Back to Journals » Patient Preference and Adherence » Volume 15
Understanding the Profiles of Blood Glucose Monitoring Among Patients with Type 2 Diabetes Mellitus: A Cross-Sectional Study in Shandong, China
Authors Yao J, Wang H , Yan J , Shao D, Sun Q, Yin X
Received 17 November 2020
Accepted for publication 22 January 2021
Published 22 February 2021 Volume 2021:15 Pages 399—409
DOI https://doi.org/10.2147/PPA.S292086
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Naifeng Liu
Jiansen Yao,1,2 Haipeng Wang,1,3 Jingjing Yan,4 Di Shao,1,3 Qiang Sun,1,3 Xiao Yin5
1Center for Health Management and Policy Research, School of Public Health, Shandong University, Jinan, People’s Republic of China; 2Institute for Hospital Management, Tsinghua University, Shenzhen, People’s Republic of China; 3NHC Key Laboratory of Health Economics and Policy Research, Shandong University, Jinan, People’s Republic of China; 4School of International and Public Affairs, Shanghai Jiao Tong University, Shanghai, People’s Republic of China; 5Endocrinology Department, Jinan Central Hospital Affiliated to Shandong University, Jinan, People’s Republic of China
Correspondence: Xiao Yin
Jinan Central Hospital Affiliated to Shandong University, Jinan, 250012, People’s Republic of China
Email [email protected]
Background: Blood glucose monitoring is essential in diabetic care and management. Monitoring using glucometers in home and in laboratories by professionals in certain health institutes were the common methods of blood glucose monitoring in clinical practice. This study aimed to characterize the profiles of blood glucose monitoring in the view of the discrepancy in methods and frequency conducted by the patients with type 2 diabetes mellitus (T2DM) in China, and to explore factors influencing the profiles.
Methods: A cross-sectional, community-based study was conducted in Shandong province, China, with a multi-stage stratified sampling. A total of 2166 T2DM patients completed the structured questionnaires about the real-world status of blood glucose monitoring and other questions composed of demographic and clinical characteristic as well as the diabetes-related cognitive scales. Latent profile analysis (LPA) was used to identify the underlying profiles of blood glucose monitoring based on self-reported frequency of blood glucose monitoring through different methods. Univariate and multivariate logistic regression were used to analyze the characteristics of the profiles and to explore the factors associated with it.
Results: Among the 2166 participants, the mean frequency of blood glucose monitoring was 2.77 times (standard deviation: 7.67) per month. LPA indicated that five-class model was the best solution for classifying the latent groups of blood glucose monitoring: Class 1 “Low frequency in all”, Class 2 “High frequency in hospitals”, Class 3 “High frequency in primary health institutes”, Class 4 “High frequency in pharmacies”, and Class 5 “High frequency in self-monitoring”. The proportions of the patients in class 1, class 2, class 3, class 4, and class 5 were 88.1% (n=1909), 1.3% (n=28), 3.1% (n=67), 6.1% (n=133) and 1.3% (n=29), respectively. Multivariate logistic regression showed that participants who had higher income (OR: 1.58, 95% CI: 1.04∼ 2.41, p< 0.05), had diabetes complication(s) (OR=1.37, 95% CI: 1.03∼ 1.02, p=0.03) and had a good knowledge of blood glucose control (OR=1.59, 95% CI: 1.17∼ 2.16, p< 0.01) were more likely to have high frequency of blood glucose monitoring (in class 2, 3, 4, 5), and the rural patients were less likely to had high frequency of blood glucose monitoring (OR=0.47, 95% CI: 0.35∼ 0.63, p< 0.01).
Conclusion: Low frequency dominates the characteristics of the profiles of blood glucose monitoring among T2DM patients in China, though distinct blood glucose monitoring groups can be identified by LPA. Educational and financial supports were recommended to increase the frequency of blood glucose monitoring in patients with T2DM, focusing on the patients with low socioeconomic status.
