Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
Traditional Chinese Medicine Jiuwei Zhenxin Granules in Treating Depression: An Overview
Received 22 July 2020
Accepted for publication 7 September 2020
Published 2 October 2020 Volume 2020:16 Pages 2237—2255
DOI https://doi.org/10.2147/NDT.S273324
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Yuping Ning
Jing Wang, Xingmao Wu
Department of Critical Care Medicine, Shengjing Hospital of China Medical University, Shenyang, Liaoning, People’s Republic of China
Correspondence: Xingmao Wu
Department of Critical Care Medicine, Shengjing Hospital of China Medical University, Heping District, Shenyang, Liaoning 110004, People’s Republic of China
Tel/ Fax +86 24 18940251899
Email [email protected]
Abstract: Depression is known as “Yu Zheng” in traditional Chinese medicine (TCM). Jiuwei Zhenxin granules (JZG) is a type of TCM. According to TCM theory, it nourishes the heart and spleen, tonifies Qi, and tranquilizes the spirit, and may also has effects in the treatment of depression. Here, we systematically reviewed recent basic and clinical experimental studies of JZG and depression, including studies of the pharmacological mechanisms, active ingredients, and clinical applications of JZG in depression treatment. This review will deepen our understanding of the pharmacological mechanisms, drug interactions, and clinical applications of TCM prescriptions and provide a basis for the development of new drugs in the treatment of depression.
Keywords: Jiuwei Zhenxin granules, depression, Yu Zheng, pharmacological, effective substance
Introduction
Depression is a common affective mental disorder. Its primary clinical characteristics are persistent depression, pessimism, lack of interest in surroundings, sleep disorders, mental and cognitive delays, and even suicidal tendencies.1 Modern medical research suggests that chronic diseases, genetic factors, social and environmental factors can induce depression.2 For example, genetic factors can determine a person’s susceptibility to depression, and environmental factors can trigger depression.3 The pathogenesis of depression is still not fully understood, but it is generally believed to be related to changes in brain neurotransmitters such as γ-aminobutyric acid (GABA), estrogen levels, neuronal cell factors, and cell signal transduction.4,5 In recent years, there have been many theories about the pathogenesis of depression, including the norepinephrine hypothesis, the dopamine hypothesis, the neurotrophic/regenerative hypothesis, and the neural plasticity pathway regulation theory. These theories have important significance for guiding the clinical treatment of depression.6,7 Clinical practice has proven that many chemical drugs have beneficial therapeutic effects in depression, but they are also associated with adverse reactions, such as drowsiness and dystonia, and have shortcomings, such as a narrow antidepressant spectrum and a high rate of relapse.8 Increasingly, studies show that traditional Chinese medicine (TCM) and its compounded preparations exhibit unique effects in the treatment of depression. Not only are there fewer adverse reactions associated with TCM preparations, but their effects are comparable to those of chemical drugs, and patients easily accept and are compliant with such treatment.9,10
TCM is a general term that includes medicine and practices of the Han nationality, as well as the minority nationality in China. TCM reflects the Chinese people’s understanding of life, health, and disease: it has a long history of use and incorporates unique theories and technologies. Jiuwei Zhenxin granules (JZG) are a classical Chinese medicine prescription that has been used in the prevention and treatment of various depressive, anxiety, and bipolar disorders in China.11 JZG originated from Pingbu Zhenxin and was first recorded in Tai Ping He pharmacy prescriptions in the Northern Song Dynasty. In the 1990s, the formula was revised by TCM Professor TuYa of Beijing University and Professor Xiaolan Cui of the Institute of Pharmacology of the Chinese Academy of Sciences. The main functions of JZG are to nourish and regulate the heart and spleen, replenish Qi, tranquilize the mind, and replenish blood. It is mainly used for the treatment of anxiety and depression, insomnia and dreaminess, palpitations, anorexia, mental fatigue, dizziness, and sweating. Recently, studies have confirmed that JZG has a significant effect on depression. In this review, we highlight the recent studies noting the progress of JZG in treating depression in order to facilitate an understanding of the pharmacology of JZG and provide a basis for the development of novel drugs for the treatment of depression.
The Understanding of Depression According to Traditional Chinese Medicine
According to TCM theory, it is believed that the five internal organs (heart, liver, spleen, lungs and kidneys), the body and all the orifices form an organic whole. The essence of the five internal organs is the material basis of emotion (Table 1). The main causes of depression are loss of liver drainage, healthy spleen function, and Heart nourishment. Although the three organs of the liver, spleen, and heart are related, each has its own emphasis and plays a different role in the development of depression. Liver Qi stagnation is mostly related to Qi, blood, and fire; while food, wetness, and sputum are mainly related to the spleen; manifestations of Heart deficiency include a loss of nourishment in the Heart, insufficient blood supply, and deficiency in Heart yin. The initial symptoms of depression are related to Qi and blood. Therefore, the symptoms of Qi stagnation and blood stasis are very common in clinical practice. Therefore, depression is categorized as “Yu Zheng,” which, in TCM, refers to a type of disease or syndrome caused by the blockage of the five internal organs due to emotional depression and Qi stagnation. Its name comes from the “Inner Canon”12
 |
Table 1 Summarizing the Theory of Five Internal Organs in Traditional Chinese Medicine |
In TCM, it is believed that “Yu Zheng” is primarily caused by long-term emotional stress and liver Qi stagnation: this causes the internal organs to function inconsistently, leading to disorders of Qi and blood, as well as the heart, spleen, and liver. The pathogenesis of “Yu Zheng” in TCM is mainly due to emotional failure, which leads to Qi stagnation, Qi and blood Yin and Yang disorders, and visceral dysfunction, and the core pathogenesis is the blockage of the Qi mechanism. Zhu Danxi in the Yuan Dynasty first proposed the theory of “Six Yu”, which divided “Yu Zheng” into six features: Qi stagnation, blood stagnation, phlegm stagnation, fire stagnation, Shi stagnation, and food stagnation. Hu et al believe that “Yu Zheng” is primarily dominated by Qi stagnation, with secondary influences of phlegm stagnation, cold coagulation, and food stagnation, leading to different aspects of heart, spleen, liver, and kidney deficiencies.
Shen jing shuai ruo, also known neurasthenia, which was introduced in China in the early 1900s and commonly understood by the Chinese to mean “neurological weakness”. These words translate as a weakness of the channels carrying vital energy, or Qi, through the body.13 Shen jing shuai ruo became widely used diagnostically by psychiatrists and other medical practitioners, who viewed it as a state determined by the interaction between an inherited neurotic tendency and environmental stress and by the 1980s, as many as 80% of psychiatric outpatients in mainland China were diagnosed as primarily “neurasthenic”, and up to one-half of general and psychiatric Chinese outpatients sought treatment for self-diagnosed neurasthenia. This is still relevant and applicable today.14
In the worldwide, the use of herbal medicine to treat mental disorders that has increased markedly. Incorporating TCM into conventional medicine to treat depression has various benefits such as better accessibility, acceptability and lesser stigma.14 There are also evidence-based herbal therapies that have shown promising results in clinical trials, when compared to standard Selective Serotonin Reuptake Inhibitors (SSRIs), such as St John’s wort15 and Curcuma longa, which is a major constituent of the ancient herbal medicine Xiao Yao San and has demonstrated antidepressant activity in clinical trials.16 JZG, a classical Chinese medicine prescription, nourishes the Heart and spleen, supplements Qi, and calms nerves: it is the main prescription for anxiety and depression caused by deficiency of both the Heart and spleen. Clinically, it can be used to treat generalized anxiety disorder with heart and spleen deficiency syndrome, symptomatic thinking, incomprehension, insomnia and dreams, heart palpitations, loss of appetite and fatigue. However, patients with yin deficiency and fire excess should use JZG with caution. The formula is composed of nine kinds of Chinese herbal medicines: Panax ginseng C.A. Mey, Ziziphus jujuba Mill, Schisandra chinensis (Turcz.) Baill, Smilax glabra Roxb, Polygala tenuifolia Willd, Corydalis yanhusuo (Y.H.Chou & Chun C.Hsu) W.T.Wang ex Z.Y.Su & C.Y.Wu, Asparagus cochinchinensis (Lour.) Merr, Rehmannia glutinosa (Gaertn.) DC., and Cinnamomum cassia (L.) J. Presl. According to traditional medicine, P. ginseng (PG) and S. glabra Roxb (SGR) invigorate vital energy, strengthen the blood vessels and spleen, benefit the lungs, calm the nerves, and improve intelligence. Z. jujuba Mill (ZJM) and S. chinensis (SC) nourish the blood of the heart and liver, calm the nerves and settle the mind, and delay aging. P. tenuifolia (PT) and A. cochinchinensis (AC) calm the mind, relieve cough, eliminate phlegm, and reduce swelling. C. yanhusuo (CYHS) and C. cassia (CS) promote blood circulation, remove blood stasis, regulate Qi, and relieve pain. R. glutinosa (RG) nourishes the blood and replenishes essence and filling marrow. The combination of these nine TCM components in proper proportions can have the greatest therapeutic effect on “Yu Zheng”.
