Back to Journals » International Journal of Women's Health » Volume 14
Uterine Artery Rupture Caused by IUD Extraction: A Case Report
Received 16 November 2021
Accepted for publication 24 May 2022
Published 27 June 2022 Volume 2022:14 Pages 831—836
DOI https://doi.org/10.2147/IJWH.S345712
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Elie Al-Chaer
Nan Wang,1,2 HongZhan Sun1
1Department of Obstetrics and Gynecology, Taizhou People’s Hospital, Taizhou, Jiangsu Province, People’s Republic of China; 2Department of Graduate College, Dalian Medical University, Dalian, Liaoning Province, People’s Republic of China
Correspondence: HongZhan Sun, Email [email protected]
Abstract: It’s been 100 years since the first intrauterine device (IUD) was invented in Germany in 1920. IUDS are widely favored by women of childbearing age for their efficiency, convenience, cheapness, and variety of materials. According to incomplete statistics, about 26 million people worldwide will need to have their IUDS removed from 2015 to 2025 alone. With such a large case base, we have to pay attention to the safety of IUD removal. There are few international guidelines on IUD placement and removal. Therefore, this paper will report a case of uterine artery rupture caused by ring removal, and discuss the important steps of ring removal surgery, so as to enhance the attention of physicians to the standard and safety of ring removal and improve the quality of medical treatment.
Keywords: IUD removal, acute complications, case report
Introduction
Since the introduction of the intrauterine device (IUD) in the previous century, it has been a popular method of contraception given its high efficiency, convenience, low cost, and long-term effectiveness. It is a long-acting reversible contraceptive (LARC) and one of the most effective and safest contraceptive methods,1 and almost all women are suitable for IUDs.2 Copper-containing IUDs can last 10–12 years, while levonorgestrel-releasing IUDs last between 3 and 7 years.3 IUDs can also serve as good delivery carriers, delivering drugs precisely to the endometrium. When used as a progesterone carrier, it is effective in the treatment of endometrial cancer and abnormal uterine bleeding. The use of intrauterine IUDs has increased dramatically in the past 30 years, with 20% of women of childbearing age worldwide choosing IUD as a method of contraception, the article reported.4 Although previous data have shown that IUD-related procedures are easy and complications during IUD insertion and removal are extremely rare5,6 such a high volume of use makes it essential to be vigilant about the complications and potential risks associated with their installation and removal. Common complications include pain,7 induced abortion syndrome caused by cervical, traction tissue damage from improper manipulation, bleeding, and intrauterine infection. We report herein a case of uterine artery rupture caused by IUD removal and describe our experience and propose some ideas for IUD removal. The IUD was not removed for fear of secondary damage if it was forcibly removed again. The patient provided informed consent to publish this report.
Case Study
A 47-year-old woman was admitted to our hospital because of a vaginal bleeding for 4 h after IUD removal.
The patient had a regular 30-day menstruation cycle, with menstruation lasting approximately 7 days; the menstrual volume was medium. There was no history of dysmenorrhea. The patient’s last menstrual period was on 26 March, 2021, at which time she had profuse dark-colored dripping for 17 days, but no abdominal pain. She was admitted to a local hospital, where her bleeding was suspected to have been caused by the IUD. So she was administered oral hemostatic agents and anti-inflammatory drugs and plans to remove the IUD once the bleeding ceased. The patient refused to take hemostatic agents and anti-inflammatory drugs and underwent IUD removal directly in the local hospital. There was massive vaginal bleeding with red color and spray shape during the IUD removal. She was given gauze filling, misoprostol 2 tablets to the anus, and intravenous drip support treatment. The patient was referred to our hospital for further treatment and was admitted to the outpatient department for acute hemorrhagic anemia. In the course of the disease, the patient presents with acute symptoms and lethargy.
Medical History
The patient had no underlying disease or infection or endemic areas of life history. In 2011, lithotomy was performed to treat ureteral stones.
Personal and Family History
The patient had no personal or family history of other illnesses.
Physical Examination
Gynecological examination:
- Vulva: delivered;
- Vagina: smooth, bleeding heavily bright red;
- Cervix: mild erosion;
- Uterine body: anterior enlargement such as pregnant 50 days of size, gentle tenderness;
- Attachments: bilateral attachments are not abnormal.
