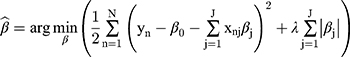Back to Journals » Neuropsychiatric Disease and Treatment » Volume 17
A Random Forest Model for Predicting Social Functional Improvement in Chinese Patients with Schizophrenia After 3 Months of Atypical Antipsychotic Monopharmacy: A Cohort Study
Authors Li Y , Zhang L, Zhang Y, Wen H, Huang J, Shen Y , Li H
Received 25 November 2020
Accepted for publication 2 March 2021
Published 19 March 2021 Volume 2021:17 Pages 847—857
DOI https://doi.org/10.2147/NDT.S280757
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Taro Kishi
Yange Li,1 Lei Zhang,1 Yan Zhang,1 Hui Wen,1 Jingjing Huang,1,2 Yifeng Shen,1– 3 Huafang Li1– 3
1Mental Health Center, Shanghai Jiao Tong University School of Medicine, Shanghai, People’s Republic of China; 2Shanghai Key Laboratory of Psychotic Disorders, Mental Health Center, Shanghai Jiao Tong University School of Medicine, Shanghai, People’s Republic of China; 3Shanghai Clinical Research Center for Mental Health, Shanghai, People’s Republic of China
Correspondence: Huafang Li
Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine, 600, Wan Ping Nan Lu, Shanghai, 200030, People’s Republic of China
Tel +86 21 34773122
Fax +86-21-54240982
Email [email protected]
Purpose: Impaired social functions contribute to the burden of schizophrenia patients and their families, but predictive tools of social functioning prognosis and specific factors are undefined in Chinese clinical practice. This article explores a machine learning tool to identify whether patients will achieve significant social functional improvement after 3 months of atypical antipsychotic monopharmacy and finds the defined risk factors using a multicenter clinical study.
Patients and Methods: A multicenter study on atypical antipsychotic (AAP) treatment in Chinese patients with schizophrenia (SALT-C) was conducted from July 2011 to August 2018. Data from 550 patients with AAP monopharmacy from their baseline to their 3-month follow-up were used to establish machine learning tools after screening. The positive outcome was an increase in the Personal and Social Performance (PSP) scale score by ≥ 10 points. The predictors were a range of investigator-rated assessments on symptoms, functioning, the safety of AAPs and illness history. The Least Absolute Shrinkage and Selection Operator (LASSO) was used for the feature screening and ranking of the predicted variables. The random forest algorithm and five-fold cross-validation for optimizing the model were selected to ensure the generalizability and precision.
Results: There were 137 patients (mean [SD] age, 41.1 [16.8] years; 77 [58.8%] female) who had a good social functional prognosis. A lower PSP score, taking a mood stabilizer, a high total Positive and Negative Symptom Scale (PANSS) and PANSS general subscale score, unemployment, a hepatic injury with medication, comorbid cardiovascular disease and being male predicted poor PSP outcomes. The generalizability of the PSP predictive tool was estimated with the precision–recall curve (accuracy of 79.5%, negative predictive value of 92.6% and positive predictive value of 57.1%) and receiver operating characteristic curve (ROC) (specificity of 81.8% and sensitivity of 78.7%).
Conclusion: The machine learning tool established using our current real-world data could assist in predicting PSP outcome by several clinical factors.
Keywords: schizophrenia, atypical antipsychotics, social functional improvement, Personal and Social Performance, PSP
Introduction
The ideal prognosis for schizophrenia includes both the relief of core symptoms and functional improvement as this severe disorder contributes to the burden of patients’ families and society.1,2 Social dysfunction represents the deterioration of functioning in one or more fields – interpersonal contacts, work or school, self-care, disturbing and aggressive behavior – in the Diagnostic and Statistical Manual of Mental Disorders (DSM-5). In most patients with schizophrenia onset in adolescence, social functioning is severely affected and progressively declines as the number of acute episodes increases.3,4
Many studies have focused on factors associated with social functioning and previously identified potential predictors including sociodemographic factors, illness duration, Positive and Negative Symptom Scale (PANSS) scores, cognition, antipsychotic use and others. Of these predictors, patients with a poor functional prognosis were found to be older, have a longer illness duration, have more severe psychiatric symptoms, have worse cognitive functioning, be unemployed, be poorly educated, and be male.5–14 However, the comprehensive effect of these factors on social functioning is still unclear since individual patients with schizophrenia demonstrate great heterogeneity in disease conditions, adverse drug reactions, family economy and other characteristics; and predicting their social functional outcomes using traditional methods is difficult. There are many factors related to social functional improvement, which is why we chose machine learning.
