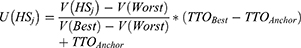Back to Journals » Patient Related Outcome Measures » Volume 12
A Discrete Choice Experiment to Derive Health Utilities for Aromatic L-Amino Acid Decarboxylase (AADC) Deficiency
Authors Smith AB ![]() , Hanbury A
, Hanbury A ![]() , Whitty JA
, Whitty JA ![]() , Buesch K
, Buesch K
Received 29 November 2020
Accepted for publication 13 March 2021
Published 12 May 2021 Volume 2021:12 Pages 97—106
DOI https://doi.org/10.2147/PROM.S294628
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Lynne Nemeth
Adam B Smith,1 Andria Hanbury,1 Jennifer A Whitty,2 Katharina Buesch3
1York Health Economics Consortium, University of York, York, UK; 2Norwich Medical School, University of East Anglia, Norwich, UK; 3PTC Therapeutics, Zug, 6300, Switzerland
Correspondence: Adam B Smith
York Health Economics Consortium, Enterprise House, Innovation Way, University of York, York, YO10 5NQ, UK
Tel +44 1904 323620
Fax +44 1904 323628
Email [email protected]
Purpose: Deriving health utilities for rare medical conditions such as aromatic L-amino acid decarboxylase (AADC) deficiency poses challenges. The rarity of AADC deficiency and the fact that this genetic condition often presents in very young children means that robust utility values cannot be derived from the child or their parent/caregiver. Alternative approaches, eg, discrete choice experiments (DCE), are required in order to provide health utilities. The aim of the study was to generate health utilities for AADC deficiency using a DCE.
Methods: The DCE was completed online by panel participants from a UK representative sample. The DCE comprised 6 AADC deficiency attributes (2– 6 levels): mobility, muscle weakness, oculogyric crises, feeding ability, cognitive impairment and screaming. These were identified from published literature, clinician input, parent interviews and expert opinion. Participants were presented with 10 choice sets specified using an orthogonal design, including a repeat task to evaluate choice consistency. Participants were presented with 5 health state vignettes prior to the DCE. These were used to elicit time trade-off (TTO) utilities. Multinomial logit models were estimated for the DCE data. The TTO utilities for the worst/best health states were used as anchors to convert indirect DCE part-worth utilities to health utilities.
Results: A total of 1596 participants completed the DCE. The majority (70.7%) gave consistent responses to the repeated choice task; only 1.7% (27) always chose the same alternative for every choice set. Five models were evaluated. There was one preference reversal (“sitting unaided”/“standing with assistance”) occurring in all models; these two mobility level coefficients were set to be equal in the final model. Rescaled utilities ranged from 0.494 to 0.7279, corresponding to the worst (633233) and best (111111) health states.
Conclusion: Health utilities were derived for AADC deficiency through a DCE. These will be used for a cost-effectiveness model of an AADC deficiency treatment.
Keywords: AADC deficiency, discrete choice experiment, health state utilities, health utilities
Introduction
Aromatic L-amino decarboxylase (AADC) deficiency is a rare, genetic condition typically presenting in infancy or early childhood with only around 150 reported cases worldwide.1 Common symptoms including hypotonia, developmental delay, movement disorders, and oculogyric crises.2 Health-related quality of life assessments (HRQoL) are critical to understanding the impact of this condition on the individual affected and in evaluating the cost-effectiveness of treatments aimed at AADC deficiency. In countries such as the United Kingdom (UK), regulatory bodies require HRQoL to be captured through preference-based instruments where responses to the patient-reported outcome measures are converted into health utilities based on societal preferences for a given health condition. For instance, in the UK, the National Institute for Health and Care Excellence (NICE) stated preferred instrument3 is the EuroQol 5D-3L (EQ-5D-3L).4
In rare diseases, particularly paediatric populations, the process of patients completing these instruments is, by definition, more problematic given the nature of the condition and patients affected. Therefore, deriving robust health state utilities, from either the child or their parent/caregiver becomes difficult.5 Other approaches have been used to generate health utilities for these populations, such as proxy-ratings from clinicians.6,7 However, these methods are not without their own shortcomings, eg, the number of clinicians involved in these studies is usually small, as well as the intrinsic issues in proxy-ratings (for both clinicians and parents and caregivers) in the form of unwitting biases.8 Time trade-off (TTO) and standard gamble tasks (SG)9 have been used to derive health state utilities for rare conditions.10,11 However, as these approaches are often used to provide input to health states in economic models, they tend to provide global utilities for the health states, rather than utilities or disutilities for individual levels of the key symptoms. Discrete choice experiments (DCE) offer a potential solution to this. In a DCE participants are sequentially presented with choice sets comprising the symptoms (referred to as attributes) and levels (degrees of severity) for the condition in question; typically, each choice consists of two (or more) health state profiles with differing combinations of levels and the participant is asked to select one of these profiles.12 This process allows preferences to be derived for the given health states. DCEs have been used in rare diseases previously.13,14 However, in most instances (with the exception such as time trade-off TTOs), the parameters derived from DCEs are not health utilities, as they are not aligned with the conventional utility scale that is used to derive quality-adjusted life years (QALYs). In order to derive health utilities from DCE parameters, these need to be anchored to utilities from preference-based methods which are aligned to the conventional utility scale used to derive QALYs such as TTO/SG utilities.15
The aim of this study, therefore, was to derive health state utilities for the key attributes of AADC deficiency using a DCE to complement a vignette study.
