Back to Journals » Vascular Health and Risk Management » Volume 10
Update on ranolazine in the management of angina
Authors Codolosa JN, Acharjee S, Figueredo V
Received 10 April 2014
Accepted for publication 12 May 2014
Published 24 June 2014 Volume 2014:10 Pages 353—362
DOI https://doi.org/10.2147/VHRM.S40477
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
J Nicolás Codolosa,1 Subroto Acharjee,1 Vincent M Figueredo1,2
1Einstein Center for Heart and Vascular Health, Einstein Medical Center, 2Jefferson Medical College, Philadelphia, PA, USA
Abstract: Mortality rates attributable to coronary heart disease have declined in recent years, possibly related to changes in clinical presentation patterns and use of proven secondary prevention strategies. Chronic stable angina (CSA) remains prevalent, and the goal of treatment is control of symptoms and reduction in cardiovascular events. Ranolazine is a selective inhibitor of the late sodium current in myocytes with anti-ischemic and metabolic properties. It was approved by the US Food and Drug Administration in 2006 for use in patients with CSA. Multiple, randomized, placebo-controlled trials have shown that ranolazine improves functional capacity and decreases anginal episodes in CSA patients, despite a lack of a significant hemodynamic effect. Ranolazine did not improve cardiovascular mortality or affect incidence of myocardial infarction in the MERLIN (Metabolic Efficiency with Ranolazine for Less Ischemia in Non-ST-Elevation Acute Coronary Syndrome)-TIMI (Thrombolysis In Myocardial Infarction) 36 trial, but significantly decreased the incidence of recurrent angina. More recently, ranolazine has been shown to have beneficial and potent antiarrhythmic effects, both on supraventricular and ventricular tachyarrhythmias, largely due to its inhibition of the late sodium current. Randomized controlled trials testing these effects are underway. Lastly, ranolazine appears to be cost-effective due to its ability to decrease angina-related hospitalizations and improve quality of life.
Keywords: ranolazine, chronic stable angina, coronary artery disease
Introduction
Mortality rates attributable to cardiovascular disease have declined in recent decades, but the burden of disease remains high. From 1999 to 2009, the relative rate of death attributable to cardiovascular disease declined by 32.7%. However, in 2009, cardiovascular disease still accounted for 32.3% of all deaths, or one in every three deaths in the USA.1
According to the most recent data published by the American Heart Association, coronary heart disease (CHD) alone caused approximately one of every six deaths in the USA in 2009. On the basis of data from the National Health and Nutrition Examination Survey 2007–2010, an estimated 15.4 million Americans have CHD, with a prevalence of 6.4% in the US population. From 1999 to 2009, the annual death rate attributable to CHD declined by 40.3% and the actual number of deaths declined by 27.1%.2 The decline in CHD deaths partly reflects a shift in the pattern of clinical presentation of acute myocardial infarction. In the past decade, there has been a marked decline in ST-segment elevation myocardial infarction.3 Further, after 1980 and 2000 data were compared, it was estimated that approximately 47% of the decrease in CHD deaths was attributable to treatments, including secondary preventive therapies, initial treatment after acute myocardial infarction, and revascularization for chronic angina.4
Angina pectoris, ie, chest pain or discomfort that results from insufficient blood flow to the heart muscle, was accurately reported by William Heberden in 1772.5 Chronic stable angina (CSA) is usually described as predictable chest pain on exertion or when under mental or emotional stress. Only 18% of acute coronary syndromes are preceded by longstanding CSA. Estimates from the National Health and Nutrition Examination Survey 2007–2010 show a prevalence of CSA of 7.8 million people (3.2% of the US population). The annual rates per 1,000 population of new episodes of CSA for nonblack men are 28.3 for those aged 65–74 years, 36.3 for those aged 75–84 years, and 33.0 for those aged ≥85 years. For nonblack women in the same age groups, the rates are 14.1, 20.0, and 22.9, respectively. For black men, the rates are 22.4, 33.8, and 39.5, and for black women, the rates are 15.3, 23.6, and 35.9, respectively.6
The two aims of pharmacological management for CSA are to obtain relief of symptoms and to prevent cardiovascular events. These are usually achieved by pharmacological or lifestyle interventions that reduce plaque progression, stabilize plaque by reducing inflammation, and prevent thrombosis, should plaque rupture or erosion occur.
