Back to Journals » Therapeutics and Clinical Risk Management » Volume 11
Transtracheal single-point stent fixation in posttracheotomy tracheomalacia under cone-beam computer tomography guidance by transmural suturing with the Berci needle – a perspective on a new tool to avoid stent migration of Dumon stents
Authors Hohenforst-Schmidt W, Linsmeier B, Zarogoulidis P, Freitag L, Darwiche K, Browning R, Turner JF, Huang H, Li Q, Vogl T, Zarogoulidis K, Brachmann J, Rittger H
Received 20 February 2015
Accepted for publication 20 March 2015
Published 22 May 2015 Volume 2015:11 Pages 837—850
DOI https://doi.org/10.2147/TCRM.S83230
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Garry Walsh
Wolfgang Hohenforst-Schmidt,1,9 Bernd Linsmeier,2 Paul Zarogoulidis,3 Lutz Freitag,4 Kaid Darwiche,4 Robert Browning,5 J Francis Turner,6 Haidong Huang,7 Qiang Li,7 Thomas Vogl,8 Konstantinos Zarogoulidis,3 Johannes Brachmann,9 Harald Rittger1
1Medical Clinic I, “Fuerth” Hospital, University of Erlangen, Fuerth, Germany; 2Department of Thoracic Surgery, Medinos Clinic Sonneberg, Sonnerberg, Germany; 3Pulmonary Department-Oncology Unit, “G. Papanikolaou” General Hospital, Aristotle University of Thessaloniki, Thessaloniki, Greece; 4Department of Interventional Pneumology, Ruhrlandklinik, University Hospital Essen, University of Essen-Duisburg, Tueschener Weg, Essen, Germany; 5Pulmonary and Critical Care Medicine, Interventional Pulmonology, National Naval Medical Center, Walter Reed Army Medical Center, Bethesda, MD, USA; 6Division of Interventional Pulmonology and Medical Oncology, Cancer Treatment Centers of America, Western Regional Medical Center, Goodyear, AZ, USA; 7Department of Respiratory Diseases Shanghai Hospital, II Military University Hospital, Shanghai, People’s Republic of China; 8Department of Diagnostic and Interventional Radiology, Goethe University of Frankfurt, Frankfurt, Germany; 9II Medical Clinic, “Coburg” Hospital, University of Wuerzburg, Coburg, Germany
Abstract: Tracheomalacia or tracheobronchomalacia (TM or TBM) is a common problem especially for elderly patients often unfit for surgical techniques. Several surgical or minimally invasive techniques have already been described. Stenting is one option but in general long-time stenting is accompanied by a high complication rate. Stent removal is more difficult in case of self-expandable nitinol stents or metallic stents in general in comparison to silicone stents. The main disadvantage of silicone stents in comparison to uncovered metallic stents is migration and plugging. We compared the operation time and in particular the duration of a sufficient Dumon stent fixation with different techniques in a patient with severe posttracheotomy TM and strongly reduced mobility of the vocal cords due to Parkinson’s disease. The combined approach with simultaneous Dumon stenting and endoluminal transtracheal externalized suture under cone-beam computer tomography guidance with the Berci needle was by far the fastest approach compared to a (not performed) surgical intervention, or even purely endoluminal suturing through the rigid bronchoscope. The duration of the endoluminal transtracheal externalized suture was between 5 minutes and 9 minutes with the Berci needle; the pure endoluminal approach needed 51 minutes. The alternative of tracheobronchoplasty was refused by the patient. In general, 180 minutes for this surgical approach is calculated. The costs of the different approaches are supposed to vary widely due to the fact that in Germany 1 minute in an operation room costs on average approximately 50–60€ inclusive of taxes. In our own hospital (tertiary level), it is nearly 30€ per minute in an operation room for a surgical approach. Calculating an additional 15 minutes for patient preparation and transfer to wake-up room, therefore a total duration inside the investigation room of 30 minutes, the cost per flexible bronchoscopy is per minute on average less than 6€. Although the Dumon stenting requires a set-up with more expensive anesthesiology accompaniment, which takes longer than a flexible investigation estimated at 1 hour in an operation room, still without calculation of the costs of the materials and specialized staff that the surgical approach would consume at least 3,000€ more than a minimally invasive approach performed with the Berci needle. This difference is due to the longer time of the surgical intervention which is calculated at approximately 180 minutes in comparison to the achieved non-surgical approach of 60 minutes in the operation suite.
Keywords: stent, airway, cone-beam computer tomography, Berci needle, Dumon stents, tracheobronchomalacia
Introduction
Intratracheal/intrabronchial stent implantation has been used in the treatment of adult tracheobronchomalacia (TBM) since 1965, especially in severe cases where medical treatments have failed.1–3
Intratracheal stents are divided into two major types: silicone stents and shape-memory alloy stents. Silicone stents are easy to implant and remove, but have complications including infection, expectoration, and tend to migrate. Furthermore, they require a high-cost endoscopy involving rigid bronchoscopy and general anesthesia.4 The limitations of silicone stents mentioned earlier are partly overcome by shape-memory alloy stents in regards to migration; however, the use of these in benign airway stenosis is controversial because they are known to be associated with complications, especially nitinol stent fracture after a longer period, the excessive growth of granulation tissue (expected to occur initially after 2 weeks to 3 weeks after stent deployment),5 and stent migration.6 Of note is the fact that metallic stents (91% uncovered in the study of Gottlieb et al)7 have shown, in 65 posttransplantation patients with airway stenosis, a high symptom relief in short-term observation (with a short-term success of 80%) but were associated with a significant higher mortality rate in a long-term 5-year analysis (60% vs 76% without metallic stent, P=0.02).
