Back to Journals » Cancer Management and Research » Volume 11
Undifferentiated Pleomorphic Sarcoma: Long-Term Follow-Up from a Large Institution
Authors Chen S ![]() , Huang W, Luo P
, Huang W, Luo P ![]() , Cai W, Yang L
, Cai W, Yang L ![]() , Sun Z, Zheng B, Yan W, Wang C
, Sun Z, Zheng B, Yan W, Wang C ![]()
Received 12 August 2019
Accepted for publication 12 November 2019
Published 27 November 2019 Volume 2019:11 Pages 10001—10009
DOI https://doi.org/10.2147/CMAR.S226896
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Seema Singh
Shiqi Chen,1,2 Wending Huang,1,2 Peng Luo,1,2 Weiluo Cai,1,2 Lingge Yang,1,2 Zhengwang Sun,1,2 Biqiang Zheng,1,2 Wangjun Yan,1,2 Chunmeng Wang1,2
1Department of Musculoskeletal Oncology, Fudan University Shanghai Cancer Center, Shanghai 200032, People’s Republic of China; 2Department of Oncology, Shanghai Medical College, Fudan University, Shanghai 200032, People’s Republic of China
Correspondence: Chunmeng Wang; Wangjun Yan
Department of Musculoskeletal Oncology, Fudan University Shanghai Cancer Center, 270 Dong’an Road, Xuhui District, Shanghai 200032, People’s Republic of China
Email [email protected]; [email protected]
Purpose: Our study aimed to describe the clinical features of undifferentiated pleomorphic sarcoma (UPS) and identify the predictors of poor outcomes.
Patients and methods: The clinicopathological variables and treatment strategies of 100 UPS patients who underwent surgical resections at a single institution between November 2004 and July 2016 were reviewed. Kaplan–Meier and Cox regression method were conducted for survival analysis.
Results: The median follow-up time was 94 months (range, 1.5–154 months). R0 resection was applied for 72 cases, and the median tumor size was 5.75cm (range, 1–30cm). Tumor grades of 45 patients were intermediate grade (G2), and 54 patients were with advanced stage (stage III/IV). Twenty-seven patients presented with tumors involving important structures, in which the nerve was the most frequently invaded structure (n=12). During the follow-up, 40 patients suffered from postoperative local recurrence, and distant metastasis was observed in 25 patients which mainly metastasized to the lung (n=14). The 5-year OS rate, 5-year LRFS rate, and 5-year MFS rate was 53%, 55%, and 70%, respectively. Multivariate analysis revealed that tumor presentation, tumor size, and important structures involved (p=0.033, p=0.004, and p=0.033, respectively) were independent prognostic factors associated with OS. Meanwhile, age, resection quality and tumor grade were independent prognostic factors for LRFS (p=0.033, p=0.045, and p=0.007, respectively) and tumor depth was significantly associated with MFS (p=0.050) in multivariate analysis.
Conclusion: Primary treatment of UPS should be conducted by experts in large sarcoma center. Wide surgical margin provides sufficient control of the disease recurrence.
Keywords: undifferentiated pleomorphic sarcoma, Asian, long-term follow-up, prognostic factor
Introduction
Undifferentiated pleomorphic sarcoma (UPS), which used to be described as malignant fibrous histiocytoma (MFH), presenting a diagnostic and therapeutic challenge. MFH was first described in 1964,1 and it previously represented a group of soft tissue sarcoma (STS) which was considered to be of probable fibrohistiocytic or fibroblastic lineage. However, MFH classification was eliminated according to the World Health Organization (WHO) classification guidelines for STS in 2002 because of lacking true histiocytic origins and had been replaced by the term of UPS.2–5 The diagnosis of UPS was exclusively based on the absence of a specific line of differentiation and it was challenging despite advanced techniques and careful histologic examination.6
UPS was one of the most common STS which mainly occurred in patients between 50 and 70 years old.7 The majority of UPS occurred in extremities, it could also be found in other parts of the body such as the chest wall, retroperitoneum, head and neck and so on.7 Aggressive surgical resection remained the principal treatment. Local recurrence occurred in 13–42% of patients and distant metastasis could be found in 31–35% of patients despite aggressive surgery.7–9 According to previous reports, chemotherapy might be palliative although it had shown some benefits.10 And, radiotherapy could be used for local control, however, approximately 3–5% of UPS occurred in a prior site of therapeutic radiation for an unrelated malignancy making radiotherapy controversial.11
Currently, it is urgent to clarify and update the prognosis of patients suffering from UPS with a long period of follow-up owing to changes in classification criteria. Therefore, we conducted this study aiming to analyze the clinicopathologic features of UPS and identify the prognostic factors with a long period of follow-up.