First-line treatment is indicated with beta-blockers and/or calcium channel blockers to control heart rate and symptoms, as well as nitrates for symptomatic relief. Prevention of cardiovascular events is usually accomplished by use of antiplatelet agents, statins, and possibly angiotensin-converting enzyme inhibitors or angiotensin II receptor antagonists. Coronary revascularization generally improves survival among certain subgroups of patients, particularly those with severe left main coronary stenosis.7 Ranolazine is a novel agent with anti-ischemic and metabolic properties, and was approved by the US Food and Drug Administration in 2006 for the treatment of patients with CSA.8
Overview of pharmacology of ranolazine
Ranolazine is a selective inhibitor of the late sodium current (INaL) in cardiomyocytes and has anti-ischemic and metabolic properties. Its extended-release (ER) form was approved in doses of 500 mg twice daily and 1,000 mg twice daily, based on clinical symptoms. After oral administration, peak plasma concentrations of ranolazine are reached between 2 and 5 hours. Ranolazine is extensively metabolized in the gut and liver, and its absorption is highly variable. The elimination half-life of ranolazine is 1.4–1.9 hours, but is apparently prolonged, on average, to 7 hours for the ER formulation as a result of extended absorption (flip-flop kinetics). Steady state is generally achieved within 3 days of twice-daily dosing.9,10
The drug is metabolized mainly by cytochrome P450 (CYP) 3A4 and, to a lesser extent, by CYP2D6. This occurs rapidly and extensively in the liver and intestine; less than 5% is excreted unchanged in urine and feces.9 Ranolazine is a weak inhibitor of CYP3A4 and CYP2D6. The interactions of ranolazine with inhibitors and substrates of the CYP3 family have been studied.11 Ketoconazole was associated with the largest effect, increasing the area under the plasma concentration time curve of ranolazine by 3.9-fold, followed by diltiazem (1.5-fold), whereas simvastatin was too weak an inhibitor to significantly affect the pharmacokinetic parameters of ranolazine. Further, ranolazine is a substrate for and transported by P-glycoprotein. Verapamil is a potent P-glycoprotein inhibitor and also inhibits CYP3A4, and can lead to a 2–3-fold increase in the area under plasma concentration time curve for ranolazine.9 Concomitant use of ranolazine and drugs transported by P-glycoprotein (such as digoxin) results in increased exposure to the latter and the dose may have to be adjusted.
Ranolazine is contraindicated in patients receiving strong CYP3A4 inhibitors or inducers and those with liver cirrhosis. It is also recommended to limit the dose to 500 mg daily in those receiving moderate CYP3A4 inhibitors, (diltiazem, verapamil, erythromycin), and to limit the dose of simvastatin to 20 mg when used in combination with ranolazine.10
Inhibition of the rapidly activating delayed rectifier potassium current (IKr) by ranolazine explains the effect of the drug to prolong ventricular and atrial action potential duration, as well as the small QTc interval prolongation observed in clinical trials. Despite this effect, ranolazine does not induce torsades de pointes and possibly suppresses long QT-related ventricular arrhythmias, as shown in long QT experimental trials.12,13
The antianginal mechanism of action of ranolazine has not been fully elucidated. Initial studies proposed myocardial metabolic modulation as the predominant mechanism of action, with minimal or no effect on heart rate, coronary blood flow, and systemic arterial blood pressure.14 In the healthy heart, fatty acids are the primary fuel source for the generation of adenosine triphosphate (ATP).15 During ischemia, fatty acid and pyruvate oxidation both decrease as glycolysis becomes the predominant route for ATP production. Glycolytic regeneration of ATP leads to intracellular acidosis, intracellular calcium overload, and contractile dysfunction.16 Results of early studies suggested that ranolazine improved myocardial energy metabolism by reducing fatty acid oxidation and increasing glucose oxidation.17 However, these effects require relatively high concentrations of ranolazine, whereas improvement in cardiac function and clinical status occur in the presence of much lower concentrations.18
In recent years, the research focus has shifted toward INaL as a contributor to the pathogenesis of angina.19,20 Activation of cardiac myocytes in normal hearts leads to rapid cell depolarization (the upstroke of the action potential) through brief opening of membrane sodium channels (INa). Most sodium channels are inactivated within a few milliseconds, but a few may continue to open and close spontaneously during the action potential plateau for reasons that are not understood. This current has been referred to as INaL. Under normal conditions, INaL constitutes only 1% of peak INa. Voltage-dependent calcium channels then open and initiate a large outflow of sarcoplasmic calcium into the cytoplasm, initiating myocardial contraction through the coupling of actin and myosin filaments.19–21
Several pathological conditions, including ischemia and heart failure, can significantly increase the late component of the sodium channel current.22,23 This may be followed by increases in myocyte membrane sodium calcium exchange, resulting intracellular increases of calcium, and excessive calcium loading of the sarcoplasmic reticulum. Calcium overload of myocardial cells is associated with electrical instability, increased diastolic and reduced systolic force generation, and an increase in oxygen consumption.24 The increase in diastolic tension may cause compression of the vascular space and further reduction of blood flow and oxygen delivery (Figure 1).25–27
  | Figure 1 Increase in intracellular sodium concentration ([Na+]i) in pathological conditions linked to imbalances between oxygen supply and demand causes calcium entry through the Na+/Ca2+ exchanger (NCX). |
Ranolazine has been shown to selectively inhibit INaL in canine and pig models without affecting the fast sodium current responsible for the upstroke of the action potential.12–28 The effect of ranolazine on INaL is more pronounced in ischemic or failing myocytes in which the current is amplified. Therefore, ranolazine has the potential to disrupt the consequences of myocardial ischemia by reducing excess sodium influx, reducing calcium overload, and the noxious increase in left ventricular wall tension.
Further, ranolazine significantly inhibits the rapidly activating component of the delayed rectifier or IKr. Although inhibition of IKr by ranolazine prolongs the action potential duration, inhibition of INaL and the late ICa2+ (which is also affected) shorten the duration of the action potential such that the net effect on its duration, or QTc, is usually modest (Figure 2).12 Experimental and clinical studies have shown that ranolazine has antiarrhythmic effects in both the ventricles and atria.13
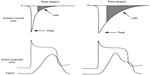  | Figure 2 Relation between peak and late sodium current and ventricular action potential (AP) and contraction. |
Use of ranolazine in chronic stable angina
The efficacy of ranolazine in CSA patients has been demonstrated in several randomized trials. Early trials used the immediate-release formulation administered three times daily and failed to show superior antianginal and anti-ischemic effects over placebo.29,30 Subsequently, the ER version was tested in several clinical studies, the major results of which are summarized in Table 1.
