Back to Journals » Neuropsychiatric Disease and Treatment » Volume 17
Traditional Chinese Formula Xiaoyaosan Alleviates Depressive-Like Behavior in CUMS Mice by Regulating PEBP1-GPX4-Mediated Ferroptosis in the Hippocampus
Authors Jiao H , Yang H, Yan Z , Chen J, Xu M, Jiang Y, Liu Y, Xue Z, Ma Q , Li X, Chen J
Received 17 January 2021
Accepted for publication 15 March 2021
Published 6 April 2021 Volume 2021:17 Pages 1001—1019
DOI https://doi.org/10.2147/NDT.S302443
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Yuping Ning
Haiyan Jiao,1 Hongjun Yang,1 Zhiyi Yan,1 Jianbei Chen,1 Mengbai Xu,1 Youming Jiang,1 Yueyun Liu,1 Zhe Xue,1 Qingyu Ma,2 Xiaojuan Li,2 Jiaxu Chen1,2
1School of Traditional Chinese Medicine, Beijing University of Chinese Medicine, Beijing, 100029, People’s Republic of China; 2Formula-Pattern Research center, School of Traditional Chinese Medicine, Jinan University, Guangzhou, 510632, Guangdong, People’s Republic of China
Correspondence: Jiaxu Chen Tel +86 1064286656
Email [email protected]
Background: At present, the pathogenesis of depression is not fully understood, and nearly half of depression patients experience no obvious effects during treatment. This study aimed to establish a depression mouse model to explore the possible role of ferroptosis in the pathogenesis of depression, and observe the effects of Xiaoyaosan on PEBP1-GPX4-mediated ferroptosis in the hippocampus.
Methods: Forty-eight male C57BL/6 mice were randomly divided into a control group, CUMS group, Xiaoyaosan group and fluoxetine group, and the model was established by chronic unpredictable mild stress (CUMS) for a successive 6 weeks. The medication procedure was performed from the 4th to the 6th week of modeling. The behavioral evaluations were measured to evaluate depressive-like behaviors. The expressions of GPX4, FTH1, ACSL4 and COX2 were detected as ferroptosis-related indicators. Then, the total iron and ferrous content in the hippocampus were measured. The levels of PEBP1 and ERK1/2 were observed, and the expressions of GFAP and IBA1 were also detected to measure the functions of astrocytes and microglia in the hippocampus.
Results: Eight herbs of Xiaoyaosan had 133 active ingredients which could regulate the 43 ferroptosis-related genes in depression. After 6 weeks of modeling, the data showed that mice in the CUMS group had obvious depressive-like behaviors, and medication with Xiaoyaosan or fluoxetine could significantly improve the behavioral changes. The expressions of GPX4, FTH1, ACSL4, COX2, PEBP1, ERK1/2, GFAP and IBA1 changed in the CUMS group mice, while the total iron and ferrous content also changed. Xiaoyaosan and fluoxetine had obvious curative effects that could significantly alleviate the above changes in the hippocampus.
Conclusion: Our results revealed that the activation of ferroptosis might exist in the hippocampi of CUMS-induced mice. The PEBP1-GPX4-mediated ferroptosis could be involved in the antidepressant mechanism of Xiaoyaosan. It also implied that ferroptosis could become a new target for research into the depression mechanism and antidepressant drugs.
Keywords: chronic unpredictable mild stress, depression, Xiaoyaosan, ferroptosis, GPX4, PEBP1
Introduction
Depression is a mental illness that seriously endangers the physical and mental health of human beings. It is a common mood disorder characterized by significant and persistent low mood, delayed thinking, impaired cognitive function, decreased volitional activity, and somatic symptoms.1 The etiology of depression is complex and has not yet been clarified. It is generally believed that it may be related to a variety of factors, including genetics, biology and social psychology. With the increase of pressure in modern human society, the accelerated pace of life is accompanied by an increase in multiple complex stress factors. The incidence of depression also shows an increasing trend. Depression affects at least 264 million people worldwide, with an increase of 18.4% between 2005 and 2015.2 The World Health Organization’s forecast shows that depression will become the world’s most important cause of disability by 2030. Furthermore, the longer depression is not treated, the worse the patient’s condition will be, and it will also increase the pressure on the national disability budget.3 The adverse consequences of depression have placed a heavy burden on all societies, so there is an urgent need to solve this problem; however, its biological mechanism is still unclear.
