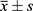Back to Journals » Drug Design, Development and Therapy » Volume 13
Adenosine Inhibits Ovarian Cancer Growth Through Regulating RhoGDI2 Protein Expression
Received 11 June 2019
Accepted for publication 18 October 2019
Published 8 November 2019 Volume 2019:13 Pages 3837—3844
DOI https://doi.org/10.2147/DDDT.S219028
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Anastasios Lymperopoulos
Bing Xia, Jing Wang
Hunan Cancer Hospital and the Affiliated Tumor Hospital of Xiang-Ya School of Medicine, Central South University, Changsha 410078, People’s Republic of China
Correspondence: Jing Wang
Hunan Cancer Hospital and the Affiliated Tumor Hospital of Xiang-Ya School of Medicine, Central South University, 283 Tongzipo Road, Yuelu District, Changsha 410078, People’s Republic of China
Tel +86 135 4865 1043
Email [email protected]
Objective: This study aimed to investigate the effect of adenosine (Ado) on the growth of ovarian cancer and to explore the related mechanisms.
Methods: The effect of Ado on the proliferation of A2780 human ovarian cancer cells was examined according to the MTT method. Moreover, the nude mouse model of subcutaneous A2780 xenograft was constructed, and then, Ado and cisplatin were administered intraperitoneally to investigate the effect of Ado on tumor growth in vivo. Immunohistochemistry (IHC) was carried out to study the effect of Ado on the expression of Rho-specific guanine nucleotide dissociation inhibitor 2 (RhoGDI2) in the subcutaneous xenografts. Afterwards, the commercially constructed RhoGDI2 siRNA plasmid was transfected into A2780 cells, and tube formation assay was conducted to determine the effect of down-regulating RhoGDI2 expression on the regulation of angiogenesis in ovarian cancer by Ado. Besides, Western blotting was performed to detect the effect of RhoGDI2 down-regulation on the regulation of matrix metalloproteinase 2 (MMP-2), MMP-9, vascular endothelial growth factor (VEGF), transforming growth factor beta (TGF-β), tumor necrosis factor (TNF-α), and platelet endothelial cell adhesion molecule-1 (PECAM-1 or CD31) expression in ovarian cancer cells by Ado.
Results: The relative viability of cells subsequent to Ado treatment proved to be both concentration- and time dependent. IHC results showed that Ado evidently enhanced the RhoGDI2 protein expression. In addition, interference with RhoGDI2 outstandingly attenuated the ability of Ado to suppress tumor cell invasion and induce angiogenesis in vitro. Furthermore, molecular mechanism studies indicated that Ado remarkably inhibited the expression of MMP-2, MMP-9, VEGF, TGF-β, TNF-α, and CD31, while interference with RhoGDI2 restored the expression of the above-mentioned angiogenic factors.
Conclusion: Ado inhibits the growth of A2780 human ovarian cancer cells through inhibiting tumor cell invasion and angiogenesis in a RhoGDI2-dependent manner.
Keywords: adenosine, ovarian cancer, RhoGDI2, invasion, growth, angiogenesis
Adenosine (Ado) is a purine nucleoside, which functions through binding to four corresponding Ado receptors, namely, A1, A2A, A2B, and A3.1 Ado is at a low extracellular level, which is markedly increased under metabolic stress, such as inflammation or cancer.2 Ado removes the harmful substances in cells through reducing tissue energy consumption, which thereby plays a protective role in the body.3 Ado is shown in plenty of studies to possess a wide range of pharmacological effects, including anti-inflammation,4 anti-oxidation,5 anti-pulmonary hypertension,6 and anti-tumor.7 Moreover, existing studies suggest that Ado exhibits anti-cancer activity in gastric cancer (GC),8 lung cancer,9 and colon cancer.10 In addition, Ado enhances the cisplatin activity in ovarian cancer.11 However, the mechanism of Ado in inhibiting ovarian cancer remains unclear so far, which should be further investigated. Recent studies show that Rho-specific guanine nucleotide dissociation inhibitor 2 (RhoGDI2) plays a critical part in regulating tumor cell invasion and metastasis.12,13 Nonetheless, it is unknown whether RhoGDI2 mediates the regulating process of Ado on ovarian cancer. Therefore, this study investigated the effects of Ado on ovarian cancer cell proliferation, growth, and angiogenesis using a subcutaneous xenograft nude mouse model and a tumor cell-induced tube formation model. Typically, this study focused on the role of RhoGDI2 in this process, as well as its molecular mechanisms of action, so as to provide a theoretical basis for further research and the clinical application of Ado.
