Back to Journals » Journal of Pain Research » Volume 13
Ultrasound-Guided Unilateral Transversus Abdominis Plane Combined with Rectus Sheath Block versus Subarachnoid Anesthesia in Patients Undergoing Peritoneal Dialysis Catheter Surgery: A Randomized Prospective Controlled Trial
Authors Li J , Guo W, Zhao W, Wang X, Hu W, Zhou J , Xu S , Lei H
Received 24 May 2020
Accepted for publication 1 August 2020
Published 14 September 2020 Volume 2020:13 Pages 2279—2287
DOI https://doi.org/10.2147/JPR.S264255
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Robert B. Raffa
Ji Li,1 Wenjing Guo,1 Wei Zhao,1 Xiang Wang,1 Wenmin Hu,1 Jie Zhou,2 Shiyuan Xu,1 Hongyi Lei1
1Department of Anesthesiology, Zhujiang Hospital, Southern Medical University, Guangzhou, Guangdong Province, People’s Republic of China; 2Department of Anesthesiology, Perioperative and Pain Medicine, Brigham and Women’s Hospital, Harvard Medical School, Boston, MA, USA
Correspondence: Hongyi Lei; Shiyuan Xu Department of Anesthesiology
Zhujiang Hospital, Southern Medical University, Guangzhou 510282, People’s Republic of China
Tel/Fax +86-20-62782898
Email [email protected]; [email protected]
Background: Peritoneal dialysis catheter placement can be performed under general anesthesia, local anesthesia or subarachnoid anesthesia (SA). Recently, studies have reported the successful placement of peritoneal dialysis catheters using a transversus abdominis plane (TAP) block and rectus sheath (RS) block. This study compared the TAP + RS block with SA for patients undergoing peritoneal dialysis catheter placement.
Methods: Sixty patients were randomly divided into two groups, with 30 receiving unilateral ultrasound-guided TAP + RS block anesthesia and 30 receiving SA. The demographic characteristics, anesthesia efficacy, indicators related to anesthesia or operation, hemodynamic index, postoperative pain numeric rating score (NRS), postoperative recovery indicators, complications related to anesthesia or surgery, and dosage of sedative or analgesic medication were analyzed.
Results: Anesthesia operation time was significantly shorter in the TAP + RS block group than in the SA group (P< 0.001), while there was no significant difference in success rates (TAP + RS 93.33% [95% confidence interval, 95% CI, 83.9– 102.8%] vs SA 100.00% [95% CI, 100– 100%], P=0.472). Two patients in the TAP + RS group needed extra analgesia, although the dermatome pinprick sensation test gave negative results for all patients. Patients who received the TAP + RS block expressed significantly less pain on movement or at rest at 4 h and 8 h postoperative. Fewer patients needed rescue analgesia with tramadol in the postoperative period in the TAP + RS block group than in the SA group (P< 0.05). The intraoperative MAP was more stable (P< 0.05) in the TAP + RS group compared to the SA group.
Conclusion: The TAP + RS block is a safe, effective method for use as the principal anesthesia technique in PD catheter placement. Compared to SA, it has the advantages of less influence on hemodynamics and a better postoperative analgesic effect.
Keywords: TAP block, rectus sheath block, peritoneal dialysis catheter placement, local anesthesia, subarachnoid anesthesia
Introduction
More than 808,159 Americans experience kidney failure. The unadjusted incidence rate of end-stage renal disease (ESRD) was 381.5 per million/year in the United States.1 Peritoneal dialysis (PD) is a common and effective renal replacement therapy. Although PD catheter placement can be performed in a variety of ways, including open dissection and laparoscopic, peritoneoscopic, percutaneous, and radiological implantation, no technique is uniformly superior and open-surgical procedures are still routinely performed.2–6
PD catheter placement can be performed under local anesthetic (LA) infiltration, subarachnoid anesthesia (SA) or general anesthesia (GA).7,8 Among these, SA is the most common. However, it may not be suitable for patients with poor cardiac function. The same applies to GA.