Establishing clinical predictive models using machine learning tools is a major breakthrough in medical research in recent years;15 and machine learning has been used to successfully resolve a variety of problems in biology and medicine, including predicting drug responses in cancer cell lines,16 identifying DNA-binding proteins, and locating cancer lesions in specific tissues from the result of liquid biopsies. Many articles on predicting the therapeutic outcomes of patients with mental disorders have been published.8,17 Predicting social functional outcomes with machine learning might help clinicians and patients with their decision-making and improve functional prognoses. Therefore, based on our observational study on Chinese schizophrenia patients taking atypical antipsychotics (AAPs), we aimed to establish a predictive model of social functional improvement using the baseline and 3-month follow-up information of patients with AAP monopharmacy.
Patients and Methods
Study Design and Database
An observational study on long-term AAP treatment in Chinese patients with schizophrenia (SALT-C; International Clinical Trails Registry Platform number, NCT02640911) was conducted in 13 hospitals starting in July 2011. Our sample comes from hospitals, outpatient and ward doctors who initially screened schizophrenia patients using atypical antipsychotic drugs. Then, on a voluntary basis, we evaluated them in detail using uniformly trained researchers. The goal of this real-world study was to evaluate the long-term outcomes of treatment with atypical antipsychotics in schizophrenia. The outcomes were the tolerability of AAPs and functional changes with AAP treatment. The study was approved by the Ethics Committee of the Shanghai Mental Health Center and conducted in accordance with the ethical principles of the Declaration of Helsinki. Written informed consent was obtained from all participants prior to study enrollment.
Subjects had to meet the following inclusion criteria: (a) age ≥ 18 years, (b) a DSM-IV diagnosis of schizophrenia, and (c) taking AAPs. The exclusion criteria were situations where patients were deemed unsuitable for enrollment by the investigator, such as suicidal tendencies, serious physical illnesses, and poor patient compliance. Variables were extensively collected, such as demographic characteristics, psychiatric and physical illness history, a range of investigator-rated measures of symptom severity and others. Regular assessments were conducted at the baseline, 3 months, 6 months, and then every half a year. All needed information was comprehensively assessed by trained investigators and then input into a uniform electronic data capture system.
Prior to August 2018, 779 patients completed the 3-month follow-up. Except for 229 patients with antipsychotic polypharmacy, information from 550 AAP monopharmacy patients was used to predict social functional outcomes with a machine learning approach (Figure 1).
Definition of Outcome Measures and Predictors
The Personal and Social Performance scale (PSP) is applicable to both acute and stable schizophrenia patients18–20 for assessing functioning last month, and the accuracy and reliability of the corresponding Chinese version have been confirmed.21 The PSP consists of four dimensions: (a) socially beneficial activities, including work and study; (b) personal relationships and social relationships; (c) self-care ability; and (d) disturbing and aggressive behaviors. The PSP score can be divided into 10 levels using every 10 points to represent different degrees of dysfunction, and the minimum detectable change caused by nonrandom variables in continuous measurements of the PSP was 10.7 points.22 In this study, the positive social functional outcome of machine learning was defined as an increase of ≥10 points in the PSP score after treatment.
The predictive variables were from a series of clinically available information in the SALT-C study, including demographics, psychiatric symptoms (PANSS, the Calgary depression scale for schizophrenia [CDSS], and clinical global impression-schizophrenia [CGI-S]), antipsychotic use, treatment compliance (medication satisfaction questionnaire [MSQ], drug attitude inventory [DAI-10] and subjective well-being under neuroleptics [SWN]), the tolerability of AAPs (Simpson-Angus scale [SAS], Barnes akathisia [BARS], abnormal involuntary movement scale [AIMS], electrocardiograph [ECG] and serum prolactin), comorbidities and concomitant medication use.