Materials And Methods
Animals, Cell Line, And Drugs Used
The 6–8-week-old female BALB/c nude mice (SPF grade, 18–20 g) were provided by Vital River Laboratory Animal Technology (Beijing, China) under the production license number of SCXK (Beijing) 2018-0001. The rats were fed a standard laboratory diet (Department of Laboratory Animals, Central South University, Changsha, China). Before the rats were divided into experimental groups, they acclimated for a week. The protocol for the experiments on animals was approved by the Ethics Commission, Hunan Cancer Hospital, Changsha, China (the permission number 17/2018). The National Institutes of Health (NIH) guide for the care and use of laboratory animals was followed in the current study.
The human A2780 ovarian cancer cell line and normal ovarian epithelial cells (IOSE80 cells) were purchased from Shanghai Institute of Cell Biology, Chinese Academy of Sciences. Ado (purity ≥98%) was derived from Sigma-Aldrich (St. Louis, MO, USA, Cat. #. A9251). The RhoGDI2 siRNA plasmid was constructed by Jiangsu KeyGEN BioTECH (Nanjing, China). The 10% fetal bovine serum (FBS), DMEM medium, and 0.25% trypsin were bought from Gibco BRL, Life Technologies Inc. (Rockville, MD). The other reagents used in this study were shown below: BCA Protein Quantification Kit (Pierce, US, Cat. #. 23227) and Matrigel basement membrane matrix (BD Biosciences, US, Cat. #. 356234, Lot #. 2342761, packaging: 5 mL). Meanwhile, the antibodies used in Western blotting were as follows: MMP-2 (Proteintech, Cat. #: 10373-2-AP), MMP-9 (Proteintech, Cat. #. 10375-2-AP), VEGF (CST, Cat. #: 3852S), TGF-β (Proteintech, Cat. #. 22308-1-AP), TNF-α (Cell Signaling, Cat. #: 8902 SC), CD31 (Proteintech, Cat. #. 11265-1-AP), and GAPDH (CST, Cat. #: 3683).
Methods
Effect Of Ado On The Proliferation Of A2780 Ovarian Cancer Cells Detected By MTT Assay
A2780 cells were cultured in DMEM medium containing 10% fetal bovine serum, and cells in the logarithmic growth phase were trypsinized, centrifuged, and counted. The cell concentration was adjusted to 5×105 cells/mL in DMEM complete medium, and the cells were seeded into a 96-well plate (200 μL of cell suspension per well) and placed in a 37°C incubator. When the cultured cells grew to approximately 80% confluence, 0, 1, 5, 10, 20, 40, and 80 μM of Ado was added, and incubation was continued for 24 hrs, 48 hrs, 72 hrs, and 96 hrs. After the incubation, 20 μL of MTT reagent was added to each well and incubation was continued for 3 hrs. After the completion of the incubation, the absorbance value (OD value) was measured at a wavelength of 490 nm using an ultraviolet spectrophotometer and the inhibition rate was calculated.
