Back to Journals » Open Access Rheumatology: Research and Reviews » Volume 12
Treatment with Repository Corticotropin Injection in Patients with Rheumatoid Arthritis, Systemic Lupus Erythematosus, and Dermatomyositis/Polymyositis
Authors Ho-Mahler N , Turner B, Eaddy M , Hanke ML, Nelson WW
Received 19 September 2019
Accepted for publication 14 January 2020
Published 20 February 2020 Volume 2020:12 Pages 21—28
DOI https://doi.org/10.2147/OARRR.S231667
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Chuan-Ju Liu
Nancy Ho-Mahler,1 Beni Turner,2 Michael Eaddy,2 Mark L Hanke,1 Winnie W Nelson1
1Mallinckrodt Pharmaceuticals, Bedminster, NJ, USA; 2Xcenda, Palm Harbor, FL, USA
Correspondence: Nancy Ho-Mahler
Mallinckrodt Pharmaceuticals, 1425 U.S. Route 206, Bedminster, NJ 07921, USA
Tel +1 747 239 0007
Fax +1 314 654 9440
Email [email protected]
Purpose: Repository corticotropin injection (RCI) is indicated for a number of autoimmune-mediated diseases including rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), and dermatomyositis (DM)/polymyositis (PM). To better understand the practice patterns and outcomes of RCI in patients with RA, SLE, or DM/PM, we conducted a retrospective medical record analysis.
Patients and Methods: Participating providers selected deidentified medical records of patients meeting the inclusion criteria (age ≥ 18 years; physician-reported diagnosis of RA, SLE, or DM/PM; initiation of treatment with RCI between 1/1/2011 and 2/15/2016; ≥ 3 in-office visits with same site/provider). Collected data spanned 12 months before and after the first prescription date for RCI. Analyses included patient demographics and clinical history, RCI treatment patterns, and physician’s impression of change.
Results: Data from 54 patients with RA, 30 patients with SLE, and 8 patients with DM/PM were analyzed. The most frequently reported reasons for initiating RCI were lack of efficacy with prior treatment, acute exacerbation of disease, and use as add-on to ongoing therapy. The most common initial RCI dosing, 80 U twice weekly, was used for 84% of patients with RA, 75% with SLE, and 86% with DM/PM. The mean duration of treatment was 4.8, 6.5, and 6.8 months for RA, SLE, and DM/PM, respectively. Among the 57 patients with data on physician’s impression of change with RCI, 78.1% of patients with RA, 94.7% with SLE, and 66.7% with DM/PM had a rating of “improved,” and the mean time to best impression of change was 3.4, 4.3, and 3.4 months for RA, SLE, and DM/PM, respectively.
Conclusion: This study reports the real-world patient profile, use patterns, and outcomes of patients who used RCI for the treatment of RA, SLE, and DM/PM. These data can inform appropriate use and clinical expectations when using RCI.
