Back to Journals » Vascular Health and Risk Management » Volume 15
Adherence To Lipid-Lowering Therapy In Patients With Coronary Heart Disease From The State Of Saxony-Anhalt, Germany
Authors Waßmuth S, Rohe K, Noack F , Noutsias M , Treede H, Schlitt A
Received 5 December 2018
Accepted for publication 12 September 2019
Published 31 October 2019 Volume 2019:15 Pages 477—483
DOI https://doi.org/10.2147/VHRM.S197089
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Daniel Duprez
Stephan Waßmuth,1 Katharina Rohe,1 Frank Noack,2 Michel Noutsias,1 Hendrik Treede,3 Axel Schlitt4,5
1Department of Medicine III, University Clinic, Halle, Germany; 2Emergency Department and Department of Medicine II, University Clinic, Halle, Germany; 3Department of Cardiac Surgery, University Clinic, Halle, Germany; 4Paracelsus Harz-Clinic Bad Suderode, Quedlinburg, Germany; 5Medical Faculty, Martin Luther-University Halle-Wittenberg, Halle, Germany
Correspondence: Stephan Waßmuth
Department of Internal Medicine, St. Josefs Hospital Wiesbaden, Beethovenstraße 20, Wiesbaden 65189, Germany
Tel +49 1726619946
Fax +49 6452 5350
Email [email protected]
Objectives: Treatment with lipid-lowering therapy (LLT) such as statins, cholesterol absorption inhibitors, or PCSK9 inhibitors is of major importance for the survival of patients with atherosclerotic diseases, and adherence to LLT is essential for treatment success. The intention of this study was to investigate adherence to LLT in patients with coronary heart disease (CHD) in a 12-month follow-up period in Saxony-Anhalt, the state with the highest incidence and mortality for CHD in Germany.
Patients and methods: Data were taken from 542 hospitalized patients with angiographically documented CHD who were prospectively included in this study conducted in the Department of Medicine III of the University Clinics (Halle). We collected data concerning medication at discharge and after 3 and 12 months.
Results: A total of 542 patients were included in this study. Mean age was 69.2 ± 11.8 years. In all, 68.8% were males, 165 (30.4%) were smokers, 39.7% suffered from diabetes, and 86.9% had arterial hypertension. The follow-up time of this study was 12 months. At discharge, 463 patients (85.4%) were being treated with a statin. After 3 months 409 (75.5%) and after 12 months, 395 patients (72.9%) were still on statin therapy, respectively. In total treatment, adherence for the statin medication decreased by 15.7% in 12 months. Kaplan–Meier analyses showed that survival, taken as freedom of death from any cause, decreased significantly if statin treatment was stopped (p=0.001). This was confirmed by multivariate Cox regression (HR 1.78, p=0.012). Ezetemibe was prescribed for 56 patients at discharge (10.3%). After 3 months, 40 patients (7.4%) were still taking ezetemibe. After 12 months, adherence to ezetemibe treatment decreased to 4.1% (22 patients).
Conclusion: During follow-up for 3 and 12 months, adherence for statin therapy decreased by 15.7% and for ezetemibe by 46.6%. Here, low adherence to statin therapy was associated with fatal outcome.
Keywords: coronary heart disease, lipid-lowering drugs, adherence
Introduction
Coronary heart disease (CHD) is the most common cause of death in industrial nations.1 The main risk factors are tobacco use, arterial hypertension, diabetes, and dyslipoproteinemia. Here, higher levels of low-density lipoprotein cholesterol (LDL-C) in particular can initiate and then promote progression of atherosclerosis.
In Germany, incidence and mortality of CHD have been decreasing in the past few decades; however, numbers are still higher in former East Germany than in the western German federal states, and the highest rate can be found in Saxony-Anhalt. (German Heart Foundation e.V. editors. Deutscher Herzbericht 2017[German Heart Report 2017]. Frankfurt, Germany; 2018. German).