Ferroptosis is a newly discovered programmed cell death process which is closely related to the disturbance of iron metabolism.4 In brief, the accumulation of reactive oxygen species (ROS) produced by lipid peroxidation and iron metabolism is the main inducer of ferroptosis, and ferroptosis is a regulated cell necrosis that is distinct from apoptosis, necrosis and autophagy at the morphological, biological and genetic levels, and can be triggered by a variety of substances and external conditions.5 In most cases, ferroptosis is caused by failure of the membrane lipid repair enzyme-glutathione peroxidase 4 (GPX4), which leads to the accumulation of ROS on membrane lipids, and this accumulation process requires the participation of iron.6 Many studies have found that there is an inseparable relationship between ferroptosis and neurological diseases.7–9 In addition to inflammatory changes, the process of ferroptosis in the central nervous system is often accompanied by functional changes in astrocyte and microglia. Therefore, the expressions of the glial fibrillary acidic protein (GFAP) and ionic calcium junction protein molecule-1 (IBA-1) can be used to confirm the occurrence of neuroinflammation and ferroptosis.10
Phoshaptidylethanolamine binding proteins (PEBPs) are a class of highly conserved intracytoplasmic soluble proteins that were first extracted from cow brain in 1984, and have a high affinity with phosphatidylethanolamine, with a molecular weight of 21 ~ 23kDa and more than 400 family members.11 The PEBPs family has four subclasses: PEBP1-4. PEBP1 is highly expressed in the brain, adrenal gland, thyroid and other organs,12 PEBP2 is mainly highly expressed in testis,13 PEBP3 is currently assumed to exist and its function is unclear, while PEBP4 is mainly expressed in skeletal muscle, myocardium, thyroid and other tissues.14 PEBPs have been reported to be involved in different physiological phenomena, including the origin and fluidity of biofilms, the formation of functional domains, the stimulation of acetylcholine during neurodevelopment, the inhibition of serine proteases in nerve tissue, the regulation of MAPK signaling pathways, cell proliferation and survival, sperm formation and maturity and so on.15 PEBP1 was first found in the brain, and then it was discovered that, through yeast two-hybrid experiments, it can be isolated as a Raf kinase inhibitor protein (RKIP), so it is also called RKIP.16 Changes in the extracellular regulated protein kinases (ERK) pathway are closely related to depression and other psychiatric disorders.17 Some studies have found that down-regulation of ERK levels is detected in the brains of depressed suicidal patients,18 while changes in the expression of PEBP1 may affect the ERK pathway, which may contribute to depression.19,20 In addition, PEBP1 may inhibit the expression of GPX4, and the ERK pathway is also believed to be closely related to the occurrence of iron death.21,22
Knowledge of Xiaoyaosan is derived from the medical book, Prescriptions of the Bureau of Taiping People’s Welfare Pharmacy in the Song Dynasty, and its antidepressant effect has been strongly confirmed in terms of TCM theory, clinical application and pharmacological research.23,24 As one of the classical prescriptions commonly used in the treatment of depression, Xiaoyaosan has the characteristics of multi-level, multi-pathway and multi-target pharmacological activities.25 In animal studies, it was found that Xiaoyaosan could improve the depression-like behaviors of CUMS mice by mediated modulation of the glutamate/glutamine cycle and glutamate transporter GLT-1 in the hippocampus.26 The brain metabonomics study of the antidepressive effect of Xiaoyaosan on CUMS-induced rats showed that Xiaoyaosan significantly reversed the brain metabolic perturbation caused by CUMS.27 It also found that the antidepression ingredient of Xiaoyaosan was the petroleum ether fraction of Bupleuri radix that could exert an antidepression effect by regulating the glycometabolism, amino acid metabolism, sphingolipid metabolism, glycerophospholipid metabolism and fatty acid metabolism in CUMS rats.28 Many studies have confirmed that there is a loss of hippocampal neurons and glial cells in depression patients or model animals, and the most significant loss is glial cells.29 In the central nervous system, astrocyte and microglia play an important role in modulating stress-related neuroinflammation; however, long-term chronic stress can cause astrocyte and microglia injury. Xiaoyaosan plays a therapeutic role by improving neuroinflammation and regulating the functions of hippocampal astrocytes and microglia.30,31 At the same time, some studies have pointed out that Xiaoyaosan can up-regulate the phosphorylation level of ERK and activate the TrkB pathway, thereby improving depressive behaviors.32 At present, the relationship between ferroptosis and the molecular mechanism of depression has not been verified; the antidepressant mechanism of Xiaoyaosan also needs to be further explained. Therefore, this study investigated the relationship between the mechanism of depression and the ferroptosis-related pathway, and observed whether the antidepressant mechanism of Xiaoyaosan was related to the PEBP1-GPX4-mediated ferroptosis mechanism.
