Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
Alcohol and Dementia – What is the Link? A Systematic Review
Authors Wiegmann C, Mick I, Brandl EJ, Heinz A, Gutwinski S
Received 3 October 2019
Accepted for publication 22 December 2019
Published 9 January 2020 Volume 2020:16 Pages 87—99
DOI https://doi.org/10.2147/NDT.S198772
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Roger Pinder
Caspar Wiegmann, 1 Inge Mick, 1 Eva J Brandl, 1 Andreas Heinz, 1, 2 Stefan Gutwinski 1
1Department of Psychiatry and Psychotherapy, Psychiatric Hospital of Charité at St. Hedwig Hospital, Berlin, Germany; 2Department of Psychiatry and Psychotherapy, Charité University Medicine Berlin, Berlin, Germany
Correspondence: Caspar Wiegmann
Psychiatrische Universitätsklinik der Charité im St. Hedwig-Krankenhaus, Große Hamburger Str. 5-11, Berlin-Mitte 10115, Germany
Tel +49 30 2311 2500
Email [email protected]
Background: Dementia is a globally increasing health issue and since no cure is currently available, prevention is crucial. The consumption of alcohol is a controversially discussed risk factor for dementia. While many previously published epidemiological studies reported a risk reduction by light to moderate alcohol consumption, there is no persuasive model of an underlying biochemical mechanism. The purpose of this article is to review current models on alcohol neurotoxicity and dementia and to analyze and compare studies focusing on the epidemiological link between alcohol consumption and the risk of dementia.
Methods: The electronic database Pubmed was searched for studies published between 1994 and 2019 concerning the topic.
Results: Available epidemiological studies are not sufficient to verify a protective effect of alcohol on dementia development.
Keywords: alcohol neurotoxicity, Alzheimer’s disease, vascular dementia, Korsakoff´s syndrome
Introduction
Psychotropic drugs are used in nearly every culture and alcohol is the most widely and growingly used recreational drug.1 Worldwide, only approximately 57% of the population have not consumed alcohol in the past 12 months.1 There is a broad consensus that consumption of alcohol has potential adverse effects and is of relevance to the global burden of disease. Over 200 health conditions are linked to harmful alcohol use, ranging from liver diseases, road injuries and violence,2 to cancers, cardiovascular diseases, suicides, tuberculosis, and HIV/AIDS.3
Dementia is considered a health priority by the WHO.4 It has been estimated that 50 million people suffer from dementia and 10 million people are newly diagnosed with dementia every year.4 A helpful distinction is made between primary and secondary dementia pathologies. Primary dementia forms include neurodegenerative dementia (Alzheimer’s disease, frontotemporal dementia, Lewy-body-dementia) and vascular dementia. Secondary dementias are a consequence of other underlying systemic or brain diseases (such as vitamin deficiencies, infectious diseases, brain tumors).5 To date, there is no effective cure for primary dementia forms, apart from arguable effects of acetylcholine esterase inhibitors (rivastigmine, donepezil, galantamine) and NMDA-receptor-antagonist (memantine) for Alzheimer’s disease.6 Therefore, the WHO considers the risk reduction a cornerstone of the action plan against dementia and seeks to strengthen the evidence base, share and disseminate evidence to support policy interventions for reducing potentially modifiable risk factors for dementia.4
In Alzheimer’s disease, two main pathologies are detected microscopically: intracellular aggregates of hyperphosphorylated tau protein, a microtubule-associated protein in neurofibrillary tangles and dystrophic neuritis7 and extracellular plaques of mainly insoluble A-beta-40 fragments of the protein beta-Amyloid-precursor protein (β-APP).8 The relation of cause and effect or interplay of these two pathologies with neurodegeneration and dementia is subject of ongoing debates. Vascular dementia is the result of microangiopathy and macroangiopathy. Since the etiology of the two most common forms of dementia is very different, the consumption of alcohol might interact via different biochemical pathways in dementia development and lead to variable effects dependent on the dementia type.
The effects of light to moderate alcohol consumption on cognitive performance and the risk of developing dementia are controversially discussed,9–11 especially concerning the effect of low-dose alcohol on vascular pathology and the existence of a specific alcohol-related dementia.12,13 Different dosage definitions, ages at study inceptions, risk stratifications, lengths of assessment intervals and lengths of studies lead to different outcomes in a large number of epidemiological studies concerning low-dose alcohol consumption, while high doses of alcohol have most certainly a detrimental effect on cognitive performance14 and can lead to a specific amnestic syndrome (Korsakoff´s syndrome).
The purpose of this review is to give an overview about the dose- and pattern-related effects of alcohol on the risk of developing dementia, while trying to differentiate different neurodegenerative, vascular and other forms of dementia. The first part of the review will give an overview about alcohol effects on the central nervous system and summarize findings with different methodological approaches (biochemical methods, histopathological findings, animal models, neuroimaging). In the second part, we will present the results of a systematic literature search we conducted. We will then address the question whether alcohol consumption constitutes a potential target for dementia prevention.
Methods
For the narrative review of alcohol effects and neurotoxicity, no systematic approach was applied. For the systematic review of epidemiological findings, the electronic database PubMed was searched to locate scholarly research examining the relationship between alcohol and dementia. The keywords dementia and alcohol were used as search entry. All studies with at least 250 participants, standardized dementia diagnosis protocol and published in English between the 1st of January 1994 and 1st of September 2019 were analyzed for information concerning the link between alcohol consumption and dementia risk. Gender-specific or beverage-specific studies were excluded from analysis for pragmatic reasons: discussion of gender-specific effects or beverage-specific effects (for example, wine15) are beyond the scope of this review.