Keywords: adrenocorticotropic hormone, analogue, ACTH, immunomodulatory, real-world patients
Introduction
The prolonged-release adrenocorticotropic hormone (ACTH) analogue repository corticotropin injection (RCI; Acthar Gel®, Mallinckrodt Pharmaceuticals, Bedminster, NJ, USA) acts through multiple mechanisms of action, including anti-inflammatory and immunomodulatory processes.1–3 RCI is approved by the US Food and Drug administration for 19 indications, including as adjunct therapy for short-term administration in rheumatoid arthritis (RA) and for exacerbation or maintenance therapy in selected cases of systemic lupus erythematosus (SLE) and the inflammatory myopathies dermatomyositis (DM) and polymyositis (PM).1
With reported incidence rates ranging from 31 to 45 per 100,000 person-years, RA is one of the most common human autoimmune diseases in North America.4 According to a conservative estimate, RA affected 1.28 to 1.36 million adults in the United States in 2014.5 The primary goal for the management of patients with RA is achievement of disease remission through initial treatment with synthetic disease-modifying antirheumatic drugs (DMARDs) with escalation to biological DMARDs.6,7 Published studies of RCI in patients with refractory RA support its efficacy and safety in this population.8,9
Approximately 320,000 people in the United States have definite or suspected SLE.10 Reported incidence rates for SLE in North America range from 1.2 to 8.7 per 100,000 person-years,4 and prevalence rates in studies of US populations range from 5 to 241 per 100,000 people.11 The complex management of patients with SLE entails treatment with hydroxychloroquine to reduce disease flares and improve symptoms as well as administration of low-dose glucocorticoids, immunosuppressive agents, and cytotoxic drugs to address different types of organ system involvement.12 RCI has been used successfully to treat patients with SLE exacerbations not controlled by conventional treatments.13–17
In the United States, DM has a prevalence of 1 to 6 per 100,000 persons, and PM ranges from the largest fraction with approximately 10 per 100,000 persons in the United States down to the rarest condition that should only be diagnosed by exclusion.18 Glucocorticoids are used for first-line treatment of patients with DM/PM, followed by (or in conjunction with) immunosuppressive maintenance; in case of intolerance or insufficient response, oral cyclosporine or intravenous immunoglobulin G may be used and, upon relapse or interstitial lung disease, rituximab or cyclophosphamide may be considered.18 Given that DM/PM is a rare disease, it is not surprising that there are a small number of published reports with results on the clinical efficacy and safety of RCI in patients with refractory DM/PM.19–21
RCI is not a common medication used for inflammatory conditions. In clinical settings, it is often used after other therapies have failed.22,23 Due to this unique place in therapy, the literature contains very little information about the profile of patients who would benefit most from RCI therapy or how the therapies are used. Therefore, it is imperative to describe these aspects of real-world data to help ensure appropriate use.
Additional data are desired to inform clinical decisions regarding the use of RCI to treat patients with RA, SLE, or DM/PM. We performed a retrospective medical record analysis to gain more insights into the practice patterns and outcomes associated with real-world RCI treatment in these patient populations.
Patients and Methods
Study Design
We contacted potential study sites in the United States to assess their interest and ability to participate in this study of adults with RA, SLE, or DM/PM treated with RCI. After central Institutional Review Board (IRB) approval, we asked providers at each participating site to provide deidentified medical records of all patients meeting the study protocol general inclusion criteria. Trained study abstractors entered prespecified data from each provided patient medical record into an electronic data capture system. Collected data included patient demographics, disease history, treatment history (treatments administered before, at the start of, and during RCI treatment), and dosing information for RCI and concomitant treatments. These abstracted real-world data spanned the 12 months prior (preindex period) and up to 12 months following (follow-up period) the first prescription date for RCI treatment.
The Chesapeake IRB reviewed and approved this study. The study was conducted in accordance with the principles of the Declaration of Helsinki and was consistent with International Conference on Harmonization Good Clinical Practices and with applicable regulations. Given the retrospective nature of the study, the IRB determined that patient informed consent was not required.
Patients
Patient inclusion criteria were age ≥18 years; a physician-reported diagnosis of RA, SLE, or DM/PM according to American College of Rheumatology (ACR) criteria; initiation of treatment with RCI between January 1, 2011, and February 15, 2016; and ≥3 in-office visits at the same site and with the same healthcare provider, including at least 1 visit before RCI initiation, 1 visit at first RCI prescription, and 1 visit following the first RCI prescription (within at least 12 months and including ≥1 visit during RCI treatment). Patients with RA or DM/PM had to have used RCI as adjunctive therapy. Patients with SLE had to have undergone RCI treatment during an exacerbation or as maintenance therapy.
Patients were excluded if they had not started active treatment with RCI or if they had a known allergy to RCI or any of its components; if they had treatment contraindications (eg, due to recent surgery, sensitivity to proteins of porcine origin); if they had previously participated or were currently participating in an interventional clinical trial for SLE, RA, or DM/PM; or if they were pregnant. Patients with a diagnosis of stage V or VI membranous or sclerosing lupus nephritis or central nervous system lupus were also excluded from the study.
