Back to Journals » Medical Devices: Evidence and Research » Volume 8
Treatment of sleep-disordered breathing with positive airway pressure devices: technology update
Received 12 June 2015
Accepted for publication 7 August 2015
Published 23 October 2015 Volume 2015:8 Pages 425—437
DOI https://doi.org/10.2147/MDER.S70062
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Karin Gardner Johnson, Douglas Clark Johnson
Department of Medicine, Baystate Medical Center, Springfield, MA, USA
Abstract: Many types of positive airway pressure (PAP) devices are used to treat sleep-disordered breathing including obstructive sleep apnea, central sleep apnea, and sleep-related hypoventilation. These include continuous PAP, autoadjusting CPAP, bilevel PAP, adaptive servoventilation, and volume-assured pressure support. Noninvasive PAP has significant leak by design, which these devices adjust for in different manners. Algorithms to provide pressure, detect events, and respond to events vary greatly between the types of devices, and vary among the same category between companies and different models by the same company. Many devices include features designed to improve effectiveness and patient comfort. Data collection systems can track compliance, pressure, leak, and efficacy. Understanding how each device works allows the clinician to better select the best device and settings for a given patient. This paper reviews PAP devices, including their algorithms, settings, and features.
Keywords: BiPAP, CPAP, iVAPS, AVAPS, ASV, positive pressure respiration, instrumentation, treatment algorithm
Introduction
Positive airway pressure (PAP) is the primary treatment of sleep-disordered breathing including obstructive sleep apnea, central sleep apnea, and sleep-related hypoventilation. The most common form – continuous positive airway pressure (CPAP) – maintains a continuous level of PAP in a spontaneously breathing patient. Other forms that provide noninvasive positive pressure ventilation include bilevel positive airway pressure (BiPAP), adaptive servoventilation (SV), and volume-assured pressure support (VAPS). This review will highlight the differences in the device types as well as features that help with the functioning and comfort.
PAP equipment involves three basic parts: a device with a motor, a mask that covers either or both the mouth and nose, and a tube that connects the device to the mask. PAP has improved greatly since Colin Sullivan invented CPAP in June 1980 in Australia, using a paint compressor attached to pool tubing and a mask made from a plaster cast glued to the patient’s face. Major landmarks in the evolution of PAP are the first commercially available units by Respironics in 1985 and the self-sealing “bubble mask” in 1990.1
Current noninvasive positive pressure ventilation units are much more complex and may include an air filter, sensors (motor speed, gas volumetric flow rate, pressure, snore transducer), microprocessor-based controller, data storage, multilingual displays, and humidifier with heated tubing.
We reviewed literature from the manufacturers, patents, and published literature to provide an understanding of the different algorithms, features, and clinical applications of the various PAP devices.
Overview of device types
CPAP maintains a continuous PAP throughout inspiration and expiration. Autoadjusting (Auto) CPAP can gradually increase or decrease the pressure based on respiratory events, but similarly maintains the same pressure throughout the respiratory cycle. This can be helpful for patients who may need a higher pressure in rapid eye movement (REM) or supine position, but cannot tolerate the higher pressure through the entire night. AutoCPAP can also be used diagnostically to determine a fixed pressure setting.2
BiPAP provides a higher pressure during inspiration and lower pressure during expiration. This may improve tolerance and help with ventilation. A backup rate can be added to give a breath with weak or absent respiratory effort. AutoBiPAP may adjust either the expiratory positive airway pressure (EPAP) and inspiratory positive airway pressure (IPAP) with a fixed pressure support (PS) or may adjust them independently.
SV is a bilevel system that continuously changes the inspiratory PS on a breath-by-breath basis in order to achieve a target ventilation or flow for a more constant breathing pattern, especially in the treatment of periodic breathing or Cheyne–Stokes respiration (CSR). Auto forms of SV also increase EPAP in response to obstruction.
Volume-assured pressure support (VAPS) is a variable bilevel PAP that allows the target volume or ventilation to be programmed, which allows more control of ventilation. This is useful for patients with combined periodic breathing and hypoventilation or patients with REM-related hypoventilation related to conditions like chronic obstructive pulmonary disease (COPD), neuromuscular disorders, or obesity, who may need different PS levels at different times.
Motor/flow generator and transducers
In order to provide a constant desired pressure at the patient’s airway, adjustments in flow must be made to account for the loss of pressure between the flow generator and the patient’s airway, as well as for breathing fluctuations and leak.
Transducers monitor the motor speed, airflow, and the pressure at a fixed point downstream from the flow generator. Because the sensor is within the device and not in the mask, the device must calculate the predicted pressure at the mask based on the flow measurements at a different point in the system. The pressure at two points varies with the flow. For turbulent flow, this can be calculated by Flow (q) = constant√(p1 − p2).3 The length and diameter of the tubing and mask characteristics affect the constant value, and so the technician must input the mask and tube type for the microprocessor to make the correct calculations. The mask flow can also be calculated by subtracting the flow through the exhaust of the device from the flow through the tubing.4
In order to maintain a stable mask pressure, the microprocessor must adjust the turbine speed in response to deviations in pressure that occur from leak or normal swings in air pressure from breathing. The flow signal is sent through low and high pass filters to separate the respiratory flow signal from artifacts. Low pass filters exclude large quick deviations in flow (eg, coughs or sneezes) and high pass filters exclude cardiogenic fluctuations. Feedback limits determine if the flow or pressure is beyond the expected range of flow variation at a particular motor speed (eg, break in the tubing), and prevent the device from delivering too much or too little pressure.5 With increasing altitude, fan speed needs to be increased to maintain the same pressure.6 Many devices adjust automatically to altitude.