MARISA (Monotherapy Assessment of Ranolazine In Stable Angina) was a randomized, multicenter, double-blind, dose-finding study utilizing ranolazine ER as monotherapy for angina.31 A total of 191 patients with documented coronary artery disease and exercise-limiting angina were enrolled. Existing antianginal treatment (with the exception of sublingual nitroglycerin) was discontinued prior to randomization. Ranolazine ER at doses of 500 mg, 1,000 mg, and 1,500 mg or placebo, was administered twice daily for one week in this four-period crossover study. Exercise treadmill tests were performed at the end of each study period to coincide with peak (4 hours) and trough (12 hours) plasma levels. Compared with placebo, ranolazine demonstrated significant dose-dependent improvements in total exercise duration, time to angina, and time to 1 mm ST-segment depression at both peak and trough levels, with minimal effects on heart rate and blood pressure. Safety of the drug was also studied in this trial, with some patients continuing in an open-label, observational follow-on study. One-year survival in this group was in line with the predicted rate.
Overall, the drug was well tolerated, with less than 8% of patients discontinuing ranolazine because of adverse effects. The majority of these patients were on the 1,500 mg dose, which was related to higher rates of dizziness, nausea, asthenia, and constipation. Dose-related increases in the corrected QT interval were also noted, although this was not a reason for drug discontinuation. Thus, the MARISA study demonstrated that ranolazine was effective and safe as monotherapy for angina. The optimum risk to benefit ratio was noted in the lower doses, with the authors suggesting starting at 500 mg and titrating upwards to 750 mg or 1,000 mg twice daily as needed to achieve angina control. In contemporary clinical practice, ranolazine is not used as the sole antianginal agent, making the above findings less applicable to current patients.
In contrast with MARISA, the CARISA (Combination Assessment of Ranolazine In Stable Angina) trial was intended to evaluate the efficacy of ranolazine ER when used in conjunction with standard doses of typical antianginal drugs (atenolol 50 mg/day, diltiazem 180 mg/day, or amlodipine 5 mg/day), prescribed based on physician discretion.32 This was a much larger multicenter international trial, randomizing 823 patients with severe chronic angina to receive ranolazine ER 750 mg or 1,000 mg twice daily, or placebo (with background antianginal therapy for all patients) for 12 weeks in a three-group, parallel, double-blind design. Treadmill exercise testing at peak and trough plasma levels was performed after 2, 6 (trough only), and 12 weeks of treatment. Increase in trough symptom-limited exercise duration (the primary endpoint) was significantly greater in both ranolazine groups compared with placebo. The time to onset of angina at trough and peak plasma concentrations was also greater with ranolazine. The time to ≥1 cm ST-segment depression at peak concentration was longer than with placebo in both ranolazine arms, although the same parameter at trough concentrations did not significantly differ between the two doses of ranolazine tested. Frequency of angina attacks and nitroglycerin use were significantly reduced in both ranolazine groups, and like in the MARISA trial, these benefits were noted in the absence of any significant hemodynamic effects. Importantly, treatment with other antianginals did not blunt the response to ranolazine.
Several subgroup analyses of this trial were reported. Among 258 patients considered to be treated with maximally tolerated doses of beta-blockers or calcium channel blockers (based on the parameters of systolic blood pressure ≤100 mmHg, a resting heart rate ≤60 beats per minute, and/or an electrocardiographic PR interval ≥200 msec) ranolazine improved exercise tolerance at both trough and peak concentrations, along with significant lengthening in the times to angina onset and ≥1 mm ST-segment depression at peak but not trough concentrations. Consistent significant reductions were also noted in frequency of angina and nitroglycerin consumption.33 Similarly, when patients with diabetes (n=189) were studied separately, there was no significant interaction with respect to the benefits of ranolazine on symptom-limited or ischemia-limited exercise duration, angina frequency, or nitroglycerin use.34 Additionally, in 237 patients with New York Heart Association class I or II heart failure, the effects of ranolazine ER on improving exercise tolerance prevailed.35
Overall, treatment was well tolerated at both doses. Adverse events were reported in 31.2% and 32.7% of ranolazine ER 750 mg and 1,000 mg recipients, respectively, compared with 26.4% of placebo-treated patients. Constipation, dizziness, nausea, and asthenia were most commonly reported. Five patients in the ranolazine ER 1,000 mg arm experienced syncope without evident ventricular tachyarrhythmia, although significant dose-related increases in the corrected QT interval were seen with both doses of ranolazine. Among the 750 ranolazine-treated patients who continued on to an open-label, observational follow-on study, survival rates of 98.4% and 95.9% after 1 or 2 years of follow-up, respectively, were reported, and were similar to expected rates. Thus, there was no long-term signal of significant harm.
The ROLE (Ranolazine Open Label Experience) program included 746 patients who completed the MARISA and CARISA trials and consented to participate in an open-label, 6-year follow-on extension.36 After 2.8 years of follow-up, 9.7% discontinued ranolazine due to adverse events, with age ≥64 years being the only predictor of drug discontinuation. Mean QTc interval was prolonged by a statistically significant 2.4 msec, although this did not lead to any discontinuations or reported torsades de pointes. Annual mortality was reported at 2.8% compared with >5% as predicted by the Duke Treadmill Score. Thus, long-term therapy with ranolazine appeared to be well tolerated, with no indication of a higher than predicted mortality in this population with stable ischemic heart disease.