Endpoints
We defined the index visit as the date of the first prescription for RCI. The follow-up period started the day after this index visit and continued for up to 12 months, including RCI treatment cessation, loss to follow-up, death, or the end of the study period. The study collected data on patient profiles (baseline demographic, clinical, and socioeconomic characteristics), RCI treatment patterns (dose, frequency, duration, and concomitant medications), and physicians’ impression of change as well as adverse events (AEs) and serious adverse events (SAEs) that resulted in dose reduction or treatment discontinuation.
Statistics
Because of the descriptive nature of the study objectives, the sample size was not based on formal statistical hypothesis testing. The sample size was planned to be a maximum of 70 patients with RA, SLE, or DM/PM. All patients who met the eligibility criteria were included in the analysis.
We presented descriptive statistics as mean and standard deviation (SD) for continuous data and number of patients and percentage for categorical variables. Before analysis, clinical data received from participating sites were validated for accuracy and completeness. Patients were considered to be lost to follow-up if they switched treatment sites during the follow-up period.
Results
Patients
From 14 participating US clinic sites, we obtained data for 92 patients, including 54 with RA, 30 with SLE, and 8 with DM/PM. Table 1 lists the baseline demographic and clinical characteristics of these 3 populations. Overall, patients had a mean age over 49 years, and most were women (78% for RA, 97% for SLE, and 100% for DM/PM). The mean (±SD) follow-up time was 8.5±3.3 months for patients with RA, 9.1±2.6 months for SLE, and 10.3±2.6 months for DM/PM.
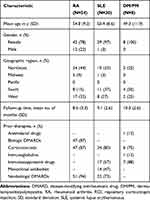 |
Table 1 Baseline Demographic and Clinical Characteristics of Patients with RA, SLE, or DM/PM Who Were Treated with RCI |
Among the 42 of 54 (78%) patients in the RA group with data, 32 (76%) had rheumatoid factor-positive disease. The most common comorbidities among patients with RA included arthralgia (96%), osteoarthritis (39%), hypertension (33%), depression (30%), and anxiety (28%). The most commonly reported symptoms among patients with RA were stiffness/pain in joints (95%), swollen joints (87%), morning stiffness (82%), pain (82%), swollen fingers (78%), fatigue (63%), muscle weakness (44%), and rash (33%).
Among the 30 patients with SLE, the most common comorbidities were arthralgia (100%), hypertension (40%), depression (33%), Sjogren’s syndrome (33%), and anxiety (27%). The most commonly reported symptoms were pain (93%), stiffness/pain in joints (90%), fatigue (87%), swollen joints (67%), muscle weakness (63%), morning stiffness (50%), shortness of breath (50%), and headaches (47%).
Among the 8 patients with DM/PM, 5 (62%) had PM and 3 (38%) had DM. The most commonly reported comorbidities among the DM/PM patients were arthralgia (100%), anxiety (50%), depression (50%), and osteoarthritis (38%). The most commonly reported symptoms were muscle weakness (88%), pain (88%), stiffness/pain in joints (88%), edema (63%), weight changes (50%), dizziness (50%), fatigue (50%), and morning stiffness (50%).
Before receiving RCI, most patients with RA had been treated with nonbiologic DMARDs (94%), corticosteroids (87%), and biologic DMARDs (87%); most patients with SLE had been treated with corticosteroids (80%), nonbiologic DMARDs (73%), and immunosuppressive drugs (57%); and most patients with DM/PM had been treated with immunosuppressive drugs (88%) and corticosteroids (75%).
