Back to Journals » Degenerative Neurological and Neuromuscular Disease » Volume 13
Ultrasound-Guided Percutaneous Neuromodulation in Multiple Sclerosis: A Case Report
Authors Javier-Ormazábal A , Herrero P , González-Platas M
Received 27 October 2022
Accepted for publication 18 January 2023
Published 27 January 2023 Volume 2023:13 Pages 15—20
DOI https://doi.org/10.2147/DNND.S395082
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Thomas Müller
Alberto Javier-Ormazábal,1,2 Pablo Herrero,3 Montserrat González-Platas1,2
1Neurology Service, Hospital Universitario de Canarias, Santa Cruz de Tenerife, Spain; 2Biomedicine Department, Universidad de Las Palmas de Gran Canaria, Las Palmas, Spain; 3IIS Aragon, Universidad de Zaragoza, Departamento de Fisiatría y Enfermería, Facultad de Ciencias de la Salud, Zaragoza, Spain
Correspondence: Pablo Herrero, IIS Aragon, Universidad de Zaragoza, Departamento de Fisiatría y Enfermería, Facultad de Ciencias de la Salud, C/ Domingo Miral s/n CP, Zaragoza, 50009, Spain, Email [email protected]
Abstract: Multiple sclerosis is a degenerative inflammatory disease that causes different musculoskeletal problems. Its impact has led to the study of treatment alternatives such as the use of invasive physiotherapy. In this study, we analyze the effects of ultrasound-guided percutaneous neuromodulation to a 51-year-old man suffering from multiple sclerosis and an associated hemiparesis in the left upper limb. A dry needling needle was placed in contact with the median nerve under ultrasound guidance and 10 trains of 10 seconds of electrostimulation with a frequency of 10 Hz and an impulse width of 240 μs were applied, with 10 seconds of pause between them. There was a significant improvement in the grip strength immediately after the treatment which increased progressively at 24 hours and at 4 days follow-up. There was also an improvement in the hand function, with a decrease in the time necessary to perform the 9 Hole Peg Test immediately after the treatment, which was maintained at 24 hours and at 4 days follow-up. Future studies with larger samples are needed to further test the effects of this invasive physiotherapy technique as well as its possible applications to other neurological conditions.
Keywords: physiotherapy, multiple sclerosis, dry needling, percutaneous neuromodulation
Introduction
Multiple sclerosis (MS) is the most common inflammatory neurological disease in young adults. The mean age of diagnosis is about 30 years, with most patients having recurrent neurological relapses. After typically 10–20 years, many of those affected develop a “progressive” clinical course.1 The estimated prevalence in Europe is 83 cases per 100,000 population, with an average incidence of 4.3 cases per 100,000.2
Symptoms of MS include fatigue, poor balance, pain, muscle weakness, and spasticity, amongst others. There is not much evidence about hand dysfunction in people with MS and how it may impact the individual’s quality of life, especially in those with more advanced disease. Hand dysfunction, which includes both strength and coordination as functional dimensions, has been associated with increased unemployment and therefore it becomes a main challenge in rehabilitation.3
In the case of rehabilitation treatments, on the one hand, different types of neuromodulation treatments such as intrathecal baclofen pump, functional electrical stimulation, deep brain stimulation, transcranial magnetic stimulation, bladder stimulation, spinal cord stimulation and brain–computer interface have been applied to treat MS-related symptoms.4 On the other hand, minimally invasive techniques such as dry needling (DN) have shown to have neuromodulating effects at different levels in the CNS5,6 and achieve positive changes in patients with MS, concretely in spasticity, quality of life, walking capacity and manual dexterity.7
In the case of functional electrical stimulation, this is applied transcutaneously to restore function and prevent muscle atrophy (e.g stimulation of the common peroneal nerve in patients with foot drop). Although the use of transcutaneous electrical stimulation has been widely used in MS patients, the effects of this stimulation performed percutaneously has been only researched in musculoskeletal conditions,6 showing to be effective to treat chronic lateral epicondylalgia,7 chronic low back pain8 and to decrease postoperative pain following ambulatory rotator cuff repair.9
Percutaneous Neuromodulation (PN), which is recommended to be applied ultrasound-guided, consists of the application of low or medium frequency percutaneous electrical stimulation seeking a sensory and/or motor response with an acupuncture-like needle, with an electrode placed near the nerve or muscle motor point.10 Considering the evidence supporting the use of ultrasound guided PN in different musculoskeletal conditions and the effectiveness of transcutaneous electrical stimulation in neurological patients, we aimed to analyze if PN may be effective to treat MS-related symptoms and become a potential treatment for this type of patients. Therefore, the objective of this case report was to analyze if a single session of ultrasound-guided PN may have any effects on grip force and hand function and how long these effects last.
