Back to Journals » Clinical Ophthalmology » Volume 9
Unilateral rhino–orbital–cerebral mucormycosis with contralateral endogenous fungal endophthalmitis
Authors Ho HC, Liew OH, Teh SS, Hanizasurana H, Ibrahim M , Shatriah I
Received 4 February 2015
Accepted for publication 26 February 2015
Published 25 March 2015 Volume 2015:9 Pages 553—556
DOI https://doi.org/10.2147/OPTH.S82204
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Hao Chi Ho,1,2 On Heong Liew,2 Swee Sew Teh,2 Hashim Hanizasurana,2 Mohtar Ibrahim,1 Ismail Shatriah1
1Department of Ophthalmology, School of Medical Sciences, Universiti Sains Malaysia, Kelantan, 2Department of Ophthalmology, Hospital Selayang, Lebuhraya Selayang-Kepong, Selangor Darul Ehsan, Malaysia
Abstract: Rhino–orbital–cerebral mucormycosis (ROCM) is an uncommon but fatal fungal infection. We report a rare case of unilateral ROCM with ipsilateral central retinal artery occlusion and contralateral choroiditis, which later progressed to endogenous fungal endophthalmitis. The patient was successfully treated with sinuses debridement, systemic liposomal amphotericin B, and intravitreal amphotericin B. The endophthalmitis completely resolved with good vision, but the ROCM eye remained blind due to central retinal artery occlusion.
Keywords: rhino–orbital–cerebral mucormycosis, central retinal artery occlusion, chorioditis, endogenous fungal endophthalmitis, Rhizopus
Introduction
Mucormycosis is a rare opportunistic infection caused by filamentous fungi of the family Mucoraceae. These fungi are commonly found in soil, decaying vegetation, animal feces, and old bread.1 There are six major clinical forms of mucormycosis – namely, rhino–orbital–cerebral mucormycosis (ROCM), pulmonary mucormycosis, cutaneous mucormycosis, gastrointestinal mucormycosis, disseminated mucormycosis, and uncommon forms such as endocarditis, osteomyelitis, peritonitis, and pyelonephritis.2 ROCM is the most common form of mucormycosis in diabetic patients, and it is the presenting manifestation in one-fourth of diabetic patients who present with ROCM.3,4 The prevalence of ROCM in diabetic patients is 0.15%.4 ROCM usually occurs in debilitated hosts and is frequently fatal. We report a case of unilateral ROCM with ipsilateral central retinal artery occlusion (CRAO), cavernous sinus thrombosis, and contralateral endogenous fungal endophthalmitis in a diabetic patient.
Case report
A 63-year-old woman with no background medical history presented with a 1-day history of sudden drooping of the right eyelid and loss of right eye vision upon waking from sleep. It was associated with intermittent fever and headache for 10 days. She had a history of right upper premolar tooth extraction 2 weeks before the onset of symptoms.
She was conscious, oriented, and afebrile. There were no signs of meningeal irritation. Her oral hygiene was good and there was no foul-smelling nasal discharge. Ophthalmic examination revealed periorbital cellulitis, complete ptosis, and total ophthalmoplegia of the right eye. No proptosis was observed. She had right infraorbital hypoesthesia.
The visual acuity in the right eye included no perception of light, with the presence of relative afferent pupillary defect in the affected eye. Anterior segment examination was unremarkable in both eyes. Fundoscopy revealed a slightly pale optic disc, a generalized pale retina, and the presence of a cherry red spot at the posterior pole, suggestive of CRAO of the right eye (Figure 1).
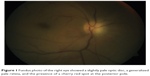  | Figure 1 Fundus photo of the right eye showed a slightly pale optic disc, a generalized pale retina, and the presence of a cherry red spot at the posterior pole. |
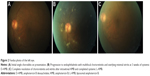
The visual acuity in the fellow eye was 6/24 correctable to 6/18 with a pinhole. The extraocular movement was full in the left eye. Fundoscopy of the left eye showed a white creamy subretinal nodule at the inferior arcade close to the optic disc, suggesting choroiditis; however, no vitritis was observed (Figure 2A).
