Back to Journals » Therapeutics and Clinical Risk Management » Volume 14
Videolaryngoscopy versus fiberoptic bronchoscope for awake intubation – a systematic review and meta-analysis of randomized controlled trials
Authors Jiang J , Ma DX, Li B , Wu AS, Xue FS
Received 1 May 2018
Accepted for publication 27 June 2018
Published 15 October 2018 Volume 2018:14 Pages 1955—1963
DOI https://doi.org/10.2147/TCRM.S172783
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Deyun Wang
Jia Jiang,1 Da-Xu Ma,1 Bo Li,2 An-Shi Wu,1 Fu-Shan Xue3
1Department of Anesthesiology, Beijing Chaoyang Hospital, Capital Medical University, Beijing 100020, China; 2Beijing Hospital of Traditional Chinese Medicine, Affiliated to Capital Medical University, Beijing Institute of Traditional Chinese Medicine, Beijing 100010, China; 3Department of Anesthesiology, Beijing Friendship Hospital, Capital Medical University, Beijing 100050, China
Background: Awake intubation with videolaryngoscopy (VL) is a novel method that is drawing more and more attention as an alternative to awake intubation with fiberoptic bronchoscope (FOB). This meta-analysis is designed to determine the performance of VL compared to the FOB for awake intubation.
Methods: The Cochrane Central Register of Controlled Trials, PubMed, Embase, and Web of science were searched from database inception until October 30, 2017. Randomized controlled trials comparing VL and FOB for awake intubation were selected. The primary outcome was the overall success rate. Rev-Man 5.3 software was used to perform the pooled analysis and assess the risk of bias for each eligible study. The GRADE system was used to assess the quality of evidence for all outcomes.
Results: Six studies (446 patients) were included in the review for data extraction. Pooled analysis did not show any difference in the overall success rate by using VL and FOB (relative risk [RR], 1.00; P=0.99; high-quality evidence). There was no heterogeneity among studies (I2=0). Subgroup analyses showed no differences between two groups through nasal (RR, 1.00; P=1.00; high-quality evidence) and oral intubations (RR, 1.00; P=0.98; high-quality evidence). The intubation time was shorter by using VL than by using FOB (mean difference, -40.4 seconds; P<0.01; low-quality evidence). There were no differences between groups for other outcomes (P>0.05).
Conclusion: For awake intubation, VL with a shorter intubation time is as effective and safe as FOB. VL may be a useful alternative to FOB.
Keywords: airway management, awake intubation, videolaryngoscopy, fiberoptic bronchoscopy, randomized controlled clinical trials, outcomes
Introduction
As difficulties in airway management contribute significantly to severe anesthesia-related morbidity and mortality,1,2 securing the airway in a safe fashion remains a concern for anesthetists. Awake intubation is one recommended option for management of difficult airway,3,4 as it can provide patients’ safety by keeping airway patency, maintaining gas exchange, and protecting against aspiration during the procedure.5 Because of its ability to be manually manipulated and see around corners, fiberoptic bronchoscope (FOB) is a common choice for awake intubation and has been regarded as a “gold standard” tool for managing difficult airway.6
Although awake intubation using FOB should be logically mastered by all anesthesiologists, its use is potentially limited by several factors. First, the technique using an FOB is difficult to learn and master, as it needs an extensive training and practice.7 In clinical setting, however, very little cases need awake intubation.5 Even practitioners can learn it through simulated training, such chance is not common in most centers, and if rarely used, the competence with fiberoptic intubation is difficult to maintain. Second, the presence of edema, excess airway tissue, secretions, or blood in the airway will obscure the image.8 Finally, it is expensive and requires disinfection between two uses.
Awake intubation with videolaryngoscopy (VL) is a new method that is drawing more and more attention as an alternative to awake intubation with FOB. In available literatures, there have been many case reports and case series regarding the successful use of VL-assisted awake intubation in patients with predicted and known difficult airways.9–20 Furthermore, several randomized clinical trials (RCTs) have compared performance of VL with FOB for awake intubation.16,17,21–24 This systematic review and meta-analysis aimed to assess whether VL can be used as a useful alternative to FOB for awake intubation according to the evidence from available RCTs.