The objective of the ERICA (Efficacy of Ranolazine In Chronic Angina) trial was to specifically examine the antianginal efficacy of ranolazine when used with maximum recommended doses of amlodipine.37 In this study, 565 patients with three or more episodes of chronic stable angina per week, despite receiving amlodipine 10 mg/day, were randomized to additional treatment with ranolazine ER 1,000 mg or placebo twice daily for 6 weeks. Patients were allowed to be on long-acting nitrates, but not on beta-blockers. Compared with placebo, ranolazine significantly reduced the weekly average frequency of self-reported angina episodes (primary endpoint) and nitroglycerin use. The therapeutic effect appeared to be more prominent in those with more severe baseline angina. In particular, significant reductions in nitroglycerin use and Seattle Angina Questionnaire angina frequency were observed for patients with more than 4.5 anginal episodes per week, but only in angina frequency for those with less frequent baseline symptoms.
Overall, ranolazine remained well tolerated and did not cause any notable change in hemodynamic parameters. Compared with 35.3% of placebo recipients, 39.9% of ranolazine ER recipients reported adverse events, mostly of mild to moderate severity. Again, constipation was commonly reported, in addition to peripheral edema, dizziness, nausea, and headache. Therefore, the ERICA trial confirmed the incremental antianginal effect of ranolazine in addition to maximal-dose amlodipine.
Data from CARISA and ERICA were pooled to compare endpoints between patients aged ≥70 years (n=363) and those <70 years of age (n=1,024). The efficacy of ranolazine remained similar in both age groups, although nonserious adverse effects were more common in the elderly.38 Patient information from two additional trials, MARISA and an early trial comparing ranolazine to atenolol,30,31 was added to study outcomes among women. Overall, data from these four trials demonstrated that the favorable effects of ranolazine were somewhat diminished in women, with less improvement than men in exercise testing, but similar improvements in angina frequency, nitroglycerin consumption, and Seattle Angina Questionnaire angina frequency scores.39
Recently, the TERISA (Type 2 Diabetes Evaluation of Ranolazine in Subjects With Chronic Stable Angina) trial was completed to evaluate the performance of ranolazine in patients with diabetes, coronary artery disease, and CSA.40 After a single-blind, 4-week placebo run-in, 949 patients (on one or two antianginal medications) were randomized to treatment with ranolazine ER 1,000 mg twice daily or placebo. After 8 weeks, the average weekly number of angina episodes (primary endpoint) was significantly reduced in the ranolazine-treated arm compared with placebo, as was the need for sublingual nitroglycerin. Therefore, ranolazine was proven to retain significant benefits among diabetic patients, although the overall magnitude of effects was somewhat diminished compared with previous studies.
Ranolazine in acute coronary syndromes
The randomized, double-blind, multinational MERLIN (Metabolic Efficiency with Ranolazine for Less Ischemia in Non-ST-Elevation Acute Coronary Syndrome)-TIMI (Thrombolysis In Myocardial Infarction) 36 trial was designed to examine the safety and efficacy of ranolazine in an acute coronary syndrome population already receiving conventional treatment.41 In all, 6,560 patients were randomized within 48 hours of angina onset to additionally receive ranolazine (intravenously for the first 96 hours and then orally 1,000 mg ER twice daily), or placebo for a median of 348 days. Although no beneficial effect of ranolazine was noted on the composite of cardiovascular death, myocardial infarction, or recurrent ischemia (primary efficacy endpoint), the incidence of recurrent ischemia alone was significantly lower in the ranolazine group, as was the rate of worsening angina requiring intensification of medical therapy.42 Mean Seattle Angina Questionnaire scores for angina frequency (at 4 and 12 months), quality of life, and treatment satisfaction (at 12 months) were significantly improved with ranolazine ER than with placebo.43 Similar to the overall trial results, the risk of recurrent ischemia, worsening angina, and intensification of antianginal therapy were all significantly reduced in patients with prior CSA. In the subgroup of CSA patients evaluated by exercise treadmill testing at 8 months, the mean total exercise duration and times to onset of angina or ≥1 mm ST-segment depression were significantly greater with ranolazine ER, consistent with prior studies.
Improvements in several domains of patient-reported angina-related health status and quality of life were seen with ranolazine ER in these CSA patients. Of note, among patients without CSA prior to their acute event, no such significant treatment effects were reported. Another subgroup analysis in those who underwent percutaneous coronary intervention within 30 days of randomization, showed a 27% reduction in the composite primary endpoint, driven by significant reductions in recurrent ischemia and cardiovascular death. Recurrent ischemia and recurrent ischemia requiring repeat revascularization were also significantly reduced.