Leak compensation
Unlike invasive ventilation, leak is an important factor that must be compensated for. Leak affects aspects of performance including pressure delivered, cycle and trigger thresholds, and respiratory event determination. Leak compensation works by constantly monitoring flow and looking for deviations from the expected respiratory flow and will compensate the motor speed for the leak. Because there are normal variations in the patient’s breathing cycle the expected leak is usually averaged over several breaths. If leak is high, auto devices may compensate by lowering the pressure, which may seal the mask and reduce leak.
The leak can be determined from the flow rate at the end of exhalation.7 Normal leak includes that from exhalation ports on the mask, which varies by mask type and pressure level, and unintentional leak from the mouth or around the mask. In general, leak should be under 24 L/min in nasal masks and under 36 L/min in full face masks.
The lips and tongue sometimes act like a one-way valve opening during exhalation, when the pharyngeal pressure is highest. This is called valve leak or expiratory puffs. Expiratory puffs may falsely imply flow limitation, which can cause auto devices to increase pressures unnecessarily. Some algorithms place less reliance on flow limitation when large leak or expiratory puffs are present.8
Respiratory cycle determination
Determination of the inspiratory and expiratory cycles is essential not only to provide bilevel PAP but also for expiratory pressure relief and determination of inspiratory flow limitation in auto algorithms. The start of inspiration is marked by a switch from negative to positive flow (relative to baseline). The point at which the flow signal switches from positive to negative flow is the start of expiration.
SV and some auto devices use more continuous respiratory cycle determination. ResMed’s servoventilator microprocessor uses fuzzy inference rules looking at the flow rate (relative to mean flow), direction, and size to determine the phase of the respiratory cycle9 (Table 1). Respironics’ servoventilator determines the length of inspiration and expiration of recent breaths to predict the future breath duration and divides the breaths into short time segments (64 ms) to determine the expected mid-inspiration and other points of the cycle.10
  | Table 1 ResMed’s fuzzy logic for phase determination |
CPAP
Most CPAP devices allow for pressure settings between 4 and 20 (all pressures in cm·H2O). An EPAP of 4 is the lowest pressure needed to provide enough flow to clear the dead space from the device, tubing, and airway to prevent rebreathing of exhaled air.11 The goal of CPAP is to increase upper airway pressure enough to provide a pneumatic splint to open the airway, which may collapse during inspiration. Typically, the pressure is set to prevent hypopnea, apnea, snoring, flow limitation, and arousals. By providing positive end expiratory pressure, CPAP may recruit alveoli and improve ventilation.
AutoCPAP
The aim of AutoCPAP is to adjust the pressure in response to respiratory events without adjustment to artifacts caused by leak or other factors. AutoCPAP from different companies and different models by the same company have varying definitions of events, responses to obstructive events, and protocols for decreasing the pressure once the breathing stabilizes. These variations are important to understand for appropriate clinical care as they affect the patient’s tolerance of the devices and the clinical efficacy.12–14 If a patient enters REM sleep or changes position, the degree of obstruction may suddenly increase and by the time the device is able to adjust to the needed pressure the patient may have had desaturations or arousals. This is why most studies reporting the equivalence of AutoCPAP to in-lab titration recommend changing the EPAP minimum to the pressure the device is at or below 90–95% of the time.2,15 In our experience, many patients left on AutoCPAP 4–20 are undertreated and may present with awakenings a couple hours into sleep, residual symptoms, or difficulty tolerating PAP. Some patients are sensitive to the pressure changes, so if patients are not doing well with AutoCPAP, fixed CPAP should be tried.
Some of the first AutoCPAPs, like Virtuoso LX smart and SOMNOsmart, only detected vibration-based snore changes making them unresponsive to many significant events.16 Most new AutoCPAP systems use snore detection in combination with flow detection. The flow is sampled many times per second, scaled with a low pass filter to remove artifact, and then a mean flow can be determined for any time length. Peak flow can be a poor measure of breath volume, which can lead to over- or underestimation of an apnea or hypopnea. Respironics uses a weighted peak flow (WPF) method to estimate ventilation, while ResMed uses a scaled low-pass-filtered absolute value of respiratory flow and uses a root mean squared (RMS) technique of the variance of the flow from the mean to compare one moving time period to another. DeVilbiss Intellipap AutoAdjust uses a scaled peak amplitude, while AutoAdjust 2 uses RMS and other filtering techniques to adjust for artifacts in peak flow.
Respironics’ WPF method first determines the inspiratory period, then the inspiratory volume and the points on the inspiratory flow curve that correspond to the 20% and 80% volume. The average flow of all points between the 20% and 80% points is used as the WPF, a measure of ventilation. This method uses WPF values over the prior 4 minutes and determines the average of values between the 80th and 90th percentile. This baseline is used to compare with the current WPF to look for decrease in amplitude, which would indicate apnea or hypopnea.17
ResMed’s RMS method determines ventilation from variance of the flow throughout the entire breath by comparing individual flow points to the mean airflow over a defined time period. The mean airflow is the zero-point between inspiration and expiration, thus variance from this mean divided by two equals the amplitude of the inspiratory flow. By taking the square root of the variance squared, outlying values receive less weight. A moving short time period (eg, one breath or 2 seconds) can be compared to a moving longer period (eg, 5 minutes) to evaluate for apnea or hypopnea.18 Apneas and hypopneas are typically defined as a reduction in ventilation below a percentage of recent breathing for at least 10 seconds, with varying methods used by different devices (Table 2).