The causes of differences between states are not well investigated. Possible factors include differing health care, lower densities of physicians in relation to populations, a less effective emergency physician system, longer time to hospital admission, especially in acute coronary syndrome patients, and different education levels among the population being associated with less health awareness.2
Lipid-lowering therapy (LLT), such as statins, ezetimibe, or PCSK9 inhibitors that mainly reduce LDL-C, reduces the risk for cardiovascular events in primary and secondary prevention.3 Since the discovery of these drugs, numerous studies have proven their effectiveness and a good cost–benefit ratio.3
Obviously, the effect of LLT depends on how patients follow through with their medication, which is commonly described by the term adherence.4 Adherence is defined as
the extent to which a person’s behavior—taking medication, following a diet, and/or executing lifestyle changes—corresponds with agreed recommendations from a healthcare provider and is literally defined as sticking to something.3,5
Good adherence is often not permanent so that after a few years at the latest adherence decreases dramatically. Mangiapane et al showed that after 5 years, only 17 percent of patients were taking the statin medication prescribed in the discharge letter after hospitalization for acute myocardial infarction.6 Ho and Magid showed that a lack of adherence to LLT translates into a significant increase in risk for rehospitalization, need for percutaneous revascularization, and cardiovascular death.7
We hypothesized that low adherence to LLT represents another factor for high CHD mortality in Saxony-Anhalt. Furthermore, we expected a decreasing adherence for statin and ezetemib therapy in the follow-up.
Methods
Aim of this monocentric, prospective cohort study was to investigate adherence to LLT in 558 patients with CHD from Saxony-Anhalt. All patients were hospitalized from 2011 to 2012 in the Department of Medicine III (Cardiology) or in the Department of Cardiac Surgery of the University Clinic Halle (Saale). CHD had to be proved by percutaneous coronary angiography during the current or previous hospitalization period before a patient was included in the study.
Collected data focused on risk factors such as body mass index (BMI), gender, dyslipoproteinemia (total cholesterol >5.2 mmol/L or LDL-C >3,9 mmol/L), diabetes mellitus (fasting blood glucose ≥ 7 mmol/L, hypertension (blood pressure >140/90 mmHg)), tobacco use, and other variables that are typical for patients with CHD. Most importantly, data on current drug treatment, including statins and ezetemibe, were collected. Before demission, patients were visited and asked about their risk factors. Also, the medication (LLT) of the discharge letters was documented.
After 3 and 12 months, patients were contacted by postal mail. Here, a standardized questionnaire was sent out, which included questions concerning medication or events such as hospital admission after discharge in the target hospitalization.
If the patients did not send back the questionnaires, a telephone interview was conducted with the patient or his/her relatives or the patient’s physician was contacted. If the information could not be obtained from these persons, civil registration offices were contacted and information was requested about current addresses or date of death.
The study was approved by the ethics committee of the Martin Luther University Halle-Wittenberg.
Patients were defined as adherent to LLT if statins and/or ezetemibe, including their effective dosage, were unchanged, newly prescribed, or increased. Patients were defined as nonadherent if statins and/or ezetemibe were stopped or dosage was decreased.
Statistical Analyses
Continuous variables were described as mean and standard deviation; skewed variables were median and 25% and 75% quartiles. Categorical variables were documented as absolute numbers and percentage. For comparison of metric, normally distributed variables, t-test was used. Mann–Whitney U-test was applied to compare skewed variables. For normally distributed, categorical variables, the chi-squared test was employed.
Survival analyses included Kaplan–Meier analyses with log rank test and multivariate Cox regression analyses. Multivariate Cox regression was applied to analyze the influence on adherence to lipid-lowering medication. P-values <0.05 were considered significant. Statistical analyses were performed using SPSS Statistics (SPSS Inc, Chicago, IL) software.
Results
Data were obtained from 548 patients as four patients were lost to follow-up and six patients withdrew their consent.
Among the 658 patients screened for this study, 542 individuals met the inclusion criteria. Mean age was 69.2 ± 11.8 years, 68.8% were males, and 165 (30.4%) were smokers. Furthermore, 39.7% suffered from diabetes and 86.9% had arterial hypertension. Three hundred and ninety-nine patients had in time of inclusion a percutaneous intervention (73.6%). The follow-up time of this study was 12 months.