Alcohol Effects on the Central Nervous System
Acute Effects of Alcohol
Alcohol usually refers to the molecule ethanol. As amphiphile, it is rapidly absorbed from the stomach and duodenum after oral consumption and passes the blood-brain barrier. The distribution and elimination show strong variability due to fed- or fasting state, drinking patterns, age, and genetics.16 In the central nervous system, ethanol modulates the function of multiple receptors: voltage-gated calcium channels and glutamate receptors are inhibited by alcohols, whereas some others, such as g-amino butyric acid type A (GABA-A) receptors, glycine receptors, n-acetylcholine- and 5-HT3-receptors, are potentiated.17,18 Prior hypotheses on the effect of alcohol on cell membrane function in the central nervous system are viewed as less relevant to its acute effects.17 The effects appear to be dose-related, since at low dosages alcohol affects monoaminergic transmission and produces disinhibition and euphoria, while at high dosages anxiolytic and sedative effects are more prominent, mediated through increasing GABA activity and inhibiting excitatory amino acids.18
Molecular Mechanisms of Central Nervous System Toxicity
In humans, chronic alcohol exposure leads to in vivo up- (glutamate) and down-regulation (D2,19 GABAA) of neuroreceptors availability related to alcohol withdrawal and craving (for a summary see Heinz et al20). Genetic constitution interacts with monoaminergic dysfunction in alcohol withdrawal.21
Adaptive up-regulation of NMDA-receptors and consecutively enhanced calcium influx is supposed to contribute to cell apoptosis (“excitotoxicity”) and link the acute and chronic effects of alcohol consumption.22,23 The alcohol metabolite acetaldehyde was shown to be directly neurotoxic.24 Other probable causes of cell death are inflammatory processes via release of pro-inflammatory cytokines and microglia activation after high dose alcohol consumption25 Alcohol activates oxidases in the central nervous system, which leads to formation of free radicals and cell membrane damage.23 Homocysteine is elevated after consumption of higher doses of alcohol and might contribute to alcohol-related brain damage.26
While these three mechanisms may underlie the more unspecific neurotoxic effects of alcohol, thiamine deficiency caused by malnutrition and decreased intestinal absorption in alcohol-dependent individuals shows distinct neuroanatomical patterns and symptomatology, which Karl Wernicke and Sergei Korsakoff already described accurately in the nineteenth century.27,28 While Wernicke specified an acute neurological syndrome (stand and gait ataxia, confusion, and ophthalmoplegia), the overlapping Korsakoff’s syndrome refers to more chronic alterations and symptoms, mainly perseveration and anterograde amnesia. Marchiafava-Bignami-Syndrome refers to thiamine deficiency-associated degeneration of the corpus callosum presenting with a wide array of symptoms including altered mental state, loss of consciousness and epileptic seizures (see Table 1 for details)29
 |
Table 1 Thiamine Deficiency Associated Neurological Syndrome |
Neurotoxicity of acetaldehyde, excitotoxicity and via NDMA-activation, neuroinflammation, and formation of free radicals as neuroanatomically less specific mechanism of neurotoxicity and thiamine deficiency with specific neuroanatomical susceptibility (corpora mammillaria, dorsal thalamic nucleus, hippocampus, periaqueductal region, corpus callosum) supposedly amplify each other, explaining the variety of neuropsychiatric symptoms in patients with high levels of alcohol consumption (for details on the correlation of etiology of neurotoxicity with neuroanatomy see Zahr et al30).
The entity of alcohol-related dementia is still controversially discussed, since there is no consensus whether it represents an entity distinct from Korsakoff syndrome and neuropsychological findings are unspecific,31 encompassing cortical and subcortical patterns.12
Neuropathology and Neuro-Imaging Studies
Autopsy studies show mild cerebral atrophy and lower mean brain weight in cases of uncomplicated alcoholism, when the individual was not affected by Wernicke-Korsakoff encephalopathy.32 The loss in brain volume is mainly attributed to white matter loss,30 diffusion tensor magnetic resonance imaging (MRI) show fiber tract degeneration.33 Histopathological studies revealed cerebellar atrophy,34 corpus callosum thinning35 and pyramidal cell loss in superior frontal and frontal association cortex,35 as well as neuronal dendritic shrinkage, presumably reversible in abstinence.32
MRI studies have generally confirmed postmortem studies by demonstrating that these patients have regional cortical volume deficits,36–38 conceptualized as accelerated aging.38 MRI cohort studies show a correlation between cerebral volume decrease and number of drinks consumed.39,40
Potentially Positive Effects of Alcohol on the Central Nervous System
A variety of positive effects on alcohol on the central nervous system have been suggested, mainly by reducing cardiovascular risk factors. Robust evidence exists for elevated high-density lipoprotein cholesterol blood levels,41 even exceeding the effect of drugs prescribed for dyslipidemia.42 The coagulation cascade is modulated by alcohol intake through down-regulation of fibrinogen, a substrate of blood clot formation.43 In vitro experiments show an inhibition of platelet aggregation.44,45 High alcohol consumption leads to higher blood pressure, while low to moderate consumption (equal or less than two drinks per day) has no effect.44,46 The definition of one standard drink and consecutively, the definition of low-risk consumption varies in-between countries.47 In the references cited, the US-American definition is used: one drink equals 14 g of pure alcohol. Nevertheless, any alleged positive aspects of drinking on cardiovascular risk factors must be weighed against seriously harmful effects, including changes in circulation, inflammatory response, oxidative stress, as well as anatomical damage to the cardiovascular system, especially the heart itself.48
Effects of Alcohol on Alzheimer’s Pathology
Regarding effects of alcohol on Alzheimer’s disease (AD)-related pathophysiology, only scarce and contradictory evidence exists: in rats, alcohol application leads to higher acetylcholine release in the hippocampus in low concentrations, while higher concentrations inhibit acetylcholine release.49 The hippocampus is affected early in Alzheimer’s disease by formation of neurofibrillary tangles and neurodegeneration, leading to the typical early symptom of disability to memorize new information.7
Findings from transgenic mouse models and cell culture models of AD are contradictory. One study on transgenic AD-mice and hippocampal cell cultures established a possible mechanism of lower Aß-toxicity through alcohol administration by reducing Aß-induced synaptic failure,50 another study argued that Aß-aggregation is reduced in cell cultures treated with alcohol.51 However, findings from experiments conducted in a different transgenic mouse model fed with alcohol showed higher expression of APP and ß-secretase with consecutively elevated amyloid deposition and neurotoxicity.52
Alcohol is supposed to enhance neuroinflammation and thereby enhancing neurotoxicity of the ß-amyloid cascade.53 In vitro studies suggest that alcohol might impede phagocytosis of ß-amyloid by microglia and thereby promote Alzheimer’s disease.54
In summary, while a number of studies have reported experimental findings to explain risk reduction through alcohol consumption for vascular dementia, data regarding the impact of alcohol on Alzheimer´s pathophysiology is more contradictory.