Pharmacological Studies of the Active Compounds of Nine Chinese Medicines Included in JZG
Panax ginseng C.A. Mey
PG is a perennial herb of Panax in the family Acanthopanax of Umbelliferae. Its main producing areas are northeast China, North Korea, South Korea, Japan, and eastern Russia. PG contains many effective ingredients, including panaxoside A, B, C, D, E, and F; monosaccharide; PG acid (a mixture of palmitic acid, stearic acid, and linoleic acid); vitamins (B1, B2, nicotinic acid, niacinamide, and pantothenic acid); amino acids; choline; enzymes (maltase, invertase, and esterase); spermine; and choleamine.17,18
Previous studies have suggested that PG has many pharmacological effects, including anticancer,19 antineuronal apoptosis,20 antidiabetic,21 anti-cognitive impairment,22 antihypertensive,23 and anti-fatigue24 activities.
Ginsenoside, an important chemical component of PG, has therapeutic effects on colorectal cancer via the SNAIL signaling axis.25 It could also induce apoptosis and downregulate PD-L1 expression by targeting the NF-κB pathway in lung adenocarcinoma.26 In addition, ginsenoside has antidepressant effects,27 prevents cognitive impairment by improving mitochondrial dysfunction in Alzheimer’s Disease (AD),28 ameliorates memory impairments,29 and has anti-anxiety and anti-stress properties.30 Previous animal experiments have suggested that stress stimulation can lead to reduced expression of brain-derived neurotrophic factor (BDNF), and they demonstrated that ginsenoside can effectively upregulate BDNF in the hippocampus, downregulate serum cortisol levels, and reduce or reverse changes in the density of synaptic spine neurons in the hippocampus caused by chronic stress.31 In addition, abnormal volume and structure of neurons was discovered in the amygdala and hippocampus;32 ginsenoside could effectively improve these pathological changes.33 Based on these results, further studies of the specific cellular and molecular mechanisms of ginsenoside’s neuroprotective effects as a human antidepressant are warranted.
Ziziphus jujuba Mil
ZJM is a dry and mature seed of Ziziphus jujuba Mil.var.spinosa (Bunge), which is distributed in most parts of China. It nourishes the Heart and liver, calms the Heart and nerves, and promotes the production of body fluid. It is often used for restlessness, palpitations, dreaminess, and sweating.34 Modern medical research shows that the chemical composition of ZJM is very complex, mainly a variety of zizyphusine, including frangufoline, nuciferine, nornuciferine, norisocorydine, coclaurine, and N-methylbarbine. It contains triterpenoids, including betulinic acid and botulin; flavonoids, including spinosin and zivulgarin;17 kinds of amino acids, including threonine, valine, methionine, leucine, isoleucine, lysine, and phenylalanine; and various metal elements such as potassium, sodium, calcium, zinc, iron, copper, and manganese.35,36 Some therapeutic functions of ZJM, including antioxidant, antihyperglycemic, hepatoprotective, antihyperlipidemic, and sedative activities, have been shown in modern pharmacological studies.37 Anticancer and anti-inflammation effects have also been observed.38,39 In addition, by dramatically diminishing the expression of 14-3-3 mRNA levels, ZJM could increase tolerance to environmental stresses.40
Jujuboside is one of the most effective chemical components of ZJM pharmacological function. The pathogenesis of depression is generally believed to be caused by the decreased activity or conduction function of noradrenaline (norepinephrine [NE]) and 5-hydroxytryptamine (5-HT) in the central nervous system.41 An existing animal model study indicated that Jujuboside exerts an antidepressant effect by reducing the content of 5-HT and dopamine in the prefrontal lobe.42 According to this analysis, Jujuboside may play an antidepressant role by regulating the 5-HT pathway. However, the exact mechanism remains to be clarified and deserves further study.
Schisandra chinensis (Turcz.) Baill
SC is the dried and mature fruit of Schisandra chinensis. Autumn fruits are picked when they are ripe and then dried or steamed to remove stems and impurities.43 The earliest references to SC in TCM appeared in Shennong’s Herbal Medicine Classic where it was listed as having the ability to nourish and strengthen the body. It has high medicinal value and is used in combination with Ganoderma to treat insomnia.
The fruit of SC contains schisandrin, vitamin C, resin, tannin, and a small amount of sugar. It has astringent effects on lungs and treats cough, nourishes astringent essence, relieves diarrhea, and decreases perspiration. Aromatic oil can be extracted from its leaves. The main function of SC is to nourish the lungs and kidneys and remove dampness.44–46
The medicinal value of SC is mainly reflected in anti-fatigue properties, prevention of mild cognitive impairment,47,48 enhanced immunity,49 anticancer effects,45 amelioration and prevention of Parkinson’s disease (PD),50 amelioration of age-related muscle wasting,51 and anti-atherosclerotic properties.52 Schisandrin is the main effective component of SC, which was previously considered to be able to treat AD;53 it has antitumor effects,54 provides protection against central ischemia recurrence,55 and treats neurodegenerative disease.56 In addition, although there is no study of the direct antidepressant effect of schisandrin, it is believed to produce sedative and hypnotic functions by elevating the level of GABA and reducing the level of glutamic acid in the peripheral blood and brain tissues, which has obvious auxiliary effects in the treatment of depression.57
Smilax glabra Roxb
SGR is the dry sclerotia of the fungus Poria Cocos (Schw.) +wolf. The sclerotia of SGR contains many components, including triterpenoids such as pachymic acid, 16α-hydroxyporic acid, methylporic acid, and poric acid; polysaccharides such aspachymaran and high (1, 3), (1,6) branched β-D-glucan H11; and other constituents such as ergosterol, caprylic acid and inorganic elements. SGR invigorates the spleen and resolves phlegm, and it also calms the Heart and nerves.58–60
Previous studies of SGR have shown that it can treat tumors,61,62 improve depression,63 relieve hyperglycemia and hyperlipidemia,64 lessen insomnia,65 and prevent dementia.66 It also has potential therapeutic effects for leukemia.67 SGR has sedative and hypnotic effects, and it has obvious therapeutic value for restlessness, palpitations, and insomnia. SGR is mainly used in the treatment of depression in compounded preparations. The antidepressant component of SGR and its mechanism require further study. Pachymic acid is one of the main active components of SGR, which may raise the expression of glucocorticoid receptor (GR) and recover the negative feedback of hypothalamus-pituitarium-adrenal gland (HPA) axis to relieve depression and illness state.68
Polygala tenuifolia Willd
PT is also known as Yuan Zhi. It mainly grows in northern, central China, and the Sichuan Province. It calms the mind and improves intelligence, eliminates phlegm, and reduces swelling. It can be used for insomnia, dreaminess, forgetfulness, palpitations, trance, and coughing up phlegm.69,70 The chemical composition of PT is relatively complicated but mainly includes: (1) a variety of saponins that are isolated from the roots of Polygala saponin, and the identified structures are Polygala saponin A, B, E, F, and G (structurally, the basic mother nucleus of Polygala saponin is oleanolic acid, a type of pentacyclic triterpene); (2) oral ketone, which exists only in the form of derivatives in plants (most of the substituents are hydroxyl and methoxy forms, and a few are methylenedioxy); (3) a large number of sugar ester components (its parent core structure has four types [A, B, C, and D], and there are 12 types of substituents); (4) alkaloids, namely R1 R2 N9-formylhalman; and (5) other ingredients, such aspolygalol, 3,4,5-trimethoxycinnamic acid, stigmasterol, a-spinasterol glucoside, and rich fatty oils and resins.71,72
Pharmacological research found that PT has protective effects on neuronal apoptosis and learning and memory impairment in mice models of AD;73 improves behavioral abnormalities in mice with depression74 and improves hippocampal-dependent learning and memory.75 One of the main active ingredients of PT is 3,4,5-trimethoxycinnamic acid (TMCA),76 which has anticonvulsant,77 antitumor,78 antiepileptic,79 and anti-stress80 properties and improves cognitive function.81 The antidepressant mechanism of PT includes regulating endocrine functions, protecting cells, increasing the expression of neurotrophic factor BDNF, improving the plasticity of monoamine neurons, and affecting the reuptake process of monoamine neurotransmitters.
Corydalis yanhusuo (Y.H.Chou & Chun C.Hsu) W.T.Wang Ex Z.Y.Su & C.Y.Wu
CYHS is the dried tuber of the Cordalisambigua Cham.et Schlecht. It grows under mountain forests or is cultivated in laboratories. CYHS activates blood, refreshes Qi, and has analgesic, hypnotic, sedative, and stabilizing properties. A total of more than 20 alkaloids have been identified from the tubers of CYHS, including corydaline, dl-tetrahydropalmatine, protopine, L-tetraline, L-tetrahydrocoptisine (L-stylopin), dl-tetrahydroberberine, L-tetrahydrocolumbamine, corybulbine, β-herbadol, β-homoche-lidonine, coptisine, dehydrocorydaline, and corydalmine.82–85
CYHS is currently used clinically, mainly for the treatment of drug addiction,86 as well as for its antiaging, anxiolytic,87 and antiplatelet aggregation88 properties and for the treatment of depression.89 Qi stagnation and blood stasis are primary causes of depression in the theory of TCM. CYHS has an obvious effect of promoting Qi and blood circulation. This effect may be the basis of its use in TCM for the treatment of depression. CYHS can be used in combination with Angelica sinensis (Oliv.) Diels and Conioselinum anthriscoides “Chuanxiong” (syn. Ligusticum chuanxiong) and could be used in combination with Campanulaceae to calm blood and Qi to achieve beneficial effects in the treatment of depression.