Laboratory examination:
- The hemoglobin (Hb) level was 90 g/L; red blood cell (RBC) count was 3.63×1012/L; hematocrit (HCT) was 29.0%; mean corpuscular hemoglobin (MCH) was 24.8 pg; and mean corpusular hemoglobin concentration (MCHC) was 310 g/L.
Imaging
An ultrasound was performed in our hospital. The uterus measured 65×44×45 mm and showed normal uterine morphology. We observed homogeneous endometrial echogenicity, endometrial line in the middle, endometrial line thickness of 6 mm, and no obvious abnormal blood flow on color Doppler flow imaging (CDFI). A strong echo of the IUD could be seen be seen in the uterine cavity, and the IUD position was normal. The lower part of the uterine cavity was separated for 9 mm. No abnormal echo was observed in the left accessory, and a liquid dark area of 23×20 mm was observed in the right accessory, with a clear boundary and fair ultrasound transmission. The pelvic cavity was characterized by a dark, fluid-filled area measuring 15 mm anterior and posterior diameter.
The final diagnosis was uterine artery rupture.
Interventions
After admission, according to the medical history and physical examination, ECG monitoring was immediately performed, two venous routes were established, hemostasis was performed by intravenous oxytocin drip, and a large gauze used to fill the vagina and apply pressure to stop the bleeding. 20 minutes later a repeat gynecological examination showed considerable bright red bleeding; hence, the big gauze was removed and replaced with five small gauze stuffing pieces. Because the estimated blood loss was nearly 1500 mL, cross matching was completed and 1.5 units of leucocyte-poor irradiated red blood cells was injected. In the meantime, the preoperative preparation was completed and UAE was performed under general anesthesia. Intraoperative angiography of the left common iliac artery showed significant extravasation of contrast media in the uterine artery. After the introduction of 3F microcatheterization, gelatin sponge embolization was performed, but the effect was not good. Then three COOK (Cook Medical Holdings LLC, USA) microspring coils with diameter of 3 mm were embolized, and three gelatin sponge strips with a diameter of 1×1×10 mm were re-embolized. The operation was completed. The results of preoperative angiography are shown in Figure 1, and those of postoperative angiography are shown in Figure 2. The equipment for interventional surgery was from SIEMENS Healthineers Artis Q ceiling. Correction of anemia and anti-inflammatory therapy were provided after surgery. Blood routine examination postoperative first day showed an RBC count of 2.27×1012/L and Hb level of 61 g/L. Irradiated white erythrocytes (2 units) was again given. On day 5 of the operation, the patient’s Hb increased to 80 g/L. Because she had no further complaints or discomfort, she was discharged.
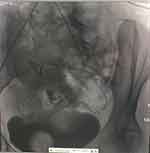 |
Figure 1 Preoperative angiography results. There is marked extravasation of uterine artery contrast. |
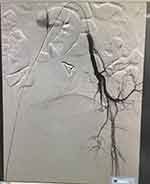 |
Figure 2 This is a radiograph after surgery. No obvious contrast extravasation was observed, indicating successful embolization. |
Follow-Up
The patient’s postoperative condition was unremarkable.
Discussion
With the wide acceptance and application of IUDs, the number of IUD removals has also increased significantly. This has led to concerns over its safety. To ensure patient safety and reduce operational complications, it is paramount to have a set of standardized and scientific surgical procedures to reduce the incidence of IUD removal-related complications. However, there is no comprehensive guide for a standardized protocol for IUD removal. Hence, we reported this case of uterine artery rupture after IUD removal and summarized our experience, hoping to improve the development of this technology. The indications for removal of the contraceptive IUD are as follows: 1) the contraceptive IUD reaches reached its expiry; 2) the contraceptive method has changed; and/or 3) the clinician thinks that removal is necessary. Contraindications are: severe pelvic inflammation. In women of childbearing age, the time to removal the IUD should be 3–7 days after menstruation has ceased. At this time, the endometrium is relatively thin, which can reduce the possibility of bleeding and endometrial malignancy, and also prevent pregnancy. As women get older, ovarian function declines, vaginal and cervical epithelium atrophy, elasticity is reduced or even disappears, the uterus shrinks and becomes smaller, the cervix stiffens and loses elasticity,8 the tissue becomes fragile, and consequently, IUD removal becomes more difficult. The traditional time for perimenopausal women is 6 months to 1 year after menopause. However, Mengxia showed that uterine shrinkage occurs before menopause. Therefore, to prevent uterine