Epidemiological Findings Concerning Alcohol Consumption and Risk of Dementia
A number of studies have been published with different study designs (cross-sectional studies, cohort studies, case–control studies), inclusion criteria, alcohol dosage standardization and dosage definitions, type of alcoholic beverages included, endpoints (only Alzheimer’s disease or all forms of dementia, mild cognitive impairment), and consumption patterns. Therefore, these studies show very heterogeneous outcomes.10,55–67
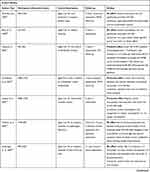
While a selection of studies is discussed below, Table 2 gives an overview of all published studies in compliance with our inclusion criteria.
Case-Control-Studies
A case-control-study of Bachmann and colleagues analyzed three assumed major risk factors (head trauma, smoking, and alcohol consumption) for dementia in African and European American individuals with dementia and their non-affected siblings.68 Alcohol consumption over an average of 0.25 US standard drinks per day was associated with a lower dementia risk in both groups (relative risk 0.88/0.82, respectively). Risk factors did not vary in African and European Americans, while exposure was different in both groups (the proportion of European Americans drinking more than 0.25 drinks was higher than in African-Americans). A study by Mukamal and colleagues showed a higher effect (OR 0.46 for 1–6 standard US drinks, 0.69 for 7–13 standard US drinks) for low to moderate consumption in a subsample analysis of the Cardiovascular Health Study. Three hundred and seventy-three incident dementia cases were compared to 373 control cases, while thoroughly controlling for various covariates (educational level, marital status, income, diabetes, physical activity, ApoE-genotype).69
Cohort Studies
In the Rotterdam Study, a large cohort study in elderly patients (over 55 years old), moderate drinking was defined as 1–3 drinks per day (no definition of grams of alcohol) and resulted in a significant risk reduction for any form of dementia, especially vascular dementia (RR 0.58/0.29, 38 patients in the group of moderate drinkers (1443) compared to 62 in the group of abstainers (1113)). The study achieved a high follow-up rate of 99.7% within an average follow-up period of 6 years. The analysis was adjusted for age, sex, systolic blood pressure, education, smoking, and body-mass index.10
The Whitehall-II-Study confirmed these findings in a younger population of civil service employees aged 35–55 years at study inclusion (9087 participants of which 397 developed dementia).70 Similar to the Rotterdam Study, the risk for developing dementia was lowest in the individuals consuming between 1 and 14 drinks per week compared to abstainers (UK standard drink equals 8 g of alcohol). The hazard ratio for abstinence compared to consumption of 1–14 drinks per week for developing any kind of dementia was 1.47, after adjusting for confounders (extensive assessment of sociodemographic data and cardiovascular health data). Diagnosis was made through linkage with mental health services data set, mortality register and national hospital episode statistics.
The 90+ study from California, USA could not establish alcohol as a risk factor for dementia for the very elderly living in a community for elderly people, after adjusting for age-, sex- and education. After an average follow-up period of 36 months, 268 of 587 had developed dementia of any kind. Neither the alcohol consumption at the first assessment (20 years earlier) nor on the second assessment at the age over 90 years correlated with the risk of dementia.58
Taking these three exemplary studies together, it may be argued that light-to-moderate alcohol consumption is protective against dementia in middle to late life, while the effect abates in the very elderly, but other cohort studies show different trends.
The HUNT study from Norway, encompassing almost the whole population of a community in Northern Norway (40,435 participants) showed no association of dementia with low or moderate alcohol consumption (only frequency of consumption measured).56 An analysis from one of the largest twin cohorts worldwide (12,326 participants, Sweden) corroborates the risk of consuming high amounts of alcohol, while showing no effect of light to moderate consumption.71 Relative to co-twins drinking light amounts, moderate-to-heavy drinking twins had a greater risk of dementia by 57% and reduced age of dementia onset by 4.76 years, while differences between lightly drinking or abstinent twins were non-significant.
An increasing number of cohort studies from different countries continue to be published. The results are heterogeneous concerning light to moderate consumption, while there is a consensus regarding high consumption and elevated dementia risk (see Table 2).