The frequency and proportion of missing data of each variable were described, and the data that were missing for different reasons were processed. The missing data with a random cause with a proportion >5% were deleted directly. Dose equivalents of antipsychotics based on oral 100 mg/d chlorpromazine were adopted from previous publications23,24 and the defined daily dose (DDD).
Machine Learning Analyses
The relationship between all predictors and outcome measures was analyzed using single-factor analysis. Continuous variables were analyzed using the t-test or nonparametric equivalent, and categorical variables were analyzed using the chi-squared test or Fisher test. This study used LASSO, namely, the L1-regularized multivariate logistic regression, for feature screening; and a random forest algorithm to establish the model. Compared with an ordinary logistic regression, LASSO adds a penalty function to the equation to establish a more refined model. The formula of the penalty function is as follows:
In this formula, N represents the number of samples, xnj represents the independent variables, J represents the number of independent variables, yn represents the dependent variable, b0 represents the intercept, and l represents the parameter of nonnegative regularization that controls the extent of regularization. 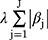 prevents the regularization from overfitting. By introducing the penalty function, the LASSO regression can eliminate the coefficients of variables that do not affect the predictive ability without having to consider the multicollinearity between variables and retain only coefficients of important variables that can improve the predictive ability.25 Therefore, LASSO is a very effective algorithm for the feature selection of high-dimensional data.
prevents the regularization from overfitting. By introducing the penalty function, the LASSO regression can eliminate the coefficients of variables that do not affect the predictive ability without having to consider the multicollinearity between variables and retain only coefficients of important variables that can improve the predictive ability.25 Therefore, LASSO is a very effective algorithm for the feature selection of high-dimensional data.
Five machine learning algorithms, including the logistic regression (LR), the stochastic gradient descent (SGD), the gradient boosting decision tree (GBDT), extreme gradient boosting (XGB) and the random forest (RF), were compared in this study. The RF algorithm achieved the best receiver operating characteristic curve (ROC) and precision–recall curve (Figure 2). The RF is a nonparametric model proposed by Leo Breiman to solve the classification (two-category or multicategory outcomes) and regression (continuous outcomes) issues. It is a combination of multiple decision trees26 and one machine learning algorithm with high predictive accuracy. Samples are classified by the decision points of each decision tree, and the classification results of all decision trees are finally collected to calculate the probability of occurrence of the outcome.27
We further divided 550 patients into a training set (70%) and a testing set (30%) and optimized the model parameters using 5-fold cross-validation. Then, for the established model, we used the accuracy, positive predicted value (PPV), negative predicted value (NPV), area under the curve (AUC), sensitivity, and specificity to describe the effectiveness of the model in predicting the outcomes of the training set and testing set. The analysis software packages involved were SPSS 22.0 and Python.
Results
There were 550 patients (mean age 48.2 years, women 41.5% and outpatients 17.1%) treated with AAP monopharmacy for 3 months in our database. In addition, 159 (28.9%) of them had an increase of ≥10 points in the PSP score, which was defined as the positive group; and the other 391 patients composed the negative group. The positive group was more likely to be younger; be female; be unmarried; be employed; be an outpatient; be experiencing their first episode; have a shorter total illness duration; have fewer relapses; have a shorter duration of their current episode; have a lower baseline PSP score; have more severe symptoms across the PANSS, CDSS and CGI-S measures; and have fewer comorbidities of the affective disorder, including liver and cardiovascular disease (Table 1).
 |
Table 1 550 Mono-Antipsychotic Patients’ Description Between PSP Improved ≥10 Points and <10 Points Groups |
The PSP score change and proportion of PSP increased by ≥10 points among the four AAP monopharmacy groups in our current database did not show significant differences. Different AAP monopharmacy groups demonstrated different characteristics in Chinese clinical treatment. The clozapine group was mainly refractory patients and showed the earliest onset age, longest illness duration, most relapses, highest proportion of inpatients, lowest proportion of employed patients, most severe psychiatric symptoms, poorest functioning in socially useful activities, poorest personal and social relationships and poorest baseline PSP total score. The aripiprazole group was characterized by the youngest age, fewer severe psychotic symptoms, fewer relapses and inpatients, and a relatively good baseline PSP total score. The olanzapine and risperidone groups were identical in most items, and the characteristics and severity of the patients were in the middle of those of the other two groups (Table 2).