One of the main active ingredients of CYHS, tetrahydropalmatine, also known as fumarate B (dl-THP),90 prevents hyperlipidemia,91 attenuates neuronal apoptosis induced by cerebral ischemia-reperfusion injury,92 offers neuroprotection,93 has antitumor effects,94 provides analgesia,95 prevents memory impairment,96 and ameliorates the development of anxiety and depression.97,98
Asparagus cochinchinensis (Lour.) Merr
AC is the root of Asparagus cochinchinensis (Lour.) Merr. AC is long and spindle-shaped, slightly curved, measuring 5 to 18 cm long and 0.5 to 2 cm in diameter. It nourishes yin, reduces heat, and nourishes the lungs and kidneys. AC contains aspartin, B-sterol, steroidal saponin, slime, furfural derivatives, and other components. AC increases blood cells, enhances the phagocytic function of the reticuloendothelial system, and extends the duration of antibody existence.99,100
Progress in the research of AC pharmacology has led to the belief that AC exhibits a wide range of pharmacological activities, including the treatment of AD101 and antidepressant, neuroprotective,102 anti-inflammatory103 and antitumor 104 effects. Depression can be clinically manifested as insomnia, early waking, memory loss, palpitations, and dizziness. In the TCM theory of Tianwang Buxin Dan, AC combined with PG, Poria, and PT has a significant effect on treating depression caused by heart, liver, and blood deficiency.
Rehmannia glutinosa (Gaertn.) DC
RG is a perennial herb of the genus Rehmannia of the genus Scrophulariaceae. Its root is a main component of TCM treatments, and its use was first published in Shennong’s Materia Medica. According to the processing method, the medicinal materials are divided into fresh RG and dry RG. The medicinal properties and efficacies of these three compounds vary greatly. According to the efficacy listed in “Chinese Materia Medica” fresh RG is used to reduce heat and cool blood, and RG is used to supplement medicine for many TCM. The chemical composition of RG is dominated by glycosides, mainly cyclic allene terpenoids, including leonuride, aucubin, catalpol, and rehmannioside A, B, C, and D; sugars, including D-glucose, D-galactose, D-fructose, and sucrose; and a variety of amino acids and inorganic elements.105–107
RG has a wide range of clinical applications. It is currently reported that RG can be used to treat attention deficit hyperactivity disorder,108 PD109, multiple sclerosis,110 anxiety,111 and memory disorders,112 and it inhibits inflammatory responses113,114 and has anti-fatigue activity.115 In the theory of TCM, liver depression and blood deficiency are manifested as depression, anxiety, insomnia, palpitations, inattention, and other depressive symptoms. For patients with deficiency of Qi and blood, the effects of Xiao Yao for treating depression can be enhanced by adding RG.
Catalpol is one of the main chemical extracts of RG. It protects dopaminergic neurons from damage in PD,116 enhances neurogenesis and inhibits apoptosis of new neurons by activating BDNF,117 ameliorates hyperactive and impulsive behavior, improves spatial learning and memory,118 improves axonal growth by regulating miR-124 after ischemia,119 relieves neuralgia,120 and has anti-depressant effects.121
Cinnamomum cassia (L.) J. Presl
CS is in the family of camphor, it is a medium-sized tree of the camphor family, and its bark is gray-brown. The main functions of CS Presl include tonifying fire and helping yang, dispersing cold and relieving pain, warming blood vessels, improving yang deficiency, and treating vertigo.122,123
The medicinal value of CS Presl is mainly reflected in antihypertensive,124 anti-allergy,125 antibacterial,126 and anticancer127 activities; it is used in the treatment of PD128 and type 2 diabetes129 and it improves cognitive dysfunction.130 In addition, it enhances blood circulation and calms and relieves pain, and it has auxiliary therapeutic effects in depression. Cinnamaldehyde is the main component of CS Presl. It improves memory deficits,131 improves subarachnoid hemorrhage nerve damage and vasospasm,132 acts as an antidepressant by inhibiting COX-2 in the central nervous system,133 reduces atherosclerosis,134 and decreases anxiety-related behavior.135 The main active compounds of nine Chinese medicines included in JZG and pharmacological mechanisms were summarized in Table 2.
 |
Table 2 Main Active Compounds of Nine Chinese Medicines Included in JZG and Pharmacological Mechanisms |
Clinical Application of JZG in the Treatment of Depression
Much evidence has confirmed the efficacy of JZG combined with western medicine in the treatment of depression. In our review, we performed a systematic review of clinical trial articles on PubMed, Embase, Cochrane, Web of Science, Wanfang (http://www.wanfangdata.com.cn/index.html), Weipu (http://www.cqvip.com/) and CNKI (https://www.cnki.net/) database during 1984–2019 (Table 3). JZG was added to duloxetine for the treatment of patients with depression and compared with duloxetine treatment alone: Wang et al found that the scores on the Hamilton Depression Scale (HAM-D) and self-ratings of depression in the combination group were significantly improved, and the levels of 5-hydroxytryptamine and cortisol in the combination treatment group were also significantly higher than those in the single-drug treatment group. Hu et al enrolled 126 patients with depression and randomly divided these patients into two groups: 62 patients were treated with bupropion combined with JZG and 64 patients were treated with bupropion alone. The HAM-D scores of the two groups were compared at weeks 2, 4, 6, and 8 of treatment, and the researchers found that the patients in the combination treatment group achieved better results than those treated with bupropion alone. Additionally, the longer the duration of JZG combination treatment, the more obvious the therapeutic effects these patients achieved. The combination of JZG and classical Western antidepressants could improve the efficacy of Western medicine alone, suggesting that JZG has a good synergistic effect with Western medicine.
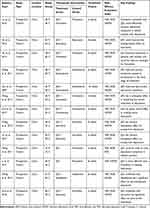 |
Table 3 Characteristics and Findings in Clinical Studies Reviewed |
Previous studies also compared the therapeutic effects of JZG with those of western medicine. Li et al compared the therapeutic effects of JZG and fluoxetine in patients with depression. The study found that after 8 weeks of treatment, the HAM-D scores of the two groups were significantly improved. The therapeutic effect of JZG was similar to that of fluoxetine, which confirmed the therapeutic effect of JZG on depression. However, the incidence of adverse reactions in the JZG treatment group was 9%, which was far lower than that of the fluoxetine group (47.7%). At present, there are few comparative studies on the effectiveness of JZG and classic antidepressants. The conclusion of this study requires more samples and multi-center clinical studies for further verification.
Moreover, JZG is widely used in diseases associated with depression. At present, the most studied of these related conditions is postpartum depression. Lin et al have achieved good results in the treatment of postpartum depression with a combination of sertraline and JZG, and, because of the decreased sertraline dosage required when used in combination with JZG, the rate of adverse reactions was significantly reduced. Wang et al achieved a similar conclusion: JZG combined with sertraline hydrochloride has a significant effect on the treatment of postpartum depression. The improvement in sex hormone levels (estradiol and progesterone) in patients receiving combined treatment with JZG was better than that in patients treated with sertraline hydrochloride alone. However, the safety of JZG for postpartum depression needs to be further evaluated, such as the concentration of the drug in breast milk and its possible effect on infants.
Taken together, these clinical studies have shown that JZG is effective in treating depression and has fewer side effects than conventional drugs. However, current research of JZG in the treatment of depression is mostly limited to Chinese patients, and the number of patients with depression included in the studies is small. In the future, large-scale randomized controlled studies will be needed to further confirm the efficacy of JZG in depression.
The Pharmacological Mechanism of JZG in the Treatment of Depression
There is currently a lack of research regarding the mechanism of JZG in the treatment of depression. However, through the study of the effective molecular structures of the nine components of JZG, it is not difficult to determine that the effective molecules in JZG play active roles in the treatment of depression through various pathways. Ginsenoside can lessen unpredictable mild stress and depression-like effects by regulating the NF-κ B/NLRP3 pathway in a rat model;136 ginsenoside may also protect depression-like behavior by inhibiting glial activation, synaptic damage, and neuronal apoptosis.137 Schisandrin protects depression-like behaviors caused by chronic unpredictable mild stress through GDNF/ERK1/2/ROS and PI3K/AKT/NOX signaling pathways.138 Tetrahydropalmatine is associated with anxiolytic- and antidepressant-like profiles in the elevated plus maze (EPM) test, probably by modifying the expression of neuropeptide Y (NPY) and corticotrophin-releasing factor (CRF) in the hypothalamus.98 Catalpol significantly protects against depression and neurodegeneration by elevating serotonin and BDNF levels; catalpol could increase mitochondrial biogenesis and activation of the PI3K/Akt pathway for insulin sensitization.139 Cinnamaldehyde shows antidepressant effects, possibly by acting on the endocannabinoid system, in the forced swim test.133
In addition to JZG, there are also classic formulations of TCM that have obvious effects in the treatment of “Yu Zheng”. Xiao Yao San (XYS) contains gardenia and paeonol; it soothes the liver and spleen, promotes Qi, and decreases stagnation. Some researchers have summarized the antidepressant mechanism of XYS in terms of neurotransmitters, neurotrophins, hypothalamic-pituitary-adrenal axis, amino acids, lipid and energy metabolism, and inflammatory factors, showing that XYS has multiple targets for producing antidepressant effects by regulating metabolic balance in the body.68,140–142 Chaihu Shugan San has a significant effect on the treatment of liver Qi stagnation syndrome, which can significantly reduce the degree of “Yu Zheng” in patients. Studies have shown that Chaihu Shugan San can better improve brainstem auditory and visual evoked potentials in patients with primary depression, reduce abnormal potential signals in the cerebral cortex, regulate the release of neurotransmitters such as serotonin, increase serum zinc levels in patients with depression, and improve patients’ cognitive function, thus achieving relief of patients’ depression symptoms. Animal experiments show that Chaihu Shugan San can lessen clinical symptoms and has an obvious therapeutic effect on depression in rats.143–146 Xiao Chaihu decoction comes from the “Treatise on Febrile Diseases” and acts to reconcile Shaoyang and regulate the stomach; it is often used to treat liver stagnation and poor vitality. Xiao Chai Hu decoction can promote the recovery of BDNF levels, improve levels of neurofunctional factors, and enhance sleep quality.147,148 Other formulas, such as Baihe Dihuang decoction, can significantly increase the content of dopamine and serotonin in the cerebral cortex, hypothalamus, hippocampus, and striatum of rats;149 Guipi decoction increases the levels of serotonin, NE, and dopamine in the hippocampus;150 and Xuefu Zhuyu decoction can significantly increase serotonin concentration and promote the expression of serotonin-1A receptors.151 These formulas also have ideal therapeutic antidepressant effects.