Keywords: blood glucose monitoring, latent profile analysis, type 2 diabetes mellitus, China
Introduction
Type 2 diabetes mellitus (T2DM) has become one of the most common non-communicable diseases and is one of the most challenging public health issues globally.1 The International Diabetes Federation estimated that 425 million people worldwide had diabetes in 2017, and this number is expected to rise to 627 million by 2045.2 China has the highest number of T2DM patients in the world, accounting for 25% of patients globally in 2013.3 According to the latest estimates, more than 104 Chinese adults have diabetes, of which, T2DM accounts for about 95% of these cases.3 T2DM is associated with increased risk of long-term microvascular and macrovascular complications caused by high blood glucose levels, and represents a fast-growing economic burden with considerable consequences for individuals, communities, and health systems.
Blood glucose monitoring is essential to the management of diabetes. The results of glucose monitoring are crucial for glycemic status assessment, prescription of the optimal treatment regimen, follow-up of patients’ glucose status, and timely therapy adjustment.4–6 Several studies have demonstrated that regular monitoring of blood glucose levels was associated with reduced blood glucose level and improved quality of life.7,8 The frequency of blood glucose monitoring negatively correlates with levels of blood glucose.9 At present, many blood glucose testing methods had been used to monitor patients’ blood glucose,10 which can provide necessary data for evidence-based clinical decision-making by healthcare professionals. Monitoring using glucometers in home, and in laboratories by professionals, are the common and affordable methods of blood glucose monitoring in clinical practice. Nevertheless, the implementation of blood glucose monitoring among diabetic patients is far from satisfactory worldwide. A study of 5104 participants showed that blood glucose monitoring adherence rates was as low as 44% for adults with T1DM and 24% for adults with T2DM.11 A study carried out in China found that only half of the 2183 participants tested their blood glucose levels more than once a month.12
Previous studies had revealed that factors that determine whether patients regularly monitor their blood glucose were complex and varied across countries and regions.13–15 Some studies showed that blood glucose monitoring was greatly affected by socioeconomic status. Diabetic patients who had higher income levels, had higher educational levels, and had health insurance would have better performance in blood glucose monitoring.16,17 Other studies indicated that diagnosis of type 1 diabetes, longer duration of diabetes, and insulin injection were associated with better adherence to blood glucose monitoring.14,18,19 In addition, previous studies also revealed that diabetic knowledge, attitude, self-efficacy, and social support were important predictors of blood glucose monitoring.20–23 However, most of these studies were conducted in west countries and regions, but findings from these studies may not apply to Asia, especially China, since the cultural, economic and healthcare status differ significantly from the west. Furthermore, most previous studies focused on self-monitoring of blood glucose,24–26 and few studies have explored blood glucose monitoring by professionals,12 not to mention distinguishing professionals from different health institutes. This study aimed to examine the comprehensive profiles of the blood glucose monitoring in terms of the certain methods conducted by the T2DM patients in China. We explored the profiles of blood glucose monitoring, identified the characteristics of the profiles, and explored the factors influencing the profiles.
Methods
Study Design and Participants
This was a cross-sectional study conducted in Shandong province in eastern China between August and December 2017. Shandong contains 17 prefectures and 140 counties (districts) and has a population of nearly 99 million (7.2% of mainland China) in 2016. There are an estimated 980,000 T2DM patients in the province in 2013 (prevalence: 9.3%).27 Nearly 30% of T2DM patients were enrolled in the Essential Public Health Services - Non-communicable Disease Management System (EPHS- NDMS) in 2016. For our study, we employed a multi-stage, stratified, randomized sampling to select participants from the EPHS-NDMS, as described in detail in a previous study.28 A total of 2520 diabetes patients in the sampling communities or villages were recruited to the survey, and 2166 diabetic patients for whom a complete dataset were available were included in the analysis. The inclusion criteria were registration in EPHS-NDMS; diagnosis of T2DM based on WHO criteria for more than 6 months;29 aged under 80 years and ability to communicate and understand instructions.
Data Collection
Participants were invited to complete a structured face-to-face questionnaire, which was developed based on a review of the literature10,30 and consultation from diabetes specialists and clinical pharmacists in endocrinology. The questionnaire contents included: (1) basic information of participants, such as residence, gender, age, marital status, education, household income, family size and health insurance status; (2) disease-related information of participants, including duration of diagnosis, diabetes complication(s), treatment regimens and blood glucose control status; (3) cognitive variables, including patients’ knowledge about blood glucose control and attitude to blood glucose monitoring; (4) healthcare utilization, such as the blood glucose tests and inpatient health services. To ensure the quality of the survey, all data collectors were rigorously trained and qualified for questionnaire delivery, and quality supervisors reviewed all completed questionnaires after each interview.