Since responding to central apneas can lead to over titration, testing for airway patency allows for differentiation of central from obstructive apneas. Two methods used to test for airway patency include cardiogenic pulsation testing and device-generated pressure oscillations. The first method looks for cardiogenic pulse artifact in the flow, which is only present if the airway is open. In the second method, the device provides single pressure pulse or small oscillation in the flow (eg, 1 cm, 4–5 Hz or forced oscillation technique), which is only reflected back to the flow sensors if the airway is closed.18–20 Respironics uses pressure pulses and also defines an obstructive apnea if there is a larger than expected breath after apnea termination. A mixed apnea can be determined if the airway is open for only part of the flow. ResMed from S9 onward uses force oscillation technique to define central apneas and defines central apneas if leak is >30 L/min.18 DeVilbiss Autoadjust 2 uses a modulating micro-oscillation to determine airway patency during apneas.
In order to evaluate flow limitations, Respironics determines roundness, flatness, skewness, and WPF to rate the most recent four breaths as better, worse, or the same compared to baseline. Roundness is determined by the similarity of the WPF between 5% and 95% values to a sine wave. Flatness is determined by the absolute value of the variance between 20% and 80% of inspiratory flow from the average of all the values in the same period, divided by 80% volume point. Skewness is determined by dividing the average of the highest 5% of flows in the mid third of the breath by the average of the highest 5% of flows in the first third of the breath.17
ResMed also determines flow limitation. S8 AutoSet defines flow limitation using flatness of an inspiratory breath. The flatness index is calculated by the RMS deviation from unit scaled flow calculated over the middle 50% of a normalized inspiratory breath.4 From the S9 onward, flow limitation is calculated using a combination of flatness index, breath shape index, ventilation change, and breath duty cycle. Ventilation change is the ratio of the current breath ventilation to recent 3-minute ventilation. Breath duty cycle is the ratio of current breath time of inspiration to total breath time of recent 5 minutes. If a breath is severely flow limited, the flow limitation index will be closer to one and when the breath is normal or round, the flow limitation index will be zero.8
Table 2 summarizes the differences between the algorithms for the ResMed AutoSet device,4,8 ResMed AutoSet for Her,8 Respironics System One REMstar Auto,17 and DeVilbiss IntelliPAP AutoAdjust and AutoAdjust 2.21 ResMed AutoSet evaluates flow every breath looking for apneas, snore, and flow limitation, but responds to flow limitation on a 3-breath average, has faster decreases in the absence of flow limitation and has a higher rate of pressure change to all responses (apnea, snore, and flow limitation) than AutoSet for Her.8,18
In comparison, ResMed AutoSet for Her evaluates the flow for every breath looking for apneas, snore, and flow limitation, and delivers a proportional increase in pressure depending on the degree of deviation of the event from normal, modulated by the current pressure setting and leak rates. If pressure is >10, then the response to flow limitation reduces and a louder snore is required to produce a response.8 Both AutoSet for Her and AutoSet respond less as leak gets higher.
Respironics REMstar Auto uses layers of control including ramp, leak, snore, apnea/hypopnea, variable breathing, and flow limitation. If there is no snore, apnea/hypopnea, variable breathing, or flow limitation breathing for 3–5 minutes, it will enter a testing period in which it will first decrease the pressure until either Pminimum or the flow characteristics (peak, flatness, roundness, and skew) worsen, which is the Pcritical, then quickly increase by 1.5 and holds for 10 minutes unless further events or flow limitation occurs. If there is snore, apnea/hypopnea, variable breathing or flow limitation, the pressure increases by 0.5/min until there is no further improvement or worsening, then decreases by 1.5, which is set as the Poptimal pressure. REMstar also uses several mechanisms to avoid over titration, which include nonresponsive apnea/hypopnea logic, variable breathing, and leak control (Table 2).17,22
Most devices start at the set EPAP minimum (EPAPmin) each night. Respironics REMstar Pro and REMstar Auto have CPAP check mode, which checks the 90% pressure every 30 hours, then decides whether to leave the EPAP unchanged, or changes the EPAP up or down by one but not more than three from set EPAP.23
DeVilbiss’s IntelliPAP AutoAdjust allows setting of amplitude and duration cut-points for apneas and hypopneas to change sensitivity. AutoAdjust does not detect flow limitation and defines central apneas as <5% of airflow for 10 seconds.21 It continuously scores events, but only decides once per minute whether to adjust pressures. AutoAdjust 2 similarly adjusts pressures once per minute, but also responds to flow limitation based on inspiratory flatness. It uses modulating micro-oscillation during apneas to test for patency and has an algorithm to define periodic breathing, looking for cyclic breathing as short as a 20-second cycle. AutoAdjust 2 holds or lowers pressures in response to central apneas and periodic breathing.
BiPAP
BiPAP provides a higher pressure during inhalation and lower pressure during exhalation. Pressures generally range from EPAP minimum of four to IPAP maximum of 25–30. Most devices use a flow trigger to determine when to change to IPAP. The trigger is set above zero flow to sense a significant patient effort. Different methods including flow, shape, and volume are used to cycle to EPAP in efforts to minimize dyssynchrony. The flow cycle algorithm changes to EPAP when the flow drops below a percentage (eg, 25%) of the peak flow so the patient will not encounter resistance to exhalation.24 Shape cycling algorithm uses shape of flow,25 and volume cycle algorithm uses exhaled volume to cycle to EPAP. There can be significant variation between devices in terms of how quickly pressure levels are met and whether a device has a delay or premature cycle, especially in the setting of leaks.26 If there is a mismatch between the patient’s respiratory cycle and the device control cycle there can be patient discomfort.