In recent years, it has been found that ketamine has the advantages of fast onset and long duration of efficacy, and it is effective for the treatment of refractory depression.152 Ketamine, as an N-methyl-D-aspartic acid (NMDA) receptor antagonist, has a different mechanism from traditional5-HT reuptake inhibitors, which provides a new target for the development of novel antidepressants.153 Ketamine may play an antidepressant role through a variety of mechanisms, including (1) blocking the effect of glutamate on NMDA receptors; (2) blocking NMDA receptors and activating α-amino-3-hydroxy-5-methyl-4-isoxazolpropionic acid receptor (AMPAR);154 (3) reducing the inhibition of the presynaptic glutamatergic pathway by inhibiting the activity of GABA’s effect, leading to increased glutamate release in the presynaptic membrane;155 and (4) upregulating the expression level of BDNF.156 By studying the mechanism of ketamine, it is not difficult to observe that some components of JZG have similar antidepressant mechanisms as ketamine. For example, CS Presl improves neuroinflammation-mediated NMDA receptor dysfunction and memory deficits by blocking the NF-kappa B pathway in presenilin1/2 conditional double knockout mice;131 PT produces antiseizure effects by modulating GABAergic systems in mice79 and increasing the expression of neurotrophic factor BDNF; and Schisandrin could elevate the level of GABA and reduce the level of glutamic acid in brain tissues, which has obvious auxiliary effects in the treatment of depression.57 These similarities may yield more clues for investigations of the pharmacological mechanisms of JZG in the treatment of depression.
Active Biomarkers of JZG in the Treatment of Depression
TCM, which has thousands of years of clinical application history, plays an extremely important role in the process of human disease prevention and treatment. The modernization of TCM is an inevitable process of scientific development. Although previous studies have extensively explored the material basis and mechanisms of the role of TCM and have accumulated a wealth of experience and data, how TCM actually works is still unclear and its clarification is an ongoing part of the modernization of this ancient practice.
The clinical application of TCM is mainly based on prescriptions (compounds), which can achieve the effect of disease prevention and treatment through the reasonable compatibility of various herbs. However, in this complex material system, how does each medicine play a role in treating diseases? In recent years, studies have shown that the efficacy of TCM is the result of multi-component and multi-target interactions. In fact, this kind of understanding is only the expression of the surface phenomenon of the action of TCM and cannot reflect the laws and mechanisms of the actions of TCM. The basis of the function of TCM should be the reasonable and organic combination of various effective ingredients. Its mode is to act on multiple targets related to diseases in the body in various ways and to play a role in the overall regulation of the body.
To further elucidate the JZG anti-depression targeting mechanism, we construct a pharmacological network and the mechanism of action of JZG against depression was studied by integrating pharmacological concepts (Figure 1).
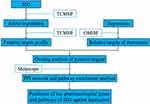 |
Figure 1 The technical strategy of this research based on network pharmacology for deciphering key pharmacological genes and pathways of JZG against depression. |
According to the parameters of absorption, distribution, metabolism and excretion, we first screened the active ingredients of JZG by searching the pharmacological database of the traditional Chinese medicine system platforms (TCMSP) (http://www.tcmspw.com/). A total of eight active biomarkers in JZG were identified, including ginsenoside, jujuboside, schisandrin, pachymic acid, 3,4,5-trimethoxycinnamic acid, tetrahydropalmatine, catalpol, and cinnamaldehyde. The 2-D structure and the Canonical SMILES of eight effective biomarkers in JZG were obtained from PubChem (https://pubchem.ncbi.nlm.nih.gov/) (Figure 2).
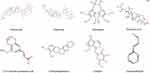 |
Figure 2 The 2-D structures of effective biomarkers in JZG. JZG, Jiuwei Zhenxin granules. |
The Swiss Target Prediction database (http://www.swisstargetprediction.ch/) is a web server for target prediction of bioactive small molecules. The targets of active biomarkers in JZG were screened in this database. The Online Mendelian Inheritance in Man (OMIM) database, a compendium of human Mendelian inheritance, is a database of human genes and genetic disorders. It mainly focuses on genetic diseases and includes text information and related reference information, sequence records, maps, and other related databases. In this study, the targets of depression were obtained from the OMIM database (http://omim.org/).
To elaborate on the multicomponent treatment system that JZG exerts in the treatment of depression, a comprehensive interaction network was applied to identify the common genes of active components in JZG and depression; in addition, a Gene Ontology (GO) analysis was performed to standardize description of the common genes. These methods are used to better construct and reveal the relationship between active ingredients in JZG and depression target genes.
A total of 854 target genes of JZG and 874 target genes of depression were obtained; as a result, 21 targets of both JZG and depression were identified (Figure 3). Cytoscape is an open-source software platform for visualizing complex networks. We used Cytoscape to construct the protein-protein network consisting of 21 nodes (Figure 4). The top 12 GO-enriched terms are listed in Table 4 and visualized in Figure 5, which was constructed using the Metascape (http://metascape.org) database.
 |
Table 4 Top 12 Clusters with Their Representative Enriched Terms of JZG on the Depression |
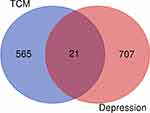 |
Figure 3 A total of 21 targets of both JZG and depression were identified. |
 |
Figure 4 A total of 21 targets were used to establish the regulation network in Cytoscape. |
 |
Figure 5 Network of enriched terms. (Size of the circle relates to p-value; terms containing more genes tended to have a more significant p-value.). |
Conclusion and Prospects
TCM may have a unique position to play in the treatment of depression: it not only has shown promise in lessening clinical symptoms, but it is also well tolerated and is associated with good compliance among patients. TCM compounds have characteristics of multi-component, multi-target, and dialectical treatments. It has the possibility be used in combination with western medicine to treat depression and this may help to reduce the occurrence of adverse reactions. Moreover, it may produce a synergistic effect, potentially enhancing the efficacy of western medicine. These are areas all in need of further research.
In recent years, much laboratory and clinical research have been conducted to reveal the treasure trove that is TCM, and many antidepressant TCMs with therapeutic effects have been identified. Still, there are problems related to TCM that need to be solved. First, there is still no unified standard for syndrome differentiation or treatment principles of depression in TCM. Doctors often rely only on clinical experience to differentiate and treat syndromes, which is also the problem with the modern DSM classification of mental illnesses. In fact, the diagnostic validity and stability of major psychiatric diagnoses is an area of perennial debate and controversy. It is known that the differential or provisional diagnosis may be rather imprecise and depends largely on a subjective assessment of the patient’s cross-sectional mental state at the specific time point 157. Second, the antidepressant effects of TCM are evaluated clinically using HAM-D (decreased scores) and psychiatric symptoms (disappeared or improved), but there are no objective standards to evaluate the therapeutic effects. Third, there is a lack of in-depth systematic research on the antidepressant effects of TCM. Some more specific mechanistic studies, such as “omics” technologies might provide greater information on the metabolite changes and possible pharmacological mechanisms of JZG.158
JZG is a type of TCM. According to TCM theory, it nourishes the heart and spleen, tonifies Qi, and tranquilizes the spirit, and it has certain auxiliary therapeutic effects in depression. Research on the components of Chinese herbal medicines have found that the effective compounds of JZG play a role in the pharmacological basis of modern medicine. In clinical practice, the results of current clinical controlled studies show that the combination of JZG and antidepressants in classic modern drugs can improve patients’ depressive symptoms better and faster than simple classic modern drugs, and can extend the effective period of drugs. However, this conclusion needs further confirmation by high quality double blinded placebo controlled randomized clinical trial. In order to further clarify the pharmacological mechanism of JZG, we predicted the possible mechanism of JZG in the treatment of depression for the first time by means of bioinformatics analysis, which can be used to accelerate studies on the mechanism of JZG in depression and will further promote the modernization of TCM.