Methods
Our review has been registered at PROSPERO (http://www.crd.york.ac.uk/PROSPERO) and the registration number is: CRD42017078280. The PRISMA guidelines were followed.25 The current issue of the Cochrane Central Register of Controlled Trials (CENTRAL; 2017, Issue 9), PubMed (1946 to October 30, 2017), Embase (1974 to October 30, 2017), and Web of Science (1900 to October 30, 2017) were searched. The search strategies of four electronic databases are provided in Supplementary material.26 The reference lists of all eligible trials were also screened for additional citations. No language restriction was imposed. If necessary, the study authors were mailed for additional information.
Only RCTs comparing the VL and FOB for awake intubation in hospital were included. Manikin study, cadaver study, simulated study, case series, and observational study were excluded. Adult patients (age >18 years) who needed an oral or nasal awake intubation for all kinds of reasons were included. Awake intubation should be performed under airway topical anesthesia and sedation, which aimed for a patient sedation equivalent to a Ramsay score of 2–4 with spontaneous breathing. Patients in the intervention group used a VL. Other maneuvers like the external laryngeal manipulation, adjustment of head and neck position, and the use of intubation stylet or introducer could be used as appropriate. Patients in the control group used an FOB. The primary outcome was overall success rate. The secondary outcomes were intubation time (confirmed by capnography measured from the advancement of the studied devices behind the teeth until the appearance of a capnography curve), first-attempt success rate, rate of low oxygen saturation (SpO2 <90%), rate of sore throat after surgery, and the operators’ and patients’ satisfaction of the studied techniques. Different methods might be used to assess these outcomes. The authors of included studies were contacted to try to obtain a binary variable, namely the number of satisfaction and dissatisfaction. If this was impossible, we planned to do a qualitative systematic review instead of pooling the results.
The titles and abstracts were independently screened by two study authors (J.J.; D.X.M.). After retrieving the full texts of any potentially relevant studies, their eligibility was determined. Any disagreements between the two review authors were resolved by discussion with other authors until a consensus was obtained. A PRISMA flow diagram was completed to record the selection process in sufficient detail.27
Data were extracted by two review authors (J.J.; D.X.M.). For continuous data, mean, SD, and sample size were extracted. Data like median that cannot be used directly were converted to SD by using the formula provided in the Cochrane Handbook.26 For the dichotomous variables, the number of events occurred and the sample size were extracted. Any disagreement on data extraction was resolved by discussion with a third author (F.S.X.) until a consensus was reached.
The risk of bias for each eligible study was independently assessed by two review authors (J.J.; D.X.M.) by using the “risk of bias” assessment tool of the Cochrane Handbook,26 and a “risk of bias” summary figure was generated by using Rev-Man 5.3. If all seven domains were assigned to “low risk” of bias, the study was classified as “low risk”; if one or more domains were assigned to “unclear risk” of bias, the study was classified as “unclear risk”; if one or more domains were assigned to “high risk” of bias, the study was classified as “high risk”.26 The criteria of the GRADE system (study limitations, consistency of effect, imprecision, indirectness, and publication bias) were used to assess the quality of the body of evidence associated with all outcomes.28,29 Then a “GRADE evidence profile” table was developed by using the GRADE software (www.guidelinedevelopment.org) to rate these outcomes as high, moderate, low, or very low quality. The quality of evidence was downgraded by one or two levels when serious or very serious deficiencies were considered in these criteria.
Weighted mean difference and 95% CI were used for continuous data. Relative risk (RR) and 95% CI were used for dichotomous data, and P<0.05 was considered statistically significant. Rev-Man 5.3 software was used to perform the pooled analysis for the outcomes from more than one study. A chi-squared test with the I2 statistic (with statistical significance set at the level of two-tailed 0.10) was used to describe the percentage of the total variance across studies from heterogeneity rather than from chance. If I2<40%, namely, there was no statistical heterogeneity among studies, a fixed-effect model was used; otherwise, a random-effects model was used instead. For the results that could not be analyzed through meta-analysis, only a qualitative systematic review was planned.