RCI Treatment Patterns
Table 2 lists the reasons for initiating treatment with RCI. Across the 3 populations, the most commonly reported reasons for initiating RCI therapy were lack of efficacy with prior treatment (12% to 30% of patients), acute exacerbation/flare of disease (12% to 26%), and as add-on therapy to prior treatment (20% to 25%). In all 3 populations, the most common starting dose and frequency were 80 U twice weekly, which was used in 84% of patients with RA, 75% of patients with SLE, and 86% of patients with DM/PM (Table 3).
 |
Table 2 Reason for Initiating RCI Treatment in Patients with RA, SLE, or DM/PM |
 |
Table 3 RCI Treatment Patterns in Patients with RA, SLE, or DM/PM |
Less than half of the patients had any RCI dose changes during treatment, including 33% of patients with RA, 43% of patients with SLE, and 25% of patients with DM/PM. The most common dose changes were dose decreases, which occurred in 33% of patients with RA, 37% of patients with SLE, and 12% of patients with DM/PM. Among patients with RA, none had >1 dose increase and 9% had >1 dose decrease. Among patients with SLE, 3% had >1 dose increase and none had >1 dose decrease. None of the patients with DM/PM had >1 dose increase or decrease (Table 3).
The mean ±SD duration of RCI treatment was 4.8±3.4 months for patients with RA, 6.5±3.3 months for patients with SLE, and 6.8±2.2 months for patients with DM/PM (Table 3). RCI treatment was discontinued in 24 (44%) of RA patients, 6 (20%) of SLE patients, and 5 (63%) of DM/PM patients. The most common reasons for discontinuation of RCI treatment for RA were AEs related to RCI (38%), disease resolution/remission/treatment no longer necessary (17%), and physician choice (17%); for SLE, the reasons were treatment-related AEs (50%); and for DM/PM, the reasons were AEs related to RCI (40%) and lack of efficacy/inadequate response/disease progression (40%).
Physician Assessment of Efficacy
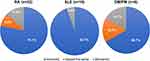
Physician’s impression of change data were available for 32 (59%) patients with RA, 19 (63%) with SLE, and 6 (75%) with DM/PM. As shown in Figure 1, physicians reported a rating of “improved” for 78.1% of patients with RA, 94.7% with SLE, and 66.7% with DM/PM. The mean time to best impression of change was 3.4±2.5 months for RA, 4.3±2.7 for SLE, and 3.4±1.6 for DM/PM.
Safety
SAEs were reported in 1 (2%) patient with RA, 4 (13%) with SLE, and 1 (12%) with DM/PM. AEs were reported in 14 (26%) patients with RA, 5 (17%) with SLE, and 2 (25%) with DM/PM. Each AE and SAE occurred in 1 patient in the RA, SLE, and DM/PM groups. The AEs reported in patients with RA included abdominal angina, bruising, diaphoresis, fatigue, gastroenteritis, heartburn, herpes flare, hypoglycemia, hypotension, injection-site reaction, irritability or mood changes, myalgia, upper respiratory infection, vomiting, and weight gain. The 1 patient with RA who had an SAE experienced both pancreatitis and urinary tract infection. AEs reported in patients with SLE included diarrhea, gastrointestinal upset, edema, neuralgia, pneumonia, and weight gain. The SAEs reported in 4 patients with SLE included altered musculoskeletal pain, dehydration, adrenal insufficiency, pneumonia, renal failure, and transient ischemic attack. AEs reported by patients with DM/PM included bruising and red or bloodshot eyes. The 1 patient with DM/PM who had an SAE experienced shortness of breath.
Discussion
In this retrospective analysis of RCI treatment in 92 patients with RA, SLE, or DM/PM, we found that the time lag in RCI treatment from prescription to first dose ranged between 1.5 and 2.4 months. The most frequently reported reasons for initiating RCI were lack of efficacy with prior treatment, acute exacerbation or flare of disease, and use as an add-on to ongoing therapy. Among patients with available prescribing information, the most common initial RCI dosing was 80 U twice weekly, which was used for 84% of patients with RA, 75% with SLE, and 86% with DM/PM. Patients with RA had the shortest duration of RCI treatment (4.8 months), while patients with SLE and DM/PM experienced similar duration of RCI treatment (6.5 and 6.8 months, respectively). Among the 57 patients with data on physician impression of change with RCI, 78.1% of patients with RA, 94.7% with SLE, and 66.7% with DM/PM had a rating of “improved.” The mean time for the best impression of change was 3.4±2.5 months for RA, 4.3±2.7 for SLE, and 3.4±1.6 for DM/PM. RCI was generally safe and well tolerated, with AE rates of 26%, 17%, and 25% and SAE rates of 2%, 13%, and 12% in patients with RA, SLE, and DM/PM, respectively.