Measurements and Variables
Latent Profile Analysis
The dependent variable in this study corresponded to the profiles of blood glucose monitoring conducted by the T2DM patients, which was measured by four questions regarding the methods and frequency of blood glucose monitoring. The first question was “How many times on average did you test your blood glucose by the professionals in the primary health institution(s) per month?”; The second question was “How many times on average did you test your blood glucose by the pharmacists in the pharmacies per month”; The third question was “How many times on average did you test your blood glucose by the professionals in the secondary or tertiary hospital (s) per month”; The fourth was “How many times on average did you test your blood glucose using glucometers at home per month”. The answers to the four questions were counted based on the frequency of capillary blood glucose monitoring (fasting blood glucose and postprandial blood glucose), ranging zero to 360 times per month. The zero means that patients never tested their blood glucose; and the 360 times was the maximum limit that the patients could not be reached. These four questions were categorized through Latent Profile Analysis (LPA), and then presented as a single variable of the profiles of blood glucose monitoring. In addition, the frequency of twice a month was used to assess the characteristics of blood glucose monitoring in different subgroups based on LPA, according to the Guideline for Blood Glucose Monitoring in China.6
LPA is a type of cluster analysis, which is used to group population into a specific (k) number of unique categories. Based on LPA, patients are most similar to each other within each category, and are most different between the categories.31 LAP estimates two parameters based on maximum-likelihood: 1) class membership probabilities, which represent individuals’ probability of belonging to each class; and 2) item-response probabilities conditional on class membership (conditional response probabilities), which refer to the conditional probability a particular response given the individual is in a certain class. Based on their highest latent class probability, individuals are assigned to one class exclusively.31 In this study, the selected four observed variables used in the LPA were the frequency of blood glucose conducted with four different methods, including by professionals in primary healthcare institutes, by pharmacists in pharmacies, by professionals in secondary or tertiary hospitals and by self-monitoring using glucometers at home. To find out the optimal number of categories, 2–6 class solutions were modelled, and the outputs were assessed; Each solution was then compared using model fit indicators (Akaike information criterion [AIC], Bayesian information criterion [BIC], entropy values),32,33 until an appropriate number of patterns was decided according to empirical and theoretical interpretations regarding simultaneously considering and interpreting multiple indices of fit. Once an optimal class solution was determined, the individual classes were characterized with respect to frequency of blood glucose tests in certain methods that defined each group.
Covariates
The independent variables in this study corresponded to participants’ demographics, disease-related variables, cognitive variables, and healthcare utilization variables.
Demographics included residence (urban or rural), gender (male or female), age group (18–44, 45–64, and ≥65 years), marital status (married or others, with others encompassing the not-married, divorced, and widowed states), educational level (no formal education, primary school, and junior school and higher), annual household income per capita (0~3000, 3~6000, 6~12,000, and ≥12,000 RMB Yuan), and health insurance (Urban and Rural Residents Basic Medical Insurance [URRBMI], Urban Employees Basic Medical Insurance [UEBMI], and Uninsured).
Disease-related variables included duration of diabetes (0~5, 6~10 and ≥10 years), diabetes complications (no or yes, yes means the presence of any other disease(s) or condition(s), such as diabetic nephropathy, diabetic eye complications, diabetic foot, diabetic cardiovascular complications, diabetic cerebrovascular disease, and diabetic neuropathy), treatment regimens (diet/exercise, oral hypoglycemic agents [OHAs], and insulin) and blood glucose control (fasting blood glucose [FBG]:<7mmol/L or ≥7mmol/L).
Cognitive variables were patients’ knowledge about blood glucose control and patients’ attitude toward blood glucose monitoring. Patients’ knowledge about blood glucose control was measured using the question: “What is the target value of blood glucose control for the patients with type 2 diabetes mellitus in China”. Patients who answered correctly to this question were considered to have a good knowledge of blood glucose control. Patients’ attitude to blood glucose monitoring was measured using the question: “How important do you think the regular blood glucose monitoring is for the blood glucose control among the patients with type 2 diabetes mellitus”. Responses for this question were rated on a 5-point Likert scale (1= very important, 2= important, 3= neutral, 4= unimportant and 5= very unimportant). Patients who answered very important or important to this question were considered to have a positive attitude to blood glucose monitoring.