Case Report
Case description: The patient is a 51-year-old Caucasian man diagnosed with herpetic encephalitis at 10 years of age and a left cerebellar stroke in 2006, with demyelinating disease type submissive-recurrent MS since 2007. His last outbreak occurred in November 2020 after receiving seven months of outpatient rehabilitation. Patients with multiple sclerosis who were previously treated in the rehabilitation service in the past years were invited to participate in a clinical trial to research the effectiveness of ultrasound-guided percutaneous neuromodulation. This patient was recruited in January 2022 and he didn´t receive any other treatments since November 2020. He had a left hemiparesis, with a muscular strength of 3 points in the Daniels-5-point scale in the left finger flexor muscles, with hypoesthesia in the 4th and 5th left hand finger, as well as dystonia in some postures.
Intervention: A single application of ultrasound-guided PN was performed. DN needles (APS, 0.30×40 with guide), an electro stimulator (Globus Génesy SII) and an imaging ultrasound device (Logiq e, GE) were used (Figure 1). The median nerve was stimulated between the two fascicles of the pronator teres muscle (Figure 2), using 10 trains of 10 seconds of electrostimulation and 10 seconds of pause between them, with a frequency of 10 Hz and an impulse width of 240 µs, as established by the protocol of Valera-Garrido et al11 The procedure was performed at the University Hospital of the Canary Islands after being approved by the Ethical Committee (code NEUROECO-2020) and after having signed the informed consent. This clinical trial was registered with reference NCT05053984 at www.clinicaltrials.gov. Informed consent was obtained from the patient to publish the case report.
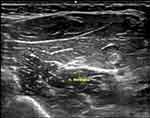 |
Figure 1 Use of ultrasound to locate the median nerve for the application of the percutaneous neuromodulation intervention. |
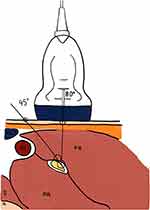 |
Figure 2 Needle approach to contact the median nerve. |
Assessment: The grip strength was measured with a dynamometer (KERN MAP 130K1, Figure 3) whereas the hand function was measured with the nine hold peg test (9HPT) (Figure 4). Three measurements were taken, choosing the highest value in the case of the grip force and the lowest one for hand function. Measurements were taken at baseline, immediately after, 24 hours and four days after the intervention.
 |
Figure 3 Dynamometry test. |
 |
Figure 4 9 Hole Peg test. |
Results
There was a significant improvement in the grip strength immediately after the treatment from baseline (5.5 Kg), which increased progressively at 24 hours (12.4 kg) and 4 days follow-up (14.1 Kg) (Table 1). There was also an improvement in hand function, with a decrease in the time necessary to perform the 9HPT of 10.61 seconds immediately after the treatment, which was maintained at 24 hours (11.87 seconds) and at 4 days follow-up (9.77 seconds) (Table 1).