Materials and Methods
Subjects
The specific-pathogen free (SPF) male C57BL/6 mice (8-week-old, SCXK (Beijing) 2016–0006) were purchased from Beijing Vital River Laboratory Animal Technology Limited Company, and then fed in a standard animal feeding room (temperature: 22 ± 2°C; relative humidity: 30–40%; light condition: 12h/12h dark/light cycle) for the study.
The animal experiment in the study was approved by the Institutional Animal Care and Use Committee of Beijing University of Chinese Medicine (No. BUCM-4-2018120401-4053) and complied with the existing current animal welfare guidelines and the Animal Management Rules of the Chinese Ministry of Health.
Preparation of Traditional Chinese Formula
The Traditional Chinese Formula used in the study was Xiaoyaosan, composed of Radix Angelicae sinensis (Chinese angelica), Radix Paeoniae alba (debark peony root), Poria (Indian bread), Radix bupleuri (Chinese thorowax root), Rhizoma Atractylodis macrocephalae (largehead atractylodes rhizome), Radix glycyrrhizae (liquorice root), Herba menthae (peppermint) and Rhizoma Zingiberis recens (fresh ginger) in a ratio of 3:3:3:3:3:1.5:1:1.33
These decoction pieces of Xiaoyaosan were identified by the HPLC-mass spectrometry analysis (LC-MS/MS), and purchased from Beijing Tongrentang (Bozhou) Pieces Co. Ltd, Bozhou, China.26 Next, the prepared drug, in pieces, was used to prepare Xiaoyaosan dry powder by Jiuzhitang Co. Ltd., Changsha, China, in accordance with the preparation process in Chinese Pharmacopoeia, 2015 edition.34
Network Pharmacology Study of Active Ingredients in Xiaoyaosan
How the active ingredients of Xiaoyaosan intervened in ferroptosis-related substances in depression weas discussed based on network pharmacology. The active ingredients of the above 8 herbs in Xiaoyaosan were screened according to absorption distribution metabolism and enhancement (ADME). The parameters of ADME were oral bioavailability (OB) and drug likeness (DL).35 According to ADME (OB ≥ 30% and DL ≥ 0.18), the active ingredients of Xiaoyaosan were collected from traditional Chinese medicine databases, including TCMSP,36 TCM Database@Taiwan37 and BATMAN-TCM.38 Target prediction was carried out using the SwissTargetPrediction database,39 the reliability of the predicted targets was screened (Probability ≥ 30%) and then the targets of active ingredients in Xiaoyaosan were collected. Table 1 shows the number of active ingredients and predicted targets.
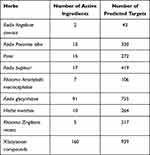 |
Table 1 Active Ingredients and Predicted Targets of Each Herb in Xiaoyaosan |
The disease databases, including GeneCards,40 OMIM,41 TTD,42 CTD43 and FerrDb,44 were used to screen the depression and ferroptosis-related targets. Then, each target of active ingredients, depression and ferroptosis were searched in the UniProt database45 to get the UniProt gene ID. Here, 12,608 depression-related targets and 260 ferroptosis-related targets were screened out.
CUMS Procedure and Drug Administration
A total of 48 mice were randomly assigned to 4 groups (n = 12): a control group (no stress + physiological saline), a CUMS group (CUMS + physiological saline), a Xiaoyaosan group (CUMS + Xiaoyaosan treatment) and a fluoxetine group (CUMS + fluoxetine treatment). The CUMS procedure, shown in Figure 1, was performed as previously described.46 Briefly, the mice in the CUMS, Xiaoyaosan and fluoxetine groups were exposed to the following stressors: restraint stress for 3 hours; crowded cages for 24 hours; food deprivation for 24 hours; water deprivation for 24 hours; 4°C cold water swimming for 5 minutes; empty cages for 11 hours; and exposure to wet and soiled cages for 24 hours. Each stressor was not repeated for two consecutive days, and CUMS was performed for 6 consecutive weeks.
To observe the behaviors of the mice in this study during the CUMS process, their body weight, food intake and sucrose preference were measured weekly, while the data from the day before the experiment was used as the baseline. The other behavioral assessments, such as the open field test, forced swimming test and novelty suppressed feeding test, were performed after 6 weeks of the CUMS procedure.
Drug intervention lasted for 3 weeks, starting from the 4th week of modeling. The Xiaoyaosan dry powder and the fluoxetine capsules were dissolved in distilled water, then the mice in the two treatment groups received Xiaoyaosan (0.254 g/kg/d) and fluoxetine (2.6 mg/kg/d) by intragastric administration after the daily CUMS procedure, respectively.30 Meanwhile, mice in the control and CUMS group received 0.5 mL physiological saline.