Based on these findings, RIVER-PCI (Ranolazine for Incomplete Vessel Revascularization Post–Percutaneous Coronary Intervention), a randomized, double-blind, Phase III, international study of approximately 2,600 participants, is currently ongoing to evaluate the efficacy of ranolazine in reducing ischemia-driven revascularization or hospitalization in patients with CSA and incomplete revascularization after percutaneous coronary intervention.44
As the MERLIN-TIMI 36 trial enrolled a higher risk cohort in comparison with previous trials among stable ischemic heart disease patients, special attention was paid to the safety of ranolazine. Overall, all-cause mortality (at 6, 12, or 18 months) was similar in the ranolazine and placebo groups, as was the incidence of death or any cardiovascular hospitalization, or sudden cardiac death. The incidence of symptomatic documented arrhythmias over the duration of the study was also similar, although clinically significant arrhythmias were observed less frequently during Holter monitoring in the first 7 days with ranolazine group versus placebo. Adverse events leading to discontinuation of medication were significantly more common in the ranolazine group (8.1% versus 4.1% for placebo), with dizziness, nausea, and constipation being most commonly reported. Unfortunately, there are not much data regarding the safety of ranolazine in special populations, especially the elderly. In summary, no significant benefit of ranolazine compared with placebo was seen in this trial of patients with non-ST-segment elevation acute coronary syndrome with respect to the primary efficacy endpoints, although ranolazine appeared to be relatively safe and well tolerated.
Role of ranolazine in patients with diastolic dysfunction
Considering its effect on the INaL, ranolazine could potentially affect diastolic function by promoting calcium extrusion through the sodium calcium exchanger, thus improving diastolic tension and relaxation. A study by Figueredo et al assessed echocardiographic parameters of systolic and diastolic function before and 2 months after initiation of ranolazine in 22 patients with CSA. There were significant improvements in global left ventricular function, as assessed by myocardial performance index and diastolic function in patients who received ranolazine.45 More recently, the RALI-DHF (RAnoLazIne for the Treatment of Diastolic Heart Failure) study was published.46 This was a small, prospective, randomized, double-blind, placebo-controlled, proof-of-concept study of 20 patients with symptoms of congestive heart failure, preserved left ventricular ejection fraction (>45%), and laboratory, echocardiographic, or hemodynamic evidence of elevated filling pressures. Treatment consisted of intravenous infusion of ranolazine for 24 hours, followed by oral treatment for 13 days versus placebo. Left ventricular end-diastolic pressure and pulmonary capillary wedge pressure decreased in the ranolazine group, but not in the placebo group. After 14 days of treatment, no significant changes were observed in echocardiographic or cardiopulmonary exercise test parameters. Thus, this study failed to provide evidence of any substantial expected clinical benefit from ranolazine in this specific population.
Ranolazine as an antiarrhythmic agent
The fairly selective blocking effect of the late sodium current led to the investigation of ranolazine as a novel antiarrhythmic agent.13–47 In a secondary analysis of the MERLIN-TIMI 36 trial, ranolazine was associated with significant reductions in supraventricular tachyarrhythmias (P<0.001) as well as a 30% reduction in new-onset atrial fibrillation (P<0.08).48 Small studies have suggested a role for ranolazine as a potential treatment for atrial fibrillation, facilitating electrical cardioversion in cardioversion-resistant patients, and as a potential “pill-in-the-pocket” approach.49–51 Additionally, one study showed a potential synergistic effect between ranolazine and dronedarone for suppressing arrhythmia.52 In an observational study by Miles et al, ranolazine was found to be more effective than amiodarone in preventing postoperative atrial fibrillation (15% versus 25%).53 Based on these findings, there are two large randomized clinical trials, ie, RAFFAELLO (Ranolazine in Atrial Fibrillation Following An ELectricaL CardiOversion), currently underway examining the efficacy of ranolazine in maintaining sinus rhythm in patients with atrial fibrillation, and HARMONY (A Study to Evaluate the Effect of Ranolazine and Dronedarone When Given Alone and in Combination in Patients With Paroxysmal Atrial Fibrillation).54,55
In the future, ranolazine may become an alternative option for treatment of ventricular arrhythmias. Data from continuous electrocardiographic monitoring in the MERLIN-TIMI 36 study showed that ranolazine significantly suppressed episodes of ventricular tachycardia of more than eight beats (P<0.001).48 In a small size, nonrandomized study, Bunch et al analyzed data from 12 patients with ventricular tachycardia refractory to antiarrhythmic drugs. Ranolazine proved effective in reducing ventricular tachycardia burden and shocks from implantable cardioverter defibrillators.56 Of note, the mean QRS and the QTc interval increased nonsignificantly.
In patients with LQT3 (long QT syndrome type 3), a monogenic disorder in which the electrophysiological phenotype is due to an increase in INaL, ranolazine has been shown to cause a dose-dependent abbreviation of the QTc.57 Lastly, a study of eight patients with symptomatic premature ventricular complexes (PVCs) with a minimum PVC burden >10% on 24-hour Holter monitoring received ranolazine which resulted in a median decrease in PVC burden of 60.2% (P=0.06).58 These findings are supportive of further studies specifically designed to address the hypothesis that ranolazine has antiarrhythmic effects in the ventricle.
Cost-effectiveness
Several cost-effectiveness analyses have been performed, with results favoring greater use of ranolazine. In one study utilizing a large US commercial insurance claims database, cost comparisons were made between three groups of patients, ie, those on long-acting nitrates, those on beta-blockers or calcium channel blockers, and those on ranolazine. After the index event (ie, change in antianginal therapy), ranolazine users had lower revascularization rates than the other groups, leading to significantly lower total costs of care.59
Another study examined the cost-effectiveness of adding ranolazine to standard-of-care antianginal therapy in patients experiencing three or more attacks per week. The incremental cost-effectiveness ratio for one patient for the addition of ranolazine was $32,682 per quality-adjusted life year (QALY), which is less than the $50,000 per QALY traditionally considered to be the threshold for cost-effectiveness.60 Finally, in a European study, the cost-utility of ranolazine versus placebo was computed as an add-on therapy for symptomatic chronic angina pectoris. The incremental cost-utility ratio was 8,455 Euros per QALY per patient, which is considerably less than the conventional willingness-to-pay threshold of 15,000 Euros per QALY.61 In summary, ranolazine therapy appears to be cost-effective over a variety of health care systems, likely by reducing angina-related hospitalization and revascularization, and improving quality of life.
