Back to Journals » OncoTargets and Therapy » Volume 16
A Primary Lung and Breast Cancer Patient with Germline EGFR R776H Mutation: A Case Report and Literature Review
Authors Li D, Liu X, Cui S, Yang D, Zhu Y, Pan E, Yang P, Dai Z
Received 29 September 2022
Accepted for publication 30 December 2022
Published 18 January 2023 Volume 2023:16 Pages 17—22
DOI https://doi.org/10.2147/OTT.S391766
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Geoffrey Pietersz
Dan Li,1,* Xin Liu,1,* SaiQiong Cui,1 DaFu Yang,1 Yue Zhu,1 Evenki Pan,2 Peng Yang,2 ZhaoXia Dai1
1The Second Department of Thoracic Medical Oncology, The Second Hospital of Dalian Medical University, Dalian, People’s Republic of China; 2Nanjing Geneseeq Technology Inc., Nanjing, People’s Republic of China
*These authors contributed equally to this work
Correspondence: ZhaoXia Dai, The Second Department of Thoracic Medical Oncology, The Second Hospital of Dalian Medical University, No. 467, Zhongshan Road, Shahekou District, Dalian, People’s Republic of China, Email [email protected]
Abstract: For advanced non-small cell lung cancer (NSCLC) patients with common epidermal growth factor receptor (EGFR) mutations (exon 19 deletions or the exon 21 L858R mutation), tyrosine kinase inhibitors (TKIs) are the standard therapies. However, EGFR germline mutations are extremely rare in lung cancer, and the effective therapy is unclear. This study reports a patient with primary breast and lung cancer carried rare germline EGFR R776H and somatic L861Q mutation, who benefit from EGFR TKIs. Her family cancer history review demonstrated that her three out of four sisters with lung cancer were positive for EGFR R776H. Interestingly, only her healthy sister had type O blood, different from other sisters with type B blood. Our study provides a meaningful insight into the potential treatment option for patients with germline EGFR R776H and somatic L861Q mutation and highlights the importance of next-generation sequencing (NGS) in discovering rare genetic alterations to guide the prevention of genetic disease.
Keywords: EGFR R776H, germline mutation, non-small cell lung cancer, Gefitinib, Osimertinib
Introduction
Lung cancer is the most frequently diagnosed cancer and also the major cause of cancer-related deaths worldwide.1 In non-small cell lung cancer (NSCLC) patients, the most frequent epidermal growth factor receptor (EGFR) mutations are point mutation L858R within exon 21 and short in-frame deletions within exon 19, which are the most common oncogenic driver mutations and hypersensitive to EGFR tyrosine kinase inhibitors (TKIs).2 Other less prevalent but clinically relevant EGFR mutations that predict a resistant response to EGFR-TKIs include G719A/S/C, S768I, T790M and L861Q. Multiple studies have revealed the involvement of inherited susceptibility in the development of cancers. Moreover, family history of lung cancer still had a significantly increased risk in never-smoker probands.3 EGFR germline mutations are rare and associated with genetic susceptibility to lung cancer, including the mutations T790M and R776H in exon 20 and V843I in exon 21.4
With the development of genetic testing techniques, rare EGFR germline alterations are increasingly observed. Here, we reported a patient with primary breast and lung cancer carried rare germline EGFR R776H and somatic L861Q mutation, who benefit from EGFR-TKIs.
Case Presentation
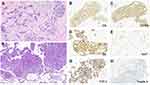
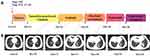
A 45-year-old Chinese female non-smoker was diagnosed with synchronous stage I (pT1cN0M0) primary breast cancer, which was surgically removed, and stage IVA (cT4N0M1a) primary lung adenocarcinoma (LUAD) in October 2015 (Figure 1A). Immunohistochemistry (IHC) staining of the resected breast tumor tissue was positive for estrogen receptor (ER, 50%), progesterone receptor (PR, 70%) and human epidermal growth factor 2 (HER2) and negative for Cytokeratin 5/6 (CK 5/6), P53, Topoisomerase I (TOPO I) and EGFR. Ki67 labeling in the tumor cells was 15% (Figure 2A–E). IHC analysis of a right lung tumor biopsy showed positive staining for LUAD markers, TTF-1 and Napsin A (Figure 2F–H), which confirming the primary LUAD. In November 2015, the patient received tamoxifen, a hormone therapy, to treat breast cancer. In December 2015, two cycles of chemotherapy with pemetrexed (0.8 g on d3) and cisplatin (50 mg on d1, 40 mg on d2,3) were administered for the treatment of LUAD (Figure 1B). Due to a positive result of sanger sequencing for exon 21 mutation (p.L861Q) and a rare EGFR exon 20 mutation (p.R776H), the patient was subsequently given Gefitinib (250 mg once daily), the first-generation EGFR TKI, in January 2016. The patient exhibited a stable disease (SD) (Figure 1B) and continued to be treated with Gefitinib until December 2017. At that time, the patient developed progressive disease (PD) with an enlarged primary lung tumor (Figure 1B). Therefore, the patient was switched to docetaxel (120 mg on d1) and carboplatin (0.55 g on d2) in January 2018. Two months later, the lung lesion increased again indicating PD (Figure 1B).