Sucrose Preference Test
The sucrose preference test (SPT), based on a two-bottle choice paradigm, was carried out weekly and performed as previously described.47 In short, each mouse was given two bottles of 1% sucrose solution for 24 h, then one filled with 1% sucrose solution and the other with fresh water for another 24 h. After 24-hour fasting and water deprivation, each mouse received two bottles filled with 1% sucrose solution and fresh water for 1 h. The sucrose preference ratio within 1 h was recorded, which could be the objective indicator of delight hedonia.
Open Field Test
The open field test (OFT) was performed in a light- and sound-attenuated room with a dim light after the CUMS process, based on a previously described procedure, and was used to observe the autonomous behavior, exploratory behavior and tension of mice in the new surroundings.48 The open field apparatus (80 cm × 80 cm × 40 cm) was made of opaque material and divided into 25 equal squares (16 outer and 9 inner). Each time, one mouse was placed in the central grid to observe the movement behavior of mice within 5 minutes, then the locomotor ability (total moving distance and number of entries into the open area) of mice was recorded and evaluated by the EthoVision 3.0 software (Noldus, Netherlands).
Forced Swimming Test
The forced swimming test (FST) was conducted after the 6-week modeling, as previously described.49 When the mice were put into a limited space to swim, they tried to escape by swimming desperately at the beginning, and soon became floating and immobile. They only showed their nostrils to keep breathing, and their limbs occasionally moved to keep their bodies from sinking. Actually, the basic principle of FST was that animals gave up hope of escape, which belongs to behavioral despair. The FST procedure lasted for two days. On the first day, the mice were forced to swim in the deep water at 25 °C for 15 minutes, then after 24 hours, FST was performed for 5 minutes under the same conditions, and the immobility time of mice was recorded.
Novelty Suppressed Feeding Test
The novelty suppressed feeding test (NSFT) was used to measure the animals' depressive behavioral changes in a novelty environment by observing their feeding latency, and it was performed according to the previously described method.50 In brief, the testing apparatus consisted of a white plastic box (50 cm × 50 cm × 40 cm) with a single pellet of regular chow placed in the center. The NSFT was conducted after food deprivation for 24 hours. The mice were individually placed in a corner of the apparatus, then the feeding latency beforet each mouse first held the food pellet was measured for up to 5 minutes.
Preparation of Tissue Samples
After 6 weeks of modeling, the mice in the four groups were anesthetized by an intraperitoneal injection of 3% sodium pentobarbital after behavioral tests. The hippocampal tissues of 6 mice in each group were separated for Western blot analysis and iron assay, then the hippocampi of the other 6 mice in each group for quantitative real-time polymerase chain reaction (qRT-PCR) assay.
Quantitative Real-Time Polymerase Chain Reaction Analysis
The levels of GPX4, PEBP1, ERK, IBA1, GFAP, ACSL4, COX2 and FTH1 mRNAs in the hippocampus were evaluated by qRT-PCR assay. The total RNA of the hippocampus was extracted using Trizol reagent (Applied Biosystems, Waltham, USA), then the quality and concentration of total RNA were determined by 1% agarose gel electrophoresis and Q3000 micro-volume spectrophotometer (Quawell Technology, San Jose, USA). The RevertAid First Strand cDNA Synthesis Kit (Thermo Fisher Scientific, Waltham, USA) was used to transfer total RNA into cDNA according to the manufacturer'sstandard protocol. The sequences for primers are shown in Table 2 and were designed based on published mRNA sequences in NCBI and synthesized by a professional biotechnology company (Sangon Biotech Co., Ltd., Shanghai, China). The qRT-PCR reaction system was prepared by SYBR® Green PCR Master Mix (Applied Biosystems, Waltham, USA) in a final volume of 25 µL, then performed on a Multicolor Real-time PCR Detection System (Bio-Rad, Hercules, USA) with the following thermal cycling conditions: preincubation at 94 °C for 3 min, followed by 40 cycles of denaturation at 94 °C for 30 s, annealing at 55°C for 30 s and extending at 72°C for 30 s. The signals were normalized to the house-keeping gene GAPDH (BBI Life Science, Amherst, USA) and the relative expression of mRNA in each sample was calculated using the 2−∆∆Ct method.