Participants and Methods
Overview
The discrete choice experiment was part of a larger study that has been described elsewhere.11 In brief, participants initially completed a time trade-off and standard gamble task designed to elicit health state utilities for AADC deficiency. The vignettes used to describe these health states ranged in severity from “bedridden” (worst) to “walking with assistance” (best). Following this, participants completed the DCE study described here. The utilities for the worst and best health states derived from the vignette study were used as anchors in the DCE to rescale the DCE parameters to health utilities. An example of the worst health state vignette is shown in Figure 1.
 |
Figure 1 Example of health state vignette. |
Participants
Participants were recruited from a panel maintained by a third party (Qualtrics, Provo, USA). There is no consensus on how to estimate the sample size for a DCE.16 Therefore, Orme’s rule of thumb was applied. This suggested a minimum sample size of 167 respondents would be expected to give precise preference estimates, assuming each respondent completes 9 choice sets, each set contains two alternative health states, and a maximum of 6 levels are used to describe each symptom.17 In order to ensure that the sample was as representative of the UK population as possible an initial sample size of 1000 respondents was selected. Basic socio-demographic details (age, biological sex (male/female/prefer not to say), parental status, and country of residence) were collected from respondents and used to screen for eligibility. Eligible respondents included UK residents, aged ≥18 years. Parents and caregivers of children with life-threatening or life-limiting conditions were screened out of the study in order to reduce any potential bias. The study was conducted online. Participants received a nominal incentive (redeemable points) if they completed the study in full. The study was submitted for review to the University of York’s Health Sciences Research Governance Committee and received ethics approval on 20 March 2020, and was conducted in accordance with the Declaration of Helsinki.
DCE Health States, Attributes and Levels
The development of the vignettes has been described in detail elsewhere.18 In brief, the vignettes were developed using a number of approaches including: a pragmatic literature review; a review of case stories provided online from AADC deficiency support groups; an advisory board with parents and caregivers of children with AADC deficiency; and an advisory board with physicians treating patients with AADC deficiency. The five health state vignettes developed reflect motor and developmental milestones and are based on an ongoing clinical trial investigating gene therapy for AADC deficiency (NCT02926066) and the associated economic model being developed to evaluate this therapy. The DCE attributes or key AADC deficiency symptoms were identified from the vignette development. Initially, 13 symptoms were identified. These were reviewed at an advisory board comprising physicians (N=5) treating patients with AADC deficiency. These physicians were requested to rank the symptoms in order of relevance, impact on patients and impact on parents and caregivers. This process led to 6 key attributes being identified: mobility; muscle weakness or floppiness; oculogyric crises; feeding support, cognitive impairment; and screaming. The levels for the attributes were chosen on a pragmatic basis. Six levels were selected for the “mobility” attribute reflecting the 5 health states in the economic model, as well as an additional level “walking unassisted”. Three levels were selected for the other attributes (except “feeding support”) to reflect mild, moderate and severe symptoms; two levels were chosen for “feeding support” (unable/able to feed themselves).