deformation, the IUD should be removed after perimenopausal stability, rather than after menopause.9 The IUD is more difficult to remove in postmenopausal women than in premenopausal women, and a short course of estrogen replacement therapy is recommended before removal of the IUD if it is removed long after menopause, which increases the risk of surgery.10 Based on our clinical experience, women should removal IUDs within 1 year when they reach menopause. It is a more appropriate time to avoid pregnancy and has relatively few operational risks. Cervical conditions should be evaluated before IUD removal. For patients with poor cervical conditions, 50 µg of vaginal misoprostol can be administered the night before surgery to reduce intraoperative pain and the incidence of cervical tears. Use of tentacles during surgery. Misoprostol does not alter the angle of the cervical canal, but works by softening the cervix, and the dose of misoprostol used has few side effects and does not overdilate the cervix.11,12 Before IUD removal, a detailed medical history should be taken to understand basic information regarding the patient’s IUD (eg, IUD types). Zhao’s doctoral thesis showed that among round, V-shaped, uterine, T-shaped, and other types of IUDs, the difficulty rate of round IUDs was the highest, followed by V-shaped ones; uterine IUDs were the easiest to remove. The differences among various types was statistically significant (P<0.05).13 At the same time, imaging examination should be clarified to understand the intrauterine state of the contraceptive IUD, determine whether the position of the contraceptive IUD is normal, and whether the contraceptive IUD is incarcerated. Bimanual examination should be performed to further understand the cervical condition and uterine curvature. Briefly, 5 mL of 2% lidocaine can be injected intrauterine before the operation. Experiments have proven that local anesthesia has a better analgesic effect than placebo during and after the operation and can greatly improve patient compliance.14 Strictly follow the principle of asepsis during IUD removal operation. IUD removal should follow the uterine curvature and position of the contraceptive device, and the device should not be forcibly pulled when it is blocked. Applying enough traction on the cervix to straighten the horizontal axis of the uterus will make it easier for the IUD to pass in and out of the uterine cavity.15 If the IUD cannot be removed, ultrasound-guided removal can be performed. When these methods are unsuccessful, hysteroscopy can be attempted; there are no postoperative complications after hysteroscopic removal of the IUDs.16 When induced abortion syndrome occurs, the operation needs to be stopped timely. It is estimated that between 2015 and 2025, about 26 million postmenopausal women required intrauterine IUD removal.17 Such a large base of operation to obstetrics and gynecology workers is an unprecedented challenge.
In recent years, many patients have shared the experience of self-removal of IUDs, which has a certain survivor bias.18 Although the uterine cavity and vagina are the natural passageways of human beings, the fact that an IUD is still a medical device should not be overlooked; moreover, its position inside the uterine cavity and its removal are not as simple and harmless as using a female condom. A study found that while endometrium doctors and patients benefit from self-removal of an IUD, some doctors worry about hasty or forced removal, and patients worry about the safety.19 So we need to be both rigorous and optimistic about IUD removal. Methods to improve the convenience of IUDs and ensure its safe removal, reduce complications, and eliminate medical accidents has gradually become a new challenge for gynecologists.
Patient Perspective
Informed consent of the patient was obtained.
Data Sharing Statement
This case report is from the Department of Obstetrics and Gynecology, Taizhou People’s Hospital. The first author can provide additional information upon reasonable request.
Ethics Approval and Informed Consent
This case report was written and published with the approval of Taizhou People’s Hospital review board and ethics committee.
Consent for Publication
Written informed consent has been provided by the patient to have the case details and any accompanying images published.
Acknowledgments
The author would like to thank the patient and her family for their support. Thanks to Charlesworth for the great edit. Thanks for Pan Shang’s help in my article writing and revision. According to GPP3 Guidelines, I can only express my thanks to Pan Shang in the Acknowledgements. Pan Shang is an excellent doctor. She has superb medical skills and all the noble character. No doubt, her noble example inspired me. I will always remember that time.
Disclosure
The authors have no conflicts of interest to declare.