Conclusion
Ranolazine is a relatively new agent added to the limited armamentarium for treatment of CSA. Its lack of significant impact on blood pressure and heart rate make it an attractive drug to utilize in different clinical scenarios. In recent years, multiple, randomized, placebo-controlled clinical trials have confirmed its benefit in patients with CSA, improving functional capacity and decreasing anginal episodes. At this time, there is no clear evidence in favor of using ranolazine in the treatment of acute coronary syndromes, as shown in MERLIN-TIMI 36. Also, there are not sufficient data to recommend its use for patients with congestive heart failure. On the other hand, its effect on the INaL is quite promising for the electrophysiology community, as some evidence exists that ranolazine can decrease the incidence of atrial fibrillation and ventricular tachyarrhythmias. In summary, ranolazine appears to be a drug with an acceptable safety profile, a clear role in the treatment of CSA, and significant potential for use in patients with arrhythmias.
Disclosure
The authors report no conflicts of interest in this work.
References
Go AS, Mozaffarian D, Roger VL, et al. Heart disease and stroke statistics – 2013 update: a report from the American Heart Association. Circulation. 2013;127(1):e6–e245. | |
Centers for Disease Control and Prevention, National Center for Health Statistics. Compressed Mortality File 1999–2009. Underlying cause-of-death 1999–2009. Available from: http://wonder.cdc.gov/mortSQl.html. Accessed February 25, 2014. | |
Yeh RW, Sidney S, Chandra M, Sorel M, Selby JV, Go AS. Population trends in the incidence and outcomes of acute myocardial infarction. N Engl J Med. 2010;362(23):2155–2165. | |
Ford ES, Ajani UA, Croft JB, et al. Explaining the decrease in US deaths from coronary disease, 1980–2000. N Engl J Med. 2007;356(23):2388–2398. | |
Silverman ME. William Heberden and some account of a disorder of the breast. Clin Cardiol. 1987;10(3):211–213. | |
National Institutes of Health. National Heart, Lung, and Blood Institute. Incidence and Prevalence: 2006 Chart Book on Cardiovascular and Lung Diseases; 2006. Available from: https://www.nhlbi.nih.gov/resources/docs/06a_ip_chtbk.pdf. Accessed May 13, 2014. | |
Montalescot G, Sechtem U, Achenbach S, et al. 2013 ESC guidelines on the management of stable coronary artery disease: the Task Force on the management of stable coronary artery disease of the European Society of Cardiology. Eur Heart J. 2013;34(38):2949–3003. | |
US Food and Drug Administration. FDA approves new treatment for chest pain. Available from: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2006/ucm108587.htm. Accessed February 25, 2014. | |
Jerling M. Clinical pharmacokinetics of ranolazine. Clin Pharmacokinet. 2006;45(5):469–491. | |
Ranexa. Full prescribing information. Available from: http://www.gilead.com/~/media/Files/pdfs/medicines/cardiovascular/ranexa/ranexa_pi.ash. Accessed February 25, 2014. | |
Jerling M, Huan BL, Leung K, et al. Studies to investigate the pharmacokinetic interactions between ranolazine and ketoconazole, diltiazem, or simvastatin during combined administration in healthy subjects. J Clin Pharmacol. 2005;45(4):422–433. | |
Antzelevitch C, Belardinelli L, Wu L, et al. Electrophysiologic properties and antiarrhythmic actions of a novel anti-anginal agent. J Cardiovasc Pharmacol Ther. 2004;9 Suppl 1:S65–S83. | |
Antzelevitch C, Burashnikov A, Sicouri S, Belardinelli L. Electrophysiologic basis for the antiarrhythmic actions of ranolazine. Heart Rhythm. 2011;8(8):1281–1290. | |
Tavazzi L. Ranolazine, a new antianginal drug. Future Cardiol. 2005;1:1–9. | |
Stanley WC, Lopaschuk GD, Hall JL, McCormack JG. Regulation of myocardial carbohydrate metabolism under normal and ischaemic conditions: potential for pharmacological interventions. Cardiovasc Res. 1997;33(2):243–257. | |
Lopaschuk GD. Optimising cardiac energy metabolism: a new approach to treating ischaemic heart disease. Eur Heart J Suppl. 1999; 1 Suppl:O32–O39. | |
McCormack JG, Barr RL, Wolff AA, et al. Ranolazine stimulates glucose oxidation in normoxic, ischemic, and reperfused ischemic rat hearts. Circulation. 1996;93(1):135–142. | |
MacInnes A, Fairman DA, Binding P, et al. The antianginal agent trimetazidine does not exert its functional benefit via inhibition of mitochondrial long-chain 3-ketoacyl coenzyme A thiolase. Circ Res. 2003;93:e26–e32. | |
Noble D, Noble PJ. Late sodium current in the pathophysiology of cardiovascular disease: consequences of sodium-calcium overload. Heart. 2006;92 Suppl 4:iv–iv5. | |
Belardinelli L, Shryock JC, Fraser H. Inhibition of the late sodium current as a potential cardioprotective principle: effects of the late sodium current inhibitor ranolazine. Heart. 2006;92 Suppl 4:iv6–iv14. | |