The Dumon stent (DS), a silicone stent, was developed in 1987. It consists of evenly spaced struts along its outside wall, which are not only to assist in maintaining placement of the stent in the airway but also allow clearance of secretions around the outer wall of the stent. The inner surface of the DS is very smooth, but problems with encrusted secretions are common and potentially life threatening. The stent is very versatile. DSs are available in various lengths and diameters for trachea and bronchi, and further onsite modification of these stents is technically possible and easy. Despite the need for rigid bronchoscopy under general anesthesia for proper placement, the DS is a tool very often used worldwide by interventionalists nowadays. The DS manages to maintain structural integrity when placed endobronchially–endotracheally, and its solid wall structure prevents the reobstruction of the airway by tumor or scar tissue. A remarkable advantage of the DS is its ease of removal, which is very important when endobronchial procedures are used early and temporarily in the management of benign diseases like TBM (possibly as a bridge to surgery), in cancer patients, or after initial endobronchial tumor debulking with provisional stent implantation to enable the initiation of radiotherapy or radiochemotherapy.
The main disadvantage of this silicone stent is the higher potential of migration8,9 in comparison to metal stents and airway obstruction by plugging. In principle, one has to accept that stents are a foreign body and disrupt mucociliary clearance. Therefore, plugging and tendency to acquire local infections are logical consequences.
Plugging is more common than migration in intratracheal or – intrabronchial stenting. The study by Gottlieb et al7 reported 65 patients followed-up in median of 777 days, stented with 111 metallic stents that even in the patients with mostly uncovered stents (91%) new bacterial colonization as a precondition for plugging occurred in 40% of recipients, whereas migration was reported only in 3%. Plugging was reported in 11% of all the cases. Saad et al10 reported in a long-term follow-up study – median range in different groups from 42 days up to 336 days – of 82 patients stented with 112 self-expandable metallic stents (partly uncovered) with different conditions (50 patients with lung carcinomas, eleven patients with posttransplant complications, and 21 patients with miscellaneous benign airway diseases) with the following complications: in 4.7% migration, in 14.6% obstructive granulomas, and in 15.9% infections, but no case of mucus plugging. In contrast to these studies Murgu and Colt4 reported 12 long-term follow-up patients (median follow-up 188 days) with expiratory central airway collapse treated with silicone stents, 12 mucus plugs, and eight migrations. Therefore, it is generally accepted that the ratio of complications (especially plugging and migration) to the number of applied stents is higher in silicone stents in comparison to self-expanding metal stents.
Plugging is a dangerous and potentially life-threatening complication due to stent obstruction followed by reduced ventilatory capacity, which could lead into asphyxia or blood gas turbulences. Plugging depends – besides other factors – on the length of the inner nonnatural surface. The shorter the stent, the lower the development of plugging. This relationship is contradictory to the fact that short unfixated stents migrate easier than longer unfixated stents due to higher resistance. Therefore, potentially reversible fixation of short and easy to remove silicone stents would solve two major problems. Migration should be nearly impossible and clinically relevant plugging should be significantly reduced due to minimalized length to overcome focal stenosis or instability. Furthermore, silicone stents are supposed to induce less local inflammatory response.11 Therapies to prevent plugging consist of repeated cleaning, bronchoscopies, antibiotics, mucolytics, and hyperosmolar agents.12,13
Interestingly, the new stent designs with biodegradable stents eluting local chemotherapy with mitomycin c, in comparative studies with commercially available silicone stents (besides other stents), showed in animal models the lowest mucus trapping and airway obstruction due to tracheal stenosis.14 Tracheal stenosis occurred only in half of the animals treated with bioabsorbable tubular stents with mitomycin c in comparison to those animals treated with silicone stents. The same concept of reversibility to overcome general stent problems by biodegradable stents has been investigated in malignant airway diseases as well.15
Tracheobronchomalacia – how to treat
Tracheo(broncho)malacia (TM)/TBM, characterized by excessive expiratory collapse due to the atrophy and/or reduction of tracheal elastic fibers of the tracheal wall or even a reduction in the integrity of tracheal cartilage, is a significant cause of morbidity in children and adults, especially at the time of airway infection.16,17 Weakening and collapse of the central airways may be propagated by the cartilaginous wall, the membranous wall, or both.
Severe TBM with complete or near-complete collapse is a pathologic entity that may result in disabling dyspnea, the inability to clear secretions, recurrent respiratory infections, and paroxysmal and frequent coughing.
Whereas focal malacia of the airway may be caused by goiter, vascular anomalies, trauma, mediastinal masses of different etiologies, severe kyphoscoliotic deformation, postradiation alteration, chronic infections like COPD, surgical anastomosis of the trachea or bronchi, tracheostomy or prolonged endotracheal intubation, the etiology of the diffuse acquired form of tracheobronchomalacia is often unknown. On the other hand this form of TBM is well related to severe COPD or some genetically defined diseases like Mounier-Kuhn disease, cystic fibrosis, Marfan syndrome, and Ehlers-Danlos syndrome.11 TBM in adults is mostly an acquired condition in which the central airway walls are weakened.
