Back to Journals » International Journal of Nanomedicine » Volume 9 » Supplement 2
Zinc oxide nanoparticles: a 90-day repeated-dose dermal toxicity study in rats
Authors Ryu HJ, Seo MY, Jung SK, Meang E, Lee S, Jang D, Lee T, Jo K, Kim Y, Cho K, Kim M, Lee BJ, Son SW
Received 20 November 2013
Accepted for publication 15 February 2014
Published 15 December 2014 Volume 2014:9(Supplement 2) Pages 137—144
DOI https://doi.org/10.2147/IJN.S57930
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Hwa Jung Ryu,1,* Mu Yeb Seo,2,* Sung Kyu Jung,1 Eun Ho Maeng,2 Seung-Young Lee,2 Dong-Hyouk Jang,2 Taek-Jin Lee,2 Ki-Yeon Jo,2 Yu-Ri Kim,3 Kyu-Bong Cho,4 Meyoung-Kon Kim,3 Beom Jun Lee,5 Sang Wook Son1
1Department of Dermatology, Korea University College of Medicine, Seoul, 2Korea Testing and Research Institute, Gyunggido, 3Department of Biochemistry and Molecular Biology, Korea University College of Medicine, Seoul, 4Department of Clinical Laboratory Science, Shinheung College, Uijeongbu, 5College of Veterinary Medicine, Chungbuk National University, Cheongju, Republic of Korea
*These authors contributed equally to this work and both should be considered first authors
Abstract: Zinc oxide (ZnO) works as a long-lasting, broad-spectrum physical sunblock, and can prevent skin cancer, sunburn, and photoaging. Nanosized ZnO particles are used often in sunscreens due to consumer preference over larger sizes, which appear opaque when dermally applied. Although the US Food and Drug Administration approved the use of nanoparticles (NPs) in sunscreens in 1999, there are ongoing safety concerns. The aim of this study was to evaluate the subchronic toxicity of ZnO NPs after dermal application according to the Organization for Economic Cooperation and Development Test Guidelines 411 using Good Laboratory Practice. Sprague Dawley rats were randomly divided into eight (one control, one vehicle control, three experimental, and three recovery) groups. Different concentrations of ZnO NPs were dermally applied to the rats in the experimental groups for 90 days. Clinical observations as well as weight and food consumption were measured and recorded daily. Hematology and biochemistry parameters were determined. Gross pathologic and histopathologic examinations were performed on selected tissues from all animals. Analyses of tissue were undertaken to determine target organ tissue distribution. There was no increased mortality in the experimental group. Although there was dose-dependent irritation at the site of application, there were no abnormal findings related to ZnO NPs in other organs. Increased concentrations of ZnO in the liver, small intestine, large intestine, and feces were thought to result from oral ingestion of ZnO NPs via licking. Penetration of ZnO NPs through the skin seemed to be limited via the dermal route. This study demonstrates that there was no observed adverse effect of ZnO NPs up to 1,000 mg/kg body weight when they are applied dermally.
Keywords: zinc oxide, nanoparticles, subchronic toxicity, dermal exposure
Introduction
Many sunscreens contain zinc oxide (ZnO) and titanium dioxide, since they not only physically reflect but also absorb ultraviolet light. When they are present as larger particles, they appear opaque and have a tendency to leave a film, which consumers dislike. Using ZnO nanoparticles (NPs), the opacity of sunscreen is decreased, resulting in increased compliance with use of sunblock. However, a significant concern with the use of nanotechnology-based products is that nanoscale materials may penetrate protective physical barriers, such as the skin, lungs, intestinal tract, and blood–brain barrier, and once internalized, may be harmful. The skin is the outermost barrier of the body and a route of entry for many foreign materials, including NPs. Recent studies have demonstrated that small amounts of zinc from ZnO particles in sunscreens applied outdoors are absorbed through human skin.1 In spite of these concerns, few studies have investigated the effects of ZnO NPs on the internal organs when they were applied to the skin repeatedly.