Materials and Methods
Patient Selection
Between November 2004 and July 2016, 130 UPS patients were treated at our hospital. All patients were entered into a computerized database during their hospitalization, and follow-up data were recorded for each patient. This study was approved by the Ethics Committee of Fudan University Shanghai Cancer Center and was performed in the light of the approved guidelines. Written informed consent was obtained from all patients. Patients would be included in this study if they met the following criteria: (1) surgical resections were performed at our hospital, patients received chemotherapy and/or radiotherapy only were excluded, (2) patients with other types of tumors synchronous were excluded, (3) histologically confirmed diagnosis of UPS, and (4) follow-up data were recorded completely. Clinical information such as patient demographics, tumor characteristics (site, size, grade, and depth), resection quality, history of previous treatment, AJCC stage, important structures (bone, vessel and/or nerve) involved or not, and adjuvant therapy could be found in our computerized database. Finally, 100 UPS patients with primary or recurrent tumors were enrolled in our study.
Resections were classified into 2 groups: gross tumor resection (R0) and palliative resection (R1/R2). R0 = referred to microscopic tumor-negative surgical margins; R1 = referred to microscopic tumor-positive surgical margins; and R2 = referred to macroscopic tumor-positive surgical margins. Tumor sizes (T-stage) were measured by the longest diameter using specimens resected after the operation and were classified into 2 groups, T1 (5cm) and T2 (>5cm) (Figure 1). Tumors that originated above the superficial fascia without invasion of the fascia and tumors that arose beneath the superficial fascia with invading through the fascia were classified as superficial and deeply located tumors, respectively. Retroperitoneal tumors were also defined as deeply situated tumors. Tumor grade (G2/G3) was defined by the French Federation of Cancer Centers Sarcoma Group (FNCLCC) grading system.12 All tumors were staged by the 2010 AJCC standards.13 Important structures involved meant that at least one structure such as bone, blood vessel or nerve was invaded by tumor. The information of tumor depth, resection quality, tumor grade and important structures involved was determined by the surgical report and/or pathology report. All histopathological specimens were confirmed at the Institute of Pathology of Fudan University Shanghai Cancer Center by two pathologists (Figure 2).
 |
Figure 1 Representative images of MRI (A&B). |
 |
Figure 2 Representative images of UPS stained with hematoxylin and eosin: (A) 100x; (B) 200x; (C) 400x. |
Follow-Up Data and Statistical Analysis
Overall survival (OS) time was calculated from the date of surgery to the date of death or the last follow-up time. Local recurrence-free survival (LRFS) time and metastasis-free survival (MFS) time were measured from the date of surgical resection to the date of pathological or radiographic determination of recurrent or metastatic disease, respectively. For patients alive or without records of local recurrence or distant metastases, follow-up was censored at the time of last follow-up. Follow-up data were collected by phone calls and medical records. All 100 patients were followed up to either January 2019 or the date of death.