 |
Table 2 Primer Sequences Used in the qRT-PCR Analysis |
Western Blot Analysis
The total protein of hippocampal tissues was extracted using the RIPA Lysis Buffer (Biomiga, Santiago, USA). Then the protein levels of GPX4 (Anti-Glutathione Peroxidase 4 antibody [EPNCIR144], ab125066, 1:500 dilution, Abcam, San Francisco, USA), PEBP1 (Anti-RKIP antibody [EPR2875Y], ab76582, 1:500 dilution, Abcam, San Francisco, USA), IBA1 (Anti-Iba1 antibody [EPR16589], ab178847, 1:100 dilution, Abcam, San Francisco, USA), GFAP (Anti-GFAP antibody, ab7260, 1:1000 dilution, Abcam, San Francisco, USA), total ERK1/2 (t-ERK1/2) (Anti-ERK1 + ERK2 antibody [EPR17526], ab184699, 1:1000 dilution, Abcam, San Francisco, USA) and phosphorylated ERK1/2 (p-ERK1/2) (Anti-ERK1 + ERK2 [phospho T202 + Y204] antibody, ab214362, 1:100 dilution, Abcam, San Francisco, USA) were measured by Simon Western Blot Analysis (ProteinSimple, California, USA), a capillary-based automated system with a standard protocol.51
Iron Determination Analysis
A microwave digestion method was used to pretreat hippocampal samples, and then the iron content was determined by Iron Assay kit (Sigma-Aldrich, St. Louis, USA) according to the manufacturer’s standard protocol. The mean optical density was detected by the Multiskan™ GO (Thermo Fisher Scientific, Waltham, USA) Detector system at a wavelength of 593 nm, and the total iron and ferrous content were calculated according to the standard curves.
Statistical Analysis
All statistics in this study were analyzed using SPSS 21.0 software and expressed as means ±SEM. The statistical analyses were performed by one-way analysis of variance (ANOVA) with least significant difference (LSD) post hoc multiple comparisons when equal variances were assumed. When the data had a normal distribution but the variances were not homogeneous, the Dunnett’s T3 test was used. Statistical significance was set at a minimum of p <0.05.
Results
Active Ingredients of Xiaoyaosan-Regulated Ferroptosis in Depression
The network pharmacology method was used to initially investigate whether the active components of Xiaoyaosan could improve ferroptosis-related genes in depression.
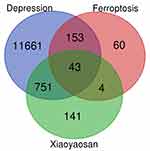
As shown in Figure 2, the 939 predicted targets, 12,608 depression-related targets and 260 ferroptosis-related targets were intersected, and we identified 43 key target genes (GPX4, PEBP1, MAPK1, MAPK3, NOX1, HRAS, ALOX12, MDM2, EGFR, G6PD, MAPK14, PTGS2, MAPK8, IDH1, ABCC1, EPAS1, SLC2A3, NOX4, FLT3, MIF, NOS2, CA9, VEGFA, DRD4, DRD5, MAPK9, TGFBR1, ATM, IL6, PIK3CA, HNF4A, HIF1A, RELA, CFTR, TLR4, AURKA, SLC2A1, SIRT1, ALOX5, DPP4, CASP8, NRAS, HSPA5) that might be associated with active ingredients of Xiaoyaosan intervening in ferroptosis-related substances in depression. Figure 3 shows the herb ingredients target genes network: 8 herbs of Xiaoyaosan compounds had 133 active ingredients which could regulate the 43 key target genes. Therefore, we can speculate that the active ingredients of traditional Chinese formula Xiaoyaosan could regulate the ferroptosis pathways in depression.
Xiaoyaosan Improved Depressive-Like Behaviors of CUMS-Exposed Mice
We observed the body weight and food intake of CUMS-exposed mice, then performed a series of behavioral tests, SPT, OFT, FST and NSFT, to evaluate the efficacy of Xiaoyaosan on depressive-like behaviors.
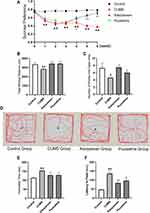
As shown in Figure 4, the body weight and food intake of mice in each group showed no significant differences before the modeling procedure began (0 week); the difference between the control group and the other three groups was gradually increased; and, finally, a significant difference was observed after 3 weeks of modeling (both p < 0.01). The treatment began at week 4 and continued for 3 weeks. The body weight and food intake of mice in the Xiaoyaosan and fluoxetine groups significantly increased compared with the mice in the CUMS group after 3 weeks of treatment (both p < 0.01), and the difference between the control group and the CUMS group was also significant (p < 0.01).