The study author of the original report was contacted for important missing statistics. If these data still could not be obtained, the available data were used. For the participants missing because of dropout, if “missing at random”, analysis was performed based on the available data, and if not, an available case analysis was performed, and the potential bias was discussed in the discussion section. If a study did not mention withdrawals, it was assumed that there was no dropout.26
Clinical and methodologic heterogeneity was considered before performing pooled analysis. In the presence of statistical heterogeneity (I2>40%) or an indication of clinical heterogeneity, subgroup analysis was planned for the primary outcome according to the following possible heterogeneous factors: different intubation pathways: oral or nasal; operator’s experience: experienced (certificated anesthesiologist, performed more than 50 successful intubations, or according to the judgment of authors) or inexperienced. Sensitivity analysis was planned to conduct to explore other potential sources of heterogeneity if necessary. Reporting bias was planned to assess by using funnel plots if the result of the primary outcome was from at least ten trials.30
To assess the reliability and conclusiveness of available evidence on adverse outcome, trial sequential analysis was planned for the rate of low oxygen saturation (SpO2 <90%), which was assumed to be a relatively low event.31,32 The information size required was calculated for this outcome, providing an estimate of how many more patients would be required to make a reliable conclusion. A conventional calculation for sample size estimation, with conventional values for α and β errors (0.05 and 0.20) and assuming a two-sided test, was used. The RR reduction was set at 50%. The control event rate from our own results was used to do calculation.
Results
Using search strategy, a total of 347 papers were identified. Of them, 335 were excluded during title and abstract screening as they were duplicate or irrelevant to our research question. Twelve studies were selected for full-text assessment using inclusion and exclusion criteria. Five studies were further removed because of non-RCTs, volunteer study, and intubation after anesthesia induction. Authors of one study were contacted for full text to confirm its eligibility;33 however, no reply was received. Due to no available data and seemly unfinished property, this study was excluded.33 Eventually, six studies with a total number of 446 patients were included in the review for data extraction.16,17,21–24 Five authors were contacted for unpublished data and detailed information on study design,16,17,21–23 and only two of them replied.17,21 Process of selection of studies has been shown in Figure 1.
  | Figure 1 Searching process of identified records. |
Description of included studies
Characteristics of included studies are listed in Table 1. Of the six included studies, four were oral intubation and the remaining were nasal intubation. In the VL group, three studies used Glidescope, and others applied C-MAC, McGrath Series 5, and Airway Scope , respectively. All awake intubations were performed by experienced operators because of predicted difficult airway or the kind of surgery that may make the intubation difficult, such as oral-facial and cervical spine surgery. All studies excluded the patients with a restricted mouth opening and all intubations were performed under airway topical anesthesia and moderate sedation (eg, target Ramsay Sedation Scale of 3–4) with spontaneous breathing. In one study, patients received airway topical anesthesia with transtracheal injection.23
The overall risk of bias of the included studies was relatively low. Three of them could be classified as “low risk” studies17,21,22 and one as “high risk” studies.23 Detailed information regarding the risk of bias of the included studies is shown in Figure 2 and summarized in Table S1. A funnel plot could not be obtained because of very limited studies. The GRADE system showed that the quality of most evidences was high or moderate because of the low level of heterogeneity and inclusion of only RCTs. The results of the evidence of outcomes are listed in Table S2.
  | Figure 2 Risk of bias summary: review authors’ judgements about each risk of bias item for each included study. |
Overall success rate
The data on the overall success rate for all six included studies were available. Pooled analysis showed no significant difference in the overall success rate between VL and FOB (RR, 1.00; 95% CI, 0.98–1.02; n=446; P=1.00; high-quality evidence). There was no heterogeneity among studies (P=0.99; I2=0%) (Figure 3).
  | Figure 3 VL vs DL for overall success rate. |
Subgroup analysis according to different intubation pathways showed no significant difference for nasal intubation (two studies; RR, 1.00; n=208; P=1.00; high-quality evidence) or oral intubation (four studies; RR, 1.06; n=238; P=0.98; high-quality evidence). All intubations were performed by the experienced operators, so subgroup analysis according to operator’s experience was impossible (Figure S1).