Methods for the treatment of severe TM/TBM in adults are limited, and there is no uniform standard. Surgical treatments like surgical lateropexia, tracheal resection, and surgical external tracheoplasty are performed for selected cases of adults and aortopexy is the most commonly performed surgery in children18 apart from stent implantation,19,20 tracheostomy with tube insertion,16 and nonsurgical external tracheal stabilization.21–25 Tracheobronchoplasty (TBP) directly compared to stenting alleviates patients suffering from TM nearly to the same extent, but TBP showed less complications than stenting26 and is therefore recommended as being superior.18 However, mortality in surgical methods like TBP show a relevant periprocedural mortality risk of 2%–3%.27 In comparison to TM where tracheal resection is one option among other possibilities, some groups tend strongly toward resection instead of stenting in benign tracheal stenosis.28 The use of all these methods requires careful consideration on an individual basis and is generally restricted to patients with localized disease. Continuous positive airway pressure in TM/TBM is rarely reported as another solution;26,29,30 sometimes even bi-level positive airway pressure is necessary.31 In contrast to the approaches mentioned earlier, different ventilation strategies are used not only for localized disease but also for general disease. However, ventilation approaches do not seem to serve as a continuous timeless solution for a severe TBM due to intermittent facial mask problems.
Suffering from severe TBM with inspiratory stridor as an urgent vital risk sign may resemble COPD or asthma in some cases.29,32–34 Furthermore, the development from COPD to COPD with TBM is not fully understood.29,35 Therapy of TBM by TBP may improve radiological airway abnormalities by reducing airway collapse, but it may not reduce the degree of air trapping in such patients, although clinical alleviation was reported.36 Therefore, understanding of choke-point physiology may be a new diagnostic approach to this problem, and TBM has to be differentiated from excessive airway collapse.37 Choke points in patients with excessive dynamic airway collapse are mainly located peripherally, while in patients with malacia they appear to be located centrally.38 Therefore, physicians caring for patients suffering from TBM must localize the flow-limiting airway segments.37 Of note is the fact that after successful stenting of a flow limitation segment, the choke point can move distally.39 However, besides others, the US Food and Drug Administration still does not recommend the use of metal stents in benign tracheobronchial disease unless absolutely necessary.40 In TBM, dynamic radial forces lead to higher stent complication rates over time, including metal fractures.41 This is the principal difference compared to stent problems in patients with malignant disease as these patients often do not live up to when the late stent complication like fracture occurs.
Depending on the degree19,42 and extent of tracheal collapse, orthopnea may occur at rest, in supine or even in upright position. Less severe cases of TBM may only hinder exercise, which can be diagnosed by the use of spiroergometry.43
The selection of patients for surgical stabilization was reported in a short-term trial of silicone stenting of the trachea or the mainstem bronchi.19 If a patient responds well to the stent trial, the assumption is made that external splinting by TBP of the same airway region should achieve the same amelioration. Stent trial should last no longer than 2 weeks, to minimize the risk of stent-related complications, such as mucus plugging, granulation tissue formation, or tracheitis-bronchitis. It is important to note that the stent trial may yield equivocal or frankly negative results in cases in which cough, recurrent infections, or secretions are the primary symptoms or in cases in which a stent-related complication ensues and obfuscates the subjective sense of improvement.19 But surgical intervention needs general anesthesia, a modified double-lumen endotracheal tube, a right posterolateral thoracotomy, and an epidural catheter for postoperative analgesia. The cost of a surgical approach is expected to be much more expensive than the minimally invasive approaches even in regards to the essential materials. TBP uses a polypropylene Y-shaped mesh to splint the posterior membrane. Posterior splinting achieves stabilization of the airway without any internal foreign body, unlike a silicone stent. It is shown to be very effective in relieving symptoms by properly selected patients. Nevertheless, it is an arduous and invasive operation with a mortality risk of 2%–3%27 which is so far not reported in short term by any form of stenting in benign airway diseases.
The optimum approach to these complex patients involves the close coordination of services provided by thoracic surgeons, interventional pulmonologists, and chest radiologists – if necessary – for a so-called minimally invasive “combined” approach. This means that thoracic surgeons and interventional pulmonologists perform a minimally invasive procedure in a sophisticated hybrid room, with cone-beam computer tomography (CBCT) as a new instrument in interventional pulmonology suites, to guide these “combined” interventions (stenting and suturing) in real time. In our suite, we use a CBCT (Artis Zee ceiling and syngo X workplace) from Siemens Healthcare AG, Forchheim, Germany. Such a CBCT has already demonstrated recently high feasibility,44 with little additional radiation load.45 For CBCT application in interventional pulmonology, there is an essential need for high-quality jet ventilation application, which is maintained in our suite by a machine from Carl Reiner, Vienna, Austria (Twinstream jet ventilator, Figure 2). We will report in an upcoming paper about the easiness and the advantages of using a CBCT for interventional bronchology procedures.
Patient and methods
A 58-year-old female, suffering from severe Parkinson’s disease and strongly reduced agility of the vocal cords due to the Parkinson’s disease, was seen in the pulmonary ambulance, with continuous stridor and severe dyspnea after her tracheostomy tube had been removed 2 weeks earlier on a surgical ward. She could neither lie down nor speak fluently in upright position. The patient refused to be tracheotomized again because previously this procedure had been performed during about of pneumonia, treated with long-term intubation followed by tracheostomy by needle puncture while weaning on an intensive care unit.
