Back to Journals » International Medical Case Reports Journal » Volume 12
Vaso-nervorumitis: responsible for pain in Buerger’s disease?
Authors Fazeli B , Farzadnia M, Taheri H
Received 11 December 2018
Accepted for publication 15 March 2019
Published 29 April 2019 Volume 2019:12 Pages 119—123
DOI https://doi.org/10.2147/IMCRJ.S197862
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Ronald Prineas
Bahare Fazeli,1,2 Mahdi Farzadnia,3 Hossein Taheri4
1Immunology Research Center, Inflammation and Inflammatory Diseases Division, School of Medicine, Mashhad University of Medical Sciences, Mashhad, Iran; 2Vascular Independent Research and Education, European Foundation, Milan, Italy; 3Department of Pathology, Emam Reza Hospital, Mashhad University of Medical Sciences, Mashhad, Iran; 4Surgery Department, Farabi Hospital, Mashhad, Iran
Abstract: Buerger’s disease (BD) is an episodic, inflammatory, and occlusive peripheral vascular disease with unknown etiology, which can lead to tissue or limb loss. BD patients usually present neurological symptoms from the early stages of the disease including numbness, cold sensation, and allodynia as the disease progresses. Pain in the late stages of BD is very severe, almost resistant to opioid pain killers, and sometimes compels the patient to pursue major amputation. Therefore, pain management in BD patients is one of the most important and, at the same time, challenging issues since its main etiology is not well understood. Recently, a 39-year-old male smoker with a diagnosis of BD underwent a below-knee amputation in his left leg. Oddly, we found that the vasa-nervorum of the sural nerve had the pathological changes usually observed in BD, including inflammation and proliferation of endothelial cells. Notably, the inflammation was limited to the vasa-nervorum and did not extend to the nerve fascicles. Our findings could provide a clue to taking the approach of managing pain in BD as if it were vasculitis neuropathy; and the inflammation of the vasa-nervorum individually might be responsible for the pain characteristic of BD. In addition, our findings could indicate that BD is a systemic vasculitis of microcirculation and, hence, a different treatment approach for BD might be needed in addition to antithrombotic and vasodilator.
Keywords: thromboangiitis obliterans, Buerger’s disease, vasa-nervorum, pain, pathophysiology
Introduction
Buerger’s disease (BD) is an episodic, inflammatory, and occlusive peripheral vascular disease with unknown etiology that can lead to tissue or limb loss. BD is usually diagnosed in young, male smokers.1
The most disturbing manifestation of BD for patients is pain. The pain in BD is a usually asymmetrical and localized burning pain of the toes that does not respond to routine pain killers.2 With progression of the disease, the pain becomes very severe and sometimes compels the patient to pursue major amputation. Therefore, pain management in BD patients is one of the most important and, at the same time, challenging issues, since its main etiology is not well understood.
We present a case of BD in which the patient underwent a below-knee amputation in his left leg. The written informed consent has been provided by the patient to have the case details and any accompanying images published and got an ethical code from Mashhad University of Medical Sciences (Ethical code: MUMS-941028). In the biopsy obtained from the sural nerve of the amputated limb, the vascular pathological changes in BD were observed in the vasa nervorum of the sural nerve, including inflammation of the vessel wall and also endothelial cell proliferation of the inner layer.
Case presentation
A 39-year-old Caucasian man reported in January 2018 complaining of experiencing burning pain at night in the first and second toes of his left foot. His past medical history included a history of cold sensitivity, foot claudication, and thrombophlebitis migrans for the 2 years prior to consulting with us. The patient was and had been a heavy smoker (two packs of cigarettes per day) for 23 years.
Upon physical examination, the left foot was colder than the right foot, and the left forefoot looked slightly oedematous, with a purple color. Also, superficial thrombophlebitis was observed on the left calf (Figure 1A,B). The pulses of the dorsalis pedis (DP) and tibialis posterior (TP) of both feet were absent. The popliteal pulses of both sides were also absent. The femoral pulses were intact.
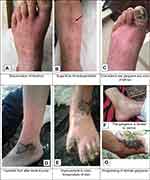 | Figure 1 The clinical manifestation of the patient with diagnosis of Buerger’s disease from early January 2018 until late May 2018. |
The laboratory tests revealed that he had normal fasting blood sugar and a normal lipid profile. The autoantibodies profile, including FANA, dsDNA, anti-phospholipid Ab (IgM, IgG), anti-cardiolipin Ab (IgM, IgG), and anti-Beta2Glyco (IgM, IgG), were evaluated and were within normal ranges.
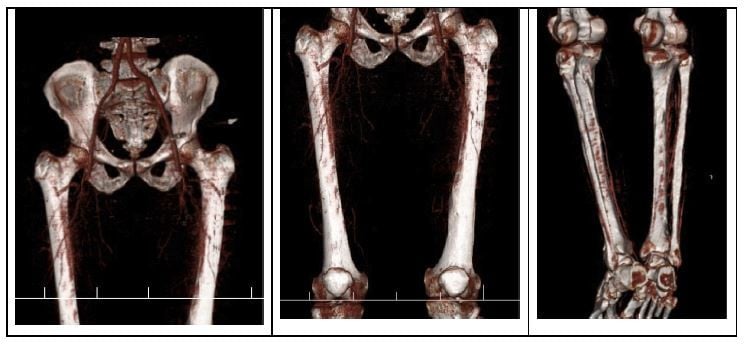
The patient underwent computed tomography (CT) angiography of his left limb (Figure 2). In the CT angiography, normal aorto-iliac and iliofemoral appearance were reported. However, a cutoff in the superficial femoral and a runoff in the middle and distal parts of the posterior tibial arteries (skip lesion) were observed.