The two groups differed significantly regarding baseline parameters: patients with peripheral artery disease (15.7% versus 23.0%, p=0.039) and patients with atrial fibrillation (30.7% versus 39.2%, p=0.040) were less adherent. Not surprisingly, patients with known dyslipoproteinemia tended to show better adherence (66.0% vs 54.7%, p=0.052). Further, patients of the nonadherent group more often had undergone coronary bypass surgery (26.1% versus 37.2%, p=0.012) (Table 1).
 |
Table 1 Baseline Parameter |
The two groups did not differ significantly regarding the laboratory results, interestingly including lipid parameters such as LDL-C at admission to the hospital (Table 2).
 |
Table 2 Laboratory Results |
Patients adherent to LLT were also more often on aspirin (73.4% versus 64.2%, p=0.038), P2Y12 inhibitors (47.7% versus 33.1%, p=0.003), and beta-blockers (93.1% versus 83.8%, p=0.020) at discharge, whereas other evidence-based treatments for CHD did not differ in the treatment groups (Table 3).
 |
Table 3 Drugs At Discharge |
At discharge, 85.5% of all patients were taking a statin. In the group of patients who were not adherent, 92 (62.2%) patients had been taking a statin when discharged. Of the adherent group, 371 patients (95.2%) were taking a statin (p=0.001) at discharge.
In the nonadherent group, 11 patients (7.4%) were taking ezetemibe. In the adherent group, 49 patients (12.4%) were on ezetemibe at discharge (p=0.282)
The LLT as presented in detail in Table 4 shows that simvastatin was the most frequently prescribed statin. The dose most commonly used was simvastatin (20mg). Ezetemibe for monotherapy was prescribed for three patients at discharge. The combination most often used was ezetemibe and simvastatin (10/40mg). After 3 and 12 months, the prescribed dosages decreased. After 3 months, 143 patients were taking simvastatin (40mg). However, the prescribed doses for simvastatin (40mg) also dropped. The combination of ezetemibe and a statin also decreased for almost all dosages (Table 4).
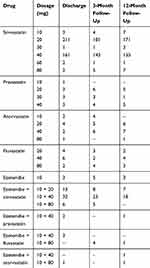 |
Table 4 LLT In Detail During Study Period |
Statin therapy differed from discharge to the 3-month follow-up (463 versus 409 patients, p=0.001). Furthermore, the prescribed statin medication dropped in the period from the 3-month follow-up to the 12-month follow-up (409 versus 395 patients, p=0.237). Particularly, the number of patients on ezetemibe decreased from 60 patients at discharge to 32 patients after 12 months (p=0.001).
The Kaplan–Meier estimate for ezetemibe medication showed an increased occurrence for cardiac death, stroke, and myocardial infarction (Figure 1) when the treatment was stopped (p=0.372 via log rank test). The Kaplan–Meier analysis for ezetemibe medication also demonstrated a nonsignificant trend for decreased survival concerning death from any cause (Figure 2) when ezetemibe therapy was stopped (p=0.146 via log rank test). Applying the Kaplan–Meier estimate for death from any cause, survival decreased significantly when statin medication was stopped (p=0.001 via log rank test). However, the Kaplan–Meier analysis for cardiac death, stroke, and myocardial infarction (Figure 3) showed no survival advantage for an unchanged statin therapy (p=0.466 via log rank test). Cox regression analysis included the factors negative adherence to LLT, coronary artery bypass surgery, atrial fibrillation, peripheral artery disease, and hyperlipidemia. An independent predictor for cardiac mortality, stroke, and myocardial infarction was a negative adherence to LLT with a HR of 1.78 (95% CI 1.13–2.79 P=0.012) (Table 5).