Currently, the transversus abdominis plane (TAP) block is widely used for analgesia after abdominal surgery.9–13 The TAP block has a good analgesic effect and little impact on the whole body without affecting lower limb sensation or movement. The rectus sheath (RS) block is a field block that can cover multiple nerves and enhance the effect of the TAP block.14,15 Although studies have shown that the TAP block, either with or without the RS block, can be used for PD catheter placement, thus far none have compared this method with SA.7,8,16-20 Hence, the primary objective of this study is to test the efficacy of the TAP + RS block as a principal anesthesia method for PD catheter surgery compared to SA. The secondary objective is to analyze the intraoperative hemodynamics and postoperative analgesic efficiency.
Methods
Study Design
The study was conducted in accordance with the Declaration of Helsinki. It was approved by the Institutional Ethics Committee of Zhujiang Hospital and registered with the Chinese Clinical Trial Registry (ChiCTR1800016320) on May 25, 2018. After giving informed consent, 60 consecutive patients who underwent PD catheter surgery between June 2018 and May 2019 were recruited (Figure 1). The patients were aged 18 to 65 years old and had American Society of Anesthesiologists (ASA) Physical Status II–IV. Exclusion criteria included a body mass index (BMI) ≥ 35 kg/m2, allergy to local anesthetics, chronic pain, opioid dependence, pregnancy or in lactation, abnormal lower limb movement, and localized infection at the TAP+RS or SA injection sites.
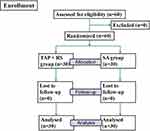 |
Figure 1 CONSORT flowchart. |
Patients were randomly allocated to receive a TAP + RS block or SA using a random number generator immediately before the procedure. The anesthesiologist in the operating room could not be blinded due to the nature of this study. The personnel in charge of postoperative care and the observer who collected the data were blinded to the randomization.
All surgical procedures were performed by the same surgeon using the open-surgical PD catheter placement technique. All TAP + RS blocks were performed using a portable ultrasound with a 10~12 Hz linear transducer (Wisonic, Shenzhen Huasheng Medical Technology co., LTD, China) and SA was performed by the same anesthesiologist, who was experienced in both techniques.
Patients were premedicated with midazolam (0.03 mg/kg) intravenously before the block. During the operation, intravenous dexmedetomidine (DEX) was used to maintain sedation at a rate of 0.2–0.7 μg/kg/h after the amount of 1μg/kg had been administered for more than 10 mins. Total dosages of midazolam and DEX were recorded.
After the anesthetic procedure, the sensory block area was assessed once per min by evaluating the pinprick sensation using a 22G needle and comparing it with the same dermatome on the opposite arm until the block segment was the same for three consecutive tests. A block region extending from T8 to T12 was considered the appropriate anesthesia effect. Once the target range of the block was reached, the point-in-time and the target or maximum range of the block were recorded.
The surgeon injected a local anesthetic of 5–10 mL 2% lidocaine into the incision site if the anesthetic effect was not satisfactory during the intervention. If a surgical level of anesthesia was not met, a conversion to GA was done through the placement of an airway device, volatile anesthetic use, and an intraoperative propofol infusion at or exceeding 100 μg/kg/min.
The primary outcome was to evaluate the proportion of patients achieving adequate anesthesia, defined as not needing an additional local anesthetic or conversion from monitored anesthesia care to GA as defined above. The secondary outcomes were the time it took to perform the block, variations in blood pressure (BP) and heart rate (HR) during the operation, and postoperative numeric rating scale (NRS) scores (indicated below). For the TAP + RS block group, the time was defined as from the skin being sterilized to the point when the needle was withdrawn from the patient as the injection was completed. This did not include the process of preparing the ultrasound machine or probe. For SA, the time began when the patient was in the lateral position for anesthesia and stopped when the patient lay on their back.
Demographic information (age, gender, ASA physical status, BMI), previous abdominal operation history (yes/no), information regarding the surgical procedure (duration of operation, laterality), and operating room time were recorded.