Most patients in this retrospective analysis had received multiple therapies before starting treatment with RCI, suggesting that they had refractory disease. Among the patients with RA, 94% had been treated with nonbiologic DMARDs, 87% with corticosteroids, and 87% with biologic DMARDs. Among patients with SLE, 80% had been treated with corticosteroids, 73% with nonbiologic DMARDs, and 57% with immunosuppressive drugs. Among patients with DM/PM, 88% had been treated with immunosuppressive drugs and 75% with corticosteroids. The patients with RA in our study may be similar to the population participating in an ongoing prospective study of RCI in patients with persistently active RA despite treatment with 1 or 2 nonbiologic or biologic DMARDs and corticosteroids (NCT02919761).24 In this 2-part clinical trial, all patients receive RCI 80 U twice weekly for 12 weeks, followed by double-blind randomization to 12 weeks of maintenance therapy with RCI or placebo. According to results presented at the European League Against Rheumatism (EULAR) 2019 annual meeting, the randomized, placebo-controlled, double-blind phase of this 2-part clinical trial demonstrated that significantly more patients with persistently active RA who met response criteria at Week 12 maintained low disease activity (LDA) when treated with RCI (62%) than with placebo (43%) at Week 24. In addition, significantly more patients in the RCI group (86%) than in the placebo group (66%) had sustained LDA at Week 24 as defined by the Clinical Disease Activity Index (CDAI), and the cumulative disease activity flare rate at Week 24 was significantly lower in the RCI continuation group (17%) than in the placebo group (30%). Another key finding of this trial was that the proportion of patients who achieved ACR 20%, 50%, and 70% criteria at Week 24 were 91%, 75%, and 47%, respectively, for the RCI group, and 84%, 70%, and 42%, respectively, for the placebo group. AEs were reported in 33% of the RCI group and 40% of the placebo group. AEs of interest included diabetes mellitus (RCI 1%, placebo 0%), increase in glycosylated hemoglobin (RCI 1%, placebo 3%), hypergyclemia (RCI 4%, placebo 3%), and hypertension (RCI 4%, placebo 0%). These AEs were consistent with those in previous trials of RCI.
There are few published clinical studies of RCI in patients with RA. A small single-center prospective trial in 8 adults with active RA refractory to ≥3 therapies with different mechanisms of action examined the effects of 12 weeks of RCI (80 U twice weekly) used as add-on therapy.8 The dose used in this trial, 80 U twice weekly, was the most commonly used in our study. The investigators reported achievement of the primary endpoint of improvements in swollen (P=0.0047) and tender joint counts (P=0.004), assessed using the Ritchie-Camp Articular Index, at the end of the 12-week treatment period and the loss of these gains 4 weeks after termination of RCI treatment. One patient had 2 SAEs, pulmonary embolism and deep vein thrombosis, requiring hospitalization. Similar to the data presented here, none of the other AEs were experienced by >1 patient. A 12-week prospective study of RCI in refractory RA reported that 6 of 9 patients achieved the primary endpoint of a >1.2 point reduction in DAS28 using C-reactive protein (DAS28-CRP) at Week 12 versus baseline.9 After starting on RCI 40 U daily for 7 days, patients with an adequate response switched to 40 U RCI twice weekly and those with an inadequate response took 80 U RCI daily for 7 days, followed by 80 U twice weekly through Week 12. There were no SAEs, and AEs included cushingoid features (2 patients) and mild hyperglycemia, herpes zoster, and pedal edema (1 patient each).