We performed three approaches with Dumon stenting in the upper third of the trachea of this patient.
Due to the worsening tracheomalacia accompanied with vocal cord hypoagility by Parkinson’s disease, we had to prepare the rigid approach with an upfront nasal jet catheter while the patient was seated. After the introduction of the nasal jet catheter, we started jet ventilation with a sophisticated dual-frequency jet ventilator (Carl Reiner) and completed sedation with relaxation for rigid bronchoscopy. We use this jet machine with different set-ups in all our CBCT approaches in interventional pulmonology as it offers unique and completely stable variations in jet ventilation technology (Figures 1–12).
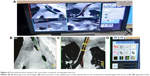
  | Figure 1 The Berci needle which is the same as the Gore Suture Passer Instrument. |
  | Figure 3 Operation suite. |
  | Figure 6 (A and B), stent processing; (C and D), stent loading. |
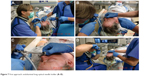  | Figure 7 First approach: endoluminal long optical needle holder (A–D). |
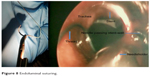  | Figure 8 Endoluminal suturing. |
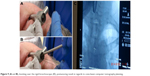  | Figure 9 (A and B), knotting over the rigid bronchoscope; (C), postsuturing result in regards to cone-beam computer tomography planning. |
  | Figure 10 Knot pusher Modell Clermont-Ferrand, Storz No 26596CL. |
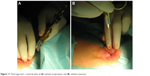  | Figure 11 Third approach – external view of (A) catheter preparation, and (B) catheter insertion. |
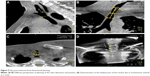
The first approach guided by CBCT visualization of the weakest location of the trachea (near the tracheotomy channel) (Figures 3–5) was a purely endoscopic approach with a transmural single-point suturing of a customized DS (18 G, 4.6 cm long, Figure 6) through the rigid bronchoscope (14 mm; Storz, Germany) by a long needle holder (Optic needle holder MEDIA 17071; Storz, Tuttlingen, Germany). We used an additional securing thread through the DS via the rigid bronchoscope for provisional longitudinal stabilization during suturing (Figures 7–9). The final node was sunk in the lumen of the rigid bronchoscope with the knot pusher tool (Figure 10). The duration of the suture performed by a very experienced thoracic surgeon consultant (13 years of experience) was 51 minutes, mainly due to the small space through the rigid bronchoscope with restricted view.
Although the symptoms were well alleviated, the suture (Ethibond Exel V-5 [17 mm, 1/2 c], size 0 [3.5 Ph. Eur.] by Ethicon, Norderstedt, Germany) ruptured after 3 days and distal migration occurred, and the symptoms relapsed very fast.
The second approach was done by a combined endoscopic transtracheal externalized suture approach performed with the Berci needle. The same stent was placed in the correct position guided by rigid bronchoscopy (Storz rigid bronchoscope 8.5 mm) 1 cm below the vocal cords. The rigid bronchoscope was removed and ventilation was easily secured by nasal jet ventilation with the machine described earlier. The stent correction was followed by a suture with the Berci needle and the following materials: Ethicon, Prolene, monofil FS 2S, USP 3-0, 1.5 metric, 45 cm, blue, nonabsorbable (needle removed before usage) (Figure 1). It was intended to use a thread with a lower metric in order to avoid overwhelming interaction with the surrounding tissue. This suture used to be stable and is well established in the hernia surgery. But this suture also ruptured after 16 days when symptoms relapsed again, and a bronchoscopy control was performed 1 day later. The duration of this second suture approach was 9 minutes.
The third approach was technically nearly the same as the second approach except for the suture material (Ethicon, Mersilene, polyfil, USP 0, 3.5 metric, 70 cm, green Sutupak EH6885, nonabsorbable) and the steering of the Berci needle below the subcutis level. We used the Berci needle with only one entrance for both passes through the skin and subcutis. Before the first pass through the tracheal and stent wall was performed, the Berci needle was assembled as a needle and was bended orally to the head in order to introduce the needle tip further down in the trachea (Figure 11). The intratracheal part of the thread was grasped by a common endoscopic tong in the trachea (Figures 12, 13 and 14). The Berci needle was then repulled out of the trachea. After securing the outer part of the thread, the Berci needle was introduced through the same skin channel bended downwards outside before puncturing a second time in order to reach a second pass into the tracheal stent wall at least one cartilage higher (more orally) than the first one (Figure 11A and B). After puncturing the stent wall, the needle was replaced by the sleeve assembly in order to grasp the thread. The main reason for changing the suture material again was to achieve a higher stability and enduring condition.
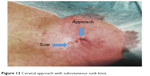  | Figure 13 Cervical approach with subcutaneous sunk knot. |
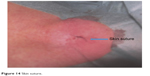  | Figure 14 Skin suture. |
The final knot was sunk subcutaneous outside of the airway lumen, far away from endoluminal loads (Figures 13 and 14). In case of incrustation of the stent, the suture is still reversible either by laser probe to cut the endoluminal part of the suture or to cut the subcutaneous knot. The patient was feeling well after the stent fixation and could breathe easily even lying down (Figure 15). Informed consent was obtained from the patient in order for her face to be shown. The duration of this third suture approach was only 5 minutes long.