The BP and HR of patients were measured every five minutes in the operating room and were recorded at the following time points: (1) entering the operating room, (2) midazolam given, (3) anesthesia procedure completed, (4) skin incision, (5) end of the surgery, and (6) leaving the operating room. Phenylephrine was administered at 1~2 µg/kg when hypotension (systolic blood pressure below 90 mmHg or more than 20% lower than the base value or mean blood pressure below 65 mmHg) occurred; ephedrine was administered at 100~200 μg/kg if bradycardia (HR ≤ 60/min) was observed at the same time.
At 2 h, 4 h, 8 h, 24 h, and 48 h after surgery, pain scores at rest and movement were assessed using NRS scores (0: no pain to 10: worst imaginable pain). When the NRS score at rest ≥4, rescue analgesia with tramadol (1 mg/kg) was administered and the dosage was recorded.
In addition, surgeon satisfaction (0: extremely dissatisfied to 10: extremely satisfied) and patient satisfaction (0: extremely dissatisfied to 10: extremely satisfied) were recorded. The points at which the lower extremity muscle strength was restored and the ureter was removed were recorded. The occurrence of nausea, vomiting, postoperative urinary retention, and other complications associated with surgery or anesthesia were also recorded.
Procedures
PD Catheter Surgery
All surgical procedures were performed by the same surgeon and utilized the open-surgical approach using mini-laparotomy, which requires a paramedian abdominal incision to expose the rectus abdominis (RA) and the posterior RS. After insertion into the peritoneal cavity, the catheter was sutured onto the posterior border of the RA and tunneled out several centimeters through subcutaneous tissue before emerging from the abdominal wall. The surgical incision and the subcutaneous tunnel are shown in Figure 2.
TAP + RS Block
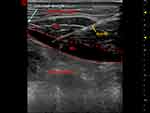
The TAP was identified using the external oblique, internal oblique, and transversus abdominis muscles as landmarks, visualized by an ultrasound linear transducer (Wisonic, Shenzhen Huasheng Medical Technology co., LTD, China) at the level of the anterior axillary line between the costal margin and the iliac crest. The TAP block was performed using an in-plane technique under real-time ultrasound sonography with an 8.5 cm 22G needle (Stimuplex® D, B. Braun Melsungen AG, Germany). Once the tip of the needle was advanced into the correct plane between the internal oblique and transversus abdominis muscles and negative aspiration was confirmed, 20 mL of 0.5% ropivacaine (10 mL: 100 mg, AstraZeneca, H20140763) was administered under ultrasonographic guidance (Figure 3).
The RA muscle and sheath were used as landmarks for the RS block, which was performed at the external margin of the rectus abdominis above the umbilicus level. An 8.5 cm 22G insulated needle (Stimuplex® D, B. Braun Melsungen AG, Germany) was inserted in-plane to the transducer in a medial to lateral direction with the endpoint being the fascial plane between the RA and posterior RS. Once the tip of the needle was advanced into the correct plane and negative aspiration was confirmed, 10 mL of 0.5% ropivacaine was administered under direct ultrasonographic guidance (Figure 4).
Subarachnoid Anesthesia
The subarachnoid puncture was performed with a 25G spinal needle (VOLTE medical company, Zhejiang, China), with patients in the right lateral position. A midline approach at the L3–4 interspace was used. Patients received 3 mL of 0.5% hyperbaric ropivacaine and were restored to the supine position after injection.
Statistical Analysis
Statistical analysis was performed with IBM SPSS 20.0. Descriptive statistics are presented as mean (SD), median (range), number or count (percentages). In the case of normal distribution, independent t-tests were carried out, whereas the Mann–Whitney U-test was performed for nonparametric variables. For categorical variables, χ2 tests were used. To assess the group differences for measures taken repeatedly over time, a general linear model (GLM) was performed. Statistical significance was accepted when the P-value was <0.05.