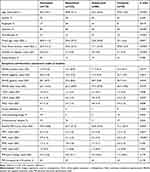 |
Table 2 Compare Among Four AAP Monopharmacy Groups |
When establishing the model, we tested five machine learning algorithms and evaluated their predictive ability using the AUC. The random forest algorithm had a stable and relatively good predictive ability when the top 13 variables were included in both the training and testing sets. The AUCs were close to 0.82 and 0.86 in the training and testing sets, respectively (Figure 2A and B). Five-fold cross-validation of the random forest model in the ROC curve and precision–recall curve (P-R curve) both showed good generalizability (Figure 2C and D).
Table 3 describes the top 13 predictors in the model for predicting an increase of ≥10 points in the PSP score. Comorbid mood stabilizers, high total PANSS and PANSS general subscale scores, unemployment, hepatic injury with medication, being male, and a comorbid cardiovascular disease predicted a poor PSP outcome. The baseline PSP score was the most important influencing factor. For patients with a baseline PSP score >70 points, the positive outcome percentage was approximately 5.5%. For patients with a baseline PSP score ≤70, the percentages were approximately 50% in first-episode patients and 25% in recurrent patients in our current study. Mood stabilizers are often used as adjuvants to antipsychotics in schizophrenia patients with agitation and aggressive behavior. In our study, 45 patients (mean illness duration 29.5 years and mean PSP score change 0.3 points) taking mood stabilizers were inpatients, and only 3 patients (6.7%) demonstrated PSP increases of ≥10 points.
 |
Table 3 Top 13 Predictive Items of PSP Improved ≥10 Points Predicted Model |
Table 4 summarizes the model’s training and validation performance, showing that the model had a high sensitivity (81.8%) and specificity (78.7%) with an accuracy as high as 79.5%, especially when predicting an increase of <10 points in the PSP score (92.6%).
 |
Table 4 Model Performance During Training and Validation |
Discussion
This observational study was conducted to make the results generalizable and maximally useful in routine clinical situations. In this observational study of Chinese schizophrenia patients, we first established a predictive model of PSP improvement using machine learning. Taking mood stabilizers, high total PANSS and PANSS general subscale scores, unemployment, a hepatic injury with medication, being male, and having a comorbid cardiovascular disease predicted poor PSP outcomes after 3 months of AAP treatment. Though the great heterogeneity among the investigated subjects was representative of the broad group of Chinese schizophrenia patients, the validated results in the testing set showed that the accuracy of the model reached 79.5%.
Using the current data to model and sort the predictive variables, the most important variables (importance≥0.15) were the PSP score at the baseline, mood stabilizers and the PANSS total score. In our study, patients with low PSP baseline scores were more likely to improve by ≥10 points after 3 months of treatment whereas patients with a baseline PSP ≥70 rarely improved by more than 10 points after 3 months of medication. Antipsychotics in the acute phase can alleviate the core symptoms of patients and obtain early improvement in the PSP, and maintenance phase treatment can ensure the continuous improvement and stability of the PSP.28 When the PSP improves to a certain degree (such as a PSP of more than 70 points), the drug cannot further significantly improve the social functioning of the patient.29 Some researchers believe that part of the theoretical basis is the change in the morphological structure of brain tissues. Only patients with slight atrophy of the marginal forebrain and the entire brain volume, small ventricles, and sufficient activity will have a better prognosis in social cognitive functions.30 With the increase in relapse times, the brain tissue progressively decreases,4,31 and the functional level decreases. After multiple relapses, a patient enters the treatment resistance period, showing chronic migration, residual symptoms, and severely impaired function.3,4 In addition, there were more studies on the correlation between the PANSS and functional outcomes. In the acute phase of schizophrenia, both the PANSS and CGI-S are negatively correlated with the PSP.19 For every 1 decrease in the PANSS total score, the PSP score increases by 0.42 points.32 The improvement of the PSP in remission was related to PANSS negative symptoms, onset age, the disability assessment scale-II (WHO) and psychological rehabilitation.33 There was no improvement in negative symptoms after 3 months of treatment (Table 1), which may be part of the reason why the PSP score could not be further increased and social function could not be fully restored. The prevalence of mood stabilizer use as adjunctive treatment with antipsychotics varied from 8.7% to 19.9% on different continents.34 Using a mood stabilizer as an adjunctive treatment was common among female gender and younger schizophrenia patients characterized by agitation or obvious hostility in clinical practice.7,35,36 According to our observational study, patients using a mood stabilizer did not show obvious improvement in the PANSS, the CGI-S, the CDSS, the PSP and other aspects after antipsychotic treatment (Table 5). This may be explained by the use of mood stabilizers to control aggressive behavior and in the treatment of resistant schizophrenia.