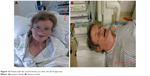  | Figure 15 Patient with the tracheostomy scar after the third approach. |
Materials
Endoluminal suturing (single-point fixation)
The endotracheal suture (single-point fixation) was managed through a 14-mm rigid tracheal bronchoscope with a long optic needle holder (Optic needle holder MEDIA 17071; Storz). The suture used was an Ethibond Exel V-5 (17 mm, 1/2 c), size 0 (3.5 Ph. Eur.), 75 cm by Ethicon. This suture was intended for permanent fixation. If transient fixation is intended, it is proper to use a 2/0 Vicryl suture with an UCLX-needle (Ethicon). This was the first approach described in the Patient and methods section.
Transtracheal suturing (single-point fixation) with the Berci needle via two skin–subcutis and transtracheal passes
The minimal invasive suturing procedure after stent placement was managed with the Berci Fascial Closure Instrument (Storz) in order to get a direct comparison for both approaches.
The puncture of the trachea and the penetration of the placed DS were managed both under CBCT guidance and fluoroscopic X-ray, respectively, accompanied simultaneously by bronchoscopic control. The puncture was supposed to be made in the middle of the DS.
Under sterile conditions, local anesthesia (5 mL Xylocaine 1%) was given on the median connecting, line between jugulum and the laryngeal cartilage, two transverse fingers above the jugulum. A very small stab incision was made. Then, an ordinary puncture cannula was used for the puncture of both trachea and the DS.
Over the well-placed cannula, a common nonabsorbable thread (Ethicon, Mersilene, polyfil, USP 0, 3.5 metric, 70 cm, green Sutupak EH6885, nonabsorbable) was introduced. This thread was intended to be more stable to external influences in order to avoid rupture or dislocation. The thread was grasped by a common endoscopic tong via the flexible bronchoscope and placed distally in the left main bronchus under bronchoscopic control.
The cannula was removed and the Berci Fascial Closure Instrument (Storz) assembled as sleeve was introduced by the same stab incision in the skin level. The puncture of the trachea was carried out under fluoroscopic X-ray and bronchoscopic control in the distance of one cartilage-clasp, through the same approach used in the first puncture. The thread was caught by the Berci Fascial Closure Instrument with the assembled sleeve and pulled out. Then several knots were made and sunk subcutaneously after a regular stability test. The suturing of the skin was made by a single-knot suture (Ethicon, Ethilon II, FS-2, UPS 4-0, 1.5 metric, 45 cm, blue).
Transtracheal suturing (single-point fixation) only with the Berci needle using different suture materials than in the second approach
The materials were different as follows to achieve an improved integrity of the suture:
- Materials used in the second approach: Ethicon, Prolene, monofil FS 2S, USP 3-0, 1.5 metric, 45 cm, blue, nonabsorbable (needle removed before usage).
- Materials used in the third approach: Ethicon, Mersilene, polyfil, USP 0, 3.5 metric, 70 cm, green Sutupak EH6885, nonabsorbable.
The slightly different technique for the third approach is described in the Patient and methods section.
Results
Single-point suturing through a rigid bronchoscope (a) is much more time consuming than transmural single-point suturing by a Berci needle (b, c).
The time duration of the complete sutures was as follows:
(a) 51 minutes
(b) 9 minutes
(c) 5 minutes
The suture integrity may be a function of the suture materials; however, the integrity time was:
(a) 3 days
(b) 16 days
(c) More than 91 days (according to last endoscopy control).
In the follow-up bronchoscopies on days 1, 3, and 7, as well as in control 6 weeks and 12 weeks after the last fixation, a regular stent placement was shown. No signs of airway induration or inflammation were described. Neither secret incrustions nor airway infection, granulation tissue, or skin cellulitis complicated the result.
In fact, at submission of this paper we have reached a suture integrity duration of 189 days. The patient is without any stent- or tracheomalacia-related problems. The patient has gained weight (5 kg) again over the last 6 months.
Frequency of different tracheal reconstruction procedures in Germany in 2013 and their remuneration
We searched the data set for 2013 of the National Institute for the Hospital Remuneration System in Germany (German abbreviation: INEK), which is responsible for the expense recompensation according to the German Diagnosis Related Group (G-DRG) system.46 Tracheal reconstruction is defined by the code 5-316. According to the data set, the overall frequency of such tracheal reconstruction in Germany in 2013 was mentioned as 1,173 times. However, the majority of those procedures were dealing with closing of fistulas by suture, tracheostoma closures, or extensions of tracheostomas. Some of them were aggravated by very long postsurgical intubation times therefore leading into a DRG coded by an A driven by long ventilation times, which is not comparable to an elective surgical or interventional approach. It was unclear which DRG was used for a pure TBP not complicated by long postsurgical intubation times. However, it appears to us that this procedure is seldom applied – otherwise, there would have been a certain number of elective surgical approaches clearly defined as TBP. Therefore, we do not know the exact recompensation value of such surgical approach, which is believed to be superior to the other approaches. We have only found 17 tracheal reconstructions with stent coded by 5-316.61 leading in eleven cases into the DRG E02C (with a mean duration time in hospital of 8.4 days), which was recompensated with 4,152.96€ and in six cases into the DRG D25D renumerated with 3,877.95€ (with a mean duration time in hospital of 5.8 days). A direct comparison of the surgical and interventional approach was not possible.