It is recognized that NPs have physicochemical characteristics that are very different from those of larger particles given that their surface area is proportionally greater, and so may be more toxic. There are worries that these NPs may escape immune surveillance and natural defense mechanisms because of their very small size. NPs could also form free radicals when internalized. ZnO NPs have been reported to elicit various adverse cellular effects, including genotoxicity2 and cytotoxicity.3 It has also been suggested that ultraviolet B irradiation causes increased dissociation of Zn2+ from ZnO and that the consequent accumulation of free or labile Zn2+ from sunscreen results in cytotoxicity and oxidative stress.3 However, the mechanisms underlying these adverse effects are not fully characterized. Decreased collagen content in the skin due to oxidative stress at the site of application of ZnO has been reported.4 It is important to know the toxic effects of ZnO when used chronically, in light of a report by Kertesz et al5 showing that penetration of nanomaterials into the skin after dermal exposure is time-dependent. We are likely to come into contact with ZnO NPs in everyday life, as they are contained in many cosmetic products. The 90-day repeated-dose toxicity test is often used to evaluate the subchronic toxicity of certain materials. In the present study, the subchronic toxicity of ZnO NPs (20 nm, negatively charged ZnOSM20(−)) was investigated in Sprague Dawley rats. The potential toxicity and safety of ZnO NPs was studied during 90 days of repeated dermal application with a 14-day recovery period according to Organization for Economic Co-operation and Development recommendations with Good Laboratory Practice. The objective was to identify potential target organ(s) for toxicity induced by ZnO NPs in rats to identify the no observed adverse effect level (NOAEL) value.
Materials and methods
Chemicals and animals
ZnO NPs (20 nm, lot number 141319) were purchased from Sumitomo Osaka Cement Co Ltd (Osaka, Japan). The surface charge was modified with coating reagents, citrate for (−) charge (ZnOSM20(−)), as reported previously.6 On scanning electron microscopy, ZnOSM20(−) were determined to be an average of 29±3 nm and to be spherical in shape. The zeta potential of ZnOSM20(−) was −44.4±1.0 mV, with a negative surface charge. One hundred and thirty Sprague Dawley rats aged 6 weeks and weighing 150−210 g were obtained from an inhouse animal facility and housed in an animal room maintained at 21.8°C±1°C and 50.8%±10% relative humidity with an alternating 12:12-hour light-dark cycle. The rats were kept in stainless steel cages, and rodent food (Cargill Agri Purina Inc., Kyunggido, Republic of Korea) and reverse osmosis water were provided ad libitum. All animal procedures were reviewed and approved by the animal ethics committee. The rats were randomly divided into five groups. Different doses of ZnOSM20(−) were dermally administered to rats in the experimental groups for 90 days. The groups and doses applied are listed in Table 1. Group 1 (normal control) animals were treated with distilled water for 90 days. Group 2 was treated dermally with HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid)-citrate buffer as a vehicle control group. Groups 3, 4, and 5 were treated dermally with ZnOSM20(−) at doses of 250, 500, and 1,000 mg/kg, respectively. In an unpublished study of 14-day repeated-dose toxicity in rats, significant toxicity was observed at doses of 2,000 mg/kg, so a maximum dose of 1,000 mg/kg was used in this study, with 500 mg/kg as an intermediate dose and 250 mg/kg as a low dose (Table 1). There were also animals in recovery groups (Table 1). They were monitored for additional 14 days after the 90 day toxicity testing was done. They were rats from control, vehicle control, and ZnO 1000 mg/kg groups.
Subchronic toxicity
A 90-day repeated-dose dermal toxicity test was conducted as per Organization for Economic Co-operation and Development Test Guideline 411 with modifications for dosage, biochemical parameters, and histopathologic evaluation. The prepared ZnO NPs were applied to the skin to evaluate the toxicity of ZnO on repeated dermal exposure. A 5 cm ×6 cm area on the back of each rat was clipped free of hair once a week. ZnO NPs were applied to approximately 10% of the total body surface area (about 4 cm ×5 cm). Sterile gauze was soaked in the ZnOSM20(−) solution and attached to the hairless portion of the back and fixed with nonirritant tape, ie, Tegaderm™ (3M, St Paul, MN, USA) and Coban™ (3M), for 6 hours to prevent disruption of the ZnO patch. After 6 hours of application, the site was cleaned with sterile water. This procedure was repeated every day for 90 days.
Clinical observations
Body weight and food and water intake were measured weekly. All 130 rats were observed daily for signs of abnormality and toxicity during the 90 days of treatment, and during the 14-day recovery period for the 30 rats in the recovery group.