Secondary outcomes
All six studies reported the intubation time and pooled analysis showed a shorter intubation time in the VL group than in the FOB group (mean difference, −40.4 seconds; 95% CI, −61.0 to −19.9 seconds; n=388; P<0.01; low-quality evidence). There was significant heterogeneity among studies (P<0.01; I2=95%) (Figure S2).
There were no significant differences in other secondary outcomes (P>0.05). Results of secondary outcomes including intubation time, first-attempt success rate, rates of low oxygen saturation, and sore throat are summarized in Table 2 and Figures S3–S5.
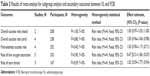  | Table 2 Results of meta-analysis for subgroup analysis and secondary outcomes between VL and FOB |
Four studies16,17,23,24 reported the patients’ satisfaction and three studies17,21,23 reported operators’ satisfaction by using quite different scales. Abdellatif et al16 and Wahba et al24 used a 3-score scale to assess patients’ satisfaction and presented the results as categorical data; Mendonca et al17 used a 0–100 mm visual analog scale (0 being “worst” and 100 “best”) to rate the ease of procedure for operators and the comfort of patients during procedure; Rosenstock et al23 also used a similar visual analog scale, but with a reversed assessment method (0 expressing “best” and 10 “worst”). Kramer et al21 used a 6-score scale to rate the operators’ satisfaction that was expressed as median and IQR. All the authors were contacted, but no original data were obtained. Thus, these data could not be transformed into a binary variable as planned. In all original reports, however, there was no significant difference in operators’ or patients’ satisfaction (P>0.05).
Data for the rate of low oxygen saturation (SpO2 <90%) were available from five trials, with 26 events and 333 participants, which gave a control event rate of 9.52%. Pooled analysis showed no significant difference between groups (RR, 0.47; 95% CI, 0.21–1.06; P=0.07; moderate-quality evidence). A 50% RR reduction would lower this event rate to 4.76%. Sample size calculation (with α=0.05 and β=0.20) gave a sample size of 919 participants (Figure S6).
Discussion
As we know, this is the first meta-analysis and systematic review of available RCTs comparing the VL and FOB for awake intubation. Pooled analysis showed that the overall success rate and the first attempt success rate were similar with two devices for awake intubation. However, the use of VL could provide a decreased intubation time by about 40 seconds. Although no significant difference was obtained, the use of VL showed a trend of lower rate of low oxygen saturation during the procedure.
Even though our results show that VL has a similar success rate to the FOB for awake intubation, VL has the following advantages. First, the use of FOB requires a long learning process. To obtain the competence with fiberoptic intubation (>90% success rate within 3 minutes), anesthetists need to perform at least 25 intubations.7 Once learned, moreover, regular practice is still required to maintain this skill. However, in clinical practice, there are very few opportunities for practical exercise. A 2003 survey of anesthetists found that only 59% of anesthetists reported to have skills in fiberoptic intubation.34 It should be noted that all the intubations included in our analysis are performed by experienced operators who probably have same experience in the use of VL and FOB. For novices, however, the result might be different. In fact, VL has been proved to be easily learned by inexperienced operators with a steep learning curve and a high success rate (>90%).35–37 Therefore, inexperienced users may find awake intubation with VL easier than awake FOB in patients with a difficult airway. Second, during the intubation, VL can create space within the upper airway, allowing effective aspiration of secretions and blood from the airway, and facilitating airway topical anesthesia under direct vision; Third, in contrast to blind passage of the tracheal tube through the FOB, the tracheal tube placement can be observed with VL, reducing the risks of the tube impingement on the glottis and airway trauma. Furthermore, the magnified airway views on VL screen help recognition of distorted airway anatomy.8,38–40 Fourth, VL is portable and more accessible and easier to set up. Fifth, most institutions still use the standard FOB, which requires disinfection after each use, delaying the device’s availability for further use and might have increasing cost.41 Finally, for the patients with an unstable cervical spine, although the use of FOB for awake intubation is always preferred as it minimizes the cervical movement, the introduction of FOB may cause further cervical spine movement because it requires some degree of jaw thrust.42,43