Results
Patient Characteristics
Demographic characteristics, duration of surgery, and operating room time were similar in both groups (Table 1).
 |
Table 1 Demographic Characteristics of the Groups |
Primary Outcome
There was no statistically significant difference (P=0.472) in the proportion of patients achieving adequate anesthesia between the groups (TAP + RS 93.33% [95% CI, 83.9–102.8%] vs SA 100% [95% CI, 100–100%]), and two patients who received the TAP + RS block needed extra analgesia during surgery (Table 2).
 |
Table 2 Data on Anesthesia or Surgical Procedure and Postoperative Outcomes for Both Groups |
Secondary Outcomes
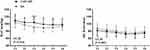
The anesthesia control time for the TAP + RS group was significantly shorter than in the SA group (8.70 ± 2.78 min vs 18.47 ± 7.36 min, respectively, with P < 0.001) (Table 2). Compared to the patients in the SA group, those in the TAP + RS block group had a significantly higher intraoperative MAP (GLM, P=0.036) and no significant difference in HR (GLM, P=0.885) (Figure 5). One patient in the TAP + RS group and 8 patients in the SA group needed vasoactive drugs during the operation, and there was a significant difference between the two groups (P<0.05) (Table 2).
The NRS scores during movement and at rest during the postoperative period are shown in Figure 6. In detail, patients in the TAP + RS group expressed significantly less pain at 4 h and 8 h compared to those in the SA group (P: mNRS at 4 h <0.01, mNRS at 8 h <0.001, rNRS at 4 h <0.01, rNRS at 8 h <0.001, GLM, PmNRS=0.001, PrNRS=0.000), whereas there was no significant difference at the other time points. Four patients (13.3% [95% CI, 0.4–26.2%]) in the TAP + RS block group needed analgesics during the postoperative period, while 13 patients (43.3% [95% CI, 24.5–62.2%]) in the SA group needed them. There was a significant difference between the two groups (P<0.05) (Table 2).
The TAP + RS block anesthesia had no effect on the motor function of the lower limbs, while the lower limb muscle strength recovery needed 194.00 ± 77.70 min after anesthesia in the SA group. There was a significant difference in the time it took for the urinary tube to be removed between the two groups (620.97 ± 425.19 min in the TAP + RS block group vs 1318.07 ± 170.84 min in the SA group, P<0.001). The patients in the TAP + RS group were significantly more satisfied than in the SA group (9 [95% CI, 9–9] vs 8 [95% CI, 7–8.25] P<0.001) (Table 2).
There was no significant difference in surgeon satisfaction, intraoperative sedative drug dosage or incidence of PONV. In addition, no complications related to anesthesia or surgery procedures occurred in either group.
Discussion
Our study indicates that the TAP + RS block can serve as the sole anesthetic modality for ESRD patients undergoing open PD catheter insertion. The TAP + RS block incurred less hemodynamic fluctuation and vasoactive agent use in the intraoperative period and provided superior analgesia and patient satisfaction post-operation compared to SA.
The area of blockage from LA infiltration is limited, which may cause a higher conversion rate to GA.7 Whereas ESRD patients may also have a substantial number of cardiac comorbidities that can be negatively influenced by GA,21,22 most anesthesiologists traditionally opt for SA.23 However, SA is also associated with complications, such as hypotension, nausea, vomiting, uroschesis, transient neurological syndrome, and headache.24–28 The concerns over patients’ coagulation status have never been reduced using SA.
The TAP block and/or RS block have been widely used as an analgesic option in postoperative abdominal analgesia.9–14 The TAP block is a regional technique for analgesia of the anterolateral abdominal wall, while the RS block has a better analgesic effect on midline incisions.14,15 With recent advances in ultrasound technology, TAP and RS blocks have become technically easier and safer. We observed that ESRD patients were less likely to be obese, which was conducive to ultrasound-guided puncture operation. Some patients were complicated by seroperitoneum, but this did not increase the difficulty in identifying abdominal muscles layers.