Median OS, LRFS, and MFS were estimated using the Kaplan–Meier method. The log rank test was used to identify potential prognostic factors such as tumor size, tumor depth, resection quality, tumor grade and so on. And, Cox proportional hazards regression modeling was applied to perform univariate analysis and multivariate analysis. The factors would be put into multivariate analysis if they were significant in the univariate analysis. All statistical analysis was conducted by SPSS 21.0 and the significance level for all statistics was set at P<0.05.
Results
Patient Characteristics
In this study, comprehensive data of 100 UPS patients who underwent surgeries at Fudan University Shanghai cancer center between November 2004 and July 2016 were analyzed. Data including patient demographics, tumor characteristics and so on are listed in Table 1. There were 60 males and 40 females and the median age was 58.5 years (range, 15–85 years). Fifty-two patients presented with primary diseases and other 48 patients presented with recurrent diseases. The most common sites were the extremities (n=55), followed by the trunk (n=35) and retroperitoneum (n=9). The median tumor size was 5.75cm (range, 1–30cm). In all 100 patients, 49 patients presented with T1 (5cm) stage and 51 patients presented with T2 (>5cm) stage. Besides, there were 2 patients with tumors occurred in the head and left atrium, respectively. Forty-nine patients presented with tumors invading deeply, which were comparable to 51 patients with the superficial tumor. According to the FNCLCC grading system, the tumor grades of 45 patients were intermediate grade (G2) and the other 55 patients’ grades were high grade (G3). R0 resections were applied for 72 cases while the other 28 patients underwent R1/R2 resections. According to the latest 2010 AJCC standard for STS, 46 patients presented with stage II and 54 patients were stage III/IV. Important structures including bone, vessel or nerve invaded by tumors could be found in 27 patients according to the surgical reports, and the most frequently invaded structure was nerve (n=12), followed by blood vessels (n=11) and bone (n=11). Postoperative adjuvant treatments were applied to 31 patients, among whom, 19 patients received adjuvant radiotherapy, 11 patients received chemotherapy, and 1 patient received both. During the follow-up, 40 patients suffered from postoperative local recurrence, and 25 patients occurred distant metastasis, in which mainly metastasized to the lung (n=14).
 |
Table 1 Patient Characteristics |
Data Analysis
Updating to January 2019, 56 patients died of disease or its complications. Median follow-up time was 94 months (range, 1.5–154 months). The 5-year OS rate, 5-year LRFS rate, and 5-year MFS rate were 53%, 55%, and 70%, respectively. The median OS was 70.5 months (95% CI, 35.5–105.5 months), while the median LRFS and MFS have not yet been reached.
Factors influencing OS, LRFS, and MFS in univariate analyses and multivariate analysis were listed in Tables 2–4, respectively. In the univariate analysis, patients with age >60 years at surgery presented markedly shorter LRFS compared to patients with age ≤60 years [hazard ratio (HR) =1.914; 95% confidence interval (CI), 1.025–3.575; p=0.0383], while the difference of OS and MFS between the two groups was not statistically significant (p=0.0634, p=0.6745, respectively). As for presentation type, recurrent diseases showed an adverse OS and LRFS over primary diseases [HR=2.147; 95% CI, 1.263–3.652; p=0.0039; Figure 3A and HR=1.888; 95% CI, 1.007–3.54; p=0.0396], while there was no significant difference between the two groups for MFS (p=0.6991). With respect to tumor size, T1 stage had a favorable outcome for OS (HR=2.552; 95% CI, 1.493–4.36; p= 0.0004; Figure 3B); however, a significant difference was not be observed between the two groups for LRFS (p=0.5211) and MFS (p=0.0570). Compared to the patients with tumors above the superficial fascia, the patients with tumors located deeply would lead to a shorter OS (HR=1.894; 95% CI, 1.116–3.214; p=0.0160) and MFS (HR=2.192; 95% CI=1.006–4.773; p=0.0438; Figure 4C) other than LRFS (p=0.5265). Similar results were also observed in resection quality, R1/R2 resections would significantly reduce OS time (HR=1.966; 95% CI, 1.072–3.608; p=0.0112) and LRFS time (HR=1.953; 95% CI, 0.9588–3.979; p=0.0328; Figure 4A) compared to R0 resections. In regard to tumor grade, the patients with intermediate grade (G2) presented an obviously longer LRFS time compared to those with high grade (G3) (HR=2.55; 95% CI, 1.369–4.748; p=0.0072; Figure 4B). Besides, AJCC stage and important structures involved were significantly associated with OS, respectively [HR=2.202; 95% CI, 1.297–3.736; p=0.0030 and HR=2.079; 95% CI, 1.094–3.951; p=0.0068; Figure 3C]. In this study, other prognostic factors such as gender, tumor location, adjuvant chemotherapy and radiotherapy had no significant differences in OS, LRFS and MFS in univariate analyses.