On the other hand, it should be emphasized that awake VL is not suitable for all types of difficult airways and thus cannot fully replace awake fiberoptic intubation for management of difficult airways.5,44 Although a slim designed McGrath MAC X-blade can be used in patients with restricted mouth opening,45 a very limited mouth opening can make the use of VL impossible, as “an inter-dental distance of at least 18–20 mm is required to insert even the narrow blades”.46,47 Moreover, some pathologic conditions of the upper airway may prevent success with VL. A large retrospective study of patients with predicted difficult airways showed that the strongest predictors of failed VL were “altered neck anatomy with the presence of a surgical scar, radiation changes, or mass”.48 This suggests that VL may not make intubation easier in severe upper airway distortion caused by malignancy or extensive oropharyngeal infection. Interestingly, in our included studies, the two approaches were nearly equivalent and complementary; that is, a failed attempt could be intubated with the alternative technique.16,21 Furthermore, awake intubation by combined VL and FOB has been successfully used in some more complicated difficult airway conditions.39,49,50 All these mean that airway training should be diversified so that there could be an alternative when one method fails.
Many of the studies included in this analysis chose intubation time as the primary outcome, probably because the success rate was not supposed to be different between VL and FOB for the experienced operators. Although our pooled analysis showed a shortened intubation time of 40 seconds by using VL, clinically significant of this time difference for patients undergoing awake intubation was still unclear. Certainly, for a sedated patient performed airway topical anesthesia, reducing intubation time must be always borne in mind to decrease the risk of aspiration.
Our analysis showed that a longer intubation time did not result in a higher incidence of hypoxemia with FOB. This may be because the included studies are carried out in elective surgical patients with a good ability to tolerate hypoxia. However, according to the trial sequential analysis, this “no difference” needs more researches to verify and there is still a trend of lower incidence of low oxygen saturation for VL. In addition, it should be noted that for less experienced intubators, the time gap may be much longer, which may result in more complications in the FOB group, such as a mild-to-severe glottis damage.
Our study only included RCTs. Although blinding was not adopted in most studies, we judged “no blinding” as “low risk”, as it seems impossible to blinding the personnel and the results are unlikely to be changed. Risk assessment of bias for the included studies showed that three of six studies could be classified as “low risk” studies and only one was classified as “high risk”. The study by Rosenstock et al had “a striking imbalance between the groups in the number of excluded patients” (two of 45 from the FOB group and seven of 48 from the VL group, most of which are due to impossible transtracheal injection).23 The postrandomization exclusions may bias or distort the conclusion of this study.51 Anyway, this kind of invasive airway topical anesthesia has been abandoned in most cases. The quality of most evidences was high or moderate because of only RCT included and very low heterogeneity.
Limitations
First, almost all included studies were performed in surgical patients by excluding patients with upper airway emergencies. Thus, we are not sure how the results might be affected when performing emergent intubations in more stressful circumstances. The use of VL in emergent intubation does not show any advantage over DL,52,53 whether VL is superior to FOB for awake emergent intubation is unclear; second, all the operators are experienced, and the results may not be applicable to less experienced operators; third, although all participants in our included studies need awake intubation for “difficult” situations, the situations are different substantially, including oropharyngeal cancer, limited neck extension, limited jaw protrusion, high Mallampati score, or a requirement to maintain the cervical spine (eg, cervical spine fixation) in the neutral position during intubation. Different intubation devices may be suitable for different “difficult” situations. Moreover, all included studies excluded the patients with restricted mouth opening, and this might underestimate the role of FOB in managing difficult airways. Finally, the results on complications remain unreliable because of limited participants, which need a much larger population.