In older BiPAP devices, the motor was braked at the transition point from higher to lower pressures and the motor was accelerated when the device transitioned from lower to higher pressure, which affected synchrony and tolerability. One method to improve comfort is to allow the blower motor to spin freely on transition between inspiration and expiration. Newer devices allow for a smoother transition in pressure changes, and waveforms can be square, exponential, ramp, or sinusoidal. ResMed has a sharkfin-shaped “Easy-Breathe” waveform.27 The shape of the waveform may be affected by the compliance and resistance of the patient’s respiratory system and the breathing effort, as well as mechanical constraints of blower momentum and propagation delays. In general, BiPAP provides a square wave of PS, but manual or automatic adjustments can give more of a smooth pressure change, which may help with comfort. Most BiPAP devices allow for adjustment of the rise time (angle of the pressure change) from 100 ms to 600 ms.
Some BiPAP devices include cycle, trigger, inspiration time, and rise time settings, which can be adjusted to enhance effectiveness and patient comfort, especially for patients with COPD or neuromuscular disorders25 (Table 3). Typically the rise time should be kept shorter in COPD patients to allow lungs to fill more quickly and give enough time to exhale. A longer rise time in patients with neuromuscular weakness helps ensure adequate tidal volume and gas exchange (Figure 1).
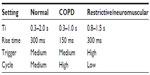  | Table 3 Suggested PAP settings |
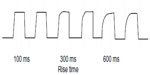  | Figure 1 Rise time. |
Because of the delay in detecting the onset of inspiration and delivery of the pressure adjustment, it is common for the inspiratory pressure needed for BiPAP settings to prevent obstruction during early inspiration to be higher than the CPAP pressure needed to prevent the same. For example, a patient requiring CPAP 8, may require BiPAP 10/6 so that a pressure of 8 is reached early enough in the inspiration to keep the airway open.
Trigger sensitivity refers to the degree of inspiratory flow change needed to change the pressure cycle from EPAP to IPAP. At high sensitivity, small changes in inspiratory flow will bring changes to IPAP. If the trigger is too sensitive (too high), then the device may force a breath in response to artifacts like leak or abnormally strong heartbeat due to cardiogenic oscillations in the breathing. Low trigger sensitivity is recommended if the patient complains the breath occurs before exhalation is complete or before the patient starts inhalation. High setting is recommended for patients with weak respiratory effort, such as in neuromuscular disorders (Figure 2).
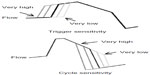  | Figure 2 Trigger and cycle sensitivities. |
Inspiration time typically ranges from 0.3 seconds to 2 seconds, often with a default of 1.5 seconds. The higher the baseline respiratory rate, the shorter the inspiratory time (Ti) recommended. Short Ti settings can be helpful for COPD patients for whom pressure does not quickly equilibrate throughout the lung, so the patient may need to actively exhale to cycle the end of expiration. Not only can this cause dyssynchrony and discomfort, it may also lead to air trapping and reduced tidal volume if expiration time is too short.25
Cycle sensitivity sets the level of inspiratory flow below which the device changes from IPAP to EPAP. High cycle setting is recommended when shortened Ti is needed, such as for COPD or if the patient complains that the breaths are too long. Low cycle is recommended when longer Ti is needed for neuromuscular diseases, weak respiratory effort, or stiff lungs, or if the patient complains that the pressure seems to switch from IPAP to EPAP too quickly.
BiPAP can be triggered by spontaneous, timed or spontaneous/timed (ST) modes. In spontaneous mode, inspiration is only triggered when the device senses a flow change. Large leak may cause the trigger to fail if the device does not appropriately adjust. Timed mode triggers at a fixed rate and makes no attempt to synchronize with the patient’s breathing, which can result in breath stacking and patient discomfort. ST mode changes pressures with spontaneous breathing efforts, but if the patient has not triggered a breath by a set respiratory rate, then the device will trigger a breath.
ST mode is most often used for primary central apnea or central apneas due to respiratory depression. ST mode may also be used for neuromuscular disease patients, whose respiratory efforts fall during REM sleep, which may make them unable to trigger inspiration.28 Timed mode is often used for patients with severe neuromuscular weakness or spinal cord injury, who are unable to trigger inspiration.
BiPAP may worsen central apneas due to CSR by increasing breath size of spontaneous breaths and forcing a triggered breath during the apneic portion, which is when the partial pressure of carbon dioxide (Pco2) level is already at its lowest.29 By further decreasing the Pco2, respiratory drive is reduced further and the duration of the apnea will often lengthen, although the oxygenation may improve with the deeper or forced breath. Sometimes the improved oxygenation and PS will help to eventually stabilize the patient’s breathing,30 but in our experience patients with CSR often find BiPAP intolerable or still have a suboptimal clinical response including fluctuations in the respirations and electroencephalogram.
AutoBiPAP
Like AutoCPAP, not all AutoBiPAP devices work in the same way. Some devices only allow a fixed PS, others set a PS maximum (PSmax), and others allow for both PS minimum (PSmin) and PSmax. Thus AutoBiPAP may not provide adequate ventilator support if PSmin cannot be set. AutoBiPAP devices generally do not have an ST option, so are not recommended for central apneas.