As shown in Figure 5A, the sucrose preference rate of the control group, CUMS group, Xiaoyaosan group and fluoxetine group showed no significant differences at the beginning of the research (0 week). Then, after 3 weeks of CUMS modeling, the mice in the CUMS group, Xiaoyaosan group and fluoxetine group demonstrated a significant drop in sucrose preference rate compared with the control group mice (both p < 0.01). The CUMS modeling continued for another 3 weeks with pharmacological intervention, during which the mice in the CUMS group still showed a low sucrose preference rate compared with the control group mice (both p < 0.01), while the sucrose preference rate of the mice in the Xiaoyaosan group and fluoxetine group significantly increased (both p < 0.01).
As shown in the OFT, after modeling for 6 weeks, the total moving distance and number of entries into the open area of the CUMS-exposed mice showed a significant difference compared to the control mice (p < 0.05 and p < 0.01, respectively; Figures 5B and C). The total moving distance and number of entries into the open area of mice in the Xiaoyaosan group were significantly increased (p < 0.05 and p < 0.01, respectively), while the total moving distance of mice in the fluoxetine group was also remarkably increased (p < 0.01). Figure 5D shows a representative moving trail of mice in each group.
The FST results showed that the CUMS group mice exhibited a significantly longer immobility time (p < 0.01; Figure 5E), while Xiaoyaosan or fluoxetine treatment could significantly reduce the immobility time in comparison with the CUMS group (both p < 0.05). For the NSFT, it was shown that mice in the CUMS group had a significantly longer latency to chewing the food pellet compared with the control mice (p < 0.01; Figure 5F). Xiaoyaosan or fluoxetine reversed the depressive-like changes in the NSF test (p < 0.05 and p < 0.01, respectively).
Xiaoyaosan Regulated the Ferroptosis in the Hippocampi of CUMS-Exposed Mice
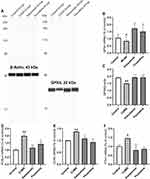
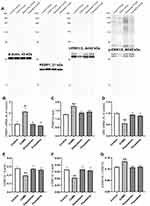
In order to explore the possible mechanism of GPX4 related ferroptosis in the mouse model of depression, the expressions of GPX4, FTH1, ACSL4 and COX2 were detected, and the total iron and ferrous content in hippocampus tissue was measured using the Iron Assay kit. As shown in Figure 6, the mRNA and protein expression levels of GPX4 were significantly down-regulated in the hippocampi of CUMS-exposed mice compared to the control group (both p < 0.01). The qRT-PCR results also showed that the expressions of FTH1, ACSL4 and COX2 mRNA in the hippocampi of CUMS group mice were significantly different compared with those of the control group micer (p < 0.05 and p < 0.01, respectively). The Xiaoyaosan or fluoxetine treatment could mitigate the stress-induced expression changes of GPX4, FTH1, ACSL4 and COX2, as shown in the comparison with CUMS group (p < 0.05 and p < 0.01, respectively).
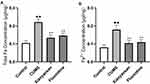
To further verify the process of GPX4-related ferroptosis in CUMS-exposed mice, the Iron Assay kit was used to determine total iron and ferrous content in the hippocampus. Figure 7 showed that total iron and ferrous content in the hippocampi of CUMS group mice obviously increased compared with that of the control group (p < 0.05 and p < 0.01, respectively). Furthermore, the treatment of Xiaoyaosan or fluoxetine could remarkably reverse the changes of hippocampal total iron and ferrous content (p < 0.05 and p < 0.01, respectively).
Xiaoyaosan Regulated the Expressions of PEBP1 and ERK1/2 in the Hippocampi of CUMS-Exposed Mice
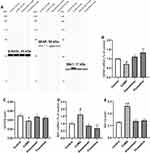
The expressions of PEBP1 and ERK1/2 were observed to further explore the possible mechanism between ferroptosis and depression. As shown in Figure 8, the mRNA and protein expression levels of PEBP1 were significantly up-regulated in the hippocampi of CUMS-exposed mice (p < 0.05 and p < 0.01, respectively), whereas the expression level of t-ERK1/2 significantly decreased (p < 0.01). The mice in the Xiaoyaosan group and fluoxetine group showed a significant increase in the levels of PEBP1 and t-ERK1/2 (p < 0.05 and p < 0.01, respectively). The CUMS modeling also decreased the level of p-ERK1/2 (p < 0.01). Moreover, the administration of Xiaoyaosan and fluoxetine noticeably increased the expression of p-ERK1/2 (both p < 0.01). Statistics also showed that the ratio of p-ERK1/2 to t-ERK1/2 in the hippocampi of CUMS group mice was higher than that of the control group (p < 0.01), while the ratio of the two treatment groups decreased significantly (p < 0.01).