Experimental Design
The levels of the symptoms were varied systematically across the alternative health states according to a main effects orthogonal design estimated in NGene statistical design software.19 This resulted in a total of 108 choice sets, which were divided into 12 blocks, each containing 9 choice sets. Each respondent was randomly allocated to complete one of 12 survey versions containing one of the 12 DCE choice set blocks. The second choice set in each block was reversed and repeated as a 10th choice task to enable an assessment of internal consistency. Thus, each respondent completed a total of 10 choice sets each similar to that shown in Figure 2. Each choice set asked respondents which of two alternative health states they would prefer their child to have. Each health state was defined according to a combination of 6 symptoms, each with between 2 and 6 levels (Table 1).
 |
Table 1 DCE Symptoms and Levels (and Dummy Code for Analysis) |
 |
Figure 2 Example choice set. |
Procedure
Participants were firstly provided a description of the study and the sponsor’s details. This description also contained a warning regarding the descriptions of AADC deficiency in the vignettes, as this is by nature a severe medical condition. Participants were also informed that they were free to withdraw from the study at any stage. Once the participant had provided their consent a screening question was presented to ensure that any parent or caregiver of a child with a potentially life-threatening or life-limiting condition was screened out of the study.
The participants were then asked to provide their home nation in the United Kingdom (England, Scotland, Wales or Northern Ireland); their age; biological gender (male, female, prefer not to say), highest level of education (lower secondary up to age 16, higher secondary up to age 18, undergraduate, postgraduate).
Subsequently, participants were taken through an explanation and examples of the time trade-off (TTO) and standard gamble (SG) tasks and were shown the five health states for these tasks in a random order. For both the TTO/SG tasks and the DCE participants were asked to imagine themselves as the parent or caregiver of the (hypothetical) child described. The TTO and SG tasks have been described in detail elsewhere.11 Having completed these tasks, the participants were then shown an explanation of the DCE (Figure 3) before completing the choice sets.
 |
Figure 3 Description of DCE. |
Analysis
Analysis of DCE Data
NLogit statistical software (version 6) was used for analysis of the DCE data.20 The relative importance of the symptom levels in driving health state choice was estimated through multinomial logit (MNL) regression analysis of the choice data. The utility function for the choice model was specified as a linear additive function of the main effects for each symptom level (Equation 1).
In Equation 1, V(j) is the systematic (observed) utility associated with health state j; ASC(A) is an alternative specific constant associated with choosing alternative A over alternative B all else equal (reflecting left-sided bias); β1–14 are the beta coefficients (also referred to as preference weights, marginal utilities or part worths) associated with each symptom level; and mobility, muscle weakness, oculogyric crisis, feeding support, cognitive impairment and screaming are the symptom levels, which were dummy coded compared to the referent (best) level as defined in Table 1.
Only the 9 choice sets included in the statistical design (and not the repeated 10th choice set included as consistency check) were included in the regression analyses. We estimated MNL models on the complete sample, and then on subsamples excluding in turn respondents who (i) always chose the same alternative (A or B) in every choice set; (ii) always chose the same alternative or gave an inconsistent answer to the tenth repeated choice set; (iii) always chose the same alternative or completed in less than the 25th percentile completion time; and (iv) always chose the same alternative, gave an inconsistent answer to the tenth repeated choice set, or completed in less than the 25th percentile completion time. The preference patterns were very similar in all models (data not presented but available from the authors on request), except that the constant associated with choosing alternative A (all else equal) was positive and significant in the model utilising the complete sample, but became insignificant in all subsample models. Therefore, to retain the integrity of the representative nature of the sample, we utilised the complete sample excluding respondents who always chose the same alternative but retaining inconsistent or fast responders, for the analyses.
Rescaling of Health States to QALY Scale
The observed utility (Vj) estimates for each health state from the DCE indicate the respondents’ preferences for being in one health state over another. However, to be able to assign a utility weight to these health states in a cost-utility analysis, it is necessary to rescale the observed utility for each health state onto the utility scale used to derive quality-adjusted life years (QALYs), in which a utility weight of 0 represents a state equivalent to being dead, and a utility weight of 1 represents full health. We followed the methods described by Gu and colleagues to do this.15 The estimated utility was rescaled to the 0 to 1 QALY scale using the time trade-off (TTO) utility values directly elicited for vignettes associated with the best (111111) and worst (633233) health states described by the DCE.