Construction Of The Nude Mouse A2780 Subcutaneous Xenograft Model To Investigate The Effect Of Ado On Tumor Growth In Vivo
In brief, the A2780 human ovarian cancer cell line was subjected to thawing, passage, and culture according to the conventional methods. After cell expansion to 10 culture dishes and 90% confluence were achieved, cells were digested with trypsin, centrifuged to remove the trypsin, and resuspended within the PBS. Later, a 10× dilution was prepared, cells were counted, and the cell suspension was adjusted to 2 × 107 cells/mL. Cells were then mixed with an equivalent volume of Matrigel so that the final concentration of cell suspension was adjusted to 1 × 107 cells/mL. Then, the cell suspension was disinfected with iodine and 75% alcohol, and 0.2 mL was collected to inoculate into the lower right limbs of 20 healthy Balb/c nude mice. On the next day of inoculation, the experimental mice were randomly divided into 4 groups (n=5 in each group) based on their body weight, including model group, low-dose Ado group (20 mg/kg), high-dose Ado group (60 mg/kg), and cisplatin group (3 mg/kg). Dosing was initiated when the average tumor volume in each group was >100 mm3. Thereafter, mice were given an intraperitoneal injection of Ado for once a day or cisplatin for once every 3 days for a total of 21 days, while the model group was given an equivalent volume of saline. The tumor volume was measured for once every 3 days during the administration period. After 21 days of administration, the mice were sacrificed, and the tumor was excised from each mouse, weighed, fixed in the 10% buffered formalin, and embedded in paraffin. Afterwards, the tissue blocks were sectioned at a thickness of 6 µm and processed for IHC detection. The tumor volume was calculated as follows: V = A × B2 × 0.5, where A represents the maximum diameter of tumor, while B is the minimum diameter of tumor.
Effect Of Ado On The Expression Of RhoGDI2 Protein Expression In Ovarian Cancer Tissues Detected By IHC
First of all, the sections were subjected to deparaffinization using xylenes, followed by immersion in ethanol at various concentrations and then in pure water for rehydration. Then, 1 L of 0.01 M sodium citrate buffer solution (pH 6.0) was added into the boiling water for high-pressure heated antigen retrieval. Thereafter, 3% H2O2 was added dropwise onto each section, with 1 drop for every section. Subsequently, 1 drop of RhoGDI primary antibody 2 (dilution 1:100) was added into each section, and all sections were placed at 4°C overnight. On the next day, 1 drop of horseradish peroxidase (HRP)-conjugated secondary antibody was added onto each section and placed at 4°C for 30−40 mins. Afterwards, 1 drop of the freshly prepared DAB solution was applied onto each section for 20 mins of color development, and the nuclei were counterstained with light hematoxylin for 30 s. After staining, all sections were quickly dehydrated with ethanol at various concentrations, transparentized with xylenes, and mounted with neutral resin. After drying, each section was observed under the optical microscope and photos were taken.
Construction Of RhoGDI2 siRNA Plasmid And Cell Transfection
First, 5 μg RhoGDI2 siRNA was added to 150 μL serum-free and antibiotic-free medium to incubate for 5 mins at room temperature. Next, 3 μL LipofectAMINE® Reagent was added into 150 μL serum-free and antibiotic-free medium to incubate for 5 mins at room temperature. Afterwards, the above two solutions were mixed, then the mixture was further incubated for 20 mins at room temperature, and approximately 300 μL transfection complex was obtained. Later, cells were incubated with the transfection complex for 8 hrs at 37°C, followed by incubation with a complete medium for 16 hrs at 37°C.
Effects Of Ado And Interference With RhoGDI2 On Angiogenesis Induced By A2780 Cell Supernatants Detected By Tube Formation Assay
The 96-well plate and sterile pipette tips were pre-cooled at −20°C overnight, and then, the Matrigel base membrane matrix stored at −20°C was placed at 4°C for slow thawing overnight. The pre-cooled 96-well plate was placed onto the ice plate, and 100 μL Matrigel TM basement membrane matrix was added into each well. Later, the 96-well plate was placed at 4°C for 10 mins and then transferred into a 37°C cell incubator at 5% CO2/95% air condition for 30 mins. HUVECs at the logarithmic phase were washed with 10 mL PBS for once, then the PBS was discarded, and 4 mL of the 0.25% trypsin–0.02% EDTA was added for cell digestion. Subsequently, the digested cells were transferred into the 15 mL centrifuge tubes to centrifuge at 1000 rpm for 3 mins. Afterwards, cells were re-suspended into the basal medium, and cell concentration was adjusted to 1.5 × 105 cells/mL. Then, 100 μL cell suspension was slowly added into each well of the 96-well plate, as described above. Later, HUVEC cells cultured in the 96-well plates were divided into four groups, including group A with 100 µL PBS, groups B and C with 100 µL A2780 cell culture supernatant, and group D with 100 µL RhoGDI2 siRNA-transfected A2780 cell culture supernatant. After reacting with Ado at various concentrations (0 and 20 μM) for 24 hrs, tube formation was observed under a microscope and photographed, and the tube length was analyzed using ImageJ.