Reddy BM, Weintraub HS, Schwartzbard AZ. Ranolazine: a new approach to treating an old problem. Tex Heart Inst J. 2010;37(6):641–647. | |
Valdivia CR, Chu WW, Pu J, et al. Increased late sodium current in myocytes from a canine heart failure model and from failing human heart. J Mol Cell Cardiol. 2005;38(3):475–483. | |
Belardinelli L, Antzelevitch C, Fraser H. Inhibition of late (sustained/persistent) sodium current: a potential drug target to reduce intracellular sodium dependent calcium overload and its detrimental effects on cardiomyocyte function. Eur Heart J. 2004;6 Suppl I:I3–I7. | |
Houser SR. Can novel therapies for arrhythmias caused by spontaneous sarcoplasmic reticulum Ca2+ release be developed using mouse models? Circ Res. 2005;96(10):1031–1032. | |
Bers DM, Barry WH, Despa S. Intracellular Na regulation in cardiac myocytes. Cardiovasc Res. 2003;57(4):897–912. | |
Murphy E, Cross H, Steenbergen C. Sodium regulation during ischemia versus reperfusion and its role in injury. Circ Res. 1999;84(12):1469–1470. | |
Chaitman BR. Ranolazine for the treatment of chronic angina and potential use in other cardiovascular conditions. Circulation. 2006;113(20):2462–2472. | |
Song Y, Shryock JC, Wu L, Belardenelli L. Antagonism by ranolazine of the proarrhythmic effects of increasing late INa in guinea pig ventricular myocytes. J Cardiovasc Pharmacol. 2004;44(2):192–199. | |
Pepine CJ, Wolff AA. A controlled trial with a novel anti-ischemic agent, ranolazine, in chronic stable angina pectoris that is responsive to conventional antianginal agents. Ranolazine Study Group. Am J Cardiol. 1999;84(1):46–50. | |
Rousseau MF, Pouleur H, Cocco G, Wolff AA. Comparative efficacy of ranolazine versus atenolol for chronic angina pectoris. Am J Cardiol. 2005;95(3):311–316. | |
Chaitman BR, Skettino SL, Parker JO, et al. Anti-ischemic effects and long-term survival during ranolazine monotherapy in patients with chronic severe angina. J Am Coll Cardiol. 2004;43(8):1375–1382. | |
Chaitman BR, Pepine CJ, Parker JO, et al. Effects of ranolazine with atenolol, amlodipine, or diltiazem on exercise tolerance and angina frequency in patients with severe chronic angina: a randomized controlled trial. JAMA. 2004;291(3):309–316. | |
Sendon JL, Lee S, Cheng ML, Ben-Yehuda O; CARISA Study Investigators. Effects of ranolazine on exercise tolerance and angina frequency in patients with severe chronic angina receiving maximally-tolerated background therapy: analysis from the Combination Assessment of Ranolazine In Stable Angina (CARISA) randomized trial. Eur J Prev Cardiol. 2012;19(5):952–959. | |
Timmis AD, Chaitman BR, Crager M. Effects of ranolazine on exercise tolerance and HbA1c in patients with chronic angina and diabetes. Eur Heart J. 2006;27(1):42–48. | |
White HD, Skettino SL, Chaitman BR, et al. Anti-anginal efficacy of ranolazine addition to beta blocker or calcium antagonist therapy in patients with a history of heart failure. Circulation. 2002;106(19 Suppl 2):349–350. | |
Koren MJ, Crager MR, Sweeney M. Long-term safety of a novel antianginal agent in patients with severe chronic stable angina: the Ranolazine Open Label Experience (ROLE). J Am Coll Cardiol. 2007;49(10):1027–1034. | |
Stone PH, Gratsiansky NA, Blokhin A, Huang IZ, Meng L; ERICA Investigators. Antianginal efficacy of ranolazine when added to treatment with amlodipine: the ERICA (Efficacy of Ranolazine in Chronic Angina) trial. J Am Coll Cardiol. 2006;48(3):566–575. | |
Rich MW, Crager M, McKay CR. Safety and efficacy of extended-release ranolazine in patients aged 70 years or older with chronic stable angina pectoris. Am J Geriatr Cardiol. 2007;16(4):216–221. | |
Wenger NK, Chaitman B, Vetrovec GW. Gender comparison of efficacy and safety of ranolazine for chronic angina pectoris in four randomized clinical trials. Am J Cardiol. 2007;99(1):11–18. | |
Kosiborod M, Arnold SV, Spertus JA, et al. Evaluation of ranolazine in patients with type 2 diabetes mellitus and chronic stable angina: results from the TERISA randomized clinical trial (Type 2 Diabetes Evaluation of Ranolazine in Subjects With Chronic Stable Angina). J Am Coll Cardiol. 2013;61(20):2038–2045. | |
Morrow DA, Scirica BM, Karwatowska-Prokopczuk E, et al. Effects of ranolazine on recurrent cardiovascular events in patients with non- ST-elevation acute coronary syndromes: the MERLIN-TIMI 36 randomized trial. JAMA. 2007;297(16):1775–1783. | |
Arnold SV, Morrow DA, Wang K, et al. Effects of ranolazine on disease-specific health status and quality of life among patients with acute coronary syndromes: results from the MERLIN-TIMI 36 randomized trial. Circ Cardiovasc Qual Outcomes. 2008;1(2):107–115. | |