Xiaoyaosan Ameliorated the Function of Neuroglial Cells in the Hippocampi of CUMS-Exposed Mice
To test the functions of astrocytes and microglia in the hippocampi of CUMS-exposed mice, the expressions of GFAP and IBA1 were identified. As shown in Figure 9, the mRNA and protein expressions of GFAP of CUMS-exposed mice were significantly reduced when compared to the control group (p < 0.05), while the mRNA and protein levels of GFAP were noticeably increased by treatment with Xiaoyaosan and fluoxetine (p < 0.05 and p < 0.01, respectively). The qRT-PCR and Western blot data also demonstrated the high level of IBA1 in the hippocampi of mice in the CUMS group compared with that of the control group mice (p < 0.05 and p < 0.01, respectively), and Xiaoyaosan or fluoxetine could significantly reverse the CUMS-induced IBA1 changes as compared to CUMS group (p < 0.05 and p < 0.01, respectively).
Discussion
Our findings revealed that ferroptosis could be involved in CUMS-induced depression, and further explained the underlying mechanism of ferroptosis in stress depression. It was further found that the antidepressant mechanism of Xiaoyaosan might be related to the improvement of ferroptosis associated with PEBP1-GPX4 related to ferroptosis. It can regulate the expressions of PEBP1, ERK1/2, GPX4, FTH1, ACSL4 and COX2, and improve the function of astrocytes and microglia in the hippocampi of CUMS model mice.
Stress occurs when individuals need to mobilize their own or external resources to adapt to life circumstances or environmental changes. Stress intensity beyond individual coping ability may lead to psychological or physical damage. A negative stress event is one of the first psychological stressors to be noticed and often causes depression, anxiety, loneliness and other negative emotional experiences.52 Many studies have found that stress can indeed lead to depression. Clinical work and life observation also reveal that many patients with depression have experienced more or more serious stressful life events than other people.53 Animal models, such as the chronic social defeat stress model,54,55 learned helplessness model56,57 and chronic mild stress model,58,59 are important tools for studying the pathogenesis of depression and screening new antidepressants. The CUMS is also an internationally accepted method of establishing an animal model of depression, which is characterized by randomness and unpredictability and more suitable for mechanism research and drug screening.60,61 Using chronic, unpredictable and low-intensity stress event modeling, the animal cannot predict the stimuli that will be received, which avoids its active adaptation and makes the stressor closer to real-life events.62 In this study, we successfully duplicated the CUMS depression model, and found, through behavioral testing, that there were obvious depression-like changes in the model mice, a finding consistent with the results of previous studies.63 Research shows that weight change in depressed patients is a factor related to depression.64 Depression modeling in research tends to cause a body weight loss in model animals.65 In the results of this study, the body weight of CUMS mice showed a significant downward trend after 6 weeks of modeling, and their food intake was also significantly different from that of control mice, a finding consistent with previous research. At the same time, SPT, FST, NSFT and OFT were also used to evaluate the behavior of model mice. The depression model established by CUMS can induce anhedonia and behavioral despair in experimental animals, which can be evaluated by SPT and FST. The depression-like behavior changes were manifested in the significant decrease of the sucrose preference coefficient and the increase in swimming immobility time.66 The NSFT used in this study reflects the latency of animal feeding, which tests the responsiveness of mice to reward and their judgment of the surrounding environment, to simulate the depressive symptoms of human beings.67 In addition, OFT is mainly used to evaluate the desire to explore and autonomous activity levels of rodents in unfamiliar environments, and can be used to evaluate their depression-like behaviors.68 The results of this study showed that the mice in the model group demonstrated a significant decrease in sucrose preference, prolonged swimming immobility time, increased latency of feeding, reduced movement distance and open area entry times in OFT, which indicated that a depression model of mice was established by the 6-week CUMS modeling. In the study, we found that treatment with Xiaoyaosan and fluoxetine could improve the depressive behavior of mice, and regulate their physical state degradation in behavioral tests. This conclusion was basically consistent with the results of previous studies, which indicated that Xiaoyaosan, a Chinese herbal compound prescription, has the same obvious antidepressant effect as fluoxetine.26,69