In addition, inpatients health service utilization was also included as the covariates in this study, which was defined as patients had been hospitalized for one or more times in hospitals during the past year.
Statistical Analysis
Descriptive statistics were used to describe the characteristics of the participants and the profiles of blood glucose monitoring through different methods. The continuous variables were presented as the mean and standard deviation (Sd), and the categorical variables were presented as the frequency and percentage. LPA was used to identify the underlying profiles of blood glucose monitoring in terms of the methods and the certain frequency of blood glucose monitoring. Based on the results of LPA, univariate comparisons and multivariate logistic regression analyses were used to identify the factors associated with the profiles of blood glucose monitoring among the T2DM patients. We conducted all LPA analyses in Mplus (version 7.3) and all other analyses in Stata (version 15.0). Statistical significance was set at a p-value of <0.05.
Ethical Statement
This study was approved by the Ethics Committee of the School of HealthCare Management, Shandong University, China (approval number: ECSHCMSDU 20,170,401). The Declaration of Helsinki on medical protocol and ethics were followed. All participants were informed of the research aims prior to participation, and informed consent forms were signed by participants themselves or by their family members if the participants could not sign their own names.
Results
The Characteristics of Participants
A total of 2166 T2DM patients were included in the analysis, including 1070 patients from urban areas and 1096 from rural areas, as shown in Table 1. Nearly half (49.9%) of the participants were ≥65 years old, and the majority of the participants were female (65.4%) and the married (86.1%). With respect to educational level, 32.8% had no formal education, 33.1% had primary school education, and 34.1% had junior school or higher education. The median annual household income per capita was 6000 Yuan (about US$869). The majority (90.3%) of the patients were covered by URRBMI, 7.1% were by UEBMI, and 2.6% were uninsured. Overall, 38.5% of the patients had been diagnosed with diabetes within the preceding 5 years, and 35.7% reported at least one diabetes complications. Approximately, two fifths of the patients control their blood glucose at FPG< 7.0mmol/L, and 71.3% were treated with OHAs. In terms of patients’ perceptions of diabetes, 59.4% had a good knowledge of blood glucose control, and 92.8% hold a positive attitude to blood glucose monitoring. In addition, 11.4% had used the inpatient services during the past year.
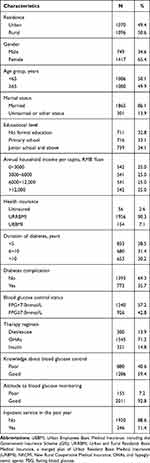 |
Table 1 The Characteristics of the Participants in Shandong Province, China (n = 2166) |
The Profile of Blood Glucose Monitoring
As shown in Table 2, of the total 2166 participants, 6.4% had their blood glucose monitored no less than twice a week; 27.1% had their blood glucose monitored less than twice a week but no less than twice a month; 54.9% had blood glucose monitored less than twice a month but no less than once per quarter; and 11.6% had blood glucose monitored less than once per quarter. The mean number of blood glucose monitoring was 2.77 times (Sd:7.67) per month. In terms of the blood glucose monitoring with specific method, the frequency of blood glucose monitoring performed by professionals in primary health institutes, in hospitals, in pharmacies and by self-monitoring using glucometer strips at home per month were 0.84, 0.03, 0.14 and 1.76, respectively.
 |
Table 2 The Frequency of Blood Glucose Monitoring Among the Participants (n=2166) |
Models with one to six latent classes were estimated, where the one-class model was deemed as a baseline. After careful review of all the models, the five-class model for blood glucose monitoring was tested to best satisfy the selection criteria, as shown in Table 3. The best model (k=5) revealed the following classes of patients, as shown in Figure 1: Class 1 “Low frequency in all”, Class 2 “High frequency in hospitals”, Class 3 “High frequency in primary health institutes”, Class 4 “High frequency in pharmacies”, and Class 5 “High frequency in self-monitoring”. In general, class 2,3,4,5, regardless of the monitoring methods, had higher frequency of blood glucose monitoring than the class 1. The proportions of the patients in class 1, class 2, class 3, class 4, and class 5 were 88.1% (n=1909), 1.3% (n=28), 3.1% (n=67), 6.1% (n=133) and 1.3% (n=29), and the proportion of the class with high monitoring frequency was 11.9%, with the sum of class 2, 3, 4, 5.