ResMed’s VPAP AutoBiPAP and DeVilbiss AutoBiPAP have a fixed set PS. Respironics Series 50 AutoBiPAP fixes PSmin at two and allows setting PSmax, while Series 60 AutoBiPAP allows setting both PSmin and PSmax. Within the limits of PSmin and PSmax, Respironics AutoBiPAP changes EPAP in response to apneas (two apneas or one apnea and one hypopnea) and snoring, and IPAP in response to hypopneas (two hypopneas) and flow limitation, with algorithms similar to REMstar AutoCPAP.31
Adaptive servoventilation
Because higher PAP pressures and high PS can induce periodic breathing and CSR, devices have been developed to try to even out the breathing over several breaths. These devices include ResMed VPAP AdaptSV (equivalent to Teijin AutoSet CS) with ASV setting, AirCurve ASV, which has been renamed AirCurve CS PaceWave with ASV and ASVAuto settings, and Respironics BiPAP AutoSV Advanced. Another device, SOMNOvent CR, is available outside of the US from Weinmann (Table 4).
ResMed’s standard ASV uses a set fixed EPAP, samples flow 50 times per second, and alters IPAP throughout inspiration to achieve target minute ventilation. The PS range can be 0–20, but default is usually a PSmin of three and PSmax of 15. The device calculates a target minute ventilation based on the recent average weighted minute ventilation, weighted toward the last 3 minutes. ASV uses fuzzy logic to determine the part of the respiratory cycle and whether the current ventilation is below or above the desired target and then adjusts the PS throughout the cycle to achieve that target, thus avoiding abrupt pressure changes. The change in pressure is calculated by multiplying a gain of 0.3 cm H2O/L/min/s by the difference between the target minute ventilation and the actual minute ventilation. An automatic backup rate starts with the current spontaneous rate based on moving average calculated over several breaths and gradually adapts during an apnea to 15 bpm.9 ASVAuto also adjusts the EPAP level similar to the AutoSet algorithm in response to obstructive apneas, flow limitation, and snoring.32
The hope is that by stabilizing breathing, the periodic breathing pattern will subside. If the device is cycling the pressures frequently to maintain the target ventilation, it indicates that the underlying periodic pattern is still present and often the patient will either not tolerate the device or there will be a suboptimal clinical response.33
Respironics BiPAP AutoSV Advanced is set with EPAP minimum and maximum, PSmin and PSmax, max pressure, and auto or fixed rate. Ti, rise time, and Bi-Flex can also be set for comfort. The level of PS is targeted based on instantaneous average inspiratory flow, which is the sum of the inspiratory flows during a time divided by the number of samples during a time in order to adjust for spurious values. The target peak flow is generally set at 90%–95% of the mean peak flow of the last 4 minutes. In an attempt to stabilize breathing in the setting of obstructive features (apnea [<20% flow], hypopnea [20%–60% flow], and snoring), the target peak flow increases from 95% of the mean inspiratory peak flows over the last 4 minutes to the 60th percentile of the inspiratory peak flow values. The target peak flow is also increased to 60th percentile if CSR features are noted based on a CSR index, which measures similarity to a CSR pattern or a flow index that measures fluctuations in breath size.34
The respiratory cycle of AutoSV Advanced is determined by using the average length of the breathing cycle over recent breaths and calculating the expected midpoint of inspiration. Compared to multiple small adjustments in the PS throughout the breath cycle with ResMed’s ASV, the inspiratory PS is determined based on the prior breath’s PS plus a gain based on a moving average of the pressure needed in the prior 30 breaths multiplied by the difference between the target peak inspiratory flow and the current breath’s peak inspiratory flow. An intrabreath PS is given if the actual flow is less than target flow in the 100 ms prior to the expected halfway point of the inspiration of the current breath. If actual flow is larger than target flow, the PS will be decreased for the following breath.34
The EPAP adjustment algorithm of AutoSV Advanced is similar to REMstar Auto algorithm, but uses a triggered breath rather than forced oscillation to differentiate obstructive from open airway events.32 The automatic backup rate of AutoSV Advanced is determined based on a moving window of the last 12 spontaneous breaths. A mandatory breath is given if the breath does not occur within certain parameters, with a minimum breath rate of 8–10 bpm. A fixed backup rate can also be set.
The older BiPAP AutoSV did not automatically titrate the expiratory pressure, and the algorithm for the automatic backup rate was not proportional to the baseline breathing rate, but it would give a breath if no spontaneous breath occurred within 8 seconds of end of expected breath length or within 4 seconds, if there was recent triggered breath.35 Respironics BiPAP AutoSV Advanced has been found to be more effective than the older BiPAP AutoSV.35 SV, which targets recent ventilation (ASV) or recent peak flow (AutoSV) is not appropriate for managing patients with respirator insufficiency, for whom BiPAP or VAPS should be considered.