 |
Table 1 Measures of Grip Force and Hand Function |
Discussion
The study results show that a single session of ultrasound-guided PN may be effective to improve hand function and grip strength in a patient with MS. Although no cause-effects can be established as this was only a case report, this study is the first one researching the effects of percutaneous needle stimulation on a peripheral nerve in a neurological patient. Grip strength followed improving at 24h and 4 days after the intervention whereas hand function improvements remained stable over time. These results are similar to the results reported by Álvarez-Prats et al in 13 patients with knee pathology, where an increase of maximal isometric strength was observed after the application of ultrasound-guided percutaneous neuromodulation of the femoral nerve.12
However, our results were not in line with a study carried out in a patient with post-stroke tremor13 and post-stroke spasticity,14 where the peak effects were achieved immediately after the DN intervention but did not maintain over time. This could be because the DN treatment performed in the aforementioned cases has demonstrated to have a mechanical effect, working through the mechanical disruption of dysfunctional endplates, which are reinnervated after 3 days,15 whereas PN acts at a central level, without mechanical effects. However, DN performed at Myofascial Trigger Points is also known to have indirect effects in the central nervous system,5,6 which makes it difficult to formulate any hypothesis that may explain the differences found. Because of this, future studies should compare different invasive techniques such as DN and PN to better understand both the effects they have and how long they last. Neuromodulation is a quality of the nervous system that regulates or modifies electrical impulses, by enhancing or inhibiting them. Even though its mechanism of action is yet to be fully determined, it is known that it lies on the ascending and descending pathways and the supraspinal regions of the central nervous system. The chronic effect of said neurophysiological process may cause central nervous system plasticity.16
Although minimally invasive techniques such as acupuncture or dry needling may have minor adverse effects such as mild bleeding, bruising, and pain,17 in this patient there were no adverse effects when performing this treatment. This was possibly due to the ultrasound-guided procedure, which decreases the risk of needling a blood vessel as well as needling the nerve. The specificity of this technique depends on placing the needle close to the nerve that is going to be stimulated without needling it, as this may provoke pain, tingling and have the risk of damaging the nerve.
This study has some strengths, such as being the first study researching the effect of PN in a patient with MS. However, it also has some limitations, which impede the generalization of results, as this is a case report and does not have a control. In this study, it was only analyzed the effects of a single session of PN, and therefore it is necessary that future studies analyze the effects of different number of PN sessions and the cost-effectiveness of them, similarly to what has been performed in other studies related to tendinous injuries,18 myofascial pain19 and spasticity,20,21 or compared to pharmacological alternatives such as BTX A.22 Moreover, future studies should be developed with larger samples and with other neurological populations, analyzing also the effect of PN as part of a multimodal treatment, in order to understand which is the additional improvement that PN may achieve in patients with neurological impairments.
Data Sharing Statement
The data that support the findings of this study are available from the first author [AJO], upon reasonable request by email to [email protected]. Clinical deidentified participant data will be made available after the manuscript publication.
Ethical Approval and Informed Consent
This study was approved by the Committee of Ethics of Hospital Universitario de Canarias, Canary Islands, Spain with code NEUROECO-2020 and is compliance with the declaration of Helsinki. The participant signed informed consent.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This research received no external funding.
Disclosure
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
1. Reich D, Lucchinetti CF, Calabresi PA. Multiple sclerosis. N Engl J Med. 2018;378:169–180. doi:10.1056/NEJMra1401483
2. Kingwell E, Marriott JJ, Jetté N, et al. Incidence and prevalence of multiple sclerosis in Europe: a systematic review. BMC Neurol. 2013;13:128. doi:10.1186/1471-2377-13-128
3. Julian LJ, Vella L, Vollmer T, et al. Employment in multiple sclerosis. J Neurol. 2008;255(9):1354–1360. doi:10.1007/s00415-008-0910-y
4. Abboud H, Hill E, Siddiqui J, Serra A, Walter B. Neuromodulation in multiple sclerosis. Multiple Sclerosis J. 2017;23(13):1663–1676. doi:10.1177/1352458517736150
5. Calvo S, Navarro J, Herrero P, Del Moral R, De Diego C, Marijuán P. Electroencephalographic changes after application of dry needling [DNHS© technique] in two patients with chronic stroke. MYOPAIN. 2017;23:3–4. doi:10.1080/24708593.2017.1291550
6. Asadi B, Fard KR, Ansari NN, Marco Á, Calvo S, Herrero P. The effect of dry needling in chronic stroke with a complex network approach: a case study. Clin EEG Neurosci. 2022;15500594221120136. doi:10.1177/15500594221120136
7. Del Pilar Pérez-Trujillo M, González-Platas M, Pérez-Martín MY, Revert-Gironés MC, González-Platas J. Dry needling for treating spasticity in multiple sclerosis. J Phys Ther Sci. 2021;33(7):505–510. doi:10.1589/jpts.33.505
8. San-Emeterio-Iglesias R, Minaya-Muñoz F, Romero-Morales C, De-la-Cruz-Torres B. Correct sciatic nerve management to apply ultrasound-guided percutaneous neuromodulation in patients with chronic low back pain: a pilot study. Neuromodulation. 2021;24(6):1067–1074. doi:10.1111/ner.1339
9. Ilfeld BM, Finneran JJ, Gabriel RA, et al. A proof-of-concept study. Reg Anesth Pain Med. 2019;44(3):310–318. doi:10.1136/rapm-2018-100121
10. de la Cruz-Torres B, Barrera-García-Martín I, Albornoz-Cabello M. Immediate effects of ultrasound-guided percutaneous neuromodulation versus physical exercise on performance of the flexor hallucis longus muscle in professional dancers: a randomised clinical trial. Acupunct Med. 2019;37(2):91–97. doi:10.1177/0964528419826103
11. Valera-Garrido F, Minaya-Muñoz F. Fisioterapia Invasiva [Invasive Physiotherapy]. Barcelona, España: Elsevier SL; 2016.