This review concludes that when awake intubation is performed by experienced operators, VL with a shorter intubation time is as effective and safe as FOB.
Author contributions
FSX and JJ designed the plan of this analysis. All authors contributed toward data collection and analysis, drafting and critically revising the paper and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
Schiff JH, Welker A, Fohr B, et al. Major incidents and complications in otherwise healthy patients undergoing elective procedures: results based on 1.37 million anaesthetic procedures. Br J Anaesth. 2014;113(1):109–121. | ||
Shiima Y, Berg RA, Bogner HR, Morales KH, Nadkarni VM, Nishisaki A; National Emergency Airway Registry for Children Investigators. Cardiac arrests associated with tracheal intubations in PICUs: a multicenter cohort study. Crit Care Med. 2016;44(9):1675–1682. | ||
Law JA, Broemling N, Cooper RM, et al; Canadian Airway Focus Group. The difficult airway with recommendations for management – part 2 – the anticipated difficult airway. Can J Anaesth. 2013;60(11):1119–1138. | ||
Apfelbaum JL, Hagberg CA, Caplan RA, et al. Practice guidelines for management of the difficult airway: an updated report by the American Society of Anesthesiologists Task Force on Management of the Difficult Airway. Anesthesiology. 2013;118(2):251–270. | ||
Law JA, Morris IR, Brousseau PA, de La Ronde S, Milne AD. The incidence, success rate, and complications of awake tracheal intubation in 1,554 patients over 12 years: an historical cohort study. Can J Anaesth. 2015;62(7):736–744. | ||
Simmons ST, Schleich AR. Airway regional anesthesia for awake fiberoptic intubation. Reg Anesth Pain Med. 2002;27(2):180–192. | ||
Heidegger T, Gerig HJ, Ulrich B, Schnider TW. Structure and process quality illustrated by fibreoptic intubation: analysis of 1612 cases. Anaesthesia. 2003;58(8):734–739. | ||
Swarbrick C, Turner M. Awake nasal intubation: from darkness into light, or a leap into the unknown? Anaesthesia. 2015;70(7):881–882. | ||
Stopar-Pintaric T, Markova L, Tomazevic M, Hodzovic I. An awake videolaryngoscope-assisted intubation in a patient with Madelung disease and a critical airway obstruction. Minerva Anestesiol. 2017;83(6):660–662. | ||
Kajekar P, Mendonca C, Danha R, Hillermann C. Awake tracheal intubation using Pentax airway scope in 30 patients: a case series. Indian J Anaesth. 2014;58(4):447–451. | ||
Jeyadoss J, Nanjappa N, Nemeth D. Awake intubation using Pentax AWS videolaryngoscope after failed fibreoptic intubation in a morbidly obese patient with a massive thyroid tumour and tracheal compression. Anaesth Intensive Care. 2011;39(2):311–312. | ||
Uslu B, Damgaard Nielsen R, Kristensen BB. McGrath videolaryngoscope for awake tracheal intubation in a patient with severe ankylosing spondylitis. Br J Anaesth. 2010;104(1):118–119. | ||
Mcguire BE. Use of the McGrath video laryngoscope in awake patients. Anaesthesia. 2009;64(8):912–914. | ||
Dimitriou VK, Zogogiannis ID, Liotiri DG. Awake tracheal intubation using the Airtraq laryngoscope: a case series. Acta Anaesthesiol Scand. 2009;53(7):964–967. | ||
Asai T. Pentax-AWS videolaryngoscope for awake nasal intubation in patients with unstable necks. Br J Anaesth. 2010;104(1):108–111. | ||
Abdellatif AA, Ali MA. GlideScope videolaryngoscope versus flexible fiberoptic bronchoscope for awake intubation of morbidly obese patient with predicted difficult intubation. Middle East J Anaesthesiol. 2014;22(4):385–392. | ||