In Equation 2, U(HSj) is the utility of health state j rescaled to the QALY scale, V(HSj) is the observed utility for health state j estimated from the DCE (per Equation 1); V(Best) and V(Worst) are the observed utility estimated from the DCE for the Best health state (which has an observed utility of 0 as its defined by all referent levels) and the Worst health state, respectively; and TTOBest and TTOAnchor are the directly elicited TTO utility weights for the best and worst health states, respectively.
Results
Sample Characteristics
There were 1598 respondents who completed the survey; however, two respondents had not completed the DCE choice sets. Therefore, 1596 respondents were included for the DCE analysis. Their sociodemographic characteristics are presented in Table 2. Just over half (58.3%) of respondents were female, the average age of respondents was 44 years, and just over a quarter (28.3%) reported having children aged 16 years or younger.
 |
Table 2 DCE Participant Characteristics (n=1596) |
Choice Data
The 12 survey versions were completed evenly across respondents (133, 8.3% respondents per version). The median survey completion time was 9.5 minutes (interquartile range 6.8 to 13.7 minutes). A minority (27, 1.7%) of respondents always chose the same alternative (A or B) for every choice set. Most (70.7%) gave a consistent response to the repeated choice task.
DCE Analysis
We excluded the 27 respondents who always chose the same alternative for every choice set. Therefore, the DCE analyses were based on 9 choices from each of 1569 respondents, giving 14,121 choice observations in total.
The MNL model results are presented in Table 3. For the unrestricted model, all symptom levels significantly impacted choice and in the direction expected with the exception of Mobility. For Mobility, the first level of detriment (MOB2) was not significant in affecting choice compared to the best level (MOB1, referent), and there was one preference reversal (MOB3 and MOB4). Therefore, we estimated a “restricted” model where the coefficient for MOB3 and MOB4 was fixed to be the same. This resolved the preference reversal and did not impact model fit; the pseudo R2 was 0.24 for both the unrestricted and restricted models compare to a constants-only model.
 |
Table 3 DCE Data MNL Model Output |
Rescaling on QALY Scale
We used the restricted model to estimate utility weights for health states associated with AADC deficiency. From the vignette study, the estimated TTO utility weights for the best and worst health states were 0.7279 and 0.494, respectively.11 Some example health states and their corresponding utility values for the DCE health states after rescaling are shown in Table 4. For instance, that the largest relative disutility (ie, with the other attributes at the lowest level) is associated with the attribute “Screaming” moving from “No screaming” to “Constant screaming”: −0.0651. This value was greater even than the disutility associated with “Mobility” moving from “No problems walking” to “Bedridden”, although the disutility moving from the best to the worst health state for this attribute was still −0.0561. Severe cognitive impairment was also associated with a high disutility value (−0.0431), relative to the best health state. The disutilities of moving from the best and worst symptom levels for the other attributes were as follows: “Muscle weakness” −0.0261; “Feeding” −0.0240; and “OGC” – 0.0194. Disutilities between adjacent health states ranged from −0.0048 between “No OGC” and “Occasional OGC” levels to −0.0303 between “Frequently screaming” and “Constantly screaming”.
 |
Table 4 DCE Health States and Utility Examples |
Discussion
The aim of this study was to generate utility values for AADC deficiency attributes using a discrete choice experiment. The results demonstrated that the DCE parameters decreased in line with expectations, that is became more negative as attribute severity levels increased. The disutilities followed a similar pattern. The main drivers for the disutilities appeared to be predominantly screaming, then followed by mobility and cognitive impairment. In fact, the greatest disutility was associated with screaming, more so even than the bedridden state. This suggests that participants may have been rating parental/caregiver health-related quality of life (HRQoL) as part of the evaluation, instead of that of the child alone. The corresponding vignette study revealed that the general population rated the HRQoL of parents/caregivers of children with AADC deficiency as very low (around 30 on a scale of 0–100).11 This may also reflect the potential difficulties a general population sample has in imagining themselves as parents of a child with a very rare condition, as well as with a general lack of familiarity or possible lack of understanding of symptoms such as oculogyric crises (OGC), compared to the relative familiarity of physical limitations and sign such as screaming.