 |
Table 3 Model Fit Statistics of the One- to Six-Class Latent Profile Analysis Models |
 |
Figure 1 The estimated item-response probabilities for blood glucose monitoring in each of the five latent classes. |
The Factors Associated with the Profiles of Blood Glucose Monitoring
As shown in Table 4, a total of eight variables were significantly associated with the profiles of blood glucose monitoring in the univariate comparisons (Low frequency [in class 1]= 0; High frequency [in class 2,3,4,5]=1), which included residence, educational level, income, health insurance, duration of diabetes, diabetes complications, knowledge about blood glucose control, utilization of inpatient services. After adjusting for the covariates, four of the variables remained significantly associated with the profiles of blood glucose monitoring in the multivariate logistic regression model. The patients who had higher income (OR=1.58, 95% CI: 1.04~2.41, p=0.03), had diabetes complication(s) (OR=1.37, 95% CI: 1.03~1.02, p=0.03) and had a good knowledge of blood glucose control (OR=1.59, 95% CI: 1.17~2.16, p<0.01) were more likely to have high frequency of blood glucose monitoring than their counterpart. The rural patients were less likely to had high frequency of blood glucose monitoring than the urban ones (OR=0.47, 95% CI: 0.35~0.63, p<0.01).
 |
Table 4 The Factors Associated with the Profiles of Blood Glucose Monitoring Among the Type 2 Diabetes Patients in Shandong Province, China |
Discussion
In this study, five clearly distinct profiles for blood glucose monitoring were found regarding the methods and frequency conducted by the T2DM patients. Class 1 had the lowest frequency of blood glucose monitoring; Class 2 had high frequency in hospitals; Class 3 had high frequency in primary health institutes; Class 4 had high frequency in primary health institutes; Class 5 had high frequency in self-monitoring. Residence, educational level, income level and knowledge about the blood glucose control were the strong indicator of blood glucose monitoring profiles.
Reliable information on patients’ glycaemia control is essential to diabetes care and management.5 However, the expected frequency of blood glucose monitoring was not inclusive among diabetic patients around the world and was largely depending on patients’ conditions. According to the 2019 edition of the Guidelines for Blood Glucose Monitoring in China,6 people who use lifestyle intervention or oral hypoglycemic drugs to manage their diabetes should monitor their blood glucose at least twice a week on average. Based on this criterion, only 6.4% participants were adherent to the requirements in this study. Our results echoed the previous studies about the adherence of blood glucose monitoring in China, which suggested that the adherence to blood glucose monitoring were suboptimal among diabetic patients.12,34 In reference to previous studies,12,35 this study used the definition of twice a month to assess the real-world frequency of blood glucose monitoring among the T2DM patients, in order to describe the characteristic of the profiles of blood glucose monitoring. Patients who tested their blood glucose less than twice a week were thought to have low frequency of blood glucose monitoring, while these who tested blood glucose no less than twice a week were thought to have high frequency of blood glucose monitoring. Based on this operational definition, of the five blood glucose monitoring profiles classified by LCA, only the class 1 members had on average less than the defined frequency of blood glucose monitoring. Interestingly, this profile represented almost ninety percent of the type 2 diabetes population in the real practice. Our study indicated more measures should be taken to narrow the gaps between the real practice and the recommended frequency of blood glucose monitoring in China.
Consistent with a previous study,34 our study showed that economic status was the most significant factor affecting blood glucose monitoring of T2DM in Shandong province in China. Although blood glucose tests were provided without charge for the T2DM patients quarterly, as one of the key contents of EPHS paid by government budget, it was still far away from the recommended frequency in the Guidelines for Blood Glucose Monitoring in China. Moreover, the fees of monitoring blood glucose and of acquiring a glucose meter and strips for diabetes, except for the blood glucose monitoring covered in EPHS, has to be paid by the patients themselves to a great extent, hindering patients to conduct regularly blood glucose monitoring. Therefore, we recommend that the national health insurance agency would reimburse the cost of monitoring blood glucose in laboratories and the costs related to glucose testing equipment used at home, so as to reduce the economic burden among diabetic patients.