Effects Of Ado On The Expression Of MMP-2, MMP-9, VEGF, TGF-β, TNF-α, And CD31 In A2780 Cells After Interference With RhoGDI2 Detected By Western Blotting
A2780 cells were inoculated into the 6-well plates and cultured. After achieving 80% cell confluence, Ado treatment was initiated, and a vehicle control group (PBS) was set up. Then, cells were cultured for 24 hrs, the total protein was extracted using the RIPA lysis buffer, and the protein content was quantified according to the Bradford method. The Western blotting samples were prepared and subjected to sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE). After terminating electrophoresis, the samples were transferred onto the polyvinylidene fluoride (PVDF) membranes at a constant pressure of 100 V for 1.5 hrs. Subsequently, the PVDF membranes were blocked with 5% skim milk in TBST for 2 hrs, then corresponding antibodies (MMP-2, MMP-9, VEGF, TGF-β, TNF-α, and CD31) were added, and membranes were incubated at 4°C overnight. On the following day, the secondary antibody was added after membrane washing to incubate for 1.5 hrs; afterwards, the membrane was washed for 10 mins for 4 times and exposed to the gel imaging system. The experiment was repeated for three times.
Statistical Methods
One-way analysis of variance (ANOVA) was performed using the Graphpad 5.0 software, and the sample means were compared by Fisher’s LSD test. The results were presented as x̅ ± SD, and a difference of P < 0.05 was deemed as statistically significant.
Results
Ado Inhibited A2780 Ovarian Cancer Cell Proliferation In Vitro
To investigate the ability of Ado to inhibit the proliferation of A2780 ovarian cancer cells, we treated A2780 with different concentrations of Ado for 24 hrs, 48 hrs, 72 hrs, and 96 hrs. As shown in Figure 1, the relative viability of cells subsequent to Ado treatment proved to be both concentration- and time dependent (P< 0.05).
Ado Restrained The Growth Of A2780 Subcutaneous Xenograft In Nude Mice
Results of subcutaneous xenograft assay in nude mice suggested no significant difference in the body weight between the Ado-treated group and the model group; meanwhile, cisplatin treatment remarkably reduced the body weight of mice, indicating that Ado caused no systemic toxicity in nude mice (Figure 1B). In addition, compared with model group, Ado at the doses of 20 mg/kg and 60 mg/kg dramatically reduced the volume of A2780 xenograft in a dose-dependent manner (Figure 1C and D, Table1). These results indicated that Ado inhibited the growth of A2780 cells in vivo.
 |
Ado Enhanced RhoGDI2 Expression In The A2780 Subcutaneous Xenografts In Nude Mice
IHC results showed that RhoGDI2 expression was remarkably lower in tumor cells than in paracancerous tissues of model group. Moreover, it was found after Ado and cisplatin treatment that Ado enhanced RhoGDI2 protein expression in a dose-dependent manner relative to that in model group (Figure 2). These results indicated that Ado enhanced RhoGDI2 expression in the A2780 subcutaneous xenografts in nude mice.