 |
Table 5 Compare Between Comorbid and Non-Comorbid Mood Stabilizer Groups |
Our results also suggest that male and unemployed patients are more likely to show poor social functional improvement, which was identified with previous results. As early as 1919, Kraepelin found that male patients were more likely to relapse frequently with more serious conditions.10 This gender difference was thought to be due to the differences in social cognition between men and women. Women have an advantage in nonwritten and oral learning and memory, emotional management, and the perception of happiness.37,38 In this way, female patients may have better social functioning.
Other indicators, such as liver injury treated with medication and cardiovascular disease, negatively affected the social functional prognosis in our current study. Most antipsychotics are metabolized by the liver, and liver toxicity in individuals delays or exacerbates conditions and increase the difficulty of antipsychotic treatment. Cardiovascular diseases can be induced by genetic susceptibility, antipsychotics, lifestyle of decreased physical and social activities attributable to negative symptoms, alcohol and tobacco abuse, and limited medical resources available.39 In our study, patients with cardiovascular disease were older and had a greater BMI and a longer duration of illness. They suffer a heavier burden under the dual pressure of physical comorbidities and severe mental disorder; thus, a worse prognosis according to the PSP seems to be inevitable.
According to our current data, olanzapine, risperidone, aripiprazole and clozapine did not show significant differences in PSP improvement, and olanzapine and aripiprazole showed a slight advantage. Patients treated with antipsychotics usually achieve a PSP improvement in an early stage of treatment, but they achieve only slightly or moderately improved social functioning.28,29 Though their action on alleviating psychotic symptoms improved with the continuous development of antipsychotics, this progress had not been fully reflected in functional improvement.40 In other studies, different AAPs also showed no statistically significant difference in PSP improvement.41,42 One possible reason is the change in the morphology of the brain – brain tissue is progressively lost and cerebral function declines with schizophrenia relapses.4,31 Based on this study, the result may be uncorrected owing to the unbalanced subject numbers and short observational duration. PSP improvement could be influenced by many factors, and AAPs may be just one of the ordinary factors. In addition, the AAPs that some SALT-C patients took were the ultimate treatment after weighting antipsychotic efficacy and safety through trial and error. Moreover, the effects of different factors, including AAPs, on PSP improvement probably counteracted each other.
The study has some limitations. First, the tool can only predict the probability of PSP improvement by ≥10 points after 3 months of treatment with AAPs but cannot predict long-term PSP. However, the database was from a real-world study; and due to its diverse medical history and courses of disease among the modeled patients, the model is suitable for repeated use in any disease course in any individual. Schizophrenia relapses easily, and 3 months is a commonly used treatment cycle in clinical practice, making the model more conducive to clinical application. Second, the number of patients in the eight AAP subgroups was relatively small and not sufficient to further clarify the effects of different drugs on the PSP, especially the effect of quetiapine on functional improvement; therefore, a larger sample size is needed. Given that the model was designed to explore the factors influencing an increase of ≥10 points in the PSP score among a large number of clinical variables, the distinction among the eight AAPs may not be recognized due to insignificant differences. Third, the effects of other treatments (such as rTMS and psychosocial therapy) on PSP outcomes were not quantified and included in the analysis. Last, this is an observational study, which does not impose too many restrictions on the subjects when collecting samples. The current follow-up period is only 3 months, which is relatively short; therefore, no other methods, including long-short term memory networks (LSTMs), have been applied.