Ophthalmologic examination and urinalysis
Ophthalmic appearance was observed with the naked eye before treatment and during the last week of the experiment. An ophthalmoscope (Genesis, Kowa Ltd, Nagoya, Japan) was used to observe the fundus oculi in the control group and in the G5 group after using a mydriatic agent (OcuTropine®; Samil Pharmaceutical Co Ltd, Seoul, Republic of Korea) at the end of the experiment. Urine tests were performed during the last week of the study using Multistix® 10SG strips (Siemens AG, Erlangen, Germany) and a urine analyzer (Clinitek 500, Bayer Inc., Pittsburgh, PA, USA) to determine specific gravity, pH, leukocyte count, nitrite, protein, glucose, ketones, urobilinogen, bilirubin, and blood in urine. Urinary sediments were analyzed from a fresh 3-hour collection of urine, and the amount of urine was measured from the 24-hour collected urine from rats of the control and G5 group.
Hematology and biochemistry
Blood samples were taken from an abdominal artery after each animal was deeply anesthetized with isoflurane. The animals were fasted for 18 hours before blood sampling. Hematologic analyses were performed using an automatic hematologic analyzer (Advia® 120E; Siemens AG). Parameters measured in the blood samples were: white blood cell count, differential counts (neutrophils, lymphocytes, monocytes, eosinophils, basophils), red blood cell count, hemoglobin, hematocrit, mean cell volume, mean corpuscular hemoglobin, mean cell hemoglobin, mean cell hemoglobin concentration, and platelet count. The reticulocyte ratio was assessed for all animals using staining and microscopy techniques. Prothrombin time and activated partial thromboplastin time were performed using blood samples collected into a 9NC BD Vacutainer® (BD Biosciences, San Jose, CA, USA) containing 3.2% sodium citrate as an anticoagulant with centrifugation at 3,000 rpm for 10 minutes.
Biochemical analyses of serum samples were performed using an automatic chemistry analyzer (Hitachi 7060; Hitachi Ltd, Tokyo, Japan, and EasyLyte; Werfen Medical, Newtonville, IL, USA). Biochemical parameters measured were total protein, albumin, albumin/globulin ratio, total bilirubin, alkaline phosphatase, alanine aminotransferase, aspartate aminotransferase, creatinine, blood urea nitrogen, total cholesterol, triglycerides, blood glucose, calcium, inorganic phosphorus, creatine kinase, sodium, potassium, and chlorine.
Necropsy findings, organ weight measurement, and pathologic examination
After collection of blood samples, the animals were sacrificed under deep anesthesia with isoflurane. After exsanguination, the external surface, all orifices, the cranial cavity, and the thoracic and abdominal cavities and their contents were visually observed for any signs of gross abnormality. Absolute and relative organ weights (organ weight/body weight) were measured for each rat. For paired organs, the sum of those organs was used.
After collection, the organs were preserved in 10% phosphate-buffered formalin solution, with the testes preserved in Davidson’s solution in preparation for histopathologic examination. Organs collected included the skin, lymph nodes, salivary glands (submandibular), bone, bone marrow (sternum, femur), thymus, trachea, bronchus, lungs, heart, thyroid gland, parathyroid glands, tongue, esophagus, stomach, small intestine, large intestine, liver, spleen, pancreas, kidneys, adrenal glands, bladder, seminal vesicles, prostate gland, testes, epididymis, ovaries, uterus, vagina, brain, pituitary gland, spinal cord, eyes, sciatic nerve, and skeletal muscle.
Tissue distribution
A 1 mL blood sample was drawn from the caudal vein, and feces were collected before necropsy. Next, the brain, liver, kidneys, testes (or ovaries), spleen, lung, stomach, small intestine, large intestine, and skin were collected and measured after removing the fat and washing with normal saline. The collected organs were preprocessed before weighing and temporarily stored in an ice box with dry ice. They were then analyzed using inductively coupled plasma atomic emission spectroscopy (Ultima 2; Horiba Jobin Yvon Inc., Tokyo, Japan) and an ME254 analytical balance (Sartorius AG, Göttingen, Germany).
Statistical analysis
The data obtained for body weight, food and water consumption, hematology and biochemistry, and organ weight were evaluated by one-way analysis of variance after Levene’s test to examine the homogeneity of their variance. If the data were determined to be homogeneous, the data were analyzed using Scheffe’s multiple comparison test, and if not homogeneous, by Dunnett’s T3 multiple comparison test. The data were analyzed using Statistical Package for the Social Sciences software version 12.0 (SPSS Inc, Chicago, IL, USA), and differences were considered to be statistically significant at P-values less than 0.05.