Volume-assured pressure support
Respironics AVAPS (average VAPS) and ResMed iVAPS (intelligent VAPS) adjust the PS in order to maintain target average ventilation over several breaths (Table 5). This mode of PAP is often helpful for patients with respiratory insufficiency due to neuromuscular and restrictive conditions in which respiratory effort varies during sleep or who need PAP during the day, COPD patients with risk of hypoventilation, and for obesity hypoventilation patients who may need compensation based on positional and sleep stage changes. Because many patients have much worse hypoventilation in REM, BiPAP with a fixed PS may provide too much pressure in NREM, which may lead to intolerance or complex sleep apnea and may not provide enough PS in REM to control Pco2 levels. Benefits of VAPS over BiPAP can include maintaining volumes in the setting of altered patient effort based on sleep stage or altered lung mechanics related to position. Lesser PS during wake may increase comfort and aid sleep onset, reduce risk of barotrauma, and lower pressures for most of the time.36 A randomized trial of iVAPS vs BiPAP found that iVAPS delivered a lower mean PS for the oxygenation and transcutaneous Pco2 level and had better adherence than BiPAP.37
Both AVAPS and iVAPS adjust PS and respiratory rate to reach a defined target with the goal of stabilizing the Pco2, which relates directly to alveolar ventilation. With a target tidal volume (eg, AVAPS), if there is a large variance in the respiratory rate, there can be fluctuations in the alveolar ventilation and thus Pco2. By targeting estimated alveolar ventilation (minute ventilation–estimated dead space ventilation; eg, iVAPS), variations in respiratory rate should not affect alveolar ventilation or Pco2 as long as the estimated dead space equals physiologic dead space. The device estimates the anatomic dead space using height.38 However, patients with lung diseases, such as emphysema, have increased physiologic dead space that would be underestimated by the above equation, and thus their alveolar ventilation can be much lower than the estimated alveolar ventilation. Thus, emphysema patients may require high target alveolar ventilation to achieve an adequate actual alveolar ventilation. Alternatively, the “height” can be entered artificially high for emphysema patients, thus the calculated dead space will be closer to physiologic dead space, and therefore iVAPS more closely maintains the actual alveolar ventilation.
Respironics AVAPS targets an average tidal volume over several breaths. Typically, the target tidal volume is set based on 6–10 mL/kg ideal body weight. It calculates the average PS provided to the patient over the prior 2 minutes in order to achieve a particular tidal volume. If average recent ventilation is less than target volume, IPAP for the next breath is increased. PS will change at a rate of 2/min if there is unstable breathing and 1/min if there is stable breathing. AVAPS-AE model (and also AVAPS on Trilogy 100 Ventilator) can set maximum rate of pressure change from 1/min to 5/min. AVAPS uses square waveform with Ti and rise time settings. EPAP is fixed with AVAPS, but AVAPS-AE adjusts EPAP similar to Respironics REMstar Auto with a searching protocol between EPAP minimum and maximum. AVAPS either uses a fixed rate or auto backup rate set at 2 bpm lower than the rate of the last six spontaneous breaths.39
ResMed iVAPS uses a similar servoventilator with fuzzy logic for determining respiratory phase as adaptive SV. Unlike adaptive SV, the goal ventilation is set to a target alveolar ventilation, which is defined as minute ventilation minus anatomical dead space ventilation. iVAPS alters the gain every 8/50th second throughout the inspiratory cycle to achieve the target ventilation with a smooth parabolic transition that can be modified with rise time, Ti, and cycle and trigger sensitivities. EPAP is fixed like adaptive SV.40
iVAPS’ intelligent backup rate function uses two-third of the set target patient rate as the backup rate during spontaneous breathing and switches during apneas to the set patient target rate apnea. This reduces potential dyssynchrony during spontaneous breathing, while providing ventilation during apneas at a lower PS than if the backup rate remained lower.41
The iVAPS ventilation and rate targets can be configured using the Learn Target feature. While the patient is awake and comfortably breathing at rest, the clinician initiates the Learn Target session, which typically lasts between 15 minutes and 20 minutes and monitors the breathing while on a base EPAP pressure of 6. It uses the average respiratory rate, and since metabolic rate decreases in sleep, it uses 90% of the average estimated alveolar ventilation of the last 5 minutes of the session to propose a target patient rate and target alveolar ventilation, which can then be set by the clinician.42
Other features of PAP
Humidification, heated tubing, ramp, automatic start and stop functions, alarms, and expiratory pressure relief are device functions that are designed to improve patient comfort and compliance (Table 6).
  | Table 6 Other PAP features |
Humidifiers include a water chamber with a heating plate through which the airflow is blown. Increasing the warmth of the heating plate increases humidification. Temperature sensors or humidification sensors allow for regulation of temperature or humidification level. Increasing humidification in the air helps reduce nasal irritation and congestion that can result from the airflow on the nasal passages.43 If the humidified air cools in the tubing, water may condense in the tube or mask, commonly referred to as “rainout”. Insulation sleeves that wrap around the tubing or heated tubing, which includes a heating element in the tubing, can increase humidification and eliminate “rainout”. The controls for the heated tubing are often linked to the warming level of the plate, but some devices allow for dissociation of the controls, which may further help with comfort in some patients. Different amounts of humidification may be needed depending on the external humidity and temperature.
For travel, standard PAP devices can be used with external battery packs or with converters to allow them to be powered by a car, boat, or other vehicles. There are also small battery-powered units weighing around 10 oz, such as Z1 CPAP and Z1 Auto from HDM, compared to 48 oz for Respironics System One and 44 oz for ResMed AirSense S10.
Data collection and display
Data from the device can be retrieved on the interface with a data card, cable, and wireless or by cellular and bluetooth to an online platform. Data cards can either be used to download locally or to an online website, which allows for sharing. Summary data from the last month is often viewable on the device. DeVilbiss IntelliPAP devices use codes that represent different data sets that their website uses to generate a report.