Author Contributions
J.W. contributed to the literature search, data extraction, and data analysis. J.W. and X.W. contributed to the project design and writing of the manuscript. All authors have read and approved the final version of the manuscript. All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
There is no funding to report.
Disclosure
The authors declare that they have no competing interests for this work.
References
1. Smith K. Mental health: a world of depression. Nature. 2014;515(7526):181.
2. Raic M. Depression and heart diseases: leading health problems. Psychiatr Danub. 2017;29 Suppl 4(Suppl 4):770–777.
3. Figueiredo FP, Parada AP, de Araujo LF, Silva WA
4. Yang R, Zhang B, Chen T, Zhang S, Chen L. Postpartum estrogen withdrawal impairs GABAergic inhibition and LTD induction in basolateral amygdala complex via down-regulation of GPR30. Eur Neuropsychopharmacol. 2017;27(8):759–772.
5. Wang Z, Zhang A, Zhao B, et al. GABA+ levels in postmenopausal women with mild-to-moderate depression: a preliminary study. Medicine. 2016;95(39):e4918.
6. Grace AA. Dysregulation of the dopamine system in the pathophysiology of schizophrenia and depression. Nat Rev Neurosci. 2016;17(8):524–532.
7. Felger JC. The role of dopamine in inflammation-associated depression: mechanisms and therapeutic implications. Current Topics Behav Neurosci. 2017;31:199–219.
8. Boer J, Ederveen E, Grundmark B. Desloratadine and depression, a drug safety signal based on worldwide spontaneous reporting of side effects. Upsala J Med Sci. 2018;123(3):174–178.
9. Huang W, Liao X, Tian J, Wu J, Shan Y, Zhou W. Traditional Chinese medicine for post-stroke depression: a systematic review and network meta-analysis (Protocol). Medicine. 2018;97(52):e13840.
10. Wang Y, Li M, Liang Y, et al. Chinese herbal medicine for the treatment of depression: applications, efficacies and mechanisms. Current Pharm Design. 2017;23(34):5180–5190.
11. Wang S, Zhao LL, Qiu XJ, et al. Efficacy and safety of a formulated herbal granula, Jiu Wei Zhen Xin, for generalized anxiety disorder: a meta-analysis. Evid Based Compl Altern Med. 2018;2018:9090181.
12. Liu F, Li R. [Discussion on relationship between “living alone with closed windows and doors” and depression]. Zhongguo zhen jiu = Chinese acupuncture & moxibustion. 2018;38(3):315–318. Chinese.
13. Parker G, Gladstone G, Chee KT. Depression in the planet’s largest ethnic group: the Chinese. Am J Psychiatry. 2001;158(6):857–864.
14. Hall BJ, Chang K, Chen W, Sou KL, Latkin C, Yeung A. Exploring the association between depression and shenjing shuairuo in a population representative epidemiological study of Chinese adults in Guangzhou, China. Transcul Psychiatry. 2018;55(6):733–753.
15. Ng QX, Venkatanarayanan N, Ho CY. Clinical use of Hypericum perforatum (St John’s wort) in depression: a meta-analysis. J Affect Disord. 2017;210:211–221.
16. Ng QX, Koh SSH, Chan HW, Ho CYX. Clinical use of curcumin in depression: a meta-analysis. J Am Med Directors Assoc. 2017;18(6):503–508.
17. Xu W, Choi HK, Huang L. State of Panax ginseng research: a global analysis. Molecules (Basel, Switzerland. 2017;22:9.
18. Lee IS, Kang KS, Kim SY. Panax ginseng pharmacopuncture: current status of the research and future challenges. Biomolecules. 2019;10:1.
19. Ma L, Wang X, Li W, et al. Synthesis and anti-cancer activity studies of dammarane-type triterpenoid derivatives. Eur J Med Chem. 2019;187:111964.
20. Wan JZ, Wang R, Zhou ZY, et al. Saponins of Panax japonicus attenuate neuronal apoptosis through oxidative stress-related pathways and autophagy in natural aging rats. Curr Pharmaceutical Biotechnol. 2019.
21. Chen W, Balan P, Popovich DG. Review of Ginseng anti-diabetic studies. Molecules (Basel, Switzerland. 2019;24:24.
22. Hu JR, Chun YS, Kim JK, Cho IJ, Ku SK. Ginseng berry aqueous extract prevents scopolamine-induced memory impairment in mice. Exp Ther Med. 2019;18(6):4388–4396.
23. Moon JN, Kim JK, Lee S, Kwon JH. Antihypertensive effects of Korean wild simulated ginseng (Sanyangsam) extracts in spontaneously hypertensive rats. Food Sci Biotechnol. 2019;28(5):1563–1569.
24. Zhang L, Chen X, Cheng Y, et al. Safety and antifatigue effect of Korean Red Ginseng: a randomized, double-blind, and placebo-controlled clinical trial. J Ginseng Res. 2019;43(4):676–683.
25. Phi LTH, Wijaya YT, Sari IN, et al. 20(R)-Ginsenoside Rg3 influences cancer stem cell properties and the epithelial-mesenchymal transition in colorectal cancer via the SNAIL signaling axis. OncoTargets Ther. 2019;12:10885–10895.
26. Hu M, Yang J, Qu L, et al. Ginsenoside Rk1 induces apoptosis and downregulates the expression of PD-L1 by targeting the NF-kappaB pathway in lung adenocarcinoma. Food Funct. 2019.
27. Wang YS, Shen CY, Jiang JG. Antidepressant active ingredients from herbs and nutraceuticals used in TCM: pharmacological mechanisms and prospects for drug discovery. Pharmacol Res. 2019;150:104520.
28. Zhang Y, Yang X, Wang S, Song S. Ginsenoside Rg3 prevents cognitive impairment by improving mitochondrial dysfunction in the rat model of alzheimer’s disease. J Agri Food Chem. 2019;67(36):10048–10058.
29. Chen C, Zhang H, Xu H, Zheng Y, Wu T, Lian Y. Ginsenoside Rb1 ameliorates cisplatin-induced learning and memory impairments. Journal of Ginseng Research. 2019;43(4):499–507.
30. Kim CY, Kim YG, Sin SJ, Koo H, Cheon K, Kim D. Preventive effect of mebicar and ginsenoside Rg1 on neurobehavioral and immunological disruptions caused by intermittent unpredictable stress in mice. Neuroimmunomodulation. 2018;25(1):49–58.
31. Cheng Y, Shen LH, Zhang JT. Anti-amnestic and anti-aging effects of ginsenoside Rg1 and Rb1 and its mechanism of action. Acta Pharmacol Sin. 2005;26(2):143–149.
32. Alo R, Avolio E, Mele M, et al. Role of Leptin and Orexin-A within the suprachiasmatic nucleus on anxiety-like behaviors in hamsters. Mol Neurobiol. 2017;54(4):2674–2684.
33. Li H, Wang T, Shi C, et al. Inhibition of GALR1 in PFC alleviates depressive-like behaviors in postpartum depression rat model by upregulating CREB-BNDF and 5-HT levels. Front Psychiatry. 2018;9:588.
34. Alfarhan AH, Rajakrishnan R, Al-Shehri MA, Al-Tamimi A, Al-Obaid S, Khalaf S. Analysis of the cuticular wax composition and ecophysiological studies in an arid plant - Ziziphus nummularia (Burm.f.) Wight & Arn. Saudi J Biol Sci. 2020;27(1):318–323.
35. Song L, Zheng J, Zhang L, et al. Phytochemical profiling and fingerprint analysis of Chinese Jujube (Ziziphus jujuba Mill.) Leaves of 66 Cultivars from Xinjiang Province. Molecules (Basel, Switzerland. 2019;24:24.
36. Ji X, Yan Y, Hou C, Shi M, Liu Y. Structural characterization of a galacturonic acid-rich polysaccharide from Ziziphus Jujuba cv. Muzao. Int J Biol Macromol. 2019.
37. Ban Z, Zhang J, Li L, et al. Ginger essential oil-based microencapsulation as an efficient delivery system for the improvement of Jujube (Ziziphus jujuba Mill.) fruit quality. Food Chem. 2020;306:125628.
38. Huang YL, Yen GC, Sheu F, Chau CF. Effects of water-soluble carbohydrate concentrate from Chinese jujube on different intestinal and fecal indices. J Agri Food Chem. 2008;56(5):1734–1739.
39. Gao QH, Wu CS, Wang M. The jujube (Ziziphus jujuba Mill.) fruit: a review of current knowledge of fruit composition and health benefits. J Agri Food Chem. 2013;61(14):3351–3363.
40. Ghimire S, Kim MS. Jujube (Ziziphus Jujuba Mill.) fruit feeding extends lifespan and increases tolerance to environmental stresses by regulating aging-associated gene expression in Drosophila. Biogerontology. 2017;18(2):263–273.
41. Xu X, Wei Y, Guo Q, et al. Pharmacological characterization of H05, a novel serotonin and noradrenaline reuptake inhibitor with moderate 5-HT2A antagonist activity for the treatment of depression. J Pharmacol Exp Ther. 2018;365(3):624–635.
42. Wang C, Hou J, Du H, et al. Anti-depressive effect of Shuangxinfang on rats with acute myocardial infarction: promoting bone marrow mesenchymal stem cells mobilization and alleviating inflammatory response. Biomed Pharmacother/Biomedecine & Pharmacotherapie. 2019;111:19–30.