This DCE complements a vignette study.11 The latter focused on health states associated with AADC deficiency. These vignettes included the primary symptoms described in the DCE, ie, mobility, oculogyric crises, feeding problems and screaming. Although all DCEs potentially suffer from a degree of artificialness given the inherent nature of the task, these symptoms were selected on the basis of extensive content validation by parents/caregivers and physicians.18 Nevertheless, for pragmatic considerations alone some levels of symptoms had to be compromised in order to ensure both that all attributes were presented in equal number and that participants were not overburdened by the number of choice sets.
The level of severity across the health states in the vignettes was reduced for all symptoms as the states moved from worst (“bedridden”) to best (“walking with assistance”). This was in line with the literature,1 which suggests that where improvement in symptoms AADC deficiency is observed under gene therapy, this is manifested at a global level, ie, all symptoms tend to improve (albeit not at the same rate). Although this vignette study provided health state utilities, it meant that the individual symptom drivers could not be isolated. The DCE, therefore, adds to this enabling the impact of key individual symptoms to be identified. These in turn could be utilised as markers to evaluate the treatment effects in clinical trials.
This study derived utility weights for a wide range of health states associated with AADC deficiency and is the first study the authors are aware of to attempt to derive health state utilities in this rare condition. The anchors used in the current study were derived from a TTO vignette task that did not allow any health states to be considered worse than dead (WTD).11 A number of approaches are available to derive anchors to recalibrate DCE parameters, eg, some studies7 in rare paediatric conditions, for instance using clinician input, have allowed participants to rate health states as being WTD resulting in lower utility values for the most severe health states. The mean health state utility used as the anchor in the current study, 0.494, suggests that participants had rated the worst health state as severe, but not sufficiently severe to classify as WTD, which presumably would have resulted in a mean utility closer to 0 (“death”). A point further emphasised by the fact that only 23% of the sample11 rated this health state (“bedridden”) at 0. Nevertheless, the use of an anchor from a vignette without the option to rate the health state as being WTD may have resulted in the DCE estimates lying on a much narrower range (ie, smaller disutilities for health states) than would be the case had worse than dead been allowed. This may also have been reflected in the upper anchor used to convert the DCE parameters into utility values. In this study, the upper anchor was based on the “best” health study from the vignette study, ie, walking with assistance. However, the “best” level for the DCE for mobility was “walking unassisted”. It may therefore be argued that the best health state could be perfect health (111111). Re-anchoring the parameters in line with perfect health still results in “screaming”, “mobility” and “cognitive impairment” being the main drivers for utilities, although the magnitude of the disutilities increases: −0.141, −0.1214 and −0.0933 for screaming, mobility and cognitive impairment, respectively (relative to the worst level). A further point for consideration is that this study did not focus on latent classes or differences between socioeconomic groups. It may, for instance, be assumed that parental status could have a potential impact on choice. That said, although the number of participants who were parents of a child <16 was low, the accompanying TTO vignette study11 found no statistically significant effect on health state utilities by parental status.
Conclusion
To the authors' knowledge, this is the first study to derive health utilities for AADC deficiency symptoms using a large sample drawn from the general population. These data will be utilised to enable improvements in health-related quality of life to be considered in an economic evaluation of a gene therapy for AADC deficiency.
Disclosure
This work was supported by PTC Therapeutics (“PTC”). KB is an employee of PTC. ABS and AH are employees of York Health Economics Consortium (YHEC). YHEC was commissioned by PTC to undertake the study. JAW received a consulting fee from YHEC for her contribution to the study.
An abstract of this paper was presented at the Virtual Conference of the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) – Europe 2020 (November 16–19) as a poster presentation. The poster’s abstract was published in Value in Health: https://www.valueinhealthjournal.com/article/S1098-3015(20)34127-9/fulltext.