Data collected varies among devices and can include pressure settings, leak, average, 90%–95% pressures and maximum pressures, PS, tidal volumes, minute ventilation, and apnea hypopnea index. Some devices report more detailed data about respiratory events, which may include central, mixed, obstructive and undetermined apneas, hypopneas, flow limitation, snoring, expiratory puffs, percentage of time in periodic breathing, tidal volume, and minute ventilation. Reports can show summary data over days and months as well as detailed data with the timing of events over the course of one night. Often data can be searched for a 30 day compliance period having over 4 hours of use on 70% of the nights. Respironics System One devices can also show full night waveform tracings with scored events and pressure settings. An oximetry module can be connected to ResMed’s AirSense device to allow for saturation data. More data about adherence tracking systems are available in a review by Schwab et al.44
Conclusion
There are a wide range of PAP devices and a wide range of different algorithms used to provide PAP to treat sleep-disordered breathing. For this reason, devices in the same category may differ greatly in their clinical efficacy and comfort. Understanding how PAP devices function can help the clinician select the best PAP device, appropriately titrate, troubleshoot, and optimize settings for a particular patient. Many comfort features have improved the function and performance of devices, which allows many patients who have been unable to tolerate PAP in the past to become compliant. Many devices track compliance and provide important clinical data to help care for the patients.
Acknowledgments
We thank ResMed and DeVilbiss for providing information about their algorithms.
Disclosure
The authors report no conflicts of interest in this work.
References
Hansford A. Thirty Years of CPAP: A Brief History of OSA; 2011. ResMedica Clinical Newsletter, Vol 14. | |
Rosen CL, Auckley D, Benca R, et al. A multisite randomized trial of portable sleep studies and positive airway pressure autotitration versus laboratory-based polysomnography for the diagnosis and treatment of obstructive sleep apnea: the HomePAP study. Sleep. 2012;35(6):757–767. | |
Farrugia SP, Finn SD, inventor; ResMed Ltd, assignee. Flow estimation and compensation of flow-induced pressure swings in CPAP treatment and assisted respiration. United States Patent US 6332463. 2001 Dec 25. | |
Berthon-Jones ML, Farrugia SP, inventor; ResMed Ltd, assignee. Administration of CPAP treatment pressure in presence of apnea. United States Patent US 8684000. 2014 Apr 1. | |
Colla GA, Kenyon BJ, inventor; ResMed Ltd, assignee. Fault diagnosis in CPAP and NIPPV devices. United States Patent US 8485182. 2013 Jul 16. | |
Fromm RE Jr, Varon J, Lechin AE, Hirshkowitz M. CPAP machine performance and altitude. Chest. 1995;108(6):1577–1580. | |
Zdrojkowski RJP, inventor; Respironics Inc., assignee. Leak compensation method and apparatus for a breathing system. United States Patent US 5313937. 1994 May 24. | |
Armitstead JP, Bateman PE, Bassin DJ, inventor; ResMed Ltd, assignee. Automated control for detection of flow limitation. United States Patent US 20110203588. 2011 Aug 25. | |
Berthon-Jones ML, inventor; ResMed Ltd, assignee. Ventilatory assistance for treatment of cardiac failure and Cheyne-Stokes breathing. United States Patent US 8857430. 2014 Oct 14. | |
Hill PDM, inventor; Respironics, Inc., assignee. Method and apparatus for providing variable positive airway pressure. United States Patent US 6752151. 2004 Jun 22. | |
Ferguson GT, Gilmartin M. CO2 rebreathing during BiPAP ventilatory assistance. Am J Respir Crit Care Med. 1995;151(4):1126–1135. | |
Zhu K, Roisman G, Aouf S, Escourrou P. All APAPs are not equivalent for the treatment of sleep disordered breathing: a bench evaluation of eleven commercially available devices. J Clin Sleep Med. 2015;11(7):725–734. | |
Stammnitz A, Jerrentrup A, Penzel T, Peter JH, Vogelmeier C, Becker HF. Automatic CPAP titration with different self-setting devices in patients with obstructive sleep apnoea. Eur Respir J. 2004;24(2):273–278. | |
Shi HB, Cheng L, Nakayama M, et al. Effective comparison of two auto-CPAP devices for treatment of obstructive sleep apnea based on polysomnographic evaluation. Auris Nasus Larynx. 2005;32(3):237–241. | |
To KW, Chan WC, Choo KL, Lam WK, Wong KK, Hui DS. A randomized cross-over study of auto-continuous positive airway pressure versus fixed-continuous positive airway pressure in patients with obstructive sleep apnoea. Respirology. 2008;13(1):79–86. | |
Sullivan CE, Lynch C, inventor; Sullivan CE, Lynch C, assignee. Device for monitoring breathing during sleep and control of CPAP treatment that is patient controlled. United States Patent US 5199424. 1993 Apr 6. | |
Matthews GP, Kane MT, Duff WK, et al. inventor; RIC Investments, LLC, assignee. Auto-titration pressure support system and method of using same. United States Patent US 7827988. 2010 Nov 9. | |
Berthon-Jones ML, inventor; ResMed Ltd, assignee. Determination of patency of the airway. United States Patent US 7730886. 2010 Jun 8. | |