As discussed earlier, only according to the time duration in theater, the costs of a TBP, in comparison to the minimal invasive approach with the Berci needle, is in our opinion at least more than 3,000€ more expensive. We did not calculate other factors like materials, specialized staff for the surgical approach, or more duration of time spent in hospital, including possible ventilation costs.
Discussion
To the best of our knowledge, this is the first description in TM as a miniseries reporting about time duration, suture integrity, and economical cost approximation in different fixation techniques. Furthermore, it is the first report about a successful use of the Berci needle in this interventional area. Additionally, we used a quite new guiding technique with CBCT for simultaneous stenting and suturing. Although already published for suturing of posterior tracheal lacerations,47 it is the first report of an application of the long optic needle holder for anterior single-point fixation through the tracheal wall after Dumon stenting.
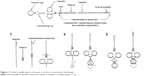
External tracheal stent fixation is well described21–25 and occasionally applied (Figure 16). In general, the principle is a loop wire-loop approach. With one needle access, one introduces the suture thread, which is grasped by a wire-loop through a second needle accessed intratracheally. So far the described techniques use two anterior passes through the tracheal wall and skin including the subcutis. This procedure may be alleviated by a fixation apparatus.24 This principle of two passes adds a certain amount of damage to cartilage and skin. In our second approach, we also used two passes through skin and subcutis, but in our third approach, there was only one pass in the subcutis level due to the fact that the Berci needle was not pulled out of the subcutis level. We expect that infection problems are less in this approach and optical result was better. The main difference to the so far described loop wire-loop techniques is the easiness of grasping the suture thread, which we believe is much faster and easier with the Berci needle than with a second wire-loop. In comparison to the Berci needle, the Endo Close suturing device (Covidien, Boston, MA, USA) used as well for external tracheal stent fixation22 is an open (unstable) grasping tool resembling a crochet hook. We believe that one may loosen the thread much easier in comparison to the closed grasping mechanism of the Berci needle. Furthermore, grasping by a Berci needle is very easy when the thread is steered into the grasp mouth by the endotracheal tong holding the thread directed by a flexible bronchoscope (Figures 11 and 12).
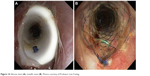  | Figure 16 Silicone stent (A); metallic stent (B). Photos courtesy of Professor Lutz Freitag. |
For any technique using a single-point fixation, it is essential to pass the stent and tracheal wall two times. However, there is recent literature48 that a common technique used in gastroenterology – percutaneous endoscopic gastrostomy tube fastener – was applied successfully for the prevention of tracheal silicone stent migration. It was reported that complications like localized cellulitis and suture breakage occurred. We believe that this method besides the advantage of only one pass through the stent and tracheal wall has considerable disadvantages. First the pass through the skin is not sutured and closed. Therefore, infection – even more than the reported cellulitis – is an obvious danger. Furthermore, having in mind the endoluminal resistance mechanism of this technique, which means an endoluminal obstacle of ventilation through a stent due to essential material volume, we expect that this technique will have more problems with plugging or incrustations. The intratracheal component of such a percutaneous endoscopic gastrostomy tube fastener could be a condensation point for plug development. Therefore, we believe that this kind of fixation is easier but only better to use in shorter fixation terms. The percutaneous endoscopic gastrostomy tube fasteners were left from 1 to 25 weeks, in this study.
The first approach in our study was a completely endoluminal suture fixation of the placed Dumon stent using a 14-mm tracheoscope and an endoscopic optic needle holder. This instrument is used for suturing posterior wall lacerations.47 Stent fixation by endotracheal suturing is feasible to avoid DS migration after placement. But in comparison to the transtracheal approaches with the Berci needle, it is much more time consuming, complicated in execution, and even more expensive in regards to the essential optical long needle holder in comparison to the cheaper Berci needle. It may have an advantage in regards to infection as the pretracheal exterior skin is not opened.
In addition, the risk of thread rupture or tearing the seam by continuous endoluminal loads cannot be negated. Therefore, higher metric threads are recommended in general.
CBCT was a useful tool,44,45 as it offers the advantage of realtime control. Optimal stent position and positioning of the needle instruments in regards to the weakest area of tracheal wall (Figure 5A) could be visualized pre- and postfixation. For this purpose, sophisticated jet ventilation technology is necessary. We are very well accustomed to the double-frequency jet ventilator model (Twinstream from Carl Reiner; Figure 2). Computed tomography technologies are used to visualize choke-point physiology.39 So far it is unclear if a CBCT application will be able to visualize this physiology too. However, by examining the different movements and changes in diameters on apnea in comparison to stable hyperinflation under jet ventilation with a flexible jet catheter, we could easily see the weakest area in this case of tracheomalacia. Furthermore, there is a need for sophisticated fixation techniques of shorter stents bridging these choke points more distally in the nontracheal airways.37 If this will be possible only by a surgical minimally invasive approach, CBCT technology with visualization techniques for choke-point physiology situated in a hybrid room will be the place of choice to deal with this type of disease.
Conclusion
After stent placement via rigid bronchoscopy in general anesthesia guided by Dyna-CT, we were able to achieve a permanent fixation of the DS endotracheally by a single-point suture in a female suffering from severe TM. This transmural fixation was managed using extraluminal suture by a Gore Suture Passer Instrument/Berci needle (Storz).