References
1. Abu – Zaid A, Alshahrani MS, Albezrah NA, et al. Vaginal dinoprostone versus placebo for pain relief during intrauterine device insertion: a systematic review and meta – analysis of randomised controlled trials. Eur J Contracept Reprod Health Care. 2021;26(5):357–366. doi:10.1080/13625187.2021.1891411
2. Philliber AE, Hirsch H, Brindis CD, Turner R, Philliber S. The use of ACOG guidelines: perceived contraindications to IUD and implant use among family planning providers. Matern Child Health J. 2017;21(9):1706–1712. doi:10.1007/s10995-017-2320-1
3. Castillo K, Zambrano K, Barba D, et al. Long – acting reversible contraceptives effects in abnormal uterine bleeding, a review of the physiology and management. Eur J Obstet Gynecol Reprod Biol. 2022;270:231–238. doi:10.1016/j.ejogrb.2022.01.020
4. Vitale SG, Di Spiezio Sardo A, Riemma G, De Franciscis P, Alonso Pacheco L, Carugno J. In – office hysteroscopic removal of retained or fragmented intrauterine device without anesthesia: a cross – sectional analysis of an international survey. Updates Surg. 2022. doi:10.1007/s13304-022-01246-0
5. Long S, Colson L. Intrauterine device insertion and removal. Prim Care. 2021;48(4):531–544. doi:10.1016/j.pop.2021.07.001
6. Wakrim S, Lahlou L. Spontaneously expelled IUD and missing fragments in the uterine cavity. Radiol Case Rep. 2020;15(9):1654–1656. doi:10.1016/j.radcr.2020.07.005
7. O’ Donohue S. The ripples of trauma caused by severe pain during IUD procedures. BMJ. 2021;374:n1910. doi:10.1136/bmj.n1910
8. Simó Alari F, Gutierrez I. Intrauterine device extraction through laparoscopic hysterotomy. Eur J Contracept Reprod Health Care. 2021;26(3):261–263. doi:10.1080/13625187.2020.1862080
9. MengXia LI. Factors of difficulty in ring removal in postmenopausal women. Pract Electron J Gynecol Endocrinol. 2019;6(34):35–42.
10. Serfaty D, Bénézech JP, Heckel S, De Reilhac P. Consensus of best practice in intrauterine contraception in France. Eur J Contracept Reprod Health Care. 2019;24(4):305–313. doi:10.1080/13625187.2019.1625325
11. Rasheedy R, Tamara TF, Allam IS, et al. A Vaginal misoprostol before copper IUD insertion after previous insertion failure: a double – blind, placebo – controlled, parallel – group, randomised clinical trial. Eur J Contracept Reprod Health Care. 2019;24(3):222–226. doi:10.1080/13625187.2019.1610871
12. New EP, Sarkar P, Mikhail E, Plosker S, Imudia AN. Use of low dose vaginal misoprostol in office hysteroscopy: a pre – post interventional study. J Obstet Gynaecol. 2021;41(6):972–976. doi:10.1080/01443615.2020.1820968
13. Zhao X. 《Technical guide for the removal of intrauterine contraceptive devices during menopause》master. Peking Union Medical College; 2019.
14. Vidal F, Paret L, Linet T, Tanguy le Gac Y, Guerby P. Contraception intra-utérine. RPC Contraception CNGOF [Intrauterine contraception: CNGOF contraception guidelines]. Gynecol Obstet Fertil Senol. 2018;46(12):806–822. French. doi:10.1016/j.gofs.2018.10.004
15. Mhlanga FG, Balkus JE, Singh D, et al. Feasibility and safety of IUD insertion by mid – level providers in Sub – Saharan Africa. Int Perspect Sex Reprod Health. 2019;45:61–69. doi:10.1363/45e8019
16. Sarver J, Cregan M, Cain D. Fractured copper intrauterine device (IUD) retained in the uterine wall leading to hysterectomy: a case report. Case Rep Womens Health. 2021;29:e00287. doi:10.1016/j.crwh.2021.e00287
17. Zhang YY. Expert consensus on removal of intrauterine menopausal iUD under anesthesia and analgesia. Chin J Pract Gynecol Obstetr. 2018;34(09):1024–1027.
18. Stimmel S, Hudson SV, Gold M, Amico JR. Exploring the experience of IUD self – removal in the United States through posts on internet forums. Contraception. 2022;106:34–38. doi:10.1016/j.contraception.2021.10.013
19. Amico JR, Bennett AH, Karasz A, Gold M. Taking the provider “out of the loop”: ‘patients and physicians’ perspectives about IUD self – removal. Contraception. 2018;98(4):288–291. doi:10.1016/j.contraception.2018.05.021
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