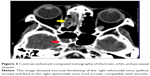
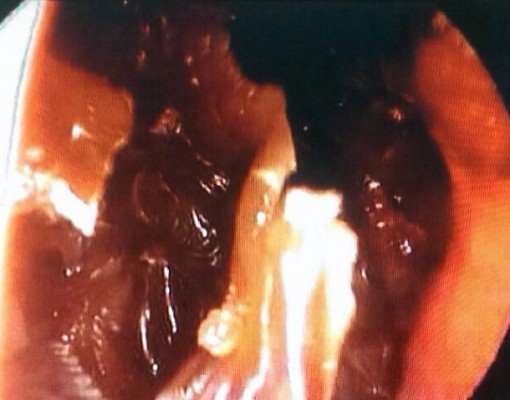
Her random blood sugar level was 23.0 mmol/L, her urine glucose was 4+ (110 mmol/L), but her urine ketone was negative. Hemogram revealed leukocytosis (17.24×10−3/μL); blood culture was negative. Computed tomography scan of the brain, orbit, and paranasal sinuses showed mucosal thickening and fluid in the right ethmoidal and sphenoidal sinuses compatible with sinusitis (Figure 3). Nasal and sinus endoscopy by the otorhinolaryngology team showed black eschar in the right sinuses (Figure 4). A biopsy was taken from the necrotic tissue for microbiological examination. Further cranial imaging with magnetic resonance imaging, magnetic resonance arteriogram, and venogram showed evidence of right cavernous sinus thrombosis.
  | Figure 4 Black eschar seen in the right nasal sinus through sinus endoscopy. |
Based on the clinical and radiological findings, she was diagnosed with right ROCM with ipsilateral CRAO and cavernous sinus thrombosis, as well as choroiditis in the left eye. She was also diagnosed with diabetes mellitus. She was started on intravenous amphotericin B deoxycholate (D-AMB), 50 mg daily, and her blood sugar was controlled with subcutaneous insulin. She underwent emergency functional endoscopic sinus surgery with debridement of the sinuses. Culture of the biopsy specimen from the sinuses grew Rhizopus spp.
At 3 weeks of treatment, the choroiditis in the left eye progressed to endogenous fungal endophthalmitis with multifocal chorioretinitis and overlying minimal vitritis (Figure 2B). Vitreous tap and single-administration intravitreal amphotericin B (AMB), 0.005 mg, diluted with sterile water to 0.1 mL was injected into the left eye. At the same time, she developed nephropathy secondary to intravenous D-AMB treatment. Her treatment was then changed to liposomal amphotericin B (L-AMB), 250 mg daily.
She was discharged well after 10 weeks of hospitalization and had completed intravenous L-AMB for a total duration of 7 weeks. Subsequent follow-up revealed that her right eye remained blind, with complete ptosis and total ophthalmoplegia. There was complete resolution of chorioretinitis and vitritis in the left eye (Figure 2C), with a final best-corrected visual acuity of 6/9.
Discussion
CRAO and endophthalmitis are rare complications of ROCM with few published reports.4,5 Ipsilateral endophthalmitis to the ROCM eye has been reported in the literature.4,5 Contralateral endophthalmitis following ROCM has not been reported before based on our PubMed search. Our patient presented with right ROCM, ipsilateral CRAO, and contralateral choroiditis, which later progressed to endogenous fungal endophthalmitis.
ROCM following tooth extraction has been reported in the literature.6,7 Following tooth extraction, the extraction socket acts as a port for the spread of mucormycosis from the oral cavity into the palate and sinuses.6 This pathogenesis contradicts the usual route of infection, which begins in the nose and paranasal sinuses from the inhalation of fungal spores.8 Thereafter, orbital involvement is due to contiguous spread from the sinuses or via the nasolacrimal duct. Further intracerebral spread extends from the orbit via the orbital apex, orbital vessels, or cribriform plate.
Talmi et al9 reported that the most common clinical feature of ROCM is black nasal eschar in a review of 19 cases. Other clinical features are malaise, periorbital cellulitis, ptosis, ophthalmoplegia, infraorbital hypoesthesia, visual loss, fever, and headache.9 Our patient presented with all of these aforementioned features. Other clinical features include conjunctival chemosis, nasal discharge, proptosis, deterioration of mental status, palatal involvement, facial palsy, hemiplegia, and seizure.9
CRAO is a rare complication of ROCM with an incidence of 16%–20%.4,5 The pathogenesis of CRAO in ROCM is due to the contiguous spread of angioinvasive fungal infection from the orbit, which infiltrates the central retinal artery, causing necrotizing vasculitis and thrombosis. In contrast, the incidence of endophthalmitis complicating ROCM has been reported as 1%–6%.4,5 Contralateral endophthalmitis may be due to hematogenous dissemination of the fungal infection forming focal infected deposits in the choroid. The choroiditis under microscopic examination consists of granulomatous inflammation with causative organisms in the center and surrounding suppuration.10 Further local spread of the lesion extends into the retina causing retinitis. Vitritis occurs when the infection breaks through the retina, seeding into the vitreous. The definition of endophthalmitis is reserved only for eyes with vitreous seeding.11 In choroiditis alone, treatment is made with a systemic antifungal.11 If it progresses to endophthalmitis, as in this case, an intravitreal antifungal is indicated.11
In the treatment of fungal choroiditis and endophthalmitis, oral fluconazole was proposed as an alternative to systemic AMB because it has better intraocular penetration and fewer side effects.11 Although oral fluconazole had been reported to be effective in clearing endophthalmitis where intravitreal AMB has failed, the causative organism in these reported cases was Candida.12 Furthermore, systemic AMB remained the mainstay treatment for ROCM. In this patient, the subsequent resolution of endophthalmitis may be attributed to the higher intravitreal concentration of antifungal due to the intravitreal injection, as well as to the change from D-AMB to L-AMB, which was shown to have better intraocular penetration.13 Only five in 19 cases of ROCM had positive blood culture.9 In endogenous fungal endophthalmitis, blood culture is rarely positive, with only 11% of patients presenting with documented systemic fungal infection.14 The vitreous culture was negative, possibly because the patient had been treated with a systemic antifungal prior to vitreous tap, and because there was only very minimal vitreous seeding.