Results
Clinical observations
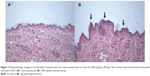
The main sign associated with application of ZnO NPs was formation of skin crusts. At first, this appeared in the G5 (1,000 mg/kg) group 2 days after initiation of ZnOSM20(−), and all groups showed crust formation in a dose-dependent manner. Skin crusts also appeared in the vehicle control group, but resolved after 7 days. Skin crusts from male rats in the 1,000 mg/kg group were biopsied, and found to contain areas of hyperkeratosis and papillomatosis when compared with the normal skin of controls (Figure 1). Scar tissue developed at the application site in one female rat in the G5 group. No difference in body weight was observed between the experimental and control groups. Food intake in male rats from the 500 mg/kg and 1,000 mg/kg groups was significantly decreased in comparison with that in the control group at week 2. Food intake was decreased in female rats from the 250 mg/kg group at week 1 and increased in the 1,000 mg/kg group at week 2 in comparison with the control group. During the recovery period, food intake was decreased in male rats in the 1,000 mg/kg group after week 2 of recovery. Male rats in the 1,000 mg/kg group consumed less water than the control group at week 11. In recovery, water consumption increased significantly in female rats from the 1,000 mg/kg group at weeks 1 and 2. All of these changes were considered to be temporary, given that body weight did not change significantly. Further, these changes were not dose-dependent, so were not considered to be toxic effects of ZnO NPs.
Ophthalmologic examination and urinalysis
No significant changes were observed on ophthalmologic examination or urinalysis.
Hematology and biochemistry
A decreased number of lymphocytes was documented in male rats from the 1,000 mg/kg group in comparison with the control group (P<0.05). Increased mean corpuscular hemoglobin was observed in female rats from the 1,000 mg/kg group in comparison with the vehicle control group (P<0.05, see Table 2).
During the recovery period, aspartate aminotransferase and creatine kinase levels were decreased in male rats from the 1,000 mg/kg group (P<0.01) in comparison with the vehicle control group. An increased albumin/globulin ratio was seen during the recovery period in female rats from the 1,000 mg/kg group in comparison with the control group (P<0.05). However, these statistically significant changes were not considered to be related to the toxic effects of ZnO, since there was no dose-dependent relationship and the results were not consistent between male and female rats (see Table 3).
Necropsy findings, organ weight, and pathologic examination
Right prostate gland hyperplasia and a yellowish mass in the head of the right epididymis was observed in one male rat each, and both were from the vehicle control group. Atrophy of the right seminal vesicle was observed in male rats from the 500 mg/kg group. A red nodule in the stomach (two cases) and a light brown change in the right kidney color (one case) occurred in female rats from the control group. A reddish stomach nodule was found in two female rats from the 250 mg/kg group. A reddish nodule in the stomach, a reddish color change in the caudal lobe of the liver, a light brown color change in the right kidney, and fragility of the right adrenal gland were observed in female rats from the 500 mg/kg group. One red nodule and a yellow mass in the stomach, a light brown color change of the right kidney, and a reddish color change in the accessory lobe of the liver were documented in female rats from the 1,000 mg/kg group (see Table 4). Given that these changes were also observed in the control group and did not show dose-dependency, they were deemed not to be due to application of ZnOSM20(−).
 
|
Table 4 Summary incidence of necropsy findings |
The weight of the epididymis decreased significantly (P<0.05) in male rats from the 500 mg/kg group. The weight of the thymus also decreased significantly in male rats from the 1,000 mg/kg recovery group in comparison with the control group (P<0.05). There was no significant difference in organ weight for female rats in comparison with the control group. Since the changes in organ weight occurred sporadically and independent of dosage, they were not considered to represent toxic effects of ZnO.
Areas of necrosis with regenerative hyperplasia and pigmentation were found in the livers of female rats in the control and 1,000 mg/kg groups; this could be the result of pressure from the bandage and repeated application of the test materials, and not from the test material itself, given that it occurred in both these groups. Also, lesions in other organs were considered as nonspecific that can be frequently seen in other repeated toxicology testing.
Analysis of tissue distribution
After repeated application for 90 days, concentrations of ZnO NPs were determined in the plasma, feces, brain, liver, kidneys, ovaries, testes, spleen, lungs, stomach, small intestine, large intestine, and skin. No differences in ZnO NP concentration were seen between females and males, with higher concentrations found in the liver, large intestine, small intestine, and feces upon increasing doses when compared with the negative and vehicle control groups. However, ZnO NP concentrations remained the same in the brain, testes, ovaries, spleen, stomach, plasma, kidneys, and lung as in the negative and vehicle control groups. The increased concentrations in the liver, large intestine, small intestine, and feces could be explained by rats ingesting the test material via licking behavior. Washing was done with distilled water prior to necropsy, but the test material seemed to remain on the skin. It was difficult to determine absorption through the skin during this experiment.