Ferroptosis is a nonapoptotic programmed cell death mode dependent on iron ions. This mode is different from other known programmed cell death in morphology, which is characterized by slightly smaller mitochondria than normal, and a slightly thickened mitochondrial membrane with slightly increased density.5 When ferroptosis occurs, free ferrous ions are introduced into cells from the extracellular environment through a transferrin and transferrin receptor,70 and the lipid hydroxide (L-OOH) is transformed into a toxic lipid free radical (L-O−) under the action of ferrous ions, which leads to the fragmentation of polyunsaturated fatty acids in cell membrane lipids and cell death.71 FTH1 plays an important role in intracellular iron metabolism, while NRF2 regulates the production and accumulation of Fe2+ by regulating FTH1. Inhibition of NRF2 or FTH1 can significantly promote the content of Fe2+ and the occurrence of ferroptosis, which indicates that the iron content in cells has a direct impact on the process of ferroptosis.72,73 The imbalance of lipid metabolism is often closely related to the accumulation of intracellular iron ions, and the content of polyunsaturated fatty acids determines the degree of lipid peroxidation, thus determining the degree of ferroptosis.74 It has been shown that phosphatidylethanolamines (PE) containing arachidonic acid or adrenalic acid is the key phospholipid to drive the cell toward ferroptosis, while ACSL4 can esterify coenzyme A to acyl coenzyme A in polyunsaturated fatty acids, which is used for fatty acid oxidation or the biosynthesis of polyunsaturated fatty acids.75 In addition, the occurrence of ferroptosis is related to the increase of PTGS2. The COX-2 gene encoded by PTGS2 is significantly expressed under the action of a ferroptosis inducer, and the increase in this study was not reversed by PTGS inhibitors, which indicated that COX-2 could regulate the ferroptosis process; the change in its content was one of the appropriate criteria for ferroptosis.76 GPX4 is a selenoprotein for repairing oxidative damage of lipid cells in mammals. It can prevent the development of ferroptosis by inhibiting the transformation of iron-dependent lipid peroxidation (IDLOP) into a lipid peroxy radical, thereby reducing the accumulation of L-ROS.77 It has been found that lipid peroxidation and accumulation of intracellular reactive oxygen species can be promoted by knocking out GPX4 or directly using GPX4 inhibitor RSL3, so GPX4 may be the key regulator of ferroptosis.78
In this study, it was found that total iron and ferrous ion content in the hippocampi of CUMS model mice increased significantly. GPX4, FTH1, ACSL4 and COX-2 also changed significantly, which proved that there was ferroptosis in the hippocampus of depression model mice and thus indicated that the incidence of depression may be associated with ferroptosis-related pathways. The excessive accumulation of iron causes the deactivation of GPX4 and the occurrence of iron-dependent programmed cell death, which leads to the accumulation of ROS on membrane lipids and the activation of the MEK/ERK signaling pathway.6 In the central nervous system, the ERK pathway is considered to be one of the key material bases for ferroptosis. The nuclear translocation of ERK1/2 plays a vital role in promoting ferroptosis, and the phosphorylation level of ERK is closely related to the degree of ferroptosis.79 In addition, ERK1/2, as a member of the mitogen activated protein (MAP) kinase family, mediates the pathophysiological process of cell proliferation, differentiation and death, and is also closely related to cognitive, learning, memory and other functional activities. The abnormal expression of ERK1/2 may play a key role in the pathophysiology of depression.17 In our study, decreased expression of t-ERK1/2 and p-ERK1/2 was detected in the hippocampus of depressed mice, while the ratio of p-ERK1/2 to t-ERK1/2 (phosphorylation/total) was elevated. The results showed that some of the ERK pathway was inhibited in the hippocampus of mice with depression, but the phosphorylation level of ERK increased, a finding consistent with previous experimental results.21,80 Therefore, this implied that the depression mechanism caused by stress may be related to the occurrence of ferroptosis involved in GPX4.
PEBP1 is a highly expressed cytosolic soluble protein in the brain,12 adrenal gland and thyroid gland. It is a key factor in Raf-1/MEK/ERK, IKK/IKB/NF-kB, GRK-2 and other signaling pathways.81 PEBP1 is distributed in the hippocampus as a precursor protein of a hippocampal cholinergic neurostimulating peptide (HCNP).82 The up-regulation of PEBP1 may cause the increase in HCNP, then increase the content of choline acetyltransferase (ChAT) in the medial septal nucleus, induce the synthesis of acetylcholine and cause depression-like changes.83 Further study found that the up-regulation of PEBP1 can inhibit the expression of ERK and promote the occurrence of depression.84 In addition, PEBP1 can increase the production of hydroperoxy-PE by forming a complex with 15LO1 and 15LO2, which will cause the synthesis of GPX4 to be blocked and lead to ferroptosis.85 In our study, the expression of PEBP1 was upregulated in the hippocampus of model mice. It suggested that PEBP1 might be a potