasets used to the study are available within the manuscript.
Ethics Approval and Consent to Participate
Ethical clearance and approval was obtained from the University of Gondar, College of Medicine and Health Sciences, and Department of psychiatry. The ethics committee had understood and approved the ethics that the participants were 18 and above years of age, and can provide informed consent on their own behalf. This study was conducted in accordance with the Declaration of Helsinki. Before data collection, the participants who were unable to read and write, the consent form was read by the data collectors and if they are interested a fingerprint was obtained and for those who can read and write, written consent was obtained. So an informed written consent was obtained from the participants, and confidentiality was maintained by omitting their identification.
Acknowledgments
The authors acknowledge data collectors, supervisors and study participants for their time and effort.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
The authors have no received any funds from the external body to conduct the study.
Disclosure
The authors declare that they have no conflict of interest.
References
1. World Health Organization. Global Status Report: Alcohol Policy. World Health Organization; 2004.
2. Cuffel BJ. Comorbid substance use disorder: prevalence, patterns of use, and course. New Dir Ment Health Serv. 1996;1996(70):93–105. doi:10.1002/yd.23319960209
3. Nery FG, Soares JC. Comorbid bipolar disorder and substance abuse: evidence based options. Curr Psychiatry. 2011;10:57–66.
4. Whiteford HA, Degenhardt L, Rehm J, et al. Global burden of disease attributable to mental and substance use disorders: findings from the Global Burden of Disease Study 2010. Lancet. 2013;382(9904):1575–1586. doi:10.1016/S0140-6736(13)61611-6
5. Swendsen J, Conway KP, Degenhardt L, et al. Mental disorders as risk factors for substance use, abuse and dependence: results from the 10-year follow-up of the national comorbidity survey. Addiction. 2010;105(6):1117–1128. doi:10.1111/j.1360-0443.2010.02902.x
6. Subramaniam M, Mahesh MV, Peh CX, et al. Hazardous alcohol use among patients with schizophrenia and depression. Alcohol. 2017;65:63–69. doi:10.1016/j.alcohol.2017.07.008
7. Cobb S, Assari S. Psychiatric disorders and alcohol consumption among low-income African Americans: gender differences. Brain Sci. 2019;9(4):86. doi:10.3390/brainsci9040086
8. Davis L, Uezato A, Newell JM, Frazier E. Major depression and comorbid substance use disorders. Curr Opin Psychiatry. 2008;21(1):14–18. doi:10.1097/YCO.0b013e3282f32408
9. Farren CK, Hill KP, Weiss RD. Bipolar disorder and alcohol use disorder: a review. Curr Psychiatry Rep. 2012;14(6):659–666. doi:10.1007/s11920-012-0320-9
10. Hapangama A, Kuruppuarachchi K, Pathmeswaran A. Substance use disorders among mentally ill patients in a General Hospital in Sri Lanka: prevalence and correlates. Ceylon Med J. 2013;58(3):111. doi:10.4038/cmj.v58i3.6103
11. Gammeter R, Nay C, Bissery A, et al. Frequency of alcohol use disorders in patients admitted in a psychiatric hospital according to admission diagnosis. Schweizer Archiv für Neurologie und Psychiatrie. 2006;157(6):290–296.
12. Akinsulore A, Fatoye FO, Awaa O, Aloba O, Mapayi B, Ibigbami O. Psychoactive substance use among psychiatric out-patients in a Nigerian teaching hospital. Niger J Psychiatry. 2012;10:2–7.