Conclusion
In summary, this is the first attempt to use machine learning to establish a probabilistic predictive model of an increase of ≥10 points in the PSP after atypical antipsychotic treatment in a real-world study. Four AAPs (olanzapine, risperidone, aripiprazole and clozapine) did not show significant differences in social functional improvement according to our current data. Furthermore, physical comorbidities might play an important role. External validation using an independent database to gain insight into the reliability of the model is critical. With the deepening of psychiatric research, it is possible to include genetic markers, EEG images, and laboratory-identified biochemical or inflammatory markers to establish more accurate predictive models.
Data Sharing Statement
The data that support the findings of this study are from a malleable research project that we are still working on. The data are still being analyzed, and so are not publicly available. The specific time of data sharing will be determined by research progress and data analysis.
Acknowledgments
We gratefully acknowledged Fei Gu and Dan Tang of the Shanghai Null Hypothesis Information and Technology Ltd., for their technological support in this study. And this research was supported by Clinical Research Center, Shanghai Jiao Tong University School of Medicine (DLY201620), Shanghai Municipal Hospital Emerging Frontier Technology Joint Research Project (SHDC12017128), Shanghai Clinical Research Center for Mental Health [19MC1911100(SCRC-MH)], Collaborative Innovation Center for Translational Medicine at Shanghai Jiao Tong University School of Medicine (TM201727) and SMHC clinical research center (CRC2018DSJ01-2). Neither of them was involved in the study design, execution, data analysis, interpretation of the data, the writing of the paper, nor in the decision for publication.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Lehman AF, Lieberman JA, Dixon LB, et al. Practice guideline for the treatment of patients with schizophrenia, second edition. Am J Psychiatry. 2004;161(2 Suppl):1–56.
2. Liang S. Clinical guidelines for treatment of schizophrenia; 2004.
3. Birchwood M, Todd P, Jackson C. Early intervention in psychosis. The critical period hypothesis. Br J Psychiatry Suppl. 1998;172(33):53–59. doi:10.1192/S0007125000297663
4. Keshavan MS, Benes FM, Braff DL, et al. Proceedings and data from The Schizophrenia Summit: a critical appraisal to improve the management of Schizophrenia. J Clin Psychiatry. 2009;70(Suppl 1):4–46. doi:10.4088/JCP.7124su1cj
5. Tsang HW, Leung AY, Chung RC, et al. Review on vocational predictors: a systematic review of predictors of vocational outcomes among individuals with schizophrenia: an update since 1998. Aust N Z J Psychiatry. 2010;44(6):495–504. doi:10.3109/00048671003785716
6. Keefe RS, Seidman LJ, Christensen BK, et al. Comparative effect of atypical and conventional antipsychotic drugs on neurocognition in first-episode psychosis: a randomized, double-blind trial of olanzapine versus low doses of haloperidol. Am J Psychiatry. 2004;161:985–995. doi:10.1176/appi.ajp.161.6.985
7. Ba MB, Zanello A, Varnier M, Koellner V, Merlo MC. Deficits in neurocognition, theory of mind, and social functioning in patients with schizophrenic disorders: are they related? J Nerv Ment Dis. 2008;196(2):153–156. doi:10.1097/NMD.0b013e318162aa08
8. Koutsouleris N, Kahn RS, Chekroud AM, et al. Multisite prediction of 4-week and 52-week treatment outcomes in patients with first-episode psychosis: a machine learning approach. Lancet Psychiatry. 2016;3(10):935–946. doi:10.1016/S2215-0366(16)30171-7
9. Norlelawati AT, Kartini A, Norsidah K, Ramli M, Wan Azizi WS, Tariq AR. Relationship of psychological symptoms, antipsychotics and social data with psychosocial function in schizophrenia patients in Malaysia. Asia Pac Psychiatry. 2015;7(1):45–53. doi:10.1111/appy.12089