 |
Table 2 Factors Influencing OS in Univariate Analysis and Multivariate Analysis |
 |
Table 3 Factors Influencing LRFS in Univariate Analysis and Multivariate Analysis |
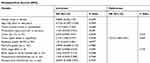 |
Table 4 Factors Influencing MFS in Univariate Analysis and Multivariate Analysis |
 |
Figure 4 (A) Survival outcome according to the extent of resection for LRFS; (B) survival outcome according to tumor grade for LRFS; and (C) survival outcome according to tumor depth for MFS. |
In multivariate analysis, tumor presentation (HR=1.821; 95% CI, 1.050–3.157; p=0.033), tumor size (HR=2.254; 95% CI, 1.290–3.938; p=0.004), and important structures involved (HR=1.836; 95% CI, 1.050–3.210; p=0.033) were independently prognostic factors associated with OS. Meanwhile, age (HR=2.005; 95% CI, 1.058–3.801; p=0.033), resection quality (HR=1.920; 95% CI, 1.014–3.638; p=0.045) and tumor grade (HR=2.717; 95% CI, 1.322–5.585; p=0.007) were independent prognostic factors for LRFS and tumor depth was an independent prognostic factor for MFS (HR=2.219; 95% CI, 1.000–4.921; p=0.050).
Discussion
Undifferentiated pleomorphic sarcoma (UPS), which was called malignant fibrous histiocytoma (MFH) previously, was the most common soft tissue sarcoma (STS). However, the diagnosis and treatment for this subtype of STS remained challenging. Currently, the available literature of UPS was limited because of the updating of the reclassification of STS. In order to better understand the biological behavior and clinical manifestations of UPS, we conducted this study for the purpose of describing the clinical features of UPS and identified predictors of poor outcomes.
In our study, gender, including 60 males and 40 females, which presented a slight gender bias, showed no significant differences associated with OS, LRFS, and MFS, which was in accordance with previous reports.14–17 As for the age of patients, the optimal cutoff value differed from 40 to 70 in different reports.9,18–20 In this study, the cutoff value of age was 60, and the age over 60 years showed a significantly worse LRFS in univariate analysis, however, multivariate analysis revalued that age was not an independent predictor for LRFS, which was in line with previous reports despite the slight bias on the optimal cutoff value of age.
Compared with tumors that occurred in the extremities, trunk tumors showed no statistically significant difference for OS, LRFS, and MFS in our cohort. However, one report21 showed that tumor located in the head and neck had a favorable prognosis due to a smaller size and a lower grade contrast with Sabesan, who reported that head and neck tumor would have a poor outcome because of the inadequate resection.22 To sum up, the tumor size, grade, depth, or important structures involvement may play a crucial role in patient prognosis.
Because of differences in the level of diagnosis and treatment, almost half of the patients presented with recurrent diseases in our study. And, we found patients with recurrent diseases had a significantly worse outcome for OS and LRFS in univariate analysis. The result was consistent with the studies of Lehnhardt and Guo.18,23 What’s more, tumor presentation was an independent predictor associated with OS in multivariate analysis. This discovery suggested that initial treatment should be careful by experts in the field of sarcoma.