13. Paruk S, Ramlall S, Burns J. Adolescent-onset psychosis: a 2-year retrospective study of adolescents admitted to a general psychiatric unit. S Afr J Psychiatr. 2009;15(4):7. doi:10.4102/sajpsychiatry.v15i4.203
14. Pengpid S, Peltzer K, Heever H. Prevalence of alcohol use and associated factors in urban hospital outpatients in South Africa. Int J Environ Res Public Health. 2011;8(7):2629–2639. doi:10.3390/ijerph8072629
15. Taukoor B, Paruk S, Karim E, Burns JK. Substance use in adolescents with mental illness in Durban, South Africa. J Child Adolesc Ment Health. 2017;29(1):51–61. doi:10.2989/17280583.2017.1318395
16. Hauli KA, Ndetei DM, Jande MB, Kabangila R. The prevalence of substance use among psychiatric patients: the Case Study of Bugando Medical Centre, Mwanza (Northern Tanzania). Substance Abuse. 2011;32(4):238–241. doi:10.1080/08897077.2011.599253
17. Kuria MW, Ndetei DM, Obot IS, et al. The association between alcohol dependence and depression before and after treatment for alcohol dependence. ISRN Psychiatry. 2012;2012:1–6. doi:10.5402/2012/482802
18. Duko B, Ayano G, Bekana L, Assefa D. Prevalence and correlates of co-occurring substance use disorder among patients with severe mental disorder at Amanuel Mental Specialized Hospital, Addis Ababa, Ethiopia. J Neuropsychopharmacol Ment Health. 2015;1:101.
19. Zenebe Y, Negash A, Feyissa G, Krahl W Alcohol use disorders and its associated factors among psychiatric outpatients in Jimma University Specialized Hospital, Southwest Ethiopia. 2015.
20. Tensae DW, Haddis Solomon BB, Getinet W, Getinet W. Prevalence and correlates of alcohol use disorders among bipolar patients at Amanuel Mental Specialized Hospital, Addis Ababa (Ethiopia): a Cross-Sectional Institution Based Study. J Public Health Africa. 2018;9(3). doi:10.4081/jphia.2018.829
21. Chakraborty R, Chatterjee A, Chaudhury S. Impact of substance use disorder on presentation and short-term course of schizophrenia. Psychiatry J. 2014;14(2).
22. Dixon L. Dual diagnosis of substance abuse in schizophrenia: prevalence and impact on outcomes. Schizophr Res. 1999;35:S93–S100. doi:10.1016/S0920-9964(98)00161-3
23. Lynskey MT. The comorbidity of alcohol dependence and affective disorders: treatment implications. Drug Alcohol Depend. 1998;52(3):201–209. doi:10.1016/S0376-8716(98)00095-7
24. Menezes PR, Johnson S, Thornicroft G, et al. Drug and alcohol problems among individuals with severe mental illnesses in South London. Br J Psychiatry. 1996;168(5):612–619. doi:10.1192/bjp.168.5.612
25. Rehm J, Baliunas D, Borges GL, et al. The relation between different dimensions of alcohol consumption and burden of disease: an overview. Addiction. 2010;105(5):817–843.
26. Rehm J. The risks associated with alcohol use and alcoholism. Alcohol Res Health. 2011;34(2):135.
27. Opreanu RC, Kuhn D, Basson MD. Influence of alcohol on mortality in traumatic brain injury. J Am Coll Surg. 2010;210(6):997–1007. doi:10.1016/j.jamcollsurg.2010.01.036
28. Egbe CO, Dakum PS, Ekong E, Kohrt BA, Minto JG, Ticao CJ. Depression, suicidality, and alcohol use disorder among people living with HIV/AIDS in Nigeria. BMC Public Health. 2017;17(1):542. doi:10.1186/s12889-017-4467-5
29. Oquendo MA, Currier D, Liu S, Hasin D, Grant B, Blanco C. Increased risk for suicidal behavior in comorbid bipolar disorder and alcohol use disorders. J Clin Psychiatry. 2010;71(7):902. doi:10.4088/JCP.09m05198gry
30. Sadock BJ, Sadock VA. Kaplan and Sadock’s Synopsis of Psychiatry: Behavioral Sciences/Clinical Psychiatry. Lippincott Williams & Wilkins; 2011.