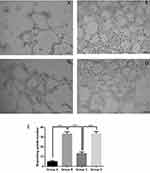
Interference With RhoGDI2 Disrupted The Ability Of Ado To Inhibit Tube Formation
Results of tube formation assays suggested that, compared with control group (PBS), the culture supernatant of A2780 cells markedly stimulated tube formation (P < 0.001). After Ado treatment, the tumor-induced tube formation was dramatically reduced (P < 0.001) compared with the 0 μM group. However, interference with RhoGDI2 greatly reduced the ability of Ado to inhibit tube formation compared with that in the non-interference group (P < 0.001, Figure 3). Results of this assay suggested that interference with RhoGDI2 disrupted the ability of Ado to inhibit tube formation.
Ado Suppressed The Protein Expression Of MMP-2, MMP-9, VEGF, TGF-β, TNF-α, And CD31 In A RhoGDI2-Dependent Manner
Results of Western blotting showed that Ado restrained the expression of invasion-related proteins MMP-2 and MMP-9, as well as the angiogenesis-related proteins VEGF, TGF-β, TNF-α, and CD31. Moreover, interference with RhoGDI2 evidently decreased the ability of Ado to inhibit the expression of the above-mentioned proteins (Figure 4). The above findings revealed that Ado inhibited the expression of angiogenesis-related factors in a RhoGDI2-dependent manner.
Discussion
In this study, the effects of Ado on the growth and angiogenesis of A2780 ovarian cancer cells were investigated through constructing a subcutaneous xenograft model in nude mice and a tumor cell-induced tube formation model, so as to examine the effect of interference with RhoGDI2 on Ado regulation. Our experimental results showed that Ado suppressed the proliferation and growth of A2780 ovarian cancer cells both in vitro and in vivo; in addition, Ado restrained the angiogenesis induced by treatment with A2780 ovarian cancer cell supernatant in HUVECs. In addition, it was discovered that RhoGDI2 was lowly expressed in ovarian cancer, but Ado treatment markedly up-regulated its expression. Besides, the effect of high-dose Ado on up-regulating RhoGDI2 expression was almost equivalent to that of cisplatin. Interference with RhoGDI2 dramatically reduced the ability of Ado to inhibit tube formation, suggesting that RhoGDI2 was involved in regulating tumor angiogenesis.
RhoGDI2 is suggested in mechanistic study to mediate tumor invasion and metastasis,14,15 and MMP-2 and MMP-9 are identified as the key proteins in regulating tumor cell invasion.16 Therefore, this study first determined the effect of interference with RhoGDI2 on the expression of MMP-2 and MMP-9. Our results showed that Ado inhibited the expression of MMP-2 and MMP-9, whereas interference with RhoGDI2 evidently reduced the inhibition of Ado on these proteins. These findings indicated that RhoGDI2 participated in regulating tumor cell invasion, which was achieved through regulating the expression of MMP-2 and MMP-9, suggesting that RhoGDI2 was a potential target for anti-tumor invasion and metastasis strategies. In addition, based on the critical role of angiogenic factors (such as VEGF, TGF-β, TNF-α, and CD31) in regulating tumor angiogenesis, inhibiting the expression of these factors effectively controls the angiogenesis process.17 CD31 is closely related to angiogenesis, and vascular system in tumor microenvironment plays an important role in tumor cell proliferation and invasion. CD31 can be a target for tumor therapy. Therefore, this study focused on the effect of interference with RhoGDI2 on the expression of these angiogenic factors. It was found that Ado evidently suppressed the expression of VEGF, TGF-β, TNF-α, and CD31, while interference with RhoGDI2 reversed the inhibition of angiogenesis by Ado. These findings suggested that RhoGDI2 participated in controlling angiogenesis through regulating the expression of these angiogenic factors. Thus, RhoGDI2 was also a potential target protein for regulating angiogenesis.