43. Wang J, Jiang B, Shan Y, et al. Metabolic mapping of Schisandra chinensis lignans and their metabolites in rats using a metabolomic approach based on HPLC with quadrupole time-of-flight MS/MS spectrometry. J Sep Sci. 2019.
44. Zhang M, Xu L, Yang H. Schisandra chinensis fructus and its active ingredients as promising resources for the treatment of neurological diseases. Int J Mol Sci. 2018;19:7.
45. Rybnikar M, Smejkal K, Zemlicka M. Schisandra chinensis and its phytotherapeutical applications. Ceska a Slovenska Farmacie: Casopis Ceske Farmaceuticke Spolecnosti a Slovenske Farmaceuticke Spolecnosti. 2019;68(3):95–118.
46. Li Z, He X, Liu F, Wang J, Feng J. A review of polysaccharides from Schisandra chinensis and Schisandra sphenanthera: properties, functions and applications. Carbohydrate Polymers. 2018;184:178–190.
47. Park E, Ryu MJ, Kim NK, et al. Synergistic neuroprotective effect of Schisandra chinensis and ribes fasciculatum on neuronal cell death and scopolamine-induced cognitive impairment in rats. Int J Mol Sci. 2019;20:18.
48. Xu M, Zhang X, Ren F, et al. Essential oil of Schisandra chinensis ameliorates cognitive decline in mice by alleviating inflammation. Food Funct. 2019;10(9):5827–5842.
49. Yang S, Dong W, Li G, et al. A recombinant vaccine of Riemerella anatipestifer OmpA fused with duck IgY Fc and Schisandra chinensis polysaccharide adjuvant enhance protective immune response. Microbial Pathogenesis. 2019;136:103707.
50. Li CL, Tsuang YH, Tsai TH. Neuroprotective effect of Schisandra chinensis on methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced parkinsonian syndrome in C57BL/6 Mice. Nutrients. 2019;11:7.
51. Kim JS, Takanche JS, Kim JE, Jeong SH, Han SH, Yi HK. Schisandra chinensis extract ameliorates age-related muscle wasting and bone loss in ovariectomized rats. Phytotherapy Research: PTR. 2019;33(7):1865–1877.
52. Chen X, Cao J, Sun Y, et al. Ethanol extract of Schisandrae chinensis fructus ameliorates the extent of experimentally induced atherosclerosis in rats by increasing antioxidant capacity and improving endothelial dysfunction. Pharm Biol. 2018;56(1):612–619.
53. Zhao ZY, Zhang YQ, Zhang YH, et al. The protective underlying mechanisms of Schisandrin on SH-SY5Y cell model of Alzheimer’s disease. J Toxicol Environ Health Part A. 2019;82(19):1019–1026.
54. Bi Y, Fu Y, Wang S, Chen X, Cai X. Schizandrin A exerts anti-tumor effects on A375 cells by down-regulating H19. Braz J Med Biol Res/Revista Brasileira de Pesquisas Medicas e Biologicas. 2019;52(10):e8385.
55. Zhou F, Wang M, Ju J, et al. Schizandrin A protects against cerebral ischemia-reperfusion injury by suppressing inflammation and oxidative stress and regulating the AMPK/Nrf2 pathway regulation. Am J Transl Res. 2019;11(1):199–209.
56. Jiang EP, Li H, Yu CR, et al. Schisandrin B protects PC12 cells against oxidative stress of neurodegenerative diseases. Neuroreport. 2015;26(6):360–366.
57. Li N, Liu J, Wang M, et al. Sedative and hypnotic effects of Schisandrin B through increasing GABA/Glu ratio and upregulating the expression of GABAA in mice and rats. Biomed Pharmacother. 2018;103:509–516.
58. Zhu LX, Xu J, Wang RJ, et al. Correlation between quality and geographical origins of poria cocos revealed by qualitative fingerprint profiling and quantitative determination of triterpenoid acids. Molecules (Basel, Switzerland. 2018;23:9.
59. Oh J, Yoon DH, Han JG, Choi HK, Sung GH. 1)H NMR based metabolite profiling for optimizing the ethanol extraction of Wolfiporia cocos. Saudi J Biol Sci. 2018;25(6):1128–1134.
60. Li Y, Wang Y. Differentiation and comparison of Wolfiporia cocos raw materials based on multi-spectral information fusion and chemometric methods. Sci Rep. 2018;8(1):13043.
61. Li Y, Li X, Lu Y, et al. Co-delivery of Poria cocos extract and doxorubicin as an ‘all-in-one’ nanocarrier to combat breast cancer multidrug resistance during chemotherapy. Nanomedicine. 2020;23:102095.
62. Li X, He Y, Zeng P, et al. Molecular basis for Poria cocos mushroom polysaccharide used as an antitumour drug in China. J Cell Mol Med. 2019;23(1):4–20.
63. Liu X, Liu C, Tian J, et al. Plasma metabolomics of depressed patients and treatment with Xiaoyaosan based on mass spectrometry technique. J Ethnopharmacol. 2020;246:112219.
64. Sun SS, Wang K, Ma K, Bao L, Liu HW. An insoluble polysaccharide from the sclerotium of Poria cocos improves hyperglycemia, hyperlipidemia and hepatic steatosis in ob/ob mice via modulation of gut microbiota. Chin J Nat Med. 2019;17(1):3–14.
65. Singh A, Zhao K. Treatment of insomnia with traditional chinese herbal medicine. International Review of Neurobiology. 2017;135:97–115.
66. May BH, Feng M, Zhou IW, et al. Memory impairment, dementia, and Alzheimer’s disease in classical and contemporary traditional chinese medicine. J Alter Compl Med. 2016;22(9):695–705.
67. Choi YH. Induction of apoptosis by an ethanol extract of Poria cocos Wolf. in human leukemia U937 cells. Oncol Rep. 2015;34(5):2533–2540.
68. Lu J, Fu L, Qin G, Shi P, Fu W. The regulatory effect of Xiaoyao San on glucocorticoid receptors under the condition of chronic stress. Cell Mol Biol. 2018;64(6):103–109.
69. Peng YQ, Fan LL, Mao FY, et al. Genetic diversity and population structure of a protected species: polygala tenuifolia Willd. C R Biol. 2018;341(3):152–159.
70. Jiang H, Liu T, Li L, Zhao Y, Pei L, Zhao J. Predicting the potential distribution of polygala tenuifolia Willd. under climate change in China. PLoS One. 2016;11(9):e0163718.
71. Tsujimoto T, Nishihara M, Osumi Y, et al. Structural analysis of polygalaxanthones, C-Glucosyl Xanthones of polygala tenuifolia roots. Chem Pharm Bull. 2019;67(11):1242–1247.
72. Xu R, Mao F, Zhao Y, et al. UPLC quantitative analysis of multi-components by single marker and quality evaluation of polygala tenuifolia wild. extracts. Molecules (Basel, Switzerland. 2017;22:12.
73. Wang L, Jin GF, Yu HH, et al. Protective effects of tenuifolin isolated from Polygala tenuifolia Willd roots on neuronal apoptosis and learning and memory deficits in mice with Alzheimer’s disease. Food Funct. 2019;10(11):7453–7460.
74. Chen H, Huang Q, Zhang S, et al. The Chinese Herbal Formula PAPZ ameliorates behavioral abnormalities in depressive mice. Nutrients. 2019;11:4.
75. Xue W, Hu JF, Yuan YH, et al. Polygalasaponin XXXII from Polygala tenuifolia root improves hippocampal-dependent learning and memory. Acta Pharmacol Sin. 2009;30(9):1211–1219.
76. Zhao Z, Song H, Xie J, et al. Research progress in the biological activities of 3,4,5-trimethoxycinnamic acid (TMCA) derivatives. Eur J Med Chem. 2019;173:213–227.
77. Zhao Z, Bai Y, Xie J, et al. Excavating precursors from the traditional Chinese herb Polygala tenuifolia and Gastrodia elata: synthesis, anticonvulsant activity evaluation of 3,4,5-trimethoxycinnamic acid (TMCA) ester derivatives. Bioorganic Chemistry. 2019;88:102832.
78. Islam MT, Khalipha ABR, Bagchi R, et al. Anticancer activity of thymol: a literature-based review and docking study with Emphasis on its anticancer mechanisms. IUBMB Life. 2019;71(1):9–19.
79. Chen CY, Wei XD, Chen CR. 3,4,5-Trimethoxycinnamic acid, one of the constituents of Polygalae Radix exerts anti-seizure effects by modulating GABAAergic systems in mice. J Pharm Sci. 2016;131(1):1–5.
80. Kawashima K, Miyako D, Ishino Y, Makino T, Saito K, Kano Y. Anti-stress effects of 3,4,5-trimethoxycinnamic acid, an active constituent of roots of Polygala tenuifolia (Onji). Biol Pharm Bull. 2004;27(8):1317–1319.
81. Wang XJ, Zhang AH, Kong L, et al. Rapid discovery of quality-markers from Kaixin San using chinmedomics analysis approach. Phytomedicine. 2019;54:371–381.