A handful of other studies have reported results of RCI treatment in patients with SLE. In a retrospective case series, 7 of 9 women with SLE treated with RCI and followed for 3 to 6 months had improvements measured by the ability to taper steroids, decreases in Systemic Lupus Erythematosus Disease Activity Index 2000 (SLEDAI-2K) score, and the extent of unresolved active clinical manifestation.13 These patients had received RCI 80 U biweekly dosing, and none had reported AEs while receiving RCI therapy. A short-term, 10-day trial of daily RCI 80 U in 10 women with SLE (exhibiting symptomatic SLE exacerbations despite conventional treatments) noted no SAEs and statistically significant improvements (P<0.05) at the 14-day and 28-day follow-up visits in different measures including SLEDAI-2K.14 All 5 participants in the 28-day trial who elected to enter an extension study of biweekly RCI 80 U experienced statistically significant improvements in various measures including SLEDAI-2K (P=0.0007), tender joint counts (P=0.0020), and swollen joint counts (P=0.0041) at 6 months.15 These patients experienced no clinically significant changes in mean systolic and diastolic blood pressure, and no other minor or major AEs were reported. In a Phase 4 pilot study, 38 patients with SLE were randomized 2:1:2:1 to 8 weeks of double-blind, placebo-controlled treatment with RCI (40 or 80 U or volume-matched placebo, once every other day) followed by a 44-week open-label extension phase.16 While this exploratory study found that numerical differences were not statistically significant in the primary endpoint, a composite responder index, secondary endpoints and post hoc analyses linked RCI treatment to sustained improvements in SLE signs and symptoms and a tolerable safety profile.16,17 These results supported the design of a well-powered, ongoing, randomized, double-blind placebo-controlled study (NCT02953821) to evaluate 24 weeks of treatment with RCI in patients with persistently active SLE despite treatment with moderate-dose corticosteroids.
In addition to our retrospective analysis, 4 other studies have evaluated RCI treatment in patients with DM/PM. A retrospective case review noted that symptoms improved in women with DM/PM treated for 12 weeks with concomitant RCI 80 U twice weekly (n=4) or once weekly (n=1).19 The investigators reported that none of the patients had significant AEs. An interim analysis of the Acthar in Dermatomyositis and Polymyositis Treatment (ADAPT) registry reported responses to RCI treatment in 14 (58%) of 24 enrolled patients with refractory DM/PM.20 Most of the patients (92%) received RCI 80 U twice weekly, and the mean duration of RCI treatment was 9.7±4.0 months for responders and 3.5±1.4 months for nonresponders. Ten (42%) patients had mild-to-moderate AEs, including worsening of diabetes (12%) and lower extremity edema (8%), which were the only AEs experienced by >1 patient. In a prospective, single-arm clinical trial in patients with refractory active DM/PM, 7 (70%) of 10 patients who completed the minimum 8 weeks of RCI treatment (80 U twice weekly) met the primary definition of improvement.21 Three patients had 5 SAEs, and 8 patients had 22 AEs. Among the SAEs, 2 herpes zoster infections were considered to be related to study drug. In addition, patients who discontinued RCI after 6 months of clinical efficacy experienced a slow increase in disease activity and disability index and a decrease in muscle strength; their first flare occurred 4 months after discontinuation of RCI.25 Interim results from an open-label study of RCI in 9 adults with refractory cutaneous DM reported that at 3 months, 7 patients had improved Cutaneous Dermatomyositis Disease Area and Severity Index activity scores and 8 patients had improved Physician Global Assessment activity scores.26
The current study is limited by its retrospective design and the small sample size yielded by our recruitment efforts. Another limitation is its reliance on patient medical records, which had missing data, as noted in our analyses, and may have had other unaccounted for errors and omissions (such as care received at other clinics). Reliance on medical records also makes it difficult to assess disease activity, which is not routinely recorded in the medical records or recorded with the appropriate timing to allow evaluation of clinical response to therapy. Future research should consider this limitation and adjust the study design to accommodate. We implemented quality control measures to minimize the occurrence of data entry errors during medical record abstraction. Our study used physician’s impression of change as a descriptive endpoint, a subjective measure that relies on an individual clinician’s own standards of improvement. Physician's impression of change was not available for 38% of the records evaluated. Also, the retrospective noncomparative study design did not allow us to determine if patients were responding to other therapies. Thus, our results are exploratory and should be interpreted with these limitations in mind.