With respect to tumor size, the optimal cutoff value differed from 5cm to 10cm in various literature. We classified patients into two groups, the T1 (5cm) group and T2 (>5cm) group. Multivariate analysis revealed that tumor size was an independent prognostic factor for OS, which was in line with Peiper and Ozcelik.16,24 Besides, Roland identified that patient with a tumor smaller than 10cm had a favorable outcome.14 What’s more, Roland and Doussal reported that larger tumor size was significantly associated with poor prognosis for MFS in spite of the slight bias on the optimal cutoff value, which could not be observed in our study.14,20 We believed that these different outcomes in different kinds of literature could attribute to the difference in the optimal cutoff value of tumor size according to Grimer’s research on size matters for sarcomas.25 Due to the deep location, the generally large size of UPS, the tumor often invaded the important structures such as blood vessels, nerve and bone. In this cohort, important structures involvement was an independent prognostic factor on OS. Therefore, we advocated plan resection in professional sarcoma center at the first visit.
According to previous studies, some studies reported that deep tumor was not related to worse OS,14,17,23 while some studies showed that significant difference between deep tumor and superficial tumor on OS or DSS.16,24 In our study, we found that patients with deep tumor had a worse prognosis for OS. There was no significant effect of the deep tumor associated with LRFS in some literature, and our study was in line with them. However, we observed that deep tumor had a worse outcome on MFS, which was consistent with Ozcelik’s study.24
As for resection quality, our study revealed that R0 resection was a favorable prognostic factor for overall survival and local recurrence survival, while there was no significant difference for MFS between patients with R0 resections and patients with R1/R2 resections. Our results were supported by previous studies,14,16,23,24,26 and these data manifested that clear surgical margin was a favorable prognostic factor for local recurrence. So we tried our best to obtain a clear surgical margin and advocated extended resection for patients who had unplanned resections in our center.
The influence of tumor grade on prognosis was reported by the majority of the literature. According to the French Federation of Cancer Centers Sarcoma Group (FNCLCC) grading system,12 we classified patients into two groups [intermediate grade (G2) vs. high grade (G3)], and our results revealed patients with high-grade tumor showed an adverse outcome associated with LRFS. The patients’ AJCC stage was also evaluated in our study, and we found a significant difference between the AJCC stage and OS. Hsu revealed that stage III/IV was correlated with worse OS and MFS,17 while Winchester found an association between AJCC stage and LRFS and MFS.27 Generally, advanced patients presented a worse outcome in any kind of tumors, so we advocated early diagnosis and early treatment.
In terms of postoperative adjuvant therapy, adjuvant chemotherapy remained controversial in UPS and our study showed no benefit of it, which was consistent with some reports.10,28,29 However, it was found that the malignant behavior of UPS may be related to epithelial–mesenchymal transition (EMT) in one transformation study, and the expression of some genes such as laptm4a and laptm4b related to chemotherapy resistance was also found, which has been reported in other chemoresistant tumors such as breast cancer.30 This research helped us to understand the molecular characteristics of UPS more deeply, and given us a certain understanding of UPS chemotherapy resistance. In addition, some new fusion genes including PDGFRA-MACROD2, NCOR1-MAP2K1 were found in Zheng’s research, which provided an important clue for targeted therapy.31 As for adjuvant radiotherapy, Belal9 reported that adjuvant radiotherapy was related to a decreased risk of local recurrence and Hsu17 showed it could improve local control, while our data showed no benefit in prolong overall survival time, local recurrence time and metastasis time. Besides, radiotherapy could lead to radiation-associated UPS, which was associated with worse clinical outcomes than sporadic lesions.11 So the dose and time of radiotherapy should be controlled strictly.