10. Kraepelin E. Dementia Praecox and Paraphrenia 1919. Huntington, New York: Publishing Co. Inc; 1971.
11. Walther S, Eisenhardt S, Bohlhalter S, et al. Gesture performance in schizophrenia predicts functional outcome after 6 months. Schizophr Bull. 2016;42(6):1326–1333. doi:10.1093/schbul/sbw124
12. Lang FU, Kosters M, Lang S, Becker T, Jager M. Psychopathological long-term outcome of schizophrenia – a review. Acta Psychiatr Scand. 2013;127(3):173–182. doi:10.1111/acps.12030
13. Zipursky RB, Reilly TJ, Murray RM. The myth of schizophrenia as a progressive brain disease. Schizophr Bull. 2013;39(6):1363–1372. doi:10.1093/schbul/sbs135
14. Park IJ, Chung Jung D, Suk-Hyun Hwang S, et al. Longitudinal relationship between Personal and Social Performance (PSP) and anxiety symptoms in schizophrenia. J Affect Disord. 2016;190:12–18. doi:10.1016/j.jad.2015.09.048
15. Suvisaari J, Mantere O, Keinanen J, et al. Is it possible to predict the future in first-episode psychosis? Front Psychiatry. 2018;9:580. doi:10.3389/fpsyt.2018.00580
16. Riddick GSH, Song H, Ahn S, et al. Predicting in vitro drug sensitivity using random forests. Bioinformatics. 2011;27(2):220–224. doi:10.1093/bioinformatics/btq628
17. Chekroud AM, Zotti RJ, Shehzad Z, et al. Cross-trial prediction of treatment outcome in depression: a machine learning approach. Lancet Psychiatry. 2016;3(3):243–250. doi:10.1016/S2215-0366(15)00471-X
18. Nasrallah H, Morosini P, Gagnon DD. Reliability, validity and ability to detect change of the Personal and Social Performance scale in patients with stable schizophrenia. Psychiatry Res. 2008;161(2):213–224. doi:10.1016/j.psychres.2007.11.012
19. Patrick DL, Burns T, Morosini P, et al. Reliability, validity and ability to detect change of the clinician-rated Personal and Social Performance scale in patients with acute symptoms of schizophrenia. Curr Med Res Opin. 2009;25(2):325–338. doi:10.1185/03007990802611919
20. Figueira ML, Brissos S. Measuring psychosocial outcomes in schizophrenia patients. Curr Opin Psychiatry. 2011;24(2):91–99. doi:10.1097/YCO.0b013e3283438119
21. Tianmei S, Liang S, Yun’ai S, et al. The Chinese version of the Personal and Social Performance Scale (PSP): validity and reliability. Psychiatry Res. 2011;185(1–2):275–279. doi:10.1016/j.psychres.2010.05.001
22. Lee SC, Tang SF, Lu WS, et al. Minimal detectable change of the Personal and Social Performance scale in individuals with schizophrenia. Psychiatry Res. 2016;246:725–729. doi:10.1016/j.psychres.2016.10.058
23. Inada T, Inagaki A. Psychotropic dose equivalence in Japan. Psychiatry Clin Neurosci. 2015;69(8):440–447. doi:10.1111/pcn.12275
24. Leucht S, Samara M, Heres S, et al. Dose equivalents for second-generation antipsychotic drugs: the classical mean dose method. Schizophr Bull. 2015;41(6):1397–1402. doi:10.1093/schbul/sbv037
25. Klyuzhin IS, Fu JF, Hong A, et al. Data-driven, voxel-based analysis of brain PET images: application of PCA and LASSO methods to visualize and quantify patterns of neurodegeneration. PLoS One. 2018;13(11):e0206607. doi:10.1371/journal.pone.0206607
26. Breiman L. Random forests. Mach Learn. 2001;45:5–32. doi:10.1023/A:1010933404324
27. Denisko D, Hoffman MM. Classification and interaction in random forests. Proc Natl Acad Sci U S A. 2018;115(8):1690–1692. doi:10.1073/pnas.1800256115