Conclusion
The findings from this study describe the real-world patient profile, use patterns, and outcomes of patients who used RCI for the treatment of RA, SLE, and DM/PM. The data can inform appropriate use of RCI as well as the clinical expectations when using this medication. Larger clinical trials are ongoing to add to the body of evidence.
Acknowledgments
The authors thank the physicians who participated in this study. Mallinckrodt Pharmaceuticals, Bedminster, NJ, USA, funded sponsorship of this study and the article processing charges. The authors thank Monica Nicosia, PhD, of Global Outcomes Group for medical writing assistance and Esther Tazartes, MS, of Global Outcomes Group for editorial assistance. This assistance was funded by Mallinckrodt Pharmaceuticals, Bedminster, NJ, USA.
Author Contributions
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship of this manuscript. All authors contributed to data analysis, drafting, and revising the article, take responsibility for the integrity of the work as a whole, and have given approval for the final version to be published. All authors had full access to all the data used in this study and take responsibility for the integrity of the data and accuracy of the data analysis.
Disclosure
Winnie W. Nelson and Mark L. Hanke were employees of Mallinckrodt Pharmaceuticals, Bedminster, NJ, USA, at the time of this writing. Nancy Ho-Mahler is an employee of Mallinckrodt Pharmaceuticals, Bedminster, NJ, USA. Beni Turner and Michael Eaddy are employees of Xcenda, Palm Harbor, FL, USA, which has received research funding from Mallinckrodt Pharmaceuticals for the conduct of this study. The authors report no other conflicts of interest in this work.
References
1. Acthar® Gel (repository corticotropin injection) [prescribing information]; 2019. Available From: https://www.acthar.com/pdf/Acthar-PI.pdf.
2. Ross AP, Ben-Zacharia A, Harris C, Smrtka J. Multiple sclerosis, relapses, and the mechanism of action of adrenocorticotropic hormone. Front Neurol. 2013;4:21. doi:10.3389/fneur.2013.00021
3. Montero-Melendez T. ACTH: the forgotten therapy. Semin Immunol. 2015;27:216–226. doi:10.1016/j.smim.2015.02.003
4. Wang L, Wang FS, Gershwin ME. Human autoimmune diseases: a comprehensive update. J Intern Med. 2015;278:369–395. doi:10.1111/joim.12395
5. Hunter TM, Boytsov NN, Zhang X, Schroeder K, Michaud K, Araujo AB. Prevalence of rheumatoid arthritis in the United States adult population in healthcare claims databases, 2004–2014. Rheumatol Int. 2017;37:1551–1557. doi:10.1007/s00296-017-3726-1
6. Smolen JS, Aletaha D, Barton A, et al. Rheumatoid arthritis. Nat Rev Dis Primers. 2018;4:18001. doi:10.1038/nrdp.2018.1
7. Smolen JS, Landewe R, Bijlsma J, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann Rheum Dis. 2017;76:960–977. doi:10.1136/annrheumdis-2016-210715
8. Gillis T, Crane M, Hinkle C, Wei N. Repository corticotropin injection as adjunctive therapy in patients with rheumatoid arthritis who have failed previous therapies with at least three different modes of action. Open Access Rheumatol. 2017;9:131–138. doi:10.2147/OARRR
9. Fischer PA, Rapoport RJ. Repository corticotropin injection in patients with rheumatoid arthritis resistant to biologic therapies. Open Access Rheumatol. 2018;10:13–19. doi:10.2147/OARRR
10. Helmick CG, Felson DT, Lawrence RC, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part I. Arthritis Rheum. 2008;58:15–25.