In summary, the better, economically more efficient, reversible, and safer method from our point of view is the transcutaneous puncture and transmural fixation using this instrument, in comparison to endoluminal suturing or surgical interventions like TBP. From our point of view, this method is much easier to handle and cheaper than other external silicone stent fixation interventions, with even less risk of mortality in comparison to surgical approaches.
Acknowledgment
Dr Wolfgang Hohenforst-Schmidt would like to thank Mr Carl Reiner for providing the Model Twinstream jet ventilator. Moreover Dr Wolfgang Hohenforst-Schmidt would like to thank Siemens, Germany for the technical and financial support that they have provided for this project.
Disclosure
The authors report no conflicts of interest in this work.
References
Rademacher J, Suhling H, Greer M, et al. Safety and efficacy of outpatient bronchoscopy in lung transplant recipients – a single centre analysis of 3,197 procedures. Transplant Res. 2014;3:11. | ||
Ernst A, Odell DD, Michaud G, Majid A, Herth FF, Gangadharan SP. Central airway stabilization for tracheobronchomalacia improves quality of life in patients with COPD. Chest. 2011; 140(5): 1162–1168. | ||
Casiano RR, Numa WA, Nurko YJ. Efficacy of transoral intraluminal Wallstents for tracheal stenosis or tracheomalacia. Laryngoscope. 2000;110(10 pt 1):1607–1612. | ||
Murgu SD, Colt HG. Complications of silicone stent insertion in patients with expiratory central airway collapse. Ann Thorac Surg. 2007;84(6):1870–1877. | ||
Jiang AG, Gao XY, Lu HY. Diagnosis and management of an elderly patient with severe tracheomalacia: a case report and review of the literature. Exp Ther Med. 2013;6(3):765–768. | ||
Chen W, Ruan Y. Late complications of nickel-titanium alloy stent in tracheal stenosis. Laryngoscope. 2012;122(4):817–820. | ||
Gottlieb J, Fuehner T, Dierich M, Wiesner O, Simon AR, Welte T. Are metallic stents really safe? A long-term analysis in lung transplant recipients. Eur Respir J. 2009;34(6):1417–1422. | ||
Bolliger CT, Probst R, Tschopp K, Soler M, Perruchoud AP. Silicone stents in the management of inoperable tracheobronchial stenoses. Indications and limitations. Chest. 1993;104(6):1653–1659. | ||
Wood DE, Liu YH, Vallieres E, Karmy-Jones R, Mulligan MS. Airway stenting for malignant and benign tracheobronchial stenosis. Ann Thorac Surg. 2003;76(1):167–172. [discussion 173–164]. | ||
Saad CP, Murthy S, Krizmanich G, Mehta AC. Self-expandable metallic airway stents and flexible bronchoscopy: long-term outcomes analysis. Chest. 2003;124(5):1993–1999. | ||
Bacon JL, Patterson CM, Madden BP. Indications and interventional options for non-resectable tracheal stenosis. J Thorac Dis. 2014;6(3):258–270. | ||
Hurt K, Bilton D. Inhaled interventions in cystic fibrosis: mucoactive and antibiotic therapies. Respiration. 2014;88(6):441–448. | ||
Bilton D, Stanford G. The expanding armamentarium of drugs to aid sputum clearance: how should they be used to optimize care? Curr Opin Pulm Med. 2014;20(6):601–606. | ||
Zhu GH, Ng AH, Venkatraman SS, et al. A novel bioabsorbable drug-eluting tracheal stent. Laryngoscope. 2011;121(10):2234–2239. | ||
Chao YK, Liu KS, Wang YC, Huang YL, Liu SJ. Biodegradable cisplatin-eluting tracheal stent for malignant airway obstruction: in vivo and in vitro studies. Chest. 2013;144(1):193–199. | ||
Carden KA, Boiselle PM, Waltz DA, Ernst A. Tracheomalacia and tracheobronchomalacia in children and adults: an in-depth review. Chest. 2005;127(3):984–1005. | ||
Jiang A, Lu H. Early diagnosis and management of tracheomalacia with invasive bronchopulmonary aspergillosis in an adult. Braz J Infect Dis. 2012;16(2):215–216. | ||
Kugler C, Stanzel F. Tracheomalacia. Thorac Surg Clin. 2014;24(1):51–58. | ||
Ernst A, Majid A, Feller-Kopman D, et al. Airway stabilization with silicone stents for treating adult tracheobronchomalacia: a prospective observational study. Chest. 2007;132(2):609–616. | ||
Thornton RH, Gordon RL, Kerlan RK, et al. Outcomes of tracheobronchial stent placement for benign disease. Radiology. 2006;240(1): 273–282. | ||
Cho JH, Kim H, Kim J. External tracheal stabilization technique for acquired tracheomalacia using a tailored silicone tube. Ann Thorac Surg. 2012;94(4):1356–1358. | ||
Majid A, Fernandez-Bussy S, Kent M, et al. External fixation of proximal tracheal airway stents: a modified technique. Ann Thorac Surg. 2012;93(6):e167–e169. | ||
Colt HG, Harrell J, Neuman TR, Robbins T. External fixation of subglottic tracheal stents. Chest. 1994;105(6):1653–1657. | ||