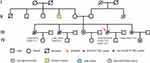
To identify a more efficient therapeutic strategy, freshly collected blood sample was subjected to targeted next-generation sequencing (NGS) of 139 cancer-related genes (Nanjing Geneseeq Technology Inc., Nanjing, China). Genomic profiling observed a rare EGFR R776H germline mutation (49.1%), EGFR L861Q (1.2%), NF1 E836X (2.6%) and TP53 P177T (2.3%). Investigation on her family history showed that her two older sisters died of lung cancer at the age of 53 years and 40 years, respectively. Her younger sister was diagnosed with lung cancer in October 2017 at the age of 44. Genetic testing revealed another three carriers of EGFR R776H aside from the proband, two elder sisters and her younger sister with lung cancer without her daughter (Figure 3). Additionally, her healthy younger sister, who was not an EGFR R776H carrier, had type O blood, which was different other sisters with type B blood.
The patient then received osimertinib (80 mg once daily) in March 2018. Six months post treatment, CT scans revealed no marked enlargement of the lesion indicating an SD (Figure 1B). Whereas, after 9 months of Osimertinib treatment, the patient refused to take it. In December 2018, the patient suffered from headache and CT scans revealed the lung lesions increased with pleural effusion, thus indicating PD (Figure 1B). The follow-up genomic testing with 139 cancer-relevant genes indicated EGFR L861Q (3.2%), NF1 E836X (6.7%) and TP53 P177T (5.3%). In January 2019, the patient received gemcitabine (1.4 g on d1,8) and nedaplatin (100 mg on d2). Unfortunately, the patient succumbed to a fourth-grade myelosuppression, then the chemotherapy was discontinued and she died in April 2019.
Discussion
Lung cancer is a major cause of death from cancer in the world. Genetic alterations are risk factors for lung cancer development, especially familial lung cancer. Germline EGFR mutations are rare but may contribute to oncogenesis,5 such as the mutations p.T790M and p.R776H in exon 20 and p.V843I in exon 21.4 Our study described a case of a female LUAD patient, who was a nonsmoker, with a germline EGFR R776H. The occurrence of germline EGFR R776H mutation is uncommon, and literature review of patients with an EGFR-R776H germline mutation identified four cases (Table 1). Including our family case (the proband and her three sisters with lung cancer), the mean age at diagnosis was 44 years (range 17–57), and two males (18%) and nine females (82%) were identified. Two out of seven patients had a personal history of another malignancy, including breast and thyroid cancers. In addition, six patients, including our proband, harbored additional somatic EGFR variations, such as G719A/S, L858R and L861Q, which is usually oncogenic. It was also observed in patients with germline EGFR T790M,6 indicating a proliferative advantage in the tumors.7 The role of germline EGFR R776H in tumor progression has not yet been elucidated. The EGFR R776H mutation is located in exon 20, which encodes the third most important kinase domain, and the mutation class encompasses in-frame insertions and indels following or within the regulatory C-helix amino acids that activates EGFR in the absence of the activating EGF ligand.8 It has been reported that these variants were selectively inhibited by second-generation TKIs, sometimes even in cases of acquired resistance to osimertinib.9 The R776H mutation activates EGFR in a dimerization-dependent manner by adopting the acceptor position in the asymmetric dimer and increased phosphorylation of monomeric EGFR in cotransfection assays using cells and activates EGFR by relieving autoinhibitory interactions with the αC-helix in computational approaches.10
 |
Table 1 Literature Review of Lung Cancer Patients with Germline EGFR R776H Mutations |
In our literature research of case reports who had germline EGFR R776H mutation (Table 1), we found one patient with a germline G776H mutation and a somatic G719S received surgery followed by erlotinib adjuvant treatment and achieved an RFS of more than 1 year.11 To the best of our knowledge, other studies also reported that cases with somatic R776H with other oncogenic EGFR mutations, such as L858R and L861Q, received EGFR TKIs and achieved a clinical benefit.12 In our case, the patient carried germline EGFR R776H and somatic L861Q mutations, who received gefitinib and achieved SD with a PFS of 23 months. Since the patient was resistant to first-generation TKI, osimertinib was administered and received an SD. Recent study also reported that osimertinib could be of benefit and may potentially be an effective treatment strategy to improve survival outcomes in patients with EGFR R776H.13
More interestingly, we also found the proband’s sisters with lung cancer all had type B blood, but only the healthy sister had type O blood. However, the mechanistic details of the relationship between blood type and germline mutations are not understood. Moreover, this study indicates a need for genetic testing for people with familial cancer history.