Conclusion
UPS was one of the most common types of STS with generally large size and frequent proximity to vital structures. R0 resection remained the mainstay of treatment. In our study, tumor presentation, tumor size and important structures involved were independent prognostic factors associated with OS, which justified the early and primary treatment was very important. We hope our study may facilitate further prospective research and clinical decision-making in UPS patients.
Disclosure
The authors report no conflicts of interest in this work.
References
1. O’Brien JE, Stout AP. Malignant fibrous xanthomas. Cancer. 1964;17:1445–1455. doi:10.1002/1097-0142(196411)17:11<1445:AID-CNCR2820171112>3.0.CO;2-G
2. Daugaard S. Current soft-tissue sarcoma classifications. Eur J Cancer. 2004;40(4):543–548. doi:10.1016/j.ejca.2003.11.009
3. Dei Tos AP. Classification of pleomorphic sarcomas: where are we now? Histopathology. 2006;48(1):51–62. doi:10.1111/his.2006.48.issue-1
4. Casali PG, Abecassis N, Aro HT, et al. Soft tissue and visceral sarcomas: ESMO-EURACAN clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018;29(Suppl 4):iv51–iv67. doi:10.1093/annonc/mdy096
5. Fletcher C, Bridge J, Hogendoorn P, editors. WHO Classification of Tumors of Soft Tissue and Bone. Pathology and Genetics of Tumours of Soft Tissue and Bone. Lyon (France): IARC Press; 2013.
6. Nascimento AF, Raut CP. Diagnosis and management of pleomorphic sarcomas (so-called “MFH”) in adults. J Surg Oncol. 2008;97(4):330–339. doi:10.1002/(ISSN)1096-9098
7. Weiss SW, Enzinger FM. Malignant fibrous histiocytoma: an analysis of 200 cases. Cancer. 1978;41(6):2250–2266. doi:10.1002/1097-0142(197806)41:6<2250::AID-CNCR2820410626>3.0.CO;2-W
8. Fletcher CDM, Gustafson P, Rydholm A, Willén H, Åkerman M. Clinicopathologic re-evaluation of 100 malignant fibrous histiocytomas: prognostic relevance of subclassification. J Clin Oncol. 2001;19:3045–3050. doi:10.1200/JCO.2001.19.12.3045
9. Belal A, Kandil A, Allam A, et al. Malignant fibrous histiocytoma. Am J Clin Oncol. 2002;25(1):16–22. doi:10.1097/00000421-200202000-00003
10. Casali PG. Adjuvant chemotherapy for soft tissue sarcoma. Am Soc Clin Oncol Educ Book. 2015;35(1):e629–e33.
11. Dineen SP, Roland CL, Feig R, et al. Radiation-associated undifferentiated pleomorphic sarcoma is associated with worse clinical outcomes than sporadic lesions. Ann Surg Oncol. 2015;22(12):3913–3920. doi:10.1245/s10434-015-4453-z
12. Trojani M, Contesso G, Coindre JM, et al. Soft-tissue sarcomas of adults; study of pathological prognostic variables and definition of a histopathological grading system. Int J Cancer. 1984;33:37–42. doi:10.1002/(ISSN)1097-0215
13. Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010;17(6):1471–1474. doi:10.1245/s10434-010-0985-4
14. Roland CL, May CD, Watson KL, et al. Analysis of clinical and molecular factors impacting oncologic outcomes in undifferentiated pleomorphic sarcoma. Ann Surg Oncol. 2016;23(7):2220–2228. doi:10.1245/s10434-016-5115-5
15. Chen K-H, Chou T-M, Shieh S-J. Management of extremity malignant fibrous histiocytoma: a 10-year experience. Formosan J Surg. 2015;48(1):1–9. doi:10.1016/j.fjs.2014.06.003
16. Peiper M, Zurakowski D, Knoefel WT, Izbicki JR. Malignant fibrous histiocytoma of the extremities and trunk: an institutional review. Surgery. 2004;135(1):59–66. doi:10.1016/S0039-6060(03)00325-8
17. Hsu HC, Huang EY, Wang CJ. Treatment results and prognostic factors in patients with malignant fibrous histiocytoma. Acta Oncol. 2004;43(6):530–535. doi:10.1080/02841860410018421
18. Guo J, Cui Q, Liu C, et al. Clinical report on transarterial neoadjuvant chemotherapy of malignant fibrous histiocytoma in soft tissue. Clin Transl Oncol. 2013;15(5):370–375. doi:10.1007/s12094-012-0933-9
19. Zagars GK, Mullen JR, Polack A. malignant fibrous histiocytoma: outcome and prognostic factors following conservation surgery and radiotherapy. Int J Radiat Oncol Biol Phys. 1996;34(5):983–994. doi:10.1016/0360-3016(95)02262-7
20. Le Doussal V, Coindre J-M, Leroux A, et al. Prognostic factors for patients with localized primary malignant fibrous histiocytoma. Am Cancer Soc. 1996;77(9):1823–1830.