82. Guo X, Gao Z, Wang J, et al. Purification of tertiary and quaternary alkaloids from Rhizoma Corydalis using reversed-phase/weak cation-exchange mixed-mode class separation combined with preparative C18 and silica based strong cation-exchange chromatography. J Chromatography B Anal Technol Biomed Life Sci. 2019;1126-1127:121742.
83. Huo W, Zhang Y, Liu Y, et al. Dehydrocorydaline attenuates bone cancer pain by shifting microglial M1/M2 polarization toward the M2 phenotype. Molecular Pain. 2018;14:1744806918781733.
84. Zheng X, Zheng W, Zhou J, et al. Study on the discrimination between Corydalis Rhizoma and its adulterants based on HPLC-DAD-Q-TOF-MS associated with chemometric analysis. J Chromatography B Anal Technol Biomed Life Sci. 2018;1090:110–121.
85. Guo Z, Cai R, Su H, Li Y. Alkaloids in processed rhizoma corydalis and crude rhizoma corydalis analyzed by GC/MS. J Anal Meth Chem. 2014;2014:281342.
86. Zhu W, Zhang Y, Huang Y, Lu L. Chinese herbal medicine for the treatment of drug addiction. Int Rev Neurobiol. 2017;135:279–295.
87. Li R, Chan W, Mat W, et al. Antiaging and anxiolytic effects of combinatory formulas based on four medicinal Herbs. Evid Based Compl Altern Med. 2017;2017:4624069.
88. Zhang Q, Chen C, Wang FQ, et al. Simultaneous screening and analysis of antiplatelet aggregation active alkaloids from Rhizoma Corydalis. Pharm Biol. 2016;54(12):3113–3120.
89. Wu H, Wang P, Liu M, et al. A 1H-NMR-based metabonomic study on the anti-depressive effect of the total Alkaloid of Corydalis Rhizoma. Molecules (Basel, Switzerland). 2015;20(6):10047–10064.
90. Zhao W, Shen C, Zhu J, et al. Identification and characterization of methyltransferases involved in benzylisoquinoline alkaloids biosynthesis from Stephania intermedia. Biotechnol Letters. 2019.
91. Sun C, Chen Z, Wang H, Ding K. Tetrahydropalmatine prevents high-fat diet-induced hyperlipidemia in golden hamsters (Mesocricetus Auratus). Med Sci Monitor. 2018;24:6564–6572.
92. Sun R, Song Y, Li S, et al. Levo-tetrahydropalmatine attenuates neuron apoptosis induced by cerebral ischemia-reperfusion injury: involvement of c-Abl activation. J Mol Neurosci. 2018;65(3):391–399.
93. Lixia H, Jun C, Song H, FaHu Y, Jinwen T. Neuroprotective effect of (-)-tetrahydropalmatine in Japanese encephalitis virus strain GP-78 infected mouse model. Microbial Pathogenesis. 2018;114:197–203.
94. Le PM, Srivastava V, Nguyen TT, et al. Stephanine from Stephania venosa (Blume) spreng showed effective antiplasmodial and anticancer activities, the latter by inducing apoptosis through the reverse of mitotic exit. Phytother Res. 2017;31(9):1357–1368.
95. Zhang MY, Liu YP, Zhang LY, et al. Levo-tetrahydropalmatine attenuates bone cancer pain by inhibiting microglial cells activation. Med Inflammation. 2015;2015:752512.
96. Qu Z, Zhang J, Yang H, et al. Protective effect of tetrahydropalmatine against d-galactose induced memory impairment in rat. Physiol Behav. 2016;154:114–125.
97. Lee B, Sur B, Yeom M, Shim I, Lee H, Hahm DH. Erratum to “l-tetrahydropalmatine ameliorates development of anxiety and depression-related symptoms induced by single prolonged stress in rats” [biomol. Ther. 22 (2014) 213-222]. Biomol Ther (Seoul). 2014;22(5):474.
98. Lee B, Sur B, Yeom M, Shim I, Lee H, Hahm DH. L-tetrahydropalmatine ameliorates development of anxiety and depression-related symptoms induced by single prolonged stress in rats. Biomol Ther (Seoul). 2014;22(3):213–222.
99. Shen Y, Xu CL, Xuan WD, et al. A new furostanol saponin from Asparagus cochinchinensis. Arch Pharmacal Res. 2011;34(10):1587–1591.
100. Fan LL, Zhu S, Chen HB, Yang DH, Cai SQ, Komatsu K. Identification of the botanical source of stemonae radix based on polymerase chain reaction with specific primers and polymerase chain reaction-restriction fragment length polymorphism. Biol Pharm Bull. 2009;32(9):1624–1627.
101. Lee HA, Kim JE, Sung JE, et al. Asparagus cochinchinensis stimulates release of nerve growth factor and abrogates oxidative stress in the Tg2576 model for Alzheimer’s disease. BMC Complement Altern Med. 2018;18(1):125.
102. Jalsrai A, Numakawa T, Kunugi H, Dieterich DC, Becker A. The neuroprotective effects and possible mechanism of action of a methanol extract from Asparagus cochinchinensis: in vitro and in vivo studies. Neuroscience. 2016;322:452–463.
103. Lee JH, Lim HJ, Lee CW, et al. Methyl protodioscin from the roots of asparagus cochinchinensis attenuates airway inflammation by inhibiting cytokine production. Evid Based Compl Altern Med. 2015;2015:640846.
104. Koo HN, Jeong HJ, Choi JY, et al. Inhibition of tumor necrosis factor-alpha-induced apoptosis by Asparagus cochinchinensis in Hep G2 cells. J Ethnopharmacol. 2000;73(1–2):137–143.
105. Xia T, Dong X, Jiang Y, et al. Metabolomics profiling reveals Rehmanniae radix preparata extract protects against glucocorticoid-induced osteoporosis mainly via intervening steroid hormone biosynthesis. Molecules (Basel, Switzerland. 2019;24:2.
106. Meng X, He M, Guo R, et al. Investigation of the effect of the degree of processing of radix Rehmanniae preparata (shu dihuang) on shu dihuangtan carbonization preparation technology. Molecules (Basel, Switzerland. 2017;22:7.
107. Liu C, Ma R, Wang L, et al. Rehmanniae Radix in osteoporosis: a review of traditional Chinese medicinal uses, phytochemistry, pharmacokinetics and pharmacology. J Ethnopharmacol. 2017;198:351–362.
108. Yuan H, Yang M, Han X, Ni X. The therapeutic effect of the chinese herbal medicine, Rehmanniae radix preparata, in attention deficit hyperactivity disorder via reversal of structural abnormalities in the cortex. Evid Based Compl Alter Med. 2018;2018:3052058.
109. Ye Q, Yuan XL, Yuan CX, Zhang HZ, Yang XM. Zishenpingchan granules for the treatment of Parkinson’s disease: a randomized, double-blind, placebo-controlled clinical trial. Neural Regeneration Res. 2018;13(7):1269–1275.
110. Song L, Zhou QH, Wang HL, et al. Chinese herbal medicine adjunct therapy in patients with acute relapse of multiple sclerosis: a systematic review and meta-analysis. Compl Ther Med. 2017;31:71–81.
111. Cui Y, Rong C, Wang J, et al. Mechanism-based anti-anxiety effects of polysaccharides extracted from shudihuang (radix rehmanniae preparata) by two-dimensional electrophoresis analysis in rat hippocampus proteins. J Trad Chin Med/Chung I Tsa Chih Ying Wen Pan. 2013;33(4):524–530.
112. Jung EY, Lee MS, Ahn CJ, Cho SH, Bae H, Shim I. The neuroprotective effect of gugijihwang-tang on trimethyltin-induced memory dysfunction in the rat. Evid Based Compl Altern Med. 2013;2013:542081.
113. Han Y, Jung HW, Lee JY, et al. 2,5-dihydroxyacetophenone isolated from Rehmanniae Radix Preparata inhibits inflammatory responses in lipopolysaccharide-stimulated RAW264.7 macrophages. J Med Food. 2012;15(6):505–510.
114. Eum HA, Lee WY, Kim SH, et al. Anti-inflammatory activity of CML-1: an herbal formulation. Am J Chin Med. 2005;33(1):29–40.
115. Tan W, Yu KQ, Liu YY, et al. Anti-fatigue activity of polysaccharides extract from Radix Rehmanniae Preparata. Int J Biol Macromol. 2012;50(1):59–62.
116. Wang LY, Yu X, Li XX, et al. Catalpol exerts a neuroprotective effect in the MPTP mouse model of Parkinson’s disease. Front Aging Neurosci. 2019;11:316.
117. Zhu HF, Shao Y, Qin L, et al. Catalpol enhances neurogenesis and inhibits apoptosis of new neurons via BDNF, but not the BDNF/Trkb pathway. Drug Design Devel Ther. 2019;13:4145–4157.
118. Yuan H, Ni X, Zheng M, Han X, Song Y, Yu M. Effect of catalpol on behavior and neurodevelopment in an ADHD rat model. Biomed Pharmacother/Biomedecine & Pharmacotherapie. 2019;118:109033.
119. Zhu H, Wang J, Shao Y, Wan D. Catalpol may improve axonal growth via regulating miR-124 regulated PI3K/AKT/mTOR pathway in neurons after ischemia. Ann Transl Med. 2019;7(14):306.