Over the past few years, some scholars had tried to apply the TAP block in PD catheter placement.7,8,16-20 In some reports, the TAP block was applied alone with a success rate of 87.50% (n=24), 91.7% (n=60) or 95% (n=20).7,8,18 However, the additional use of anesthetic drugs was needed to achieve a better analgesia or sedation effect. Eldawlatly and colleagues proposed that the combination of TAP and RS blocks provides a safe and effective anesthetic technique for patients undergoing laparoscopic PD catheter insertion.20 Wei Dai et al applied this combination of blocks for open PD catheter insertion with a 96.67% (n=30) success rate.16 They combined the two blocking methods to make it more suitable for PD catheter insertion, but they did not compare this type of anesthesia with other methods. In our setting, the TAP + RS block group achieved a 93.33% (n=30) success rate, similar to the SA group, while also taking less time to perform. The time needed to reach the target block range and the surgeon satisfaction were similar between the two groups.
We found that patients undergoing TAP + RS block anesthesia had more stable intraoperative hemodynamics and less need for vasoactive drugs. The TAP + RS block is considered to have no effect on sympathetic nerves and is suitable for ESRD patients.
In this study, the postoperative NRS score and tramadol dosage were lower in patients who received the TAP + RS block compared to those who received SA. This is consistent with the long duration of postoperative analgesia after TAP in previous studies.29 Our study also revealed earlier postoperative out-of-bed activity in the TAP + RS group since the blocks had no effect on the lower limbs. Meanwhile, early removal of the urinary catheter and the absence of lower limb dyskinesia was achieved. No urine retention occurred in either group. Less postoperative pain, earlier removal of the urinary catheter, and no effect on leg movement led to the patients in the TAP + RS group demonstrating greater satisfaction.
DEX was administered to maintain sedation during the operation because its pharmacokinetic parameters in patients with end-stage renal failure are similar to those in patients with normal renal function.30 More importantly, it has no adverse effect on kidney function. We used tramadol as rescue analgesia for postoperative analgesia insufficiency because nonsteroidal drugs are inappropriate for these patients, and potent opioids are not necessary because of the small incision.31 Due to the anatomical characteristics and the application of ultrasound, the TAP block and RS block are relatively safe and there have been no reports of local anesthetic toxicity related to either. Meanwhile, it has been shown that the plasma concentration of ropivacaine reaches a maximum of 2.5 µg/mL at 15 min after a bilateral TAP block with 120 mg (1.8 mg/kg, 0.15%, 40 mL each side) of ropivacaine in a 66-year-old man with cardiac failure and renal dysfunction, a concentration that was potentially neurotoxic.32 This suggests that we need to pay attention to the anesthesia management process in these patients. On the other hand, unilateral TAP combined with an RS block can reduce the amount of local anesthetics when compared to a bilateral TAP block as the RS is relatively narrow.
Our results reflect the treatment practices and patient characteristics of our single medical center. Nevertheless, some limitations of this study should be considered. First, postoperative hospital stay was not observed because numerous factors influence the length of hospital stay in such patients. Second, renal function was not tracked after surgery. The drugs used during the study theoretically do not affect renal function. However, more convincing results could be obtained if laboratory tests could be submitted. Lastly, our study contains a relatively small number of patients, which further limits definitive conclusions.
Conclusions
Collectively, TAP + RS block anesthesia can be used as an alternative to intraspinal anesthesia for PD catheter placement and is more conducive to intraoperative hemodynamic stability and postoperative analgesic effect maintenance as well as early activities and rehabilitation.
It is important to note that a TAP block combined with an RS block should be practiced as postoperative analgesia before being applied as an isolated anesthesia method because the effects of the technique are closely related to the anesthesiologist’s proficiency.
Data Sharing Statement
Data pertaining to this study, including the individual test results and demographic characteristics data, are available for sharing for a period of 3 years after the date of publication of this manuscript by writing to the corresponding authors.
Acknowledgment
The authors thank Fengxian Li (Department of Anesthesiology, Zhujiang Hospital, Southern Medical University, Guangdong Province, P. R. China) for her valuable input and help with writing the article.
Disclosure
The authors report no conflicts of interest for this work.
References
1. CKD in the United States. An overview of USRDS annual data report volume2. ESRD: United States renal data system. Available from: http://www.usrds.org/reference.aspx.