Martin DC, Oates JD, inventor; ResMed Ltd, assignee. Systems, methods, and/or apparatuses for non-invasive monitoring of respiratory parameters in sleep disordered breathing. United States Patent US 8646447. 2014 Nov 13. | |
Axe JR, Bebehani K, Burk JR, Lucas EA, Yen F, inventor; Respironics, Inc., assignee. Method and apparatus for controlling sleep disorder breathing. United States Patent US 6085747. 2000 Jul 11. | |
DeVilbiss Healthcare. Clinical Overview: DeVilbiss IntelliPAP AutoAdjust; 2014. Available from: http://www.devilbisshealthcare.com/files/LT-2089_RevC_FINAL_050814_Web.pdf. Accessed June 9, 2015. | |
Remmers JE, Feroah TR, inventor; University Technologies International, Inc., assignee. Auto CPAP system profile information. United States Patent US 6550478. 2003 Apr 22. | |
Phillips Respironics. System One Spec Sheet/REMstarPro and Auto; 2012. Available from: http://www.medical.philips.com/asset.aspx?alt=&p=http://www.medical.philips.com/pwc_hc/main/homehealth/sleep/systemone/pdf/SpecSheet_REMstarProandAuto-intl.pdf. Accessed July 31, 2015. | |
Sanders MH, Zdrojkowski RJ, inventor; Respironics Inc., assignee. Breathing gas delivery method and apparatus. United States Patent US 5433193. 1995 Jul 18. | |
Zdrojkowski RJ, Estes M, inventor; Respironics, Inc., assignee. Breathing gas delivery method and apparatus. United States Patent US 6029664. 2000 Feb 29. | |
Battisti A, Tassaux D, Janssens JP, Michotte JB, Jaber S, Jolliet P. Performance characteristics of 10 home mechanical ventilators in pressure-support mode: a comparative bench study. Chest. 2005;127(5):1784–1792. | |
Douglas RN, Ujhazy AJ, Richards G, Buckley MD, Schindhelm KH, inventor; Resmed Ltd, assignee. Mechanical ventilation in the presence of sleep disordered breathing. United States Patent US 8011365. 2011 Sept 6. | |
Berry RB, Chediak A, Brown LK, et al; NPPV Titration Task Force of the American Academy of Sleep Medicine. Best clinical practices for the sleep center adjustment of noninvasive positive pressure ventilation (NPPV) in stable chronic alveolar hypoventilation syndromes. J Clin Sleep Med. 2010;6(5):491–509. | |
Johnson KG, Johnson DC. Bilevel positive airway pressure worsens central apneas during sleep. Chest. 2005;128(4):2141–2150. | |
Willson GN, Wilcox I, Piper AJ, et al. Noninvasive pressure preset ventilation for the treatment of Cheyne-Stokes respiration during sleep. Eur Respir J. 2001;17(6):1250–1257. | |
Matthews G, Duff WK, Martin D, Shankar US, Ressler H, inventors; Ric Investments, Llc, assignee. Auto-titration bi-level pressure support system and method of using the same. United States patent US 8136521 B2. 2012 Mar 20. | |
Javaheri S, Brown LK, Randerath WJ. Positive airway pressure therapy with adaptive servoventilation: part 1: operational algorithms. Chest. 2014;146(2):514–523. | |
Chokroverty S, Thomas R. Atlas of Sleep Medicine. 2nd ed. Philadelphia, PA: Elsevier Saunders; 2014:130. | |
Kane MT, Bann SL Siirola R, Duff WK, Baloa LA, inventor; RIC Investments, LLC assignee. Method and apparatus for treating Cheyne-Stokes respiration. United States Patent US 8695595. 2014 Apr 15. | |
Javaheri S, Goetting MG, Khayat R, Wylie PE, Goodwin JL, Parthasarathy S. The performance of two automatic servo-ventilation devices in the treatment of central sleep apnea. Sleep. 2011;34(12):1693–1698. | |
Oscroft NS, Ali M, Gulati A, et al. A randomised crossover trial comparing volume assured and pressure preset noninvasive ventilation in stable hypercapnic COPD. COPD. 2010;7(6):398–403. | |
Kelly JL, Jaye J, Pickersgill RE, Chatwin M, Morrell MJ, Simonds AK. Randomized trial of ‘intelligent’ autotitrating ventilation versus standard pressure support non-invasive ventilation: impact on adherence and physiological outcomes. Respirology. 2014;19(4):596–603. | |
Hart MC, Orzalesi MM, Cook CD. Relation between anatomic respiratory dead space and body size and lung volume. J Appl Physiol. 1963;18(3):519–522. | |
Hill PD, Kissel MH, Frank J, Kane MT, Bann SL, Duff WK, inventor; RIC Investments, LLC, assignee. Average volume ventilation. United States Patent US 7011091. 2006 Mar 14. | |
Berthon-Jones ML, inventor; Resmed Ltd, assignee. Assisted ventilation to match patient respiratory need. United States Patent US 6532957. 2003 Mar 18. | |
Bassin DJC, inventor; Resmed Ltd, assignee. Methods and apparatus for varying the back-up rate for a ventilator. United States Patent US 8051852. 2011 Nov 11. | |
Berthon-Jones ML, Bateman P, Bassin D, Malouf G, inventor; ResMed Ltd, assignee. Determining suitable ventilator settings for patients with alveolar hypoventilation during sleep. United States Patent US 8544467. 2013 Oct 1. | |
Massie CA, Hart RW, Peralez K, Richards GN. Effects of humidification on nasal symptoms and compliance in sleep apnea patients using continuous positive airway pressure. Chest. 1999;116(2):403–408. | |
Schwab RJ, Badr SM, Epstein LJ, et al; ATS Subcommittee on CPAP Adherence Tracking Systems. An official American Thoracic Society statement: continuous positive airway pressure adherence tracking systems. The optimal monitoring strategies and outcome measures in adults. Am J Respir Crit Care Med. 2013;188(5):613–620. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