Cross-Sectional Studies
Since Alzheimer’s disease risk rises with ageing,10 an alternative approach to study the effect of alcohol on Alzheimer’s disease is to measure the age of onset in Alzheimer’s disease patients exposed to putative risk factors. An earlier average onset in exposed individuals is an indicator of a higher relative risk. A study of Harwood and colleagues in a cohort of patients with Alzheimer’s disease showed that patients who had been drinking more than two US standard drinks a day before the onset of dementia were on average diagnosed with dementia 4.1 years earlier than patients that never drank,72 a finding corroborated by the study of Handing and colleagues.71
Epidemiological Studies of Electronic Health Data
A recent retrospective study of hospital discharge records encompassing about 80% of all discharge diagnoses in France (31624156 patient records) established an alcohol use disorder as major risk factor for developing any form of dementia, especially early-onset dementia.9 The study set the diagnosis of alcohol use disorder in the patient discharge records in relation with a later diagnosis of a dementia. While this approach relies on hospital diagnoses with probable underestimation of both dementia and alcohol use disorders and although the amount of alcohol consumed could not be measured, the sample size and setting corroborates the high external validity of the results.73 A Danish study using hospital admissions records and a German study using general health practitioners' treatment records replicated these results.10,74
Meta-Analyses
A vast number of meta-analyses and reviews exist to the subject, mostly confirming the j-shaped correlation.75–79 It has to be taken into account that meta-analysis adopts and merges the partially variable definitions of light to moderate consumption. Anstey and colleagues included 22 studies with 29,868 participants on alcohol consumption and dementia of any cause or cognitive decline.77 Low to moderate consumption correlated to a lower OR for dementia (AD: 0.72 VaD: 0.75). A meta-analysis of Peters et al showed lower OR for any form of dementia in alcohol consumer than abstainers (OR for AD: 0.57, VaD: 0.82, 23 studies included).76 There was no information given on controlled variables and the total number of participants.
In a dose–response analysis by Xhu and colleagues, 10 studies were used to calculate the weekly dose associated with the lowest risk of dementia of any cause: 6 g per day.59 Varying assessment of consumption patterns (drinks per week, grams per day, etc.) complicated the analysis. A repeated evaluation using hazard ration rather than odds ratio is being planned.80 The publication of Ilomäki and colleagues gives an overview about existing reviews and metanalyses78 and observes a positive effect of light alcohol consumption (<14 units per week) on any form of dementia.
Discussion
What can we deduct from the vast amount of studies and meta-analyses concerning alcohol consumption and dementia risk? Obviously, only correlations and not causal relationships can be deducted from cohort studies, since unmeasured factors could be mainly responsible for both drinking behavior. Nonetheless, low to average alcohol consumption does not appear to increase the risk of dementia significantly or might be even protective. For both vascular dementia and Alzheimer’s disease low- to moderately consuming individuals show lower odds for dementia than abstainers and the effects are very similar when comparing the two dementia etiologies.10,69,70
This finding evidently leads us the question of possible confounders. The difficulty of assessing the consumed amounts is apparent; all reviews face rely on personal declaration of alcohol consumption and different definitions of one drink (from 8 g to 12 g of alcohol47,69,70) and the definition of heavy, moderate or light drinking vary.78 It can be assumed that underreporting of alcohol consumption is a limitation to all studies related to alcohol consumption. In a Norwegian study, encompassing 88% of the population of a region, the average self-reported alcohol intake was lower than the statistically expected dose from other sources (statistics on sale, estimated self production and tourist import of alcohol).56,81
The group of abstainers, which is commonly used as reference group, includes former drinkers, who are abstinent only because they already suffer from consequences of their former drinking habits or take medication incompatible with alcohol consumption (“sick quitters hypothesis”).69
The majority of studies do not assess lifetime drinking trajectories. One study showed smaller effects (less cognitive decline in moderate drinkers) if former drinkers (“quitters”) where excluded from the group of abstainers.82 Another study found a 20–60% higher risk of dementia in former drinkers.69 This must be viewed as a cogent objection to supposed protective effect of light-moderate alcohol consumption.
In the patient cohorts included, alcohol use disorders might be severely underrepresented. Over 95% of patients with Alzheimer’s disease have a late onset and are over 65 years of age. People with alcohol use disorder are estimated to have a much lower life expectancy. A Scandinavian register study showed that people hospitalized with alcohol use disorder have an average life expectancy of 47–53 years (men) and 50–58 years (women) and die 24–28 years earlier than people in the general population, even though they have, presumably in contrast to other countries, wide access to the health-care system.83 In a cohort study from Norway, the group that failed to report their alcohol consumption showed the highest risk of dementia, which might be explained by a high proportion of heavy drinking and consecutive high dementia risk.56 In a study of centenarians, on the other hand, only 11% of the centenarian-males and 22% of the centenarian-females indicated to never consume alcohol.84
The retrospective cohort study of Schwarzinger et al showed the impressive increase in (mainly early onset) dementia risk in patients with alcohol use disorder.9 This patient group would not have been included in most prospective cohort studies assessing alcohol use and dementia risk in the elderly, because of their already present dementia at the date of study inception.
Most prospective cohort studies have follow-up periods of two to three years (see Table 2). Due to the adverse effects on other organ systems22 and higher mortality of alcohol-consuming individuals, these subjects may decease in-between intervals (attrition bias). It has been shown that patients with Alzheimer’s disease-consuming alcohol decease more rapidly than abstinent AD patients.59 This attrition effect could lead to an underestimation of AD cases in the group of drinkers especially in prospective cohort studies with long intervals between data acquisition.
Alcohol consumption is a marker for several lifestyle factors. Moderate drinkers are leaner, more physically active, more often married and of higher socioeconomic status. A higher socioeconomic status (highest education degree and income) is associated with higher alcohol consumption in the elderly.85 Higher education and higher socioeconomic status are known to be protective against dementia.86 The lower risk of developing dementia in moderate drinkers might therefore be, at least partially be explained by their higher socioeconomic status. Only a randomized controlled trial could sufficiently establish causality – but would be difficult to conduct concerning the consumption of a widely consumed and potentially toxic substance.
Since we excluded gender- and beverage-specific studies, no conclusions can be drawn concerning different effects for men or women or effects of specific ingredients of alcoholic beverages other than alcohol. This review is also limited by the use of only one database. An even greater amount of evidence concerning the link between dementia and alcohol consumption might be gathered by using further databases and including publications in languages other than English for a more thorough systematic review.
Conclusion
High-level alcohol consumption (>14 drink units/week) is certainly linked to an increase in dementia risk, post-mortem reduction in brain volume and MRI signs of brain damage via possibly multiple pathways.