21. Borucki RB, Neskey DM, Lentsch EJ. Malignant fibrous histiocytoma: database review suggests a favorable prognosis in the head and neck. Laryngoscope. 2018;128(4):885–888. doi:10.1002/lary.26909
22. Sabesan T, Xuexi W, Yongfa Q, Pingzhang T, Ilankovan V. Malignant fibrous histiocytoma: outcome of tumours in the head and neck compared with those in the trunk and extremities. Br J Oral Maxillofac Surg. 2006;44(3):209–212. doi:10.1016/j.bjoms.2005.06.006
23. Lehnhardt M, Daigeler A, Homann HH, et al. MFH revisited: outcome after surgical treatment of undifferentiated pleomorphic or not otherwise specified (NOS) sarcomas of the extremities – an analysis of 140 patients. Langenbecks Arch Surg. 2009;394(2):313–320. doi:10.1007/s00423-008-0368-5
24. Ozcelik M, Seker M, Eraslan E, et al. Evaluation of prognostic factors in localized high-grade undifferentiated pleomorphic sarcoma: report of a multi-institutional experience of anatolian society of medical oncology. Tumour Biol. 2016;37(4):5231–5237. doi:10.1007/s13277-015-4359-1
25. Grimer RJ. Size matters for sarcomas! Ann R Coll Surg Engl. 2006;88(6):519–524. doi:10.1308/003588406X130651
26. Kamat NV, Million L, Yao DH, et al. The outcome of patients with localized undifferentiated pleomorphic sarcoma of the lower extremity treated at Stanford University. Am J Clin Oncol. 2019;42(2):166–171. doi:10.1097/COC.0000000000000496
27. Winchester D, Lehman J, Tello T, et al. Undifferentiated pleomorphic sarcoma: factors predictive of adverse outcomes. J Am Acad Dermatol. 2018;79(5):853–859. doi:10.1016/j.jaad.2018.05.022
28. Mendenhall WM, Indelicato DJ, Scarborough MT, et al. The management of adult soft tissue sarcomas. Am J Clin Oncol. 2009;32(4):436–442. doi:10.1097/COC.0b013e318173a54f
29. Singer S, Demetri GD, Baldini EH, Fletcher CDM. Management of soft-tissue sarcomas: an overview and update. Lancet Oncol. 2000;1(2):75–85. doi:10.1016/S1470-2045(00)00016-4
30. De Vita A, Recine F, Mercatali L, et al. Primary culture of undifferentiated pleomorphic sarcoma: molecular characterization and response to anticancer agents. Int J Mol Sci. 2017;18(12):2662. doi:10.3390/ijms18122662
31. Zheng B, Zhang S, Cai W, et al. Identification of novel fusion transcripts in undifferentiated pleomorphic sarcomas by transcriptome sequencing. Cancer Genomics Proteomics. 2019;16(5):399–408. doi:10.21873/cgp.20144
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