2. Boujelbane L, Fu N, Chapla K, et al. Percutaneous versus surgical insertion of PD catheters in dialysis patients: a meta-analysis. J Vasc Access. 2015;16(6):498–505. doi:10.5301/jva.5000439
3. Cox TC, Blair LJ, Huntington CR, et al. Laparoscopic versus open peritoneal dialysis catheter placement. Surg Endosc. 2016;30(3):899–905. doi:10.1007/s00464-015-4297-4
4. Crabtree JH, Chow KM. Peritoneal dialysis catheter insertion. Semin Nephrol. 2017;37(1):17–29. doi:10.1016/j.semnephrol.2016.10.004
5. Lund L, Jonler M. Peritoneal dialysis catheter placement: is laparoscopy an option? Int Urol Nephrol. 2007;39(2):625–628. doi:10.1007/s11255-007-9193-y
6. Zhu W, Jiang C, Yan X, Sun C, Zhang M. The ureteroscope-assisted “Mini-Perc” technique of placement of peritoneal dialysis catheters with a 16-fr peel-away sheath: 3-year results in 47 patients. Int Urol Nephrol. 2013;45(1):233–237. doi:10.1007/s11255-012-0229-6
7. Li Q, Tang X, Tao T, et al. A randomized controlled trial of comparing ultrasound-guided transversus abdominis plane block with local anesthetic infiltration in peritoneal dialysis catheter implantation. Blood Purif. 2018;45(1–3):8–14. doi:10.1159/000479083
8. Markic D, Vujicic B, Ivanovski M, et al. Peritoneal dialysis catheter surgery using transversus abdominis plane block. Perit Dial Int. 2017;37(4):429–433. doi:10.3747/pdi.2016.00198
9. Baeriswyl M, Kirkham KR, Kern C, Albrecht E. The analgesic efficacy of ultrasound-guided transversus abdominis plane block in adult patients: a meta-analysis. Anesth Analg. 2015;121(6):1640–1654. doi:10.1213/ANE.0000000000000967
10. Emile SH, Abdel-Razik MA, Elbahrawy K, et al. Impact of ultrasound-guided transversus abdominis plane block on postoperative pain and early outcome after laparoscopic bariatric surgery: a randomized double-blinded controlled trial. Obes Surg. 2019;29(5):1534–1541. doi:10.1007/s11695-019-03720-y
11. Faiz SHR, Nader ND, Niknejadi S, et al. A clinical trial comparing ultrasound-guided ilioinguinal/iliohypogastric nerve block to transversus abdominis plane block for analgesia following open inguinal hernia repair. J Pain Res. 2019;12:201–207. doi:10.2147/JPR.S179506
12. Gasanova I, Alexander J, Ogunnaike B, et al. Transversus abdominis plane block versus surgical site infiltration for pain management after open total abdominal hysterectomy. Anesth Analg. 2015;121(5):1383–1388. doi:10.1213/ANE.0000000000000909
13. Gulyam Kuruba SM, Mukhtar K, Singh SK. A randomised controlled trial of ultrasound-guided transversus abdominis plane block for renal transplantation. Anaesthesia. 2014;69(11):1222–1226. doi:10.1111/anae.12704
14. Primrose M, Al Nebaihi H, Brocks DR, et al. Rectus sheath single-injection blocks: a study to quantify local anaesthetic absorption using serial ultrasound measurements and lidocaine serum concentrations. J Pharm Pharmacol. 2019;71(8):1282–1290. doi:10.1111/jphp.13110
15. Xu L, Hu Z, Shen J, McQuillan PM. Efficacy of US-guided transversus abdominis plane block and rectus sheath block with ropivacaine and dexmedetomidine in elderly high-risk patients. Minerva Anestesiol. 2018;84(1):18–24. doi:10.23736/S0375-9393.17.11538-5
16. Dai W, Lu Y, Liu J, et al. Ultrasound-guided left lateral transversus abdominis plane block combined with rectus sheath block in peritoneal dialysis catheter placement. J Anesth. 2018;32(4):645–648. doi:10.1007/s00540-018-2528-2