Concerning low-level consumption of alcohol no recommendation to begin drinking moderate doses of alcohol in higher age to reduce dementia risk can be deducted, because (1) results of studies are heterogeneous (2) the detrimental effects on other organ systems and risk of addiction; (3) the possible confounders in the studies presented and (4) because of the varying individual metabolism (gender, body weight, acetaldehyde dehydrogenase type16) and susceptibility. On the other hand, there is no rationale either, to recommend cutting down on alcohol consumption to reduce dementia risk if consumption is moderate (disregarding other risks of alcohol consumption).
A prospective, randomized, controlled trial could distinguish between direct, alcohol-related effects, statistical confounders, and lifestyle effects,87 while more insight on the pathogenesis of Alzheimer’s disease in general and more specifically the influence of alcohol on the different biochemical pathways could provide a cogent model of alcohol effects on dementia pathogenesis and progression.
Disclosure
Eva Brandl receives speaker fees from Medicine and Servier. All other authors report no other conflicts of interest in this work.
References
1. Manthey J, Shield KD, Rylett M, Hasan OSM, Probst C, Rehm J. Global alcohol exposure between 1990 and 2017 and forecasts until 2030: a modelling study. Lancet. 2019;393:2493–2502. doi:10.1016/S0140-6736(18)32744-2
2. Heinz AJ, Beck A, Meyer-Lindenberg A, Sterzer P, Heinz A. Cognitive and neurobiological mechanisms of alcohol-related aggression. Nat Rev Neurosci. 2011;12:400–413. doi:10.1038/nrn3042
3. World Health Organization. Management of substance abuse team, world health organization. Global Status Report on Alcohol and Health. 2018.
4. WHO. Global Action Plan on the Public Health Response to Dementia 2017–2025. WHO; 2017.
5. Hagg-Grün U. Basiswissen Medizin Des Alterns Und Des Älteren Menschen.
6. Bazzari FH, Abdallah DM, El-Abhar HS. Pharmacological Interventions to attenuate alzheimer’s disease progression: the story so far. Curr Alzheimer Res. 2019;16:261–277. doi:10.2174/1567205016666190301111120
7. Braak H, Braak E. Neuropathological stageing of alzheimer-related changes. Acta Neuropathol. 1991;82:239–259. doi:10.1007/BF00308809
8. Hardy JA, Higgins GA. Alzheimer’s disease: the amyloid cascade hypothesis. Science. 1992;256:184–185. doi:10.1126/science.1566067
9. Schwarzinger M, Pollock BG, Hasan OSM, et al. Contribution of alcohol use disorders to the burden of dementia in France 2008–13: a nationwide retrospective cohort study. Lancet Public Health. 2018;3:e124–e132. doi:10.1016/S2468-2667(18)30022-7
10. Ruitenberg A, Van Swieten JC, Witteman JCM, et al. Alcohol consumption and risk of dementia: the rotterdam study. Lancet. 2002. doi:10.1016/S0140-6736(02)07493-7
11. Neafsey EJ, Collins MA. Moderate alcohol consumption and cognitive risk. Neuropsychiatr Dis Treat. 2011. doi:10.2147/NDT.S23159
12. Ridley NJ, Draper B, Withall A. Alcohol-related dementia: an update of the evidence. Alzheimers Res Ther. 2013;5(1):1–8. doi:10.1186/alzrt157
13. Gutwinski S, Schreiter S, Priller J, Henssler J, Wiers CE, Heinz A. Drink and think: impact of alcohol on cognitive functions and dementia - evidence of dose-related effects. Pharmacopsychiatry. 2018. doi:10.1055/s-0043-118664
14. Chandrakumar A, Bhardwaj A, T’Jong GW. Review of thiamine deficiency disorders: wernicke encephalopathy and korsakoff psychosis. J Basic Clin Physiol Pharmacol. 2019;30(2):153–162. doi:10.1515/jbcpp-2018-0075
15. Pinder RM. Does wine prevent dementia? Int J Wine Res. 2009;41. doi:10.2147/IJWR.S4691
16. Norberg A, Jones AW, Hahn RG, Gabrielsson JL. Role of variability in explaining ethanol pharmacokinetics. Clin Pharmacokinet. 2003;42(1):1–31. doi:10.2165/00003088-200342010-00001
17. Harris RA. Ethanol actions on multiple ion channels: which are important? Alcohol Clin Exp Res. 2006;23(10):1563–1570. doi:10.1111/j.1530-0277.1999.tb04045.x
18. Narahashi T, Kuriyama K, Illes P, et al. Neuroreceptors and ion channels as targets of alcohol. Alcohol Clin Exp Res. 2006;25(s1):182S–188S. doi:10.1111/j.1530-0277.2001.tb02394.x
19. Heinz A, Siessmeier T, Wrase J, et al. Correlation between dopamine D2 receptors in the ventral striatum and central processing of alcohol cues and craving. Am J Psychiatry. 2004;161:1783–1789. doi:10.1176/ajp.161.10.1783
20. Heinz A, Beck A, Grüsser SM, Grace AA, Wrase J. Identifying the neural circuitry of alcohol craving and relapse vulnerability. Addict Biol. 2009;14(1):108–118. doi:10.1111/j.1369-1600.2008.00136.x
21. Heinz A, Goldman D, Gallinat J, Schumann G, Puls I. Pharmacogenetic insights to monoaminergic dysfunction in alcohol dependence. Psychopharmacology (Berl). 2004;174. doi:10.1007/s00213-004-1903-x
22. Sinforiani E, Zucchella C, Pasotti C, Casoni F, Bini P, Costa A. The effects of alcohol on cognition in the elderly: from protection to neurodegeneration. Funct Neurol. 2011;26(2):103–106.
23. Fadda F, Rossetti ZL, Chronic ethanol consumption: from neuroadaptation to neurodegeneration. Prog Neurobiol. 1998;56(4):385–431. doi:10.1016/S0301-0082(98)00032-X
24. Kruman II, Henderson GI, Bergeson SE. DNA damage and neurotoxicity of chronic alcohol abuse. Exp Biol Med. 2012;237:740–747. doi:10.1258/ebm.2012.011421
25. Crews FT, Bechara R, Brown LA, et al. Cytokines and alcohol. Alcohol Clin Exp Res. 2006;30:720–730. doi:10.1111/j.1530-0277.2006.00084.x
26. Kamat PK, Mallonee CJ, George AK, Tyagi SC, Tyagi N. Homocysteine, alcoholism, and its potential epigenetic mechanism. Alcohol Clin Exp Res. 2016;40:2474–2481. doi:10.1111/acer.13234
27. Korsakoff SS. Eine psychische störung combinirt mit multipler neuritis. Allgem Zschr Psychiatr. 1890;47:475–485.
28. Wernicke, C. Die acute, hämorrhagische poliencephalitis superior. In: Lehrbuch Der Gehirnkrankheiten Für Aerzte Und Studierende.
29. Hillbom M, Saloheimo P, Fujioka S, Wszolek ZK, Juvela S, Leone MA. Diagnosis and management of marchiafava-bignami disease: a review of CT/MRI confirmed cases. J Neurol Neurosurg Psychiatry. 2014;85(2):168–173. doi:10.1136/jnnp-2013-305979
30. Zahr NM, Kaufman KL, Harper CG. Clinical and pathological features of alcohol-related brain damage. Nat Rev Neurol. 2011;7(5):284–294. doi:10.1038/nrneurol.2011.42
31. Moriyama Y, Mimura M, Kato M, Kashima H. Primary alcoholic dementia and alcohol-related dementia. Psychogeriatrics. 2006;6(3):114–118.
32. Harper C. The neuropathology of alcohol-related brain damage. Alcohol Alcohol. 2009;44:136–140. doi:10.1093/alcalc/agn102
33. Rosenbloom M, Sullivan EV, Pfefferbaum A. Using magnetic resonance imaging and diffusion tensor imaging to assess brain damage in alcoholics. Alcohol Res Health. 2003;27(2):146–152.
34. Phillips SC, Harper CG, Kril J, A quantitative histological study of the cerebellar vermis in alcoholic patients. Brain. 1987;110(2):301–314. doi:10.1093/brain/110.2.301
35. Harper CG, Kril JJ. Corpus callosal thickness in alcoholics. Br J Addict. 1988;83(5):577–580. doi:10.1111/j.1360-0443.1988.tb02577.x
36. Jernigan TL, Butters N, DiTraglia G, et al. Reduced cerebral grey matter observed in alcoholics using magnetic resonance imaging. Alcohol Clin Exp Res. 1991;15:418–427. doi:10.1111/j.1530-0277.1991.tb00540.x
37. Pfefferbaum A, Lim KO, Zipursky RB, et al. Brain gray and white matter volume loss accelerates with aging in chronic alcoholics: a quantitative MRI study. Alcohol Clin Exp Res. 1992;16:1078–1089. doi:10.1111/j.1530-0277.1992.tb00702.x
38. Guggenmos M, Schmack K, Sekutowicz M, et al. Quantitative neurobiological evidence for accelerated brain aging in alcohol dependence. Transl Psychiatry. 2017;7:12. doi:10.1038/s41398-017-0037-y
39. Ding J, Eigenbrodt ML, Mosley TH, et al. Alcohol intake and cerebral abnormalities on magnetic resonance imaging in a community-based population of middle-aged adults. Stroke. 2004;35(1):16–21. doi:10.1161/01.STR.0000105929.88691.8E
40. Mukamal KJ, Longstreth WT, Mittleman MA, Crum RM, Siscovick DS. Alcohol consumption and subclinical findings on magnetic resonance imaging of the brain in older adults: the cardiovascular health study. Stroke. 2001;32:1939–1946. doi:10.1161/hs0901.095723
41. Rimm EB, Williams P, Fosher K, Criqui M, Stampfer MJ. Moderate alcohol intake and lower risk of coronary heart disease: meta-analysis of effects on lipids and haemostatic factors. BMJ. 1999;319:1523–1528. doi:10.1136/bmj.319.7224.1523
42. Stampfer MJ, Sacks FM, Salvini S, Willett WC, Hennekens CH. A prospective study of cholesterol, apolipoproteins, and the risk of myocardial infarction. J Occup Environ Med. 1992. doi:10.1097/00043764-199203000-00004
43. Meade TW, Brozovic M, Chakrabarti RR, et al. Haemostatic function and ischaemic heart disease: principal results of the northwick heart study. Lancet. 1986;328:533–537. doi:10.1016/S0140-6736(86)90111-X
44. Rubin R. Effect of ethanol on platelet function. Alcohol Clin Exp Res. 1999;23:1114–1118. doi:10.1111/j.1530-0277.1999.tb04234.x
45. Fenn CG, Littleton JM. Inhibition of platelet aggregation by ethanol in vitro shows specificity for aggregating agent used and is influenced by platelet lipid composition. Thromb Haemost. 198248(01):49–53.