Discussion
ZnO NPs may enter the body via a number of routes. The skin is a potentially important route because it is the largest organ in the body, accounting for more than 10% of body mass. Since ZnO NPs are frequently used as an ingredient in sunscreens, safety concerns persist about these products.6 This study investigated the potential toxicity of ZnO NPs in internal organs and at the dermal administration site in Sprague Dawley rats after repeated exposure for 90 days. Since Kertesz et al5 demonstrated that penetration of nanomaterials into the skin after exposure may be time-dependent, it was important to evaluate the potential toxicity of ZnO NPs over a prolonged period of exposure. Although concern regarding the potential toxicity of ZnO NPs has been mounting, only a limited number of studies are available.7,8 A study by Pasupuleti et al9 reported lesions in the liver and pancreas after oral administration of ZnO NPs in rats. The same study also suggested that ZnO NPs were more toxic than ZnO microparticles, even at a lower dose. A 28-day repeated-dose toxicity study of dermally administered ZnO NPs4 reported significant changes in the collagen content of the skin at the application site but no systemic toxicity, except for an increased clotting time. Severe damage to the liver and lung tissues was observed when ZnO NPs were inhaled.10 There is a broad consensus that the principal human health risk may be from inhalation of NPs. However, dermal contact may increase this risk. Some researchers have proposed that there would not be any toxicologic concerns with dermally applied ZnO NPs;11,12 however, others have demonstrated absorption of ZnO particles through human skin or in a model equivalent to human skin.1,13 Hence, concerns about ZnO NPs are ongoing, not only with regard to possible skin toxicity, but also systemic toxicity. When ZnO NPs were administered through the oral or inhalational route, damage to the liver was evident.4,10 In the present study of the dermal toxicity of ZnO NPs, although increased concentrations of ZnO were seen in the liver during the tissue distribution test, no important adverse effects were observed. Only temporary effects, such as local skin irritation, occurred in a dose-dependent manner. Adachi et al14 also recently reported development of spongiotic dermatitis at the site of application with increased doses of titanium dioxide NPs, which was caused by mechanical irritation and generation of reactive oxygen species by the uncoated titanium dioxide NPs on the skin surface.
Skin penetration studies showed that less than 0.03% of the zinc content in ZnO NPs penetrated the epidermis.11,12 No NPs were observed in the lower stratum corneum or viable epidermis. Even the penetration measurement using multiphoton tomography showed that 30 nm ZnO NPs cannot penetrate skin.15 However, other studies have shown that small amounts of zinc from ZnO NPs in sunscreens could pass through the protective layers of skin when exposed to the sun in a real-life environment, and were detected in blood and urine.1 In a study of the repeated-dose dermal toxicity of ZnO NPs over 28 days, significant decreases in collagen content were found in ZnO-treated groups, implying that ZnO NPs penetrate deeply into the dermis and damage the collagen layer.15 Additional concerns in that study were the penetration of NPs into diseased or sunburnt skin with decreased barrier function. Interestingly, even in ultraviolet B-damaged skin with defective barrier function, ZnO was observed to stay in the epidermis and be unable to penetrate viable skin. Finally, the formulation or coating used for ZnO NPs may affect their penetration in skin. Leite-Silva et al17 demonstrated that coated ZnO NPs dispersed in a water-in-oil emulsion penetrated into skin more easily. In the present study, we did not find any evidence of skin penetration by ZnO NPs, and the increased concentrations found in the liver, small intestines, large intestines, and feces could be interpreted as oral ingestion of the test material by licking behavior. Systemic absorption of ZnO NPs via skin absorption seemed to be very limited. Even when applied to the dermis chronically, penetration of ZnO NPs may not be increased. To our knowledge, this is the first subchronic toxicity study of dermally applied ZnO NPs.
Conclusion
This study evaluated changes in internal organs, various bodily parameters, and skin after applying ZnO NPs dermally for 90 days. We evaluated the toxicity of these NPs in target organs, but there was no study-related internal organ toxicity except for temporary, dose-dependent inflammation of the skin at the application site. Therefore, there was no observed adverse effects of ZnO (20 nm, negative charge) up to 1,000 mg/kg body weight in both sexes of rats.