Mendonca C, Mesbah A, Velayudhan A, Danha R. A randomised clinical trial comparing the flexible fibrescope and the Pentax Airway Scope (AWS)(®) for awake oral tracheal intubation. Anaesthesia. 2016;71(8):908–914. | ||
Fayolle-Pivot L, Magnin C, Tissot S, Bertin-Maghit M, Allaouchiche B. Aws-Pentax™ Lglottiscope. Alternative la fibroscopie dans deux cas d’intubation difficile prévue du brûlé grave. Ann Fr Anesth Reanim. 2013;32(5):355–357. | ||
Markova L, Stopar-Pintaric T, Luzar T, Benedik J, Hodzovic I. A feasibility study of awake videolaryngoscope-assisted intubation in patients with periglottic tumour using the channelled King Vision® videolaryngoscope. Anaesthesia. 2017;72(4):512–518. | ||
Mhyre JM, Healy D. The unanticipated difficult intubation in obstetrics. Anesth Analg. 2011;112(3):648–652. | ||
Kramer A, Müller D, Pförtner R, Mohr C, Groeben H. Fibreoptic vs videolaryngoscopic (C-MAC(®) D-BLADE) nasal awake intubation under local anaesthesia. Anaesthesia. 2015;70(4):400–406. | ||
Mahran EA, Hassan ME. Comparative randomised study of GlideScope® video laryngoscope versus flexible fibre-optic bronchoscope for awake nasal intubation of oropharyngeal cancer patients with anticipated difficult intubation. Indian J Anaesth. 2016;60(12):936–938. | ||
Rosenstock CV, Thøgersen B, Afshari A, Christensen AL, Eriksen C, Gätke MR. Awake fiberoptic or awake video laryngoscopic tracheal intubation in patients with anticipated difficult airway management: a randomized clinical trial. Anesthesiology. 2012;116(6):1210–1216. | ||
Wahba SS, Tammam TF, Saeed AM. Comparative study of awake endotracheal intubation with Glidescope video laryngoscope versus flexible fiber optic bronchoscope in patients with traumatic cervical spine injury. Egypt J Anaesth. 2012;28(4):257–260. | ||
Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol. 2009;62(10):1006–1012. | ||
Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. | ||
Shamseer L, Moher D, Clarke M, et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: elaboration and explanation. BMJ. 2015;350:g7647. | ||
Guyatt G, Oxman AD, Akl EA, et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J Clin Epidemiol. 2011;64(4):383–394. | ||
Guyatt GH, Oxman AD, Kunz R, Vist GE, Falck-Ytter Y, Schünemann HJ; GRADE Working Group. What is “quality of evidence” and why is it important to clinicians? BMJ. 2008;336(7651):995–998. | ||
Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–634. | ||
Wetterslev J, Thorlund K, Brok J, Gluud C. Trial sequential analysis may establish when firm evidence is reached in cumulative meta-analysis. J Clin Epidemiol. 2008;61(1):64–75. | ||
Wetterslev J, Thorlund K, Brok J, Gluud C. Estimating required information size by quantifying diversity in random-effects model meta-analyses. BMC Med Res Methodol. 2009;9:86. | ||
Moore A, El-Bahrawy A, El-Mouallem E, Lattermann R, Hatzakorzian R, LiPishan W, Schricker T. Videolaryngoscopy or fibreoptic bronchoscopy for awake intubation of bariatric patients with predicted difficult airways – a randomised, controlled trial. Anaesthesia. 2017;72(4):538–539. | ||
Ezri T, Szmuk P, Warters RD, Katz J, Hagberg CA. Difficult airway management practice patterns among anesthesiologists practicing in the United States: have we made any progress? J Clin Anesth. 2003;15(6):418–422. | ||
Cooper RM, Pacey JA, Bishop MJ, Mccluskey SA. Early clinical experience with a new videolaryngoscope (GlideScope) in 728 patients. Can J Anaesth. 2005;52(2):191–198. | ||