17. Hecquet E, Bonamy C, Levesque C, et al. Peritoneal dialysis catheter insertion under TAP block procedure: a pilot study. Nephrol Ther. 2015;11(3):164–168. doi:10.1016/j.nephro.2015.01.005
18. Henshaw DS, Baker ML, Weller RS, Reynolds JW, Jaffe JD. Transversus abdominis plane block as the primary anesthetic for peritoneal dialysis catheter surgery. J Clin Anesth. 2016;31:182–188. doi:10.1016/j.jclinane.2016.01.028
19. Yamamoto H, Shido A, Sakura S, Saito Y. Monitored anesthesia care based on ultrasound-guided subcostal transversus abdominis plane block for continuous ambulatory peritoneal dialysis catheter surgery: case series. J Anesth. 2016;30(1):156–160. doi:10.1007/s00540-015-2074-0
20. Eldawlatly AA, Aldohayan A. Combined transversus abdominis plane block and rectus sheath block in laparoscopic peritoneal dialysis catheter insertion. Saudi J Anaesth. 2016;10(3):251–252. doi:10.4103/1658-354X.183403
21. Georgatzakou HT, Antonelou MH, Papassideri IS, Kriebardis AG. Red blood cell abnormalities and the pathogenesis of anemia in end-stage renal disease. Proteomics Clin Appl. 2016;10(8):778–790. doi:10.1002/prca.201500127
22. Kiefer MM, Ryan MJ. Primary care of the patient with chronic kidney disease. Med Clin North Am. 2015;99(5):935–952. doi:10.1016/j.mcna.2015.05.003
23. Kowark A, Rossaint R, Coburn M. General versus spinal anesthesia for the elderly hip fractured patient. Curr Opin Anaesthesiol. 2019;32(1):116–119. doi:10.1097/ACO.0000000000000679
24. Bos EME, Haumann J, de Quelerij M, et al. Haematoma and abscess after neuraxial anaesthesia: a review of 647 cases. Br J Anaesth. 2018;120(4):693–704. doi:10.1016/j.bja.2017.11.105
25. Chambers DJ, Bhatia K. Horner’s syndrome following obstetric neuraxial blockade - a systematic review of the literature. Int J Obstet Anesth. 2018;35:75–87. doi:10.1016/j.ijoa.2018.03.005
26. Hofer JE, Scavone BM. Cranial nerve VI palsy after dural-arachnoid puncture. Anesth Analg. 2015;120(3):644–646. doi:10.1213/ANE.0000000000000587
27. Lagerkranser M. Neuraxial blocks and spinal haematoma: review of 166 case reports published 1994–2015. Part 1: demographics and risk-factors. Scand J Pain. 2017;15(1):118–129. doi:10.1016/j.sjpain.2016.11.008
28. Lee JE, George RB, Habib AS. Spinal-induced hypotension: incidence, mechanisms, prophylaxis, and management: summarizing 20 years of research. Best Pract Res Clin Anaesthesiol. 2017;31(1):57–68. doi:10.1016/j.bpa.2017.01.001
29. Stoving K, Rothe C, Rosenstock CV, et al. Cutaneous sensory block area, muscle-relaxing effect, and block duration of the transversus abdominis plane block: a randomized, blinded, and placebo-controlled study in healthy volunteers. Reg Anesth Pain Med. 2015;40(4):355–362. doi:10.1097/AAP.0000000000000252
30. Zhong W, Zhang Y, Zhang MZ, et al. Pharmacokinetics of dexmedetomidine administered to patients with end-stage renal failure and secondary hyperparathyroidism undergoing general anaesthesia. J Clin Pharm Ther. 2018;43(3):414–421. doi:10.1111/jcpt.12652
31. Kimmel PL, Fwu C-W, Abbott KC, et al. Opioid prescription, morbidity, and mortality in united states dialysis patients. Clin J Am Soc Nephrol. 2017;28(12):3658–3670. doi:10.1681/asn.2017010098
32. Ishida T, Tanaka S, Sakamoto A, Hirabayashi T, Kawamata M. Plasma ropivacaine concentration after TAP block in a patient with cardiac and renal failure. Local Reg Anesth. 2018;11:57–60. doi:10.2147/LRA.S173877
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.