Acknowledgment
This research was supported by a grant (10182MFDS991) from the Ministry of Food and Drug Safety between 2010 and 2013, and Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Science, ICT and Future Planning (NRF2013R1A2A2A01068137).
Disclosure
The authors report no conflicts of interest in this work.
References
Gulson B, McCall M, Korsch M, et al. Small amounts of zinc from zinc oxide particles in sunscreens applied outdoors are absorbed through human skin. Toxicol Sci. 2010;118(1):140–149. | ||
Smijs TG, Bouwstra JA. Focus on skin as a possible port of entry for solid nanoparticles and the toxicological impact. J Biomed Nanotechnol. 2010;6(5):469–484. | ||
Martorano LM, Stork CJ, Li YV. UV irradiation-induced zinc dissociation from commercial zinc oxide sunscreen and its action in human epidermal keratinocytes. J Cosmet Dermatol. 2010;9(4):276–286. | ||
Surekha P, Kishore AS, Srinivas A, et al. Repeated dose dermal toxicity study of nano zinc oxide with Sprague-Dawley rats. Cutan Ocul Toxicol. 2012;31(1):26–32. | ||
Kertesz Z, Szikszai Z, Gontier E, et al. Nuclear microprobe study of TiO2-penetration in the epidermis of human skin xenografts. Nucl Instrum Methods Phys Res. 2005;231(1):280–285. | ||
Burnett ME, Wang SQ. Current sunscreen controversies: a critical review. Photodermatol Photoimmunol Photomed. 2011;27(2):58–67. | ||
Jang YS, Lee EY, Park YH, et al. The potential for skin irritation, phototoxicity, and sensitization of ZnO nanoparticles. Mol Cell Toxicol. 2012;8(2):171–177. | ||
Lee SH, Pie JE, Kim YR, Lee HR, Son SW, Kim MK. Effects of zinc oxide nanoparticles on gene expression profile in human keratinocytes. Mol Cell Toxicol. 2012;8(2):113–118. | ||
Pasupuleti S, Alapati S, Ganapathy S, Anumolu G, Pully NR, Prakhya BM. Toxicity of zinc oxide nanoparticles through oral route. Toxicol Ind Health. 2012;28(8):675–686. | ||
Wang L, Ding W, Zhang F. Acute toxicity of ferric oxide and zinc oxide nanoparticles in rats. J Nanosci Nanotechnol. 2010;10(12):8617–8624. | ||
Pirot F, Millet J, Kalia YN, Humbert P. In vitro study of percutaneous absorption, cutaneous bioavailability and bioequivalence of zinc and copper from five topical formulations. Skin Pharmacol. 1996;9(4):259–269. | ||
Cross SE, Innes B, Roberts MS, Tsuzuki T, Robertson TA, McCormick P. Human skin penetration of sunscreen nanoparticles: in-vitro assessment of a novel micronized zinc oxide formulation. Skin Pharmacol Physiol. 2007;20(3):148–154. | ||
Jeong SH, Kim JH, Yi SM, et al. Assessment of penetration of quantum dots through in vitro and in vivo human skin using the human skin equivalent model and the tape stripping method. Biochem Biophys Res Commun. 2010;394(3):612–615. | ||
Adachi K, Yamada N, Yoshida Y, Yamamoto O. Subchronic exposure of titanium dioxide nanoparticles to hairless rat skin. Exp Dermatol. 2013;22(4):278–283. | ||
Darvin ME, König K, Keller-Hoefer M, et al. Safety assessment by multiphoton fluorescence/second harmonic generation/Hyper-Rayleigh scattering tomography of ZnO nanoparticles used in cosmetic products. Skin Pharmacol Physiol. 2012;25(4):219–226. | ||
Monteiro-Riviere NA, Wiench K, Landsiedel R, Schulte S, Inman AO, Riviere JE. Safety evaluation of sunscreen formulations containing titanium dioxide and zinc oxide nanoparticles in UVB sunburned skin: an in vitro and in vivo study. Toxicol Sci. 2011;123(1):264–280. | ||
Leita-Silva VR, Lamer ML, Sanchez WY, et al. The effect of formualtion on the penetration of coated and uncoated zinc oxide nanoparticles into the viable epidermis of human skin in vivo. Eur J Pharm Biopharm. 2013;84(2):297–308. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.