12. Álvarez-Prats D, Carvajal-Fernández O, Pérez-Mallada N, et al. Changes in maximal isometric quadriceps strength after the application of ultrasound-guided percutaneous neuromodulation of the femoral nerve: a case series. J Invasive Tech Phys Ther. 2019;2:39–45.
13. Ortín JA, Bravo-Esteban E, Ibáñez J, Herrero P, Gómez-Soriano J, Marcén-Román Y. Effects of deep dry needling on tremor severity and functionality in stroke: a case report. Healthcare. 2020;9(1):5. doi:10.3390/healthcare9010005
14. Calvo S, Quintero I, Herrero P. Effects of dry needling (DNHS technique) on the contractile properties of spastic muscles in a patient with stroke: a case report. Int J Rehabil Res. 2016;39:372–376. doi:10.1097/MRR.0000000000000185
15. Domingo A, Mayoral O, Monterde S, Santafé MM. Neuromuscular damage and repair after dry needling in mice. Evid Based Complement Alternat Med. 2013;2013:260806. doi:10.1155/2013/260806
16. Fidalgo-Martin I, Ramos-álvarez JJ, Murias-Lozano R, Rodríguez-López ES. Effects of percutaneous neuromodulation in neuromusculoskeletal pathologies: a systematic review. Medicine. 2022;101(41):e31016. doi:10.1097/MD.0000000000031016
17. Boyce D, Wempe H, Campbell C, et al. Adverse events associated with therapeutic dry needling. Int J Sports Phys Ther. 2020;15(1):103–113. doi:10.26603/ijspt20200103
18. Fernández-Sanchis D, López-Royo MP, Jiménez-Sánchez C, Herrero P, Gómez-Barrera M, Calvo S. A comparative study of treatment interventions for patellar tendinopathy: a secondary cost-effectiveness analysis. Acupunct Med. 2022;40:516–523. doi:10.1177/09645284221085283
19. Fernández D, Al-Boloushi Z, Bellosta-López P, Herrero P, Gómez M, Calvo S. Cost-effectiveness of two dry needling interventions for plantar heel pain: a secondary analysis of an RCT. Int J Environ Res Public Health. 2021;18(4):1777. doi:10.3390/ijerph18041777
20. Fernández-Sanchis D, Brandín-de la Cruz N, Jiménez-Sánchez C, Gil-Calvo M, Herrero P, Calvo S. Cost-effectiveness of upper extremity dry needling in chronic stroke. Healthcare. 2022;10:160. doi:10.3390/healthcare10010160
21. Fernández Sanchis D, Cuenca Zaldívar JN, Calvo S, Herrero P, Gómez Barrera M. Cost-effectiveness of upper extremity dry needling in the rehabilitation of patients with stroke. Acupunct Med. 2022;40(2):160–168. doi:10.1177/09645284211055750
22. Fernández D, Pujol C, Ruber C, et al. Economics of dry needling and botulinum toxin type A for treatment of post-stroke spasticity: a review. Explor Neuroprot Ther. 2022;2:131–140. doi:10.37349/ent.2022.00024
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