The results of our study showed that the high frequency of blood glucose monitoring profile was associated with better knowledge of blood glucose control. This finding was supported by findings from the studies in Xiamen, China,16 which demonstrated that patients with a higher educational level were more likely to regularly perform self-monitoring of blood glucose and to attain adequate glycemic control. This finding could be attributed to the fact that patients with better knowledge of blood glucose control may be easier to understand both the importance of blood glucose monitoring and the way to conduct the tests. Moreover, previous study also indicated that better knowledge of blood glucose control, an important controllable factor, was positively associated with glycemic control of diabetes.36 Our finding implied that health workers should probably focus on increasing the knowledge of blood glucose control in the population with T2DM, thus, to improve the adherence to blood glucose monitoring.
In our survey, T2DM patients who live in the rural areas were less likely to perform high frequency of blood glucose monitoring than the patients live in urban areas. This finding could be related to the fact that the annual household income and health educational level in the urban diabetic patients of our study were higher than those in rural patients. In fact, education and income are tightly linked in China,37 and may interact to influence the blood glucose monitoring. Our findings also suggested that the difference in socioeconomic development and the quality of healthcare services between rural and urban areas may explain the observed difference in the frequency of blood glucose monitoring. In China, the quality of healthcare in rural regions is far below that in urban areas. Therefore, we should improve the quality of the healthcare services in rural regions, along with providing more diabetes-related education for diabetic patients in rural regions.
This study found that patients who had longer duration of diabetes and had been hospitalized in the past year monitored their blood glucose more frequently than their counterparts. Interestingly, multivariate analysis of the logistic regression showed that disease duration and hospitalized experience were not associated with the frequency of blood glucose monitoring to any great degree. We found that patients with diabetes-related complication(s) were more likely to perform higher frequency of blood glucose monitoring. This was also confirmed in the quantitative data reported by other studies that showed that a higher level of complication(s) was associated with greater performance of blood glucose monitoring.38 One explanation for the better performance in blood glucose monitoring is that patients with diabetes complications may be more susceptible to the control of diabetes and may pay more attention to blood glucose monitoring. Another explanation is that patients with diabetes complications may visit frequently to the hospitals and get more support from the physicians, which may be of great use for the patients to perform the blood glucose monitoring.
Limitations
This study had some limitations that should be considered. First, due to the cross-sectional nature of the study, inferences about causality or temporal ordering of variables cannot be made. Second, recall and information bias might still exist when the status of blood glucose monitoring was measured using self-reported blood glucose monitoring behaviors, despite strict quality control were conducted during the interviews. Third, four distinctive groups of blood glucose monitoring profiles (Class 2,3,4,5), given their similarity in the frequency of blood glucose monitoring, were merged into one group of high frequency, when exploring the factors associated with the frequency of the blood glucose monitoring in the logistic regress model. Patients’ preference for blood glucose monitoring method was not well revealed in this study, and needs to be explored in future research.
Conclusion
In summary, low frequency dominates the characteristics of the profiles among T2DM patients in China, though distinct blood glucose monitoring groups can be identified by LPA. Patients who lived in the rural area, had lower income, and had poor knowledge about blood glucose control were found to be less likely to perform the blood glucose monitoring as recommended, though various methods were available to them. Our study implies a need for physicians to focus on the education of diabetes-related knowledge in the clinical practice, which is of great importance to improve blood glucose monitoring adherence. In addition, the government and the national health insurance agency should reimburse the cost of monitoring blood glucose in laboratories and the costs related to glucose testing equipment used at home.
Acknowledgments
This study was funded by the Key Research and Development Program of Shandong Province (2018GSF118184). We are grateful to the Shandong Department of Health officers and Shandong CDC for their support during the planning of the study and assistance with data collection. We would also like to thank the staff members of all the clinics for facilitating the data collection process.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. World Health Organization. Global Report on Diabetes. Geneva: World Health Organization; 2016.
2. International Diabetes Federation. Diabetes Atlas 8th Edition. Brussels, Belgium: International Diabetes Federation; 2017.