Wilson SR, Scirica BM, Braunwald E, et al. Efficacy of ranolazine in patients with chronic angina observations from the randomized, double-blind, placebo-controlled MERLIN-TIMI (Metabolic Efficiency With Ranolazine for Less Ischemia in Non-ST-Segment Elevation Acute Coronary Syndromes) 36 Trial. J Am Coll Cardiol. 2009;53(17):1510–1516. | |
Weisz G, Farzaneh-Far R, Ben-Yehuda O, et al. Use of ranolazine in patients with incomplete revascularization after percutaneous coronary intervention: design and rationale of the Ranolazine for Incomplete Vessel Revascularization Post-Percutaneous Coronary Intervention (RIVER-PCI) trial. Am Heart J. 2013;166(6):953–959. e953. | |
Figueredo VM, Pressman GS, Romero-Corral A, et al. Improvement in left ventricular systolic and diastolic performance during ranolazine treatment in patients with stable angina. J Cardiovasc Pharmacol Ther. 2011;16(2):168–172. | |
Maier LS, Layug B, Karwatowska-Prokopczuk E, et al. RAnoLazIne for the treatment of diastolic heart failure in patients with preserved ejection fraction: the RALI-DHF proof-of-concept study. J Am Coll Cardiol. 2013;1(2):115–122. | |
Antzelevitch C, Belardinelli L, Zygmunt AC, et al. Electrophysiological effects of ranolazine, a novel antianginal agent with antiarrhythmic properties. Circulation. 2004;110(8):904–910. | |
Scirica BM, Morrow DA, Hod H, et al. Effect of ranolazine, an antianginal agent with novel electrophysiological properties, on the incidence of arrhythmias in patients with non ST-segment elevation acute coronary syndrome: results from the Metabolic Efficiency With Ranolazine for Less Ischemia in Non ST-Elevation Acute Coronary Syndrome Thrombolysis in Myocardial Infarction 36 (MERLINTIMI 36) randomized controlled trial. Circulation. 2007;116(15):1647–1652. | |
Murdock DK, Overton N, Kersten M, Kaliebe J, Devecchi F. The effect of ranolazine on maintaining sinus rhythm in patients with resistant atrial fibrillation. Indian Pacing Electrophysiol J. 2008;8(3):175–181. | |
Murdock DK, Kersten M, Kaliebe J, Larrain G. The use of oral ranolazine to convert new or paroxysmal atrial fibrillation: a review of experience with implications for possible “pill in the pocket” approach to atrial fibrillation. Indian Pacing Electrophysiol J. 2009;9(5):260–267. | |
Vaishnav A, Vaishnav A, Lokhandwala Y. Refractory atrial fibrillation effectively treated with ranolazine. Indian Heart J. 2014;66(1):115–118. | |
Burashnikov A, Sicouri S, Di Diego JM, Belardinelli L, Antzelevitch C. Synergistic effect of the combination of dronedarone and ranolazine to suppress atrial fibrillation. J Am Coll Cardiol. 2010;56(15):1216–1224. | |
Miles RH, Passman R, Murdock DK. Comparison of effectiveness and safety of ranolazine versus amiodarone for preventing atrial fibrillation after coronary artery bypass grafting. Am J Cardiol. 2011;108(5):673–676. | |
US National Institutes of Health, ClinicalTrials.gov, Menarini Group. Ranolazine in atrial fibrillation following an electricaL cardioversion, RAFFAELLO, ClinicalTrials.gov identifier NCT01534962. Available from: http://clinicaltrials.gov/ct2/show/NCT01534962. Accessed May 13, 2014. | |
US National Institutes of Health, ClinicalTrials.gov, Gilead Sciences. A study to evaluate the effect of ranolazine and dronedarone when given alone and in combination in patients with paroxysmal atrial fibrillation HARMONY, ClinicalTrials.gov identifier NCT01522651. Available from: http://clinicaltrials.gov/show/NCT01522651. Accessed May 13, 2014. | |
Bunch TJ, Mahapatra S, Murdock D, et al. Ranolazine reduces ventricular tachycardia burden and ICD shocks in patients with drug-refractory ICD shocks. Pacing Clin Electrophysiol. 2011;34(12):1600–1606. | |
Moss AJ, Zareba W, Schwarz KQ, Rosero S, McNitt S, Robinson JL. Ranolazine shortens repolarization in patients with sustained inward sodium current due to type-3 long-QT syndrome. J Cardiovasc Electrophysiol. 2008;19(12):1289–1293. | |
Yeung E, Krantz MJ, Schuller JL, Dale RA. Ranolazine for the suppression of ventricular arrhythmia: a case series. Ann Noninvasive Electrocardiol. February 7, 2014. [Epub ahead of print.] | |
Phelps CE, Buysman EK, Gomez Rey G. Costs and clinical outcomes associated with use of ranolazine for treatment of angina. Clin Ther. 2012;34(6):1395–1407. e1394. | |
Kohn CG, Parker MW, Limone BL, Coleman CI. Cost-effectiveness of ranolazine added to standard-of-care treatment in patients with chronic stable angina pectoris. Am J Cardiol. 2014;113(8):1306–1311. | |
Hidalgo-Vega A, Ramos-Goni JM, Villoro R. Cost-utility of ranolazine for the symptomatic treatment of patients with chronic angina pectoris in Spain. Eur J Health Econ. October 12, 2013. [Epub ahead of print.] |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