11. Rees F, Doherty M, Grainge MJ, Lanyon P, Zhang W. The worldwide incidence and prevalence of systemic lupus erythematosus: a systematic review of epidemiological studies. Rheumatology (Oxford). 2017;56:1945–1961. doi:10.1093/rheumatology/kex260
12. Lam NC, Ghetu MV, Bieniek ML. Systemic lupus erythematosus: primary care approach to diagnosis and management. Am Fam Physician. 2016;94:284–294.
13. Li X, Golubovsky J, Hui-Yuen J, et al. Adrenocorticotropic hormone gel in the treatment of systemic lupus erythematosus: a retrospective study of patients. F1000Res. 2015;4:1103. doi:10.12688/f1000research
14. Fiechtner JJ, Montroy T. Treatment of moderately to severely active systemic lupus erythematosus with adrenocorticotropic hormone: a single-site, open-label trial. Lupus. 2014;23:905–912. doi:10.1177/0961203314532562
15. Fiechtner JJ, Montroy T. Six months’ treatment of moderately to severely active systemic lupus erythematosus with repository corticotropin injection: an extension of a single-site, open-label trial. J Immunol Clin Res. 2016;3:1025.
16. Furie R, Mitrane M, Zhao E, Das M, Li D, Becker PM. Efficacy and tolerability of repository corticotropin injection in patients with persistently active SLE: results of a phase 4, randomised, controlled pilot study. Lupus Sci Med. 2016;3:e000180. doi:10.1136/lupus-2016-000180
17. Furie RA, Mitrane M, Zhao E, Becker PM. Repository corticotropin injection in patients with persistently active SLE requiring corticosteroids: post hoc analysis of results from a two-part, 52-week pilot study. Lupus Sci Med. 2017;4:e000240. doi:10.1136/lupus-2017-000240
18. Schmidt J. Current classification and management of inflammatory myopathies. J Neuromuscul Dis. 2018;5:109–129. doi:10.3233/JND-180308
19. Levine T. Treating refractory dermatomyositis or polymyositis with adrenocorticotropic hormone gel: a retrospective case series. Drug Des Devel Ther. 2012;6:133–139. doi:10.2147/DDDT
20. Levine T, Malone J, Efthimiou P, et al. H.P. Acthar® Gel in dermatomyositis and polymyositis treatment registry: an interim analysis. J Neurol Disord. 2016;4:292. doi:10.4172/2329-6895.1000292
21. Aggarwal R, Marder G, Koontz DC, Nandkumar P, Qi Z, Oddis CV. Efficacy and safety of adrenocorticotropic hormone gel in refractory dermatomyositis and polymyositis. Ann Rheum Dis. 2018;77:720–727. doi:10.1136/annrheumdis-2017-212047
22. Chopra I, Qin Y, Kranyak J, et al. Repository corticotropin injection in patients with advanced symptomatic sarcoidosis: retrospective analysis of medical records. Ther Adv Respir Dis. 2019;13:1753466619888127. doi:10.1177/1753466619888127
23. Nelson WW, Lima AF, Kranyak J, et al. Retrospective medical record review to describe use of repository corticotropin injection among patients with uveitis in the United States. J Ocul Pharmacol Ther. 2019;35(3):182–188. doi:10.1089/jop.2018.0090
24. Fleischmann R, Furst DE, Connolly-Strong E, Liu J, Zhu J, Brasington R. A multicentre study assessing the efficacy and safety of repository corticotropin injection in patients with persistently active rheumatoid arthritis: Poster presented at: The European League Against Rheumatism (EULAR) Annual Congress; June 12–15, 2019; Madrid, Spain.
25. Saygin D, Marder G, Oddis CV, et al. Long term follow-up results of myositis patients treated with H. P. Acthar Gel [abstract]. Arthritis Rheum. 2018;70.
26. Fernandez A. Interim results of an open-label study assessing efficacy and safety of adrenocorticotropic hormone gel for treatment of refractory cutaneous manifestations of dermatomyositis [abstract]. Arthritis Rheum. 2018;70.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.