31. Berglund M. Suicide in alcoholism: a Prospective Study of 88 suicides: the multidimensional diagnosis at first admission. Arch Gen Psychiatry. 1984;41(9):888–891. doi:10.1001/archpsyc.1984.01790200070009
32. Margolese HC, Malchy L, Negrete JC, Tempier R, Gill K. Drug and alcohol use among patients with schizophrenia and related psychoses: levels and consequences. Schizophr Res. 2004;67(2–3):157–166. doi:10.1016/S0920-9964(02)00523-6
33. Maremmani AG, Bacciardi S, Gehring ND, et al. Substance use among homeless individuals with schizophrenia and bipolar disorder. J Nerv Ment Dis. 2017;205(3):173–177. doi:10.1097/NMD.0000000000000462
34. Hong M, Ha TH, Lee S, Oh S, Myung W. Clinical correlates of alcohol use disorder in depressed patients with unipolar and bipolar disorder. Psychiatry Investig. 2019;16(12):926. doi:10.30773/pi.2019.0182
35. Brunette MF, Mueser KT, Babbin S, et al. Demographic and clinical correlates of substance use disorders in first episode psychosis. Schizophr Res. 2018;194:4–12. doi:10.1016/j.schres.2017.06.039
36. Higuchi S, Nakamura I, Shibasaki Y. Prevalence of problem drinking in patients with depression and association between depression severity and problem drinking: a cross-sectional survey. Clin Neuropsychopharmacol Ther. 2019;10:1–9. doi:10.5234/cnpt.10.1
37. Fekadu A, Alem A, Hanlon C. Alcohol and drug abuse in Ethiopia: past, present and future. Afr J Drug Alcohol Stud. 2007;6:40–53.
38. Pagano M, Gabuvereau K. Principles of Biostatistics.
39. Babor T, Higgins-Biddle J, Saunders J, Monteiro M. Audit: The Alcohol Use Disorders Identification Test.
40. Dalgard OS, Bj S, Tambs K. Social support, negative life events and mental health. Br J Psychiatry. 1995;166(1):29–34. doi:10.1192/bjp.166.1.29
41. Cohen S, Kamarck T, Mermelstein R. Perceived stress scale. In: Measuring Stress: A Guide for Health and Social Scientists. 1994:235–283.
42. Meier BJ, El-Gabri D, Friedman K, et al. Perceptions of alcohol use among injury patients and their family members in Tanzanian society. Alcohol. 2020;83:9–15. doi:10.1016/j.alcohol.2019.06.001
43. Hall WD, Patton G, Stockings E, et al. Why young people’s substance use matters for global health. Lancet Psychiatry. 2016;3(3):265–279. doi:10.1016/S2215-0366(16)00013-4
44. Stockings E, Hall WD, Lynskey M, et al. Prevention, early intervention, harm reduction, and treatment of substance use in young people. Lancet Psychiatry. 2016;3(3):280–296. doi:10.1016/S2215-0366(16)00002-X
45. Barnes TR, Mutsatsa SH, Hutton SB, Watt HC, Joyce EM. Comorbid substance use and age at onset of schizophrenia. Br J Psychiatry. 2006;188(3):237–242. doi:10.1192/bjp.bp.104.007237
46. Galanter M, Kleber HD, Brady K. The American Psychiatric Publishing Textbook of Substance Abuse Treatment. American Psychiatric Pub; 2014.
47. D’Onofrio G, Becker B, Woolard RH. The impact of alcohol, tobacco, and other drug use and abuse in the emergency department. Emerg Med Clin. 2006;24(4):925–967. doi:10.1016/j.emc.2006.06.008
48. Soboka M, Tesfaye M, Feyissa G, Hanlon C. Alcohol use disorders and associated factors among people living with HIV who are attending services in south west Ethiopia. BMC Res Notes. 2014;7(1):828. doi:10.1186/1756-0500-7-828
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.