Ray DC, Billington C, Kearns PK, et al. A comparison of McGrath and Macintosh laryngoscopes in novice users: a manikin study. Anaesthesia. 2009;64(11):1207–1210. | ||
Rai MR, Dering A, Verghese C. The Glidescope system: a clinical assessment of performance. Anaesthesia. 2005;60(1):60–64. | ||
Johnson DM, From AM, Smith RB, From RP, Maktabi MA. Endoscopic study of mechanisms of failure of endotracheal tube advancement into the trachea during awake fiberoptic orotracheal intubation. Anesthesiology. 2005;102(5):910–914. | ||
Markova L, Stopar-Pintaric T, Luzar T, Benedik J, Hodzovic I. A feasibility study of awake videolaryngoscope-assisted intubation in patients with periglottic tumour using the channelled King Vision® videolaryngoscope. Anaesthesia. 2017;72(4):512–518. | ||
Asai T, Shingu K. Difficulty in advancing a tracheal tube over a fibreoptic bronchoscope: incidence, causes and solutions. Br J Anaesth. 2004;92(6):870–881. | ||
Gupta D, Wang H. Cost-effectiveness analysis of flexible optical scopes for tracheal intubation: a descriptive comparative study of reusable and single-use scopes. J Clin Anesth. 2011;23(8):632–635. | ||
Brimacombe J, Keller C, Künzel KH, Gaber O, Boehler M, Pühringer F. Cervical spine motion during airway management: a cinefluoroscopic study of the posteriorly destabilized third cervical vertebrae in human cadavers. Anesth Analg. 2000;91(5):1274–1278. | ||
Greenland KB, Lam MC, Irwin MG. Comparison of the Williams Airway Intubator and Ovassapian Fibreoptic Intubating Airway for fibreoptic orotracheal intubation. Anaesthesia. 2004;59(2):173–176. | ||
Lohse J, Noppens R. Awake video laryngoscopy – an alternative to awake fiberoptic intubation? Anasthesiol Intensivmed Notfallmed Schmerzther. 2016;51(11–12):656–663. | ||
Arslan Zİ, Ozdal P, Ozdamar D, Agır H, Solak M. Nasotracheal intubation of a patient with restricted mouth opening using a McGrath MAC X-Blade and Magill forceps. J Anesth. 2016;30(5):904–906. | ||
Xue FS, Liu YY, Li HX, Yang GZ. Videolaryngoscopy in airway management-what every anesthesiologist should know. J Anesth Perioper Med. 2018;5(1):23–33. | ||
Kleine-Brueggeney M, Greif R, Schoettker P, Savoldelli GL, Nabecker S, Theiler LG. Evaluation of six videolaryngoscopes in 720 patients with a simulated difficult airway: a multicentre randomized controlled trial. Br J Anaesth. 2016;116(5):670–679. | ||
Aziz MF, Healy D, Kheterpal S, Fu RF, Dillman D, Brambrink AM. Routine clinical practice effectiveness of the Glidescope in difficult airway management: an analysis of 2,004 Glidescope intubations, complications, and failures from two institutions. Anesthesiology. 2011;114(1):34–41. | ||
Gupta A, Kapoor D, Awana M, Lehl G. Fiberscope assisted videolaryngoscope intubation in the surgical treatment of TMJ ankylosis. J Maxillofac Oral Surg. 2015;14(2):484–486. | ||
Moore AR, Schricker T, Court O. Awake videolaryngoscopy-assisted tracheal intubation of the morbidly obese. Anaesthesia. 2012;67(3):232–235. | ||
Todd MM, Bayman EO. Fiberoptic versus videolaryngoscopic management of the difficult airway: problems with postrandomization patient exclusion. Anesthesiology. 2013;118(2):460. | ||
Zhao BC, Huang TY, Liu KX. Video laryngoscopy for ICU intubation: a meta-analysis of randomised trials. Intensive Care Med. 2017;43(6):947–948. | ||
Savino PB, Reichelderfer S, Mercer MP, Wang RC, Sporer KA. Direct versus video laryngoscopy for prehospital intubation: a systematic review and meta-analysis. Acad Emerg Med. 2017;24(8):1018–1026. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