Conclusion
In summary, we reported the case of a rare EGFR R776H germline mutated patient with confirmed family history developing breast and advanced LUAD who received chemotherapy, followed by TKI-treatment and achieved clinical benefit. This report highlights the importance of comprehensive genomic profiling in discovering rare gene mutations and provides a valuable clue for treatment-decision-making in patients with EGFR germline R776H and somatic L861Q mutations. More effective therapeutic strategies for these patients need to be further studied in the future.
Data Sharing Statement
All datasets generated for this study are included in the manuscript.
Ethics Approval and Consent for Publication
This research was approved by the Ethics Committee of The Second Hospital of Dalian Medical University. Written informed consent to publish the clinical details and images were obtained from the patient.
Acknowledgments
Wu Jieping Medical Foundation (No. 320.6750.2021-16-28).
Author Contributions
All authors contributed to data analysis and drafting or revising of the manuscript. All authors agreed on the journal to which the article is submitted, provided final approval of the manuscript version to be published, and agreed to be accountable for all aspects of the study.
Disclosure
Evenki Pan and Peng Yang are employed by Nanjing Geneseeq Technology Inc. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–249. doi:10.3322/caac.21660
2. Midha A, Dearden S, McCormack R. EGFR mutation incidence in non-small-cell lung cancer of adenocarcinoma histology: a systematic review and global map by ethnicity (mutMapII). Am J Cancer Res. 2015;5(9):2892.
3. Lissowska J, Foretova L, Dąbek J, et al. Family history and lung cancer risk: international multicentre case–control study in Eastern and Central Europe and meta-analyses. Cancer Causes Control. 2010;21(7):1091–1104.
4. Li C, Wang Y, Su K, et al. Presentation of EGFR mutations in 162 family probands with multiple primary lung cancer. Transl Lung Cancer Res. 2021;10(4):1734–1746. doi:10.21037/tlcr-20-1001
5. Centeno I, Blay P, Santamaría I, et al. Germ-line mutations in epidermal growth factor receptor (EGFR) are rare but may contribute to oncogenesis: a novel germ-line mutation in EGFR detected in a patient with lung adenocarcinoma. BMC Cancer. 2011;11(1):1–8. doi:10.1186/1471-2407-11-172
6. Ma W, Gong J, Shan J, et al.Safety and efficacy of osimertinib in the treatment of a patient with metastatic lung cancer and concurrent somatic EGFR L858R and germline EGFR T790M mutations. JCO Precis Oncol. 2018;(2):1–7. doi:10.1200/PO.18.00076
7. Yokoyama T, Kondo M, Goto Y, et al. EGFR point mutation in non-small cell lung cancer is occasionally accompanied by a second mutation or amplification. Cancer Sci. 2006;97(8):753–759. doi:10.1111/j.1349-7006.2006.00233.x
8. Costa DB. Kinase inhibitor-responsive genotypes in EGFR mutated lung adenocarcinomas: moving past common point mutations or indels into uncommon kinase domain duplications and rearrangements. Transl Lung Cancer Res. 2016;5(3):331. doi:10.21037/tlcr.2016.06.04
9. Robichaux JP, Le X, Vijayan RSK, et al. Structure-based classification predicts drug response in EGFR-mutant NSCLC. Nature. 2021;597(7878):732–737. doi:10.1038/s41586-021-03898-1
10. Ruan Z, Kannan N. Mechanistic insights into R776H mediated activation of epidermal growth factor receptor kinase. Biochemistry. 2015;54(27):4216–4225. doi:10.1021/acs.biochem.5b00444
11. van Noesel J, van der Ven WH, van Os TA, et al. Activating germline R776H mutation in the epidermal growth factor receptor associated with lung cancer with squamous differentiation. J Clin Oncol. 2013;31(10):e161–e164. doi:10.1200/JCO.2012.42.1586
12. Guo T, Zhu L, Li W, et al. Two cases of non-small cell lung cancer patients with somatic or germline EGFR R776H mutation. Lung Cancer. 2021;161:94–97. doi:10.1016/j.lungcan.2021.05.036
13. An G, He L, Wang X, et al. Molecular Characteristics of EGFR Exon 20 Uncommon R776H Mutation and Response to Osimertinib in NSCLC Patients. Wolters Kluwer Health; 2021.
14. Su K, Gao S, Ying J, Zou S, He J. Sequencing a super multiple synchronous lung cancer reveals a novel variant in driver gene ARID1B. J Thorac Cardiovasc Surg. 2018;155(6):e185–e191. doi:10.1016/j.jtcvs.2018.01.010
15. Sovich JL, Dan T, Gemmell A, Clark C, Gagan J, Dowell JE. Lung adenocarcinoma associated with germline EGFR R776H variant: a case report and review of the literature. JCO Precis Oncol. 2022;6:e2100559. doi:10.1200/PO.21.00559
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.