3. Wang LM, Gao P, Zhang M, et al. Prevalence and ethnic pattern of diabetes and prediabetes in China in 2013. JAMA. 2017;317(24):2515. doi:10.1001/jama.2017.7596
4. American Diabetes Association. Standards of medical care in diabetes-2017. Diabetes Care. 2017;Suppl1(40):S1–142.
5. Clinical Guidelines Task Force. Global Guideline for Type 2 Diabetes. Brussels, Belgium: International Diabetes Foundation; 2012.
6. Jia WP, Weng JP, Zhu DL, et al. Standards of medical care for type 2 diabetes in China 2019. Diabetes Metab Res Rev. 2019;35(6):6. doi:10.1002/dmrr.3158
7. Giuseppina TR, Marina S, Elena A, et al. The burden of structured self-monitoring of blood glucose on diabetes-specific quality of life and locus of control in patients with noninsulin-treated type 2 diabetes: the PRISMA Study. Diabetes Technol Ther. 2016;18(7):421–428. doi:10.1089/dia.2015.0358
8. Miller KM, Beck RW, Bergenstal RM, et al. Evidence of a strong association between frequency of self-monitoring of blood glucose and hemoglobin A1c levels in T1D exchange clinic registry participants. Diabetes Care. 2013;36(7):2009–2014. doi:10.2337/dc12-17702013-07-01
9. Ji L, Su Q, Feng B, et al. Structured self-monitoring of blood glucose regimens improve glycemic control in poorly controlled Chinese patients on insulin therapy: results from COMPASS. J Diabetes. 2017;9(5):495–501. doi:10.1111/1753-0407.12434
10. Mark E. Current methods of assessing blood glucose control in diabetes. Br J Diabetes. 2016;16:7–09. doi:10.15277/bjd.2016.0712016-03-21
11. Peyrot M, Rubin RR, Lauritzen T, et al. Psychosocial problems and barriers to improved diabetes management: results of the cross-national Diabetes Attitudes, Wishes and Needs (DAWN) Study. Diabet Med. 2005;22(10):1379–1385. doi:10.1111/j.1464-5491.2005.01644.x
12. Raoufi AM, Tang X, Jing ZY, et al. Blood glucose monitoring and its determinants in diabetic patients: a cross-sectional study in Shandong, China. Diabetes Ther. 2018;9(5):2055–2066. doi:10.1007/s13300-018-0499-9
13. Zeleke B, Solomon S, Eshetu ZT. Adherence to diabetic self-care practices and its associated factors among patients with type 2 diabetes in Addis Ababa, Ethiopia. Patient Prefer Adherence. 2018;12:963–970. doi:10.2147/PPA.S156043
14. Al-Keilani MS, Almomani BA, Al-Sawalha NA, Shhabat BA. Self-monitoring of blood glucose among patients with diabetes in Jordan: perception, adherence, and influential factors. Diabetes Res Clin Pract. 2017;126:79–85. doi:10.1016/j.diabres.2017.01.005
15. Won CL, Elise S, Barrie C, et al. Frequency of blood glucose testing among insulin-treated diabetes mellitus patients in the United Kingdom. J Med Econ. 2014;17(3):167–175. doi:10.3111/13696998.2013.873722
16. Zeng Y, Wu J, Han Y, et al. Educational disparities in the associations between self-monitoring of blood glucose and glycemic control in type 2 diabetes patients in Xiamen, China. J Diabetes. 2018;10(9):715–723. doi:10.1111/1753-0407.12651
17. Chen CC, Chen LW, Cheng SH. Rural–urban differences in receiving guideline-recommended diabetes care and experiencing avoidable hospitalizations under a universal coverage health system: evidence from the past decade. J Diabetes. 2017;151:13–22. doi:10.1016/j.puhe.2017.06.009
18. Luo YY, Bao YQ, Zhang P, et al. Self-monitoring of blood glucose in patients with type 2 diabetes before and after initiating basal insulin treatment in China. Diabetes Technol Ther. 2017;19(9):541–548. doi:10.1089/dia.2017.0040
19. Wang X, Luo J-F, Qi L, et al. Adherence to self-monitoring of blood glucose in Chinese patients with type 2 diabetes: current status and influential factors based on electronic questionnaires. Patient Prefer Adherence. 2019;13:1269–1282. doi:10.2147/PPA.S211668