28. Buchanan RW, Kreyenbuhl J, Kelly DL, et al. The 2009 schizophrenia PORT psychopharmacological treatment recommendations and summary statements. Schizophr Bull. 2010;36(1):71–93. doi:10.1093/schbul/sbp116
29. Kalin M, Kaplan S, Gould F, Pinkham AE, Penn DL, Harvey PD. Social cognition, social competence, negative symptoms and social outcomes: inter-relationships in people with schizophrenia. J Psychiatr Res. 2015;68:254–260. doi:10.1016/j.jpsychires.2015.07.008
30. Wojtalik JA, Smith MJ, Keshavan MS, Eack SM. A systematic and meta-analytic review of neural correlates of functional outcome in schizophrenia. Schizophr Bull. 2017;43(6):1329–1347. doi:10.1093/schbul/sbx008
31. Guo JY, Huhtaniska S, Miettunen J, et al. Longitudinal regional brain volume loss in schizophrenia: relationship to antipsychotic medication and change in social function. Schizophr Res. 2015;168(1–2):297–304. doi:10.1016/j.schres.2015.06.016
32. Jelastopulu E, Giourou E, Merekoulias G, Mestousi A, Moratis E, Alexopoulos EC. Correlation between the Personal and Social Performance scale (PSP) and the Positive and Negative Syndrome Scale (PANSS) in a Greek sample of patients with schizophrenia. BMC Psychiatry. 2014;14:197. doi:10.1186/1471-244X-14-197
33. He Q, Chen Y, Chow SL, et al. [Negative symptoms predict the improvement of social functioning of patients with schizophrenia]. Sichuan Da Xue Xue Bao Yi Xue Ban. 2014;45(2):284–288. Chinese.
34. Karagianis J, Novick D, Pecenak J, et al. Worldwide-Schizophrenia Outpatient Health Outcomes (W-SOHO): baseline characteristics of pan-regional observational data from more than 17,000 patients. Int J Clin Pract. 2009;63(11):1578–1588. doi:10.1111/j.1742-1241.2009.02191.x
35. Sim K, Yong KH, Chan YH, et al. Adjunctive mood stabilizer treatment for hospitalized schizophrenia patients: asia psychotropic prescription study (2001–2008). Int J Neuropsychopharmacol. 2011;14(9):1157–1164. doi:10.1017/S1461145711000563
36. Puranen A, Koponen M, Tanskanen A, Tiihonen J, Taipale H. Use of antidepressants and mood stabilizers in persons with first-episode schizophrenia. Eur J Clin Pharmacol. 2020;76:711–718. doi:10.1007/s00228-020-02830-2
37. Leger M, Neill JC. A systematic review comparing sex differences in cognitive function in schizophrenia and in rodent models for schizophrenia, implications for improved therapeutic strategies. Neurosci Biobehav Rev. 2016;68:979–1000. doi:10.1016/j.neubiorev.2016.06.029
38. Mendrek A, Mancini-Marie A. Sex/gender differences in the brain and cognition in schizophrenia. Neurosci Biobehav Rev. 2016;67:57–78.
39. Emul M, Kalelioglu T. Etiology of cardiovascular disease in patients with schizophrenia: current perspectives. Neuropsychiatr Dis Treat. 2015;11:2493–2503. doi:10.2147/NDT.S50006
40. Yoram Barak DA. Clinical and psychosocial remission in schizophrenia: correlations with antipsychotic treatment. Barak Aizenberg BMC Psychiatry. 2012;12:108–112. doi:10.1186/1471-244X-12-108
41. Lieberman JA, Stroup TS. The NIMH-CATIE Schizophrenia Study: what Did We Learn? Am J Psychiatry. 2011;168(8):770–775. doi:10.1176/appi.ajp.2011.11010039
42. Zhang C, Chen MJ, Wu GJ, et al. Effectiveness of antipsychotic drugs for 24-month maintenance treatment in first-episode schizophrenia: evidence from a community-based “Real-World” Study. J Clin Psychiatry. 2016;77(11):e1460–e1466. doi:10.4088/JCP.15m10047
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.