References
1. Kojima K, Nakajima T, Taga N, et al. Gene therapy improves motor and mental function of aromatic l-amino acid decarboxylase deficiency. Brain. 2019;142(2):322–333. doi:10.1093/brain/awy331
2. Wassenberg T, Molero-Luis M, Jeltsch K, et al. Consensus guideline for the diagnosis and treatment of aromatic l-amino acid decarboxylase (AADC) deficiency. Orphanet J Rare Dis. 2017;12(1):12. doi:10.1186/s13023-016-0522-z
3. National Institute for Health and Care Excellence (NICE). Guide to the Methods of Technology Appraisal. London: National Institute for Clinical Excellence; 2004.
4. Dolan P. Modeling valuations for EurQol health states. Med Care. 1997;35(11):1095–1108. doi:10.1097/00005650-199711000-00002
5. Landfeldt E, Edström J, Sejersen T, Tulinius M, Lochmüller H, Kirschner J. Quality of life of patients with spinal muscular atrophy: a systematic review. Eur J Paediatr Neurol. 2019;23(3):347–356. doi:10.1016/j.ejpn.2019.03.004
6. Landfeldt E, Lindgren P, Bell CF, et al. Health-related quality of life in patients with Duchenne muscular dystrophy: a multinational, cross-sectional study. Dev Med Child Neurol. 2016;58(5):508–515. doi:10.1111/dmcn.12938
7. Lloyd AJ, Thompson R, Gallop K, Teynor M. Estimation of the quality of life benefits associated with treatment for spinal muscular atrophy. Clinicoecon Outcomes Res. 2019;11:615–622. doi:10.2147/CEOR.S214084
8. Pickard AS, Knight SJ. Proxy evaluation of health-related quality of life: a conceptual framework for understanding multiple proxy perspectives. Med Care. 2005;43(5):493–499. doi:10.1097/01.mlr.0000160419.27642.a8
9. Torrance GW. Measurement of health state utilities for economic appraisal. J Health Econ. 1986;5(1):1–30. doi:10.1016/0167-6296(86)90020-2
10. Smith AB, Retzler J, Taylor MJ. Standard gamble to derive utility health states for limbal stem cell deficiency. Clinicoecon Outcomes Res. 2020;12:535–546. doi:10.2147/CEOR.S251918
11. Smith AB, Hanbury A, Buesch K. Eliciting health state utilities for Aromatic L-amino Acid Decarboxylase (AADC) deficiency: a UK vignette study. [under review]. Patient Relat Outcome Meas.
12. Ryan M, Farrar S. Eliciting preference for healthcare using conjoint analysis. BMJ. 2000;320(7248):1530–1533. doi:10.1136/bmj.320.7248.1530
13. Toumi M, Millier A, Cristeau O, Thokagevistk-Desroziers K, Dorey J, Aballéa S. Social preferences for orphan drugs: a discrete choice experiment among the French general population. Front Med. 2020;7:323. doi:10.3389/fmed.2020.00323
14. López-Bastida J, Ramos-Goñi JM, Aranda-Reneo I, Taruscio D, Magrelli A, Kanavos P. Using a stated preference discrete choice experiment to assess societal value from the perspective of patients with rare diseases in Italy. Orphanet J Rare Dis. 2019;14(1):154. doi:10.1186/s13023-019-1126-1
15. Gu NY, Botteman MF, Gerber RA, et al. Eliciting health state utilities for Dupuytren’s contracture using a discrete choice experiment. Acta Orthop. 2013;84(6):571–578. doi:10.3109/17453674.2013.865097
16. Marshall D, Bridges JFP, Hauber B, et al. Conjoint analysis applications in health - how are studies being designed and reported? Patient. 2010;3(4):249–256. doi:10.2165/11539650-000000000-00000
17. Orme BK. Getting Started with Conjoint Analysis: Strategies for Product Design and Pricing Research. Madison Wisconsin: Research Publishers LLC; 2006.
18. Hanbury A, Smith AB, Buesch K. Deriving vignettes for the rare disease AADC using parent, caregiver and clinician interviews to evaluate the impact on health-related quality of life. Patient Relat Outcome Meas. 2021;12:1–12. doi:10.2147/PROM.S278258
19. Rose JM, Collins AT, Bliemer MCJ, Hensher DA. Ngene [Computer Program]. Version 1.1.1 ed. ChoiceMetrics Pty Ltd; 2012.
20. Econometric Software Inc. NLOGIT [Computer Program]. Version 5 ed. Plainview, NY: Econometric Software, Inc.;2012.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.