120. Zhang J, Bi R, Meng Q, et al. Catalpol alleviates adriamycin-induced nephropathy by activating the SIRT1 signalling pathway in vivo and in vitro. Br Jo Pharmacol. 2019;176(23):4558–4573.
121. Wang JM, Yang LH, Zhang YY, et al. BDNF and COX-2 participate in anti-depressive mechanisms of catalpol in rats undergoing chronic unpredictable mild stress. Physiol Behav. 2015;151:360–368.
122. Vasconcelos NG, Croda J, Simionatto S. Antibacterial mechanisms of cinnamon and its constituents: a review. Microbial Pathogenesis. 2018;120:198–203.
123. Hariri M, Ghiasvand R. Cinnamon and Chronic Diseases. Advan Exp Med Biol. 2016;929:1–24.
124. Mousavi SM, Karimi E, Hajishafiee M, Milajerdi A, Amini MR, Esmaillzadeh A. Anti-hypertensive effects of cinnamon supplementation in adults: a systematic review and dose-response Meta-analysis of randomized controlled trials. Crit Rev Food Sci Nutr. 2019;1–11.
125. Ose R, Tu J, Schink A, et al. Cinnamon extract inhibits allergen-specific immune responses in human and murine allergy models. Clin Exp Allergy. 2020;50(1):41–50.
126. Liang Y, Li Y, Sun A, Liu X. Chemical compound identification and antibacterial activity evaluation of cinnamon extracts obtained by subcritical n-butane and ethanol extraction. Food Sci Nutri. 2019;7(6):2186–2193.
127. Sadeghi S, Davoodvandi A, Pourhanifeh MH, et al. Anti-cancer effects of cinnamon: insights into its apoptosis effects. Eur J Med Chem. 2019;178:131–140.
128. Patel D, Jana A, Roy A, Pahan K. Cinnamon and its metabolite protect the nigrostriatum in a mouse model of parkinson’s disease via astrocytic GDNF. J Neuroimmune Pharmacol. 2019;14(3):503–518.
129. Deyno S, Eneyew K, Seyfe S, et al. Efficacy and safety of cinnamon in type 2 diabetes mellitus and pre-diabetes patients: a meta-analysis and meta-regression. Diabetes Res Clin Pract. 2019;156:107815.
130. Yulug B, Cankaya S. Translational perspective: is cinnamon a suitable agent for cognitive impairment and Alzheimer’s disease associated with brain trauma? Neural Regeneration Res. 2019;14(8):1372–1373.
131. Zhao Y, Deng H, Li K, et al. Trans-cinnamaldehyde improves neuroinflammation-mediated NMDA receptor dysfunction and memory deficits through blocking NF-kappaB pathway in presenilin1/2 conditional double knockout mice. Brain Behav Immunity. 2019;82:45–62.
132. Gurer B, Kertmen H, Kuru Bektasoglu P, et al. The effects of Cinnamaldehyde on early brain injury and cerebral vasospasm following experimental subarachnoid hemorrhage in rabbits. Metabol Brain Dis. 2019;34(6):1737–1746.
133. Lin J, Song Z, Chen X, et al. Trans-cinnamaldehyde shows anti-depression effect in the forced swimming test and possible involvement of the endocannabinoid system. Biochem Biophys Res Commun. 2019;518(2):351–356.
134. Li W, Zhi W, Zhao J, et al. Cinnamaldehyde attenuates atherosclerosis via targeting the IkappaB/NF-kappaB signaling pathway in high fat diet-induced ApoE(-/-) mice. Food Funct. 2019;10(7):4001–4009.
135. Etaee F, Komaki A, Faraji N, et al. The effects of cinnamaldehyde on acute or chronic stress-induced anxiety-related behavior and locomotion in male mice. Stress (Amsterdam, Netherlands). 2019;22(3):358–365.
136. Zhang YQ, Wang XB, Xue RR, Gao XX, Ginsenoside LW. Rg1 attenuates chronic unpredictable mild stress-induced depressive-like effect via regulating NF-kappaB/NLRP3 pathway in rats. Neuroreport. 2019;30(13):893–900.
137. Fan C, Song Q, Wang P, Li Y, Yang M, Yu SY. Neuroprotective effects of ginsenoside-rg1 against depression-like behaviors via suppressing glial activation, synaptic deficits, and neuronal apoptosis in Rats. Front Immunol. 2018;9:2889.
138. Wan S, Xu M, Hu L, et al. Schisandrin rescues depressive-like behaviors induced by chronic unpredictable mild stress via GDNF/ERK1/2/ROS and PI3K/AKT/NOX signaling pathways in mice. Psychiatry Res. 2017;257:230–237.
139. Bhattamisra SK, Yap KH, Rao V, Choudhury H. Multiple biological effects of an iridoid glucoside, catalpol and its underlying molecular mechanisms. Biomolecules. 2019;10:1.
140. Shi BY, Rao ZL, Luo J, et al. [Protective effect and mechanism of Xiaoyao San on lipopolysaccharide-induced hippocampal neurons injury]. Zhongguo Zhong Yao Za Zhi = Zhongguo Zhongyao Zazhi = China Journal of Chinese Materia Medica. 2019;44(4):781–786.Chinese.
141. Wang M, Bi Y, Zeng S, et al. Modified Xiaoyao San ameliorates depressive-like behaviors by triggering autophagosome formation to alleviate neuronal apoptosis. Biomed Pharmacother. 2019;111:1057–1065.
142. Wang J, Li X, He S, et al. Regulation of the kynurenine metabolism pathway by Xiaoyao San and the underlying effect in the hippocampus of the depressed rat. J Ethnopharmacol. 2018;214:13–21.
143. Liu Q, Sun NN, Wu ZZ, Fan DH, Cao MQ. Chaihu-Shugan-San exerts an antidepressive effect by downregulating miR-124 and releasing inhibition of the MAPK14 and Gria3 signaling pathways. Neural Regeneration Res. 2018;13(5):837–845.
144. He Z, Fan R, Zhang C, et al. Chaihu-Shugan-San Reinforces CYP3A4 expression via pregnane X receptor in depressive treatment of liver-Qi stagnation syndrome. Evid Based Compl Altern Med. 2019;2019:9781675.
145. Ren F, Ma Z, Shen Y, et al. Effects of Chaihu-Shugan-San capsule for psychogenic erectile dysfunction: study protocol of a randomized placebo-controlled trial. Medicine. 2019;98(46):e17925.
146. Chen S, Asakawa T, Ding S, et al. Chaihu-Shugan-San administration ameliorates perimenopausal anxiety and depression in rats. PLoS One. 2013;8(8):e72428.
147. Chen JX, Ji B, Lu ZL, Hu LS. Effects of chai Hu (radix burpleuri) containing formulation on plasma beta-endorphin, epinephrine and dopamine on patients. Am J Chin Med. 2005;33(5):737–745.
148. Xiong Z, Yang J, Huang Y, et al. Serum metabonomics study of anti-depressive effect of Xiao-Chai-Hu-Tang on rat model of chronic unpredictable mild stress. J Chromatography B Anal Technol Biomed Life Sci. 2016;1029–1030:28–35.
149. Miao M, Peng M, Chen H, Liu B. Effects of Baihe Dihuang powder on chronic stress depression rat models. Saudi J Biol Sci. 2019;26(3):582–588.
150. Sheng CX, Chen ZQ, Cui HJ, et al. Is the Chinese medicinal formula Guipi Decoction () effective as an adjunctive treatment for depression? A meta-analysis of randomized controlled trials. Chin J Integrative Med. 2017;23(5):386–395.
151. Lu LL, Shen XH, Chen JX. [Xuefu zhuyu oral liquid intervened stress-stimulated depression model rats]. Zhongguo Zhong Xi yi Jie He Za Zhi Zhongguo Zhongxiyi Jiehe Zazhi = Chinese Journal of Integrated Traditional and Western Medicine. 2013;33(5):638–640. Chinese.
152. Price RB, Nock MK, Charney DS, Mathew SJ. Effects of intravenous ketamine on explicit and implicit measures of suicidality in treatment-resistant depression. Biol Psychiatry. 2009;66(5):522–526.
153. Loo CK, Galvez V, O’Keefe E, et al. Placebo-controlled pilot trial testing dose titration and intravenous, intramuscular and subcutaneous routes for ketamine in depression. Acta psychiatrica Scandinavica. 2016;134(1):48–56.
154. Pereira VS, Hiroaki-Sato VA. A brief history of antidepressant drug development: from tricyclics to beyond ketamine. Acta Neuropsychiatr. 2018;30(6):307–322.
155. Liu J, Moghaddam B. Regulation of glutamate efflux by excitatory amino acid receptors: evidence for tonic inhibitory and phasic excitatory regulation. J Pharmacol Exp Ther. 1995;274(3):1209–1215.
156. Bus BA, Molendijk ML, Tendolkar I, et al. Chronic depression is associated with a pronounced decrease in serum brain-derived neurotrophic factor over time. Mol Psychiatry. 2015;20(5):602–608.
157. Ng QX, Lim DY, Chee KT. Reimagining the spectrum of affective disorders. Bipolar Disorders. 2020.
158. Zhang B, Zhang H, Du C, et al. Metabolic responses of the growing Daphnia similis to chronic AgNPs exposure as revealed by GC-Q-TOF/MS and LC-Q-TOF/MS. Water Research. 2017;114:135–143.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