20. Marcus S, Oliver G, Ulrich S. Self-monitoring of blood glucose—psychological aspects relevant to changes in HbA1c in type 2 diabetic patients treated with diet or diet plus oral antidiabetic medication. Patient Educ Couns. 2006;62(1):104–110. doi:10.1016/j.pec.2005.06.013
21. Jeffrey SG, Molly LT, Commissariat PV. Psychosocial factors in medication adherence and diabetes self-management: implications for research and practice. Am Psychol. 2016;71(7SI):539–551. doi:10.1037/a0040388
22. Qin Z-Y, Yan J-H, Yang D-Z, et al. Behavioral analysis of Chinese adult patients with type 1 diabetes on self-monitoring of blood glucose. Chin Med J. 2017;130(1):39–44. doi:10.4103/0366-6999.196574
23. Krishnan V. Assessment of knowledge of self blood glucose monitoring and extent of self titration of anti-diabetic drugs among diabetes mellitus patients – a Cross Sectional, Community Based Study. J Clin Diagn Res. 2016. doi:10.7860/JCDR/2016/18387.7396
24. Sudore RL. Assessment of self-monitoring of blood glucose in individuals with type 2 diabetes not using insulin. JAMA Intern Med. 2019;179(2):268. doi:10.1001/jamainternmed.2018.52802019-02-01
25. Gábor V, Jamie CB, Debra L. Factors associated with adherence to self-monitoring of blood glucose among persons with diabetes. Diabetes Educ. 2016;30(1):112–125. doi:10.1177/014572170403000119
26. Charity KW, Ajay MV, Sven GH, et al. Do diabetes mellitus patients adhere to self-monitoring of blood glucose (SMBG) and is this associated with glycemic control? Experiences from a SMBG program in western Kenya. Diabetes Res Clin Pract. 2016;112:37–43. doi:10.1016/j.diabres
27. Zhang GH, Jun HU, Chen X, et al. Morbidity, awareness rate, treatment, and control among residents with diabetes (≥ 18 years old) of Shandong Province in 2013. J Chinese J Prev Contro Chron Dis. 2017;12(25):881–884.
28. Yao JJ, Wang HP, Yin X, et al. The association between self-efficacy and self-management behaviors among Chinese patients with type 2 diabetes. PLoS One. 2019;14(11):e224869. doi:10.1371/journal.pone.02248692019-11-11
29. Alberti KG, Zimmet PZ. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med. 1998;15(7):539–553. doi:10.1002/(SICI)1096-9136(199807)15:7<539::AID-DIA668>3.0.CO;2-S
30. Holt P. Blood glucose monitoring in diabetes. Nurs Stand. 2014;28(27):52–58. doi:10.7748/ns2014.03.28.27.52.e650
31. Linda MC, Stephanie TL. Latent Class and Latent Transition Analysis: With Applications in the Social, Behavioral, and Health Sciences. John Wiley & Sons; 2010.
32. Jung T, Wickrama KA. An introduction to latent class growth analysis and growth mixture modeling. Soc Personal Psychol Compass. 2008;2:302–317. doi:10.1111/j.1751-9004.2007.00054.x
33. Nagin DS. Group-Based Modeling of Development. Cambridge. London: Harvard University Press; 2005.
34. Yuan L, Guo X, Xiong Z, et al. Self-monitoring of blood glucose in type 2 diabetic patients in China: current status and influential factors. Chin Med J. 2014;127(2):201–207.
35. Sapkota S, Brien JE, Aslani P. Blood glucose monitoring in type 2 diabetes - Nepalese patients’ opinions and experiences. Glob Health Action. 2017;10(1):1322400. doi:10.1080/16549716.2017.1322400
36. Li C, Wang A, Zhang Y, et al. Knowledge of blood sugar control standard brings the higher attainment rate of HbA1c. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2013;38(8):773–778. doi:10.3969/j.issn.1672-7347.2013.08.003
37. Xiao TH. The correlation analysis of income of rural labor force and educational level in China (in Chinese). J Huazhong Agric Univ. 2005;4:24–27.
38. Guan X, Ma L, Wushouer H, et al. Determinants of self-monitoring of blood glucose with type 2 diabetes based on 496 questionnaire surveys in China. Int J Diabetes Dev Ctries. 2019;39(1):181–187. doi:10.1007/s13410-018-0627-0
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