 |
Table 5 Multivariate Cox Regression Analysis |
 |
Figure 1 Kaplan–Meier estimate. Kaplan–Meier analysis for cardiac death, stroke, and myocardial infarction, ezetemibe therapy stopped versus ezetemibe therapy unchanged. |
 |
Figure 2 Kaplan–Meier estimate. Kaplan–Meier analysis for death, ezetemibe therapy stopped versus ezetemibe therapy unchanged. |
 |
Figure 3 Kaplan–Meier estimate. Kaplan–Meier analysis for cardiac death, stroke, and myocardial infarction, statin therapy stopped versus statin therapy unchanged. |
Discussion
Cardiovascular diseases are the most common cause of death in Germany.8 However, there are regional differences in the occurrence of cardiovascular death and myocardial infarction, especially for Saxony-Anhalt. In addition to the classical risk factors (diabetes mellitus, arterial hypertension, and tobacco abuse), hyperlipidemia is quite important for the development of atherosclerosis and thus for the emergence of cardiovascular diseases. Therefore, LLT is important for the prognosis of cardiovascular diseases. More than any other lipid-lowering medication, statins and ezetemibe have been well examined and are established in daily clinical practice. The “4S-study”, accomplished in 1994, showed that the decrease in LDL-C reduces the occurrence of cardiovascular diseases. Furthermore, the “Heart protection study” demonstrated that statins also reduce the risk of cardiovascular diseases within a longer observation period (follow-up time 5 years).9,10
Ezetemibe monotherapy reduces LDL-C levels by 10 to 20 percent, but the most significant benefit was shown in the “Improve it” study (2014) for the combination of simvastatin and ezetemibe, which demonstrated an advantage for patients on that regimen in comparison to a statin monotherapy regarding cardiovascular outcome.11,12
Further adherence and persistence are cornerstones for prevention and treatment of cardiovascular disease. Good adherence reduces cardiovascular events and mortality.13
Reducing mortality and morbidity with statins provides an important economic benefit. The “Ward study” showed that therapy with statins has a better cost–benefit ratio than treatment with placebo.14 Furthermore, it is obvious that a lack of adherence increases the cost of health care.15
Especially adherence to LLT, shown in international studies, decreases after demission. In a long time trial, there was a decline of adherence to LLT from demission to a 12-month follow-up to 54%.16 Also, statin discontinuation represents a problem for successful treatment of CHD. Forty to seventy-five percent of patients treated with statins interrupt their statin therapy in a 1-year observation period.17
To effectively treat atherosclerosis-related diseases, guidelines for therapy are required. Concerning the special risk profile in Saxony-Anhalt, it is crucial to investigate whether adherence has an effect on morbidity and mortality. As described in the Results section, the two groups in part differed significantly concerning the prevalence of cardiovascular risk factors (hyperlipidemia, peripheral artery disease). The present study shows a significant difference concerning the LLT at discharge (62.2% in the nonadherent group were taking a statin vs 85.5% in the adherent group, p=0.001; 7.4% in the nonadherent group were on ezetemibe vs 12.4% in the adherent group, p=0.282). Simvastatin was the most frequently prescribed statin in this study (dose 20mg). The most often used combination was ezetemibe and simvastatin (10/40mg).
Results of other studies have shown a nonadherence rate of 13% for statin medication in a 6-month follow-up period.18 Follow-ups of 12 months report a nonadherence rate of 16%.19 Long-term studies show a decrease of up to 26% after 4 years.7
The findings of this study demonstrate that statin medication use dropped to 11.7% from discharge to the 3-month follow-up (p=0.001). After 12 months, there was a decline of 15.7% (Figure 4). Concerning the ezetemibe medication, there was a reduction of 25% after 3 months and 46.6% after 12 months (Figure 4; p=0.001).
 |
Figure 4 LLT at discharge, 3- and 12-month follow-up. Note: Values in absolute numbers; reduction in LLT in percentage from discharge to 12-month follow-up. |
The Kaplan–Meier analysis for ezetemibe indicated only a tendential increase in mortality (p=0.146; Figure 2) for patients who stopped taking ezetemibe. When applying univariate Kaplan–Meier analysis with log rank test, mortality increased after statin therapy was stopped (p=0.001; Figure 5). However, stopped statin therapy was not linked to increased occurrence of the combined endpoint (stroke, myocardial infarction, and cardiac death; p=0.466; Figure 3). In addition, multivariate Cox regression showed that patients with a negative adherence had a hazard ratio of 1.78 for the combined endpoint (Table 5).
 |
Figure 5 Kaplan–Meier estimate. Kaplan–Meier analysis for death, statin therapy stopped versus statin therapy unchanged. |
The fact that, in the present study, negative adherence to LLT was the only independent predictor in the multivariate model for worse outcome in patients with coronary heart disease emphasizes the importance of adherence to LLT (Table 5).
This study confirms results of previous investigations which demonstrated an increase in cardiovascular death after withdrawal of statins.20
As this study represents the results of “real-life” practice, many patients showed changes in medication after discharge. The causes for this could be economic status or intolerance. Unauthorized withdrawal might also explain this.
Thus, a limitation of this study is the smaller sample size of patients treated with ezetemibe in comparison to patients treated with statins.
Conclusion
Lipid-lowering therapy with statins and ezetemibe diminishes mortality in patients with cardiovascular diseases. In the case of nonadherence, this effect is lost.
Ethics And Consent
We confirm that his study was conducted in accordance with the Declaration of Helsinki. The study was approved by the ethics committee of Martin Luther University Halle-Wittenberg. The data which were collected in this study were only intended for internal use. The patients in this study didn´t sign a consent form, that data could be available for others or for any intention to share. Unfortunately, this trial was not registered in clinical trials.
Disclosure
Dr Michel Noutsias reports personal fees from Abbott, personal fees from AstraZeneca, personal fees from Bayer, personal fees from MiltenyiBiotech, personal fees from Novartis, and personal fees from Zoll, outside the submitted work. The authors report no other conflicts of interest in this work.
References
1. Montalescot G, Sechtem U, Achenbach S, et al. ESC guidelines on the management of stable coronary artery disease: the Task Force on the management of stable coronary artery disease of the European Society of Cardiology. Eur Heart J. 2013;34:2949–3003. doi:10.1093/eurheartj/eht296
2. Stang A, Stang M. An inter-state comparison of cardiovascular risk in Germany – towards an explanation of high ischemic heart disease mortality in Saxony-Anhalt. Dtsch Arztebl Int. 2014;530–536.
3. Catapano AL, Graham I, De Backer G, et al. Additional contributor. 2016 ESC/EAS guidelines for the management of dyslipidaemias. Eur Heart J. 2016;37:2999–3058.
4. Kircher T: Hrsg. Medikamentenadhärenz. Kompendium Der Psychotherapie. Berlin, Heidelberg: Springer-Verlag; 2012.
5. Available from: http://apps.who.int/medicinedocs/en/d/Js4883e/6.html.
6. Mangiapane S, Busse R. Prescription prevalence and continuing medication use for secondary prevantion after myocardial infarction: the reality of care revealed by claims data analysis. Dtsch Arztebl Int. 2011;2011:856–862.
7. Ho PM, Magid DJ, Shetterly SM, et al. Medication nonadherence is associated with a broad range of adverse outcomes in patients with coronary artery disease. Am Heart J. 2008;155:772–779. doi:10.1016/j.ahj.2007.12.011
8. Statistisches Bundesamt:2.
9. Grodos G, Tonglet R. Scandinavian simvastatin study (4s). Lancet. 1994;8939–8940.
10. HPSCG. Heart Protection Study Collaborative Group. The Lancet. 2003;2005–2016.
11. Karow T, Lang-Roth R. Allgemeine Und Spezielle Pharmakologie Und Toxikologie [General and special pharmacology and toxicology].
12. Spinar J, Spinarova L, Vitovec J. IMProved reduction of outcomes: vytorin Efficacy International Trial (studie IMPROVE-IT). Vnitr Lek. 2014;60:1095–1101.
13. Deshpande S, Quek RG, Forbes CA, et al. A systematic review to assess adherence and persistence with statins. Curr Med Res Opin. 2017;33:4. doi:10.1080/03007995.2017.1301903
14. National Institute for Health and Clinical Excellence: Guidance. BLipid Modification: Cardiovascular Risk Assessment and the Modification of Blood Lipids for the Primary and Secondary Prevention of Cardiovascular Disease; 2014.
15. Pittman DG, Chen W, Bowlin SJ, et al. Adherence to statins, subsequent healthcare costs, and cardiovascular hospitalizations. Am J Cardiol. 2011;107:1662–1666.
16. Chen ST, Huang ST, Shau WY, et al. Long term statin adherence in patients after hospital discharge for new onset of atherosclerotic cardiovascular disease. BMC Cardiovasc Disord. 2019;19:1. doi:10.1186/s12872-019-1032-4
17. Maciej B, Stulc T, Dent R, Toth PP. Statin non adherence and cardiovascular risk: there is need for substantial improvement. Int J Cardiol. 2016;225:184–196. doi:10.1016/j.ijcard.2016.09.075
18. Griffiths B, Lesosky M, Ntsekhe M. Self-reported use of evidence-based medicine and smoking cessation 6–9 months after acute coronary syndrome: a single-centre perspective. S Afr Med J. 2014;104:483–487. doi:10.7196/samj.7798
19. Lemstra M, Blackburn D. Nonadherence to statin therapy: discontinuation after a single fill. Can J Cardiol. 2012;28:567–573. doi:10.1016/j.cjca.2012.03.018
20. Kim MC, Cho JY, Jeong HC, et al. Impact of Postdischarge Statin Withdrawal on Long-Term Outcomes in Patients With Acute Myocardial Infarction.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