Our patient was treated aggressively for both complications of ROCM. She survived this fatal condition with preservation of visual acuity in her left eye. The right eye remained blind following the CRAO. The infection was controlled with an appropriate systemic and intravitreal antifungal. She was very fortunate that her left visual acuity remained good, as the fovea was spared from the lesions and responded well to the treatment.
Conclusion
In conclusion, the early diagnosis and prompt treatment of ROCM is essential to improving the survival rate of the patient. A high index of suspicion in diabetic patients with imaging and nasal endoscopic evaluation is the key to an early diagnosis of ROCM. Both ipsilateral CRAO and contralateral endogenous endophthalmitis are rare and blinding complications of ROCM. In a case of unilateral ROCM, it is important to examine the contralateral eye for signs of endogenous spread, while monitoring it closely for progression.
Acknowledgment
This case report received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Disclosure
The authors report no conflicts of interest in this work.
References
Brown SR, Shah IA, Grinstead M. Rhinocerebral mucormycosis caused by Apophysomyces elegans. American Journal of Rhinology. 1998;12(4):289–292. | ||
Petrikkos G, Drogari-Apiranthitou M. Zygomycosis in immunocompromised non-haematological patients. Mediterr J Hematol Infect Dis. 2011;3(1):e2011012. | ||
Roden MM, Zaoutis TE, Buchanan WL, et al. Epidemiology and outcome of zygomycosis: a review of 929 reported cases. Clin Infect Dis. 2005;41(5):634–653. | ||
Bhansali A, Bhadada S, Sharma A, et al. Presentation and outcome of rhino-orbital-cerebral mucormycosis in patients with diabetes. Postgrad Med J. 2004;80(949):670–674. | ||
Yohai RA, Bullock JD, Aziz AA, Markert RJ. Survival factors in rhino-orbital-cerebral mucormycosis. Surv Ophthalmol. 1994;39(1):3–22. | ||
Bakathir AA. Mucormycosis of the jaw after dental extractions: two case reports. Sultan Qaboos Univ Med J. 2006;6(2):77–82. | ||
Kim J, Fortson JK, Cook HE. A fatal outcome from rhinocerebral mucormycosis after dental extractions: a case report. J Oral Maxillofac Surg. 2001;59(6):693–697. | ||
Verma A, Singh V, Jindal N, Yadav S. Necrosis of maxilla, nasal, and frontal bone secondary to extensive rhino-cerebral mucormycosis. Natl J Maxillofac Surg. 2013;4(2):249–251. | ||
Talmi YP, Goldschmied-Reouven A, Bakon M, et al. Rhino-orbital and rhino-orbito-cerebral mucormycosis. Otolaryngol Head Neck Surg. 2002;127(1):22–31. | ||
Brod RD, Flynn HW Jr, Clarkson JG, Pflugfelder SC, Culbertson WW, Miller D. Endogenous Candida endophthalmitis. Management without intravenous amphotericin B. Ophthalmology. 1990;97(5):666–672; disc: 672–674. | ||
Smiddy WE. Treatment outcomes of endogenous fungal endophthalmitis. Curr Opin Ophthalmol. 1998;9(3):66–70. | ||
Borne MJ, Elliott JH, O’Day DM. Ocular fluconazole treatment of Candida parapsilosis endophthalmitis after failed intravitreal amphotericin B. Arch Ophthalmol. 1993;111(10):1326–1327. | ||
Bhansali A, Sharma A, Kashyap A, Gupta A, Dash RJ. Mucor endophthalmitis. Acta Ophthalmol Scand. 2001;79(1):88–90. | ||
Essman TF, Flynn HW Jr, Smiddy WE, et al. Treatment outcomes in a 10-year study of endogenous fungal endophthalmitis. Ophthalmic Surg Lasers. 1997;28(3):185–194. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.