In conclusion, RhoGDI2 is involved in the invasion and angiogenesis of ovarian cancer, and Ado inhibits ovarian cancer growth through enhancing RhoGDI2 expression. Future studies should focus on the effects of over-expressing RhoGDI2 on tumor growth in vivo, so as to determine the molecular mechanism by which RhoGDI2 regulates tumor growth. In addition, the key roles of various proliferation- and growth-regulating signaling pathways in this process should also be investigated. Findings in this study show that Ado can potentially serve as a highly valuable anti-tumor compound, which displays promising prospects for clinical application.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Manjunath S, Sakhare PM. Adenosine and adenosine receptors: newer therapeutic perspective. Indian J Pharmacol. 2009;41(3):97–105. doi:10.4103/0253-7613.55202
2. Daval JL, Nehlig A, Nicolas F. Physiological and pharmacological properties of adenosine: therapeutic implications. Life Sci. 1991;49(20):1435–1453. doi:10.1016/0024-3205(91)90043-B
3. Yap SC, Lee HT. Adenosine and protection from acute kidney injury. Curr Opin Nephrol Hypertens. 2012;21(1):24–32. doi:10.1097/MNH.0b013e32834d2ec9
4. Cronstein BN. Adenosine, an endogenous anti-inflammatory agent. J Appl Physiol (1985). 1994;76(1):5–13. doi:10.1152/jappl.1994.76.1.5
5. Lu J, Zhu SM, Zang WJ, et al. Protective mechanism of adenosine to the rat arterial endothelial dysfunction induced by hydrogen peroxide. Biol Pharm Bull. 2007;30(7):1206–1211. doi:10.1248/bpb.30.1206
6. Fullerton DA, Jones SD, Grover FL, McIntyre RC
7. Stagg J, Smyth MJ. Extracellular adenosine triphosphate and adenosine in cancer. Oncogene. 2010;29(39):5346–5358. doi:10.1038/onc.2010.292
8. Tsuchiya A, Nishizaki T. Anticancer effect of adenosine on gastric cancer via diverse signaling pathways. World J Gastroenterol. 2015;21(39):10931–10935. doi:10.3748/wjg.v21.i39.10931
9. Yang D, Song J, Wu L, et al. Induction of senescence by adenosine suppressing the growth of lung cancer cells. Biochem Biophys Res Commun. 2013;440(1):62–67. doi:10.1016/j.bbrc.2013.09.030
10. Yu S, Hou D, Chen P, et al. Adenosine induces apoptosis through TNFR1/RIPK1/P38 axis in colon cancer cells. Biochem Biophys Res Commun. 2015;460(3):759–765. doi:10.1016/j.bbrc.2015.03.103
11. Sureechatchaiyan P, Hamacher A, Brockmann N, Stork B, Kassack MU. Adenosine enhances cisplatin sensitivity in human ovarian cancer cells. Purinergic Signal. 2018;14(4):395–408. doi:10.1007/s11302-018-9622-7
12. Harding MA, Theodorescu D. RhoGDI2: a new metastasis suppressor gene: discovery and clinical translation. Urol Oncol. 2007;25(5):401–406. doi:10.1016/j.urolonc.2007.05.006
13. Stevens EV, Banet N, Onesto C, et al. RhoGDI2 antagonizes ovarian carcinoma growth, invasion and metastasis. Small GTPases. 2011;2(4):202–210. doi:10.4161/sgtp.2.4.17795
14. Cho HJ, Baek KE, Yoo J. RhoGDI2 as a therapeutic target in cancer. Expert Opin Ther Targets. 2010;14(1):67–75. doi:10.1517/14728220903449251
15. Gildea JJ, Seraj MJ, Oxford G, et al. RhoGDI2 is an invasion and metastasis suppressor gene in human cancer. Cancer Res. 2002;62(22):6418–6423.
16. Li H, Qiu Z, Li F, Wang C. The relationship between MMP-2 and MMP-9 expression levels with breast cancer incidence and prognosis. Oncol Lett. 2017;14(5):5865–5870. doi:10.3892/ol.2017.6924
17. Breier G, Blum S, Peli J, et al. Transforming growth factor-beta and Ras regulate the VEGF/VEGF-receptor system during tumor angiogenesis. Int J Cancer. 2002;97(2):142–148. doi:10.1002/ijc.1599
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.