The patient was prescribed 100 mg (mg) of cilostazol twice per day, with aspirin 81 mg daily, in addition to 75 mg of pregabalin twice per day. The patient was strongly encouraged to pursue smoking cessation.
In late January 2018, the patient reported again with gangrene and an ulcer of the left big toe, along with burning pain at rest (Figure 1C). The patient was admitted, and, for 2 weeks, he received Prostavasin. However, the toe gangrene having progressed, he underwent left toe amputation.
In March 2018, the patient reported again with a chief complaint of severe pain at rest of the left foot after his ankle blunt trauma. He was recommended for below-knee amputation after his cold and cyanotic foot was observed (Figure 1D). The patient refused amputation. Thus, he continued taking cilostazol and oral antibiotics, including clindamycin 500 mg and ofloxacin 300 mg along with a spray containing silver nanoparticles, which was added to his treatment for the management of the ulcer. He also stopped smoking completely, according to self-report. After a week, the color of the foot changed to purple, and the foot became warmer (Figure 1E). The pain also diminished, but for only 3 days. The gangrene started to progress (Figure 1F), but was limited to the derma of the forefoot skin (Figure 1G). Finally, in late May 2018, the patient underwent a left below-knee amputation.
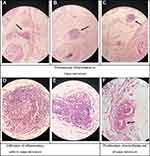
In the pathology study of the amputated leg, suppurative necrosis of the skin of the dorsal foot at the site of the amputation and at the first toe and third toe was reported. However, the necrosis was limited to the derma, whilst the infiltration of inflammatory cells was observed in the hypoderma and the connective tissue. Both acute and chronic inflammation in the thrombus and the layers of the vessel walls were observed (Figure 3). Perineural fibrosis was also reported. Notably, in the microscopic examination of the stump of the below-knee amputation, dilated arteries and arterioles were observed, a few of which had thrombosis inside. All factors being considered, the pathology report recommended a diagnosis of BD.
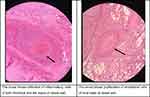 | Figure 3 H&E staining; ×10 objective lens. The biopsy obtained around the ulcer of the amputated limb of a patient with diagnosis of Buerger’s disease |
In the biopsy obtained from the sural nerve of the amputated limb, we found inflammation and also endothelial cell proliferation of the vasa nervorum without any inflammation in the fascicles of the sural nerve (Figure 4).
Discussion
BD patients usually present neurological symptoms from the early stages of the disease, including numbness, cold sensation, and allodynia as the disease progresses.3 Foot claudication during the second stage of the disease usually manifests as paraesthesia and tingling instead of a cramping pain.3 In the third stage of the disease, patients usually suffer from circadian rhythmic burning pain in the toes or fingers that begins late at night and ceases in the early morning.2 During this stage, among the patients who have intermittent claudication, 17% complain of burning pain instead of cramping after activity.3 Patients tend to bend their knees to diminish this pain. In the fourth stage of the disease, patients are usually reported to have burning pain at rest, with or without gangrene. Notably, the smoking habit influences all of the neurological symptoms of BD patients.3
Pain in the late stages of BD is very severe, almost resistant to opioid pain killers, and sometimes compels the patient to pursue major amputation. Therefore, pain management in BD patients is one of the most important and, at the same time, challenging issues, since its main etiology is not well understood.
Recently, inflammation and infiltration of neutrophils and lymphocytes in the sympathetic ganglia of BD patients who underwent sympathectomy as a pain management protocol were demonstrated.4 Sympathetic ganglia inflammation may explain the generalized vasoconstriction in BD patients and the early-stage neurological manifestation of BD, including cold sensation, numbness, and paraesthesia.4 However, it is not known if the pathological changes in the sympathetic ganglia are also responsible for the circadian rhythmic nature of the pain or its burning quality.
In this case report, we found that the vasa-nervorum of the sural nerve had the pathological changes usually observed in BD,5 including inflammation and proliferation of endothelial cells. Notably, the inflammation was limited to the vasa-nervorum and did not extend to the nerve fascicles. Therefore, the inflammation of the vasa-nervorum individually might be responsible for the pain characteristic of BD, such as localized pain, or the circadian rhythmic burning pain.
Conclusion
Our findings could provide a clue to taking the approach of managing pain in BD as if it were vasculitis neuropathy. In addition, our findings could indicate that BD is a systemic vasculitis of microcirculation and, hence, a different treatment approach for BD might be needed in addition to antithrombotic and vasodilator medications.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Fazeli B, Ravari H. Mechanisms of thrombosis, available treatments and management challenges presented by thromboangiitis obliterans. Curr Med Chem. 2015;22(16):1992–2001.
2. Fazeli B, Dadgar Moghadam M, Niroumand S. How to treat a patient with thromboangiitis obliterans: a systematic review. Ann Vasc Surg. 2018;49:219–228. doi:10.1016/j.avsg.2017.10.022
3. Fazeli B, Ravari H. A disease-specific activity score for thromboangiitis obliterans. Vascular. 2014;22(5):336–340. doi:10.1177/1708538113516315
4. Farzadnia M, Ravari H, Masoudian M, Valizadeh N, Fazeli B. Unexpected inflammation in the sympathetic ganglia in thromboangiitis obliterans. Int J Angiol. 2017;26(4):212–217. doi:10.1055/s-0037-1604410
5. Kobayashi M, Sugimoto M, Komori K. Endarteritis obliterans in the pathogenesis of Buerger’s disease from the pathological and immunohistochemical points of view. Circ J 2014;78(12):2819–2826.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.