Miwa K, Takamori S, Hayashi A, Fukunaga M, Shirouzu K. Fixation of silicone stents in the subglottic trachea: preventing stent migration using a fixation apparatus. Ann Thorac Surg. 2004;78(6):2188–2190. | ||
Mace A, Sandhu G, Howard D. Securing tracheal stents: a new and simple method. J Laryngol Otol. 2005;119(3):207–208. | ||
Ferguson GT, Benoist J. Nasal continuous positive airway pressure in the treatment of tracheobronchomalacia. Am Rev Respir Dis. 1993;147(2):457–461. | ||
Gangadharan SP, Bakhos CT, Majid A, et al. Technical aspects and outcomes of tracheobronchoplasty for severe tracheobronchomalacia. Ann Thorac Surg. 2011;91(5):1574–1580. [discussion 1580–1571]. | ||
Krajc T, Janik M, Benej R, et al. Urgent segmental resection as the primary strategy in management of benign tracheal stenosis. A single center experience in 164 consecutive cases. Interact Cardiovasc Thorac Surg. 2009;9(6):983–989. | ||
Kandaswamy C, Balasubramanian V. Review of adult tracheomalacia and its relationship with chronic obstructive pulmonary disease. Curr Opin Pulm Med. 2009;15(2):113–119. | ||
Davis S, Jones M, Kisling J, Angelicchio C, Tepper RS. Effect of continuous positive airway pressure on forced expiratory flows in infants with tracheomalacia. Am J Respir Crit Care Med. 1998;158(1):148–152. | ||
Essouri S, Nicot F, Clément A, et al. Noninvasive positive pressure ventilation in infants with upper airway obstruction: comparison of continuous and bilevel positive pressure. Intensive Care Med. 2005;31(4):574–580. | ||
Alici IO, Kar Kurt O, Dursun AB, Yilmaz A, Erkekol FO. Two cases of tracheal disease misdiagnosed as difficult-to-treat asthma. Respir Care. 2013;58(11):e133–e137. | ||
Hernandez Arauzo N, Castillo Marchuet MJ, Vinas Domingo M, Ibero Iborra M. Tracheomalacia: uncommon onset in a patient with severe asthma. J Investig Allergol Clin Immunol. 2011;21(5):412–413. | ||
Gangadharan SP. Tracheobronchomalacia in adults. Semin Thorac Cardiovasc Surg. 2010;22(2):165–173. | ||
Murgu SD, Colt HG. Treatment of adult tracheobronchomalacia and excessive dynamic airway collapse: an update. Treat Respir Med. 2006;5(2):103–115. | ||
Lee KS, Ashiku SK, Ernst A, et al. Comparison of expiratory CT airway abnormalities before and after tracheoplasty surgery for tracheobronchomalacia. J Thorac Imaging. 2008;23(2):121–126. | ||
Murgu S, Colt H. Tracheobronchomalacia and excessive dynamic airway collapse. Clin Chest Med. 2013;34(3):527–555. | ||
Murgu SD, Colt HG. Tracheobronchomalacia and excessive dynamic airway collapse. Respirology. 2006;11(4):388–406. | ||
Handa H, Miyazawa T, Murgu SD, et al. Novel multimodality imaging and physiologic assessments clarify choke-point physiology and airway wall structure in expiratory central airway collapse. Respir Care. 2012;57(4):634–641. | ||
Gaissert HA, Grillo HC, Wright CD, Donahue DM, Wain JC, Mathisen DJ. Complication of benign tracheobronchial strictures by self-expanding metal stents. J Thorac Cardiovasc Surg. 2003;126(3): 744–747. | ||
Filler RM, Forte V, Chait P. Tracheobronchial stenting for the treatment of airway obstruction. J Pediatr Surg. 1998;33(2):304–311. | ||
Freitag L, Ernst A, Unger M, Kovitz K, Marquette CH. A proposed classification system of central airway stenosis. Eur Respir J. 2007;30(1):7–12. | ||
Wassermann K, Eckel HE. Stenosen der oberen Atemwege. Lungenfunktion, lokalen Widerstand und Lastausgleich. Eine Überprüfung [Stenoses of the upper airways. Lung function, local resistance and load compensation. A review]. HNO. 1999;47(11):947–956. German. | ||
Hohenforst-Schmidt W, Zarogoulidis P, Vogl T, et al. Cone beam computer tomography (CBCT) in interventional chest medicine stenoses of the upper airways – high feasibility for endobronchial real-time navigation. J Cancer. 2014;5(3):231–241. | ||
Hohenforst-Schmidt W, Banckwitz R, Zarogoulidis P, et al. Radiation exposure of patients by cone beam CT during endobronchial navigation – a phantom study. J Cancer. 2014;5(3):192–202. | ||
Hytych V, Horazdovsky P, Stehlik L, et al. Our own method of fixation of biodegradable tracheal stent. Bratisl Lek Listy. 2015;116(5):340–342. | ||
Welter S, Krbek T, Halder R, Stamatis G. A new technique for complete intraluminal repair of iatrogenic posterior tracheal lacerations. Interact Cardiovasc Thorac Surg. 2011;12(1):6–9. | ||
Musani AI, Jensen K, Mitchell JD, Weyant M, Garces K, Hsia D. Novel use of a percutaneous endoscopic gastrostomy tube fastener for securing silicone tracheal stents in patients with benign proximal airway obstruction. J Bronchology Interv Pulmonol. 2012;19(2):121–125. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.