46. Roerecke M, Kaczorowski J, Tobe SW, Gmel G, Hasan OSM, Rehm J. The effect of a reduction in alcohol consumption on blood pressure: a systematic review and meta-analysis. Lancet Public Health. 2017;2:e108–e120. doi:10.1016/S2468-2667(17)30003-8
47. Kalinowski A, Humphreys K. Governmental standard drink definitions and low-risk alcohol consumption guidelines in 37 countries. Addiction. 2016;111:1293–1298. doi:10.1111/add.13341
48. Piano MR. Alcohol’s effects on the cardiovascular system. Alcohol Res. 2017;38(2):219.
49. Henn C, Löffelholz K, Klein J. Enhancement of hippocampal acetylcholine release by local ethanol infusion. Neurochemistry. 2011. doi:10.1007/978-1-4615-5405-9_123
50. Muñoz G, Urrutia JC, Burgos CF, et al. Low concentrations of ethanol protect against synaptotoxicity induced by Aβ in hippocampal neurons. Neurobiol Aging. 2015;36(2):845–856. doi:10.1016/j.neurobiolaging.2014.10.017
51. Ormeño D, Romero F, López-Fenner J, Avila Á, Martínez-Torres A, Parodi J. Ethanol reduces amyloid aggregation in vitro and prevents toxicity in cell lines. Arch Med Res. 2013;44(1):1–7. doi:10.1016/j.arcmed.2012.12.004
52. Huang D, Yu M, Yang S, et al. Ethanol alters APP processing and aggravates alzheimer-associated phenotypes. Mol Neurobiol. 2018;55(6):5006–5018. doi:10.1007/s12035-017-0703-3
53. Venkataraman A, Kalk N, Sewell G, Ritchie CW, Lingford-Hughes A. Alcohol and alzheimer’s disease-does alcohol dependence contribute to beta-amyloid deposition, neuroinflammation and neurodegeneration in alzheimer’s disease? Alcohol Alcohol. 2017;52(2):151–158. doi:10.1093/alcalc/agw092
54. Kalinin S, González-Prieto M, Scheiblich H, et al. Transcriptome analysis of alcohol-treated microglia reveals downregulation of beta amyloid phagocytosis. J Neuroinflammation. 2018;15. doi:10.1186/s12974-018-1184-7
55. Yoshitake T, Kiyohara Y, Kato I, et al. Incidence and risk factors of vascular dementia and alzheimer’s disease in a defined elderly japanese population: the Hisayama study. Neurology. 1995;45:1161–1168. doi:10.1212/WNL.45.6.1161
56. Langballe EM, Ask H, Holmen J, et al. Alcohol consumption and risk of dementia up to 27 years later in a large, population-based sample: the HUNT study, Norway. Eur J Epidemiol. 2015;30:1049–1056. doi:10.1007/s10654-015-0029-2
57. Heffernan M, Mather KA, Xu J, et al. Alcohol consumption and incident dementia: evidence from the sydney memory and ageing study. J Alzheimers Dis. 2016;52(2):529–538. doi:10.3233/JAD-150537
58. Paganini-Hill A, Kawas CH, Corrada MM. Lifestyle factors and dementia in the oldest-old: the 90 + study. Alzheimer Dis Assoc Disord. 2016;30:21–26. doi:10.1097/WAD.0000000000000087
59. Xue H, Sun Q, Liu L, et al. Risk factors of transition from mild cognitive impairment to alzheimer’s disease and death: a cohort study. Compr Psychiatry. 2017;78:91–97. doi:10.1016/j.comppsych.2017.07.003
60. Broe GA, Creasey H, Jorm AF, et al. Health habits and risk of cognitive impairment and dementia in old age: A prospective study on the effects of exercise, smoking and alcohol consumption. Aust N Z J Public Health. 1998;22:621–623. doi:10.1111/j.1467-842X.1998.tb01449.x
61. Huang W, Qiu C, Winblad B, Fratiglioni L. Alcohol consumption and incidence of dementia in a community sample aged 75 years and older. J Clin Epidemiol. 2002;55(10):959–964. doi:10.1016/S0895-4356(02)00462-6
62. Lindsay J, Laurin D, Verreault R, et al. Risk factors for alzheimer’s disease: a prospective analysis from the Canadian study of health and aging. Am J Epidemiol. 2002;156(5):445–453. doi:10.1093/aje/kwf074
63. Truelsen T, Thudium D, Grønbæk M. Amount and type of alcohol and risk of dementia: the copenhagen city heart study. Neurology. 2002;59(9):1313–1319. doi:10.1212/01.WNL.0000031421.50369.E7
64. Luchsinger JA, Tang MX, Siddiqui M, Shea S, Mayeux R. Alcohol intake and risk of dementia. J Am Geriatr Soc. 2004;52(4):540–546. doi:10.1111/j.1532-5415.2004.52159.x
65. Anttila T, Helkala E-L, Viitanen M, et al. Alcohol drinking in middle age and subsequent risk of mild cognitive impairment and dementia in old age: a prospective population based study. BMJ. 2004;329(September):538–539. doi:10.1136/bmj.38181.48958.BE
66. Ogunniyi A, Hall KS, Gureje O, et al. Risk factors for incident alzheimer’s disease in African Americans and Yoruba. Metab Brain Dis. 2006;21:224–229. doi:10.1007/s11011-006-9017-2
67. Deng J, Zhou DHD, Li J, Wang YJ, Gao C, Chen M. A 2-year follow-up study of alcohol consumption and risk of dementia. Clin Neurol Neurosurg. 2006;108:378–383. doi:10.1016/j.clineuro.2005.06.005
68. Bachman DL, Green RC, Benke KS, Cupples LA, Farrer LA. Comparison of alzheimer’s disease risk factors in white and African American families. Neurology. 2003;60(8):1372–1374. doi:10.1212/01.WNL.0000058751.43033.4D
69. Mukamal KJ, Kuller LH, Fitzpatrick AL, Longstreth WT, Mittleman MA, Siscovick DS. Prospective study of alcohol consumption and risk of dementia in older adults. J Am Med Assoc. 2003;289(11):1405–1413. doi:10.1001/jama.289.11.1405
70. Sabia S, Fayosse A, Dumurgier J, et al. Alcohol consumption and risk of dementia: 23 year follow-up of whitehall II cohort study. BMJ. 2018:k2927. doi:10.1136/bmj.k2927
71. Handing EP, Andel R, Kadlecova P, Gatz M, Pedersen NL. Midlife alcohol consumption and risk of dementia over 43 years of follow-up: a population-based study from the swedish twin registry. J Gerontol a Biol Sci Med Sci. 2014. doi:10.1093/gerona/glv038
72. Harwood DG, Kalechstein A, Barker WW, et al. The effect of alcohol and tobacco consumption, and apolipoprotein E genotype, on the age of onset in alzheimer’s disease. Int J Geriatr Psychiatry. 2010;25(5):511–518. doi:10.1002/gps.2372
73. Holst C, Tolstrup JS, Sørensen HJ, Becker U. Alcohol dependence and risk of somatic diseases and mortality: a cohort study in 19 002 men and women attending alcohol treatment. Addiction. 2017;112:1358–1366. doi:10.1111/add.13799
74. Weyerer S, Schäufele M, Wiese B, et al. Current alcohol consumption and its relationship to incident dementia: results from a 3-year follow-up study among primary care attenders aged 75 years and older. Age Ageing. 2011;40(4):456–463. doi:10.1093/ageing/afr007
75. Piazza-Gardner AK, Gaffud TJB, Barry AE. The impact of alcohol on alzheimer’s disease: a systematic review. Aging Ment Heal. 2013;17(2):133–146. doi:10.1080/13607863.2012.742488
76. Peters R, Peters J, Warner J, Beckett N, Bulpitt C. Alcohol, dementia and cognitive decline in the elderly: a systematic review. Age Ageing. 2008;37(5):505–512. doi:10.1093/ageing/afn095
77. Anstey KJ, Mack HA, Cherbuin N. Alcohol consumption as a risk factor for dementia and cognitive decline: meta-analysis of prospective studies. Am J Geriatr Psychiatry. 2009;17(7):542–555. doi:10.1097/JGP.0b013e3181a2fd07
78. Ilomaki J, Jokanovic N, Tan ECK, Lonnroos E. Alcohol consumption, dementia and cognitive decline: an overview of systematic reviews. Curr Clin Pharmacol. 2015;10:204–212. doi:10.2174/157488471003150820145539
79. Xu W, Wang H, Wan Y, et al. Alcohol consumption and dementia risk: a dose–response meta-analysis of prospective studies. Eur J Epidemiol. 2017;32(1):31–42. doi:10.1007/s10654-017-0225-3
80. Li J, Hui X, Gu Q, et al. Alcohol consumption and risk of dementia: a dose-response meta-analysis. Medicine (Baltimore). 2019;98(26):e16099. doi:10.1097/MD.0000000000016099
81. Nordlund S. Rusmiddelbruk i norge. Nor Epidemiol. 2009;6. doi:10.5324/nje.v6i1.269
82. Ganguli M, Vander Bilt J, Saxton JA, Shen C, Dodge HH. Alcohol consumption and cognitive function in late life: A longitudinal community study. Neurology. 2005;65:1210–1217. doi:10.1212/01.wnl.0000180520.35181.24
83. Westman J, Wahlbeck K, Laursen TM, et al. Mortality and life expectancy of people with alcohol use disorder in Denmark, Finland and Sweden. Acta Psychiatr Scand. 2015;131:297–306. doi:10.1111/acps.12330
84. Holstege H, Beker N, Dijkstra T, et al. The 100-plus study of cognitively healthy centenarians: rationale, design and cohort description. Eur J Epidemiol. 2018;3. doi:10.1007/s10654-018-0451-3
85. Platt A, Sloan FA, Costanzo P. Alcohol-consumption trajectories and associated characteristics among adults older than age 50*. J Stud Alcohol Drugs. 2010;71(2):169–179. doi:10.15288/jsad.2010.71.169
86. Cobb JL, Wolf PA, Au R, White R, D’agostino RB. The effect of education on the incidence of dementia and alzheimer’s disease in the framingham study. Neurology. 1995;45:1707–1712. doi:10.1212/WNL.45.9.1707
87. Mukamal K, Clowry C, Krystal JH. Moderate alcohol consumption and chronic disease: the case for a long-term trial. Alcohol Clin Exp Res. 2016;40(11):2283–2291. doi:10.1111/acer.13231
88. Jung YC, Chanraud S, Sullivan EV. Neuroimaging of wernicke’s encephalopathy and korsakoff’s syndrome. Neuropsychol Rev. 2012;22:170–180. doi:10.1007/s11065-012-9203-4
89. Marchiafava E, Bignami A. Sopra und’alterazione del corpo callosi osservata da sogetti alcoolisti. Riv Pathol Nerv Ment. 1903;8:544–549.
90. Järvenpää T, Rinne JO, Koskenvuo M, Räihä I, Kaprio J. Binge drinking in midlife and dementia risk. Epidemiology. 2005;16(6):766–771. doi:10.1097/01.ede.0000181307.30826.6c
91. Toure K, Coume M, Ndiaye M, et al. Risk factors for dementia in a senegalese elderly population aged 65 years and over. Dement Geriatr Cogn Dis Extra. 2012;2(1):160–168. doi:10.1159/000332022
92. Pilleron S, Desport J-C, Jésus P, et al. Diet, alcohol consumption and cognitive disorders in central Africa. J Nutr Heal Aging. 2014;7.
93. Radford K, Lavrencic LM, Delbaere K, et al. Factors associated with the high prevalence of dementia in older aboriginal Australians. J Alzheimers Dis. 2018:1–11. doi:10.3233/jad-180573
94. Anstey KJ, Peters R. Alcohol and dementia — risk or protective factor? Nat Rev Neurol. 2018;14(11):635–636. doi:10.1038/s41582-018-0073-0
95. Tyas SL, Koval JJ, Pederson LL. Does an interaction between smoking and drinking influence the risk of alzheimer’s disease? Results from three Canadian data sets. Stat Med. 2000. doi:10.1002/(SICI)1097-0258(20000615/30)19:11/12<1685::AID-SIM454<3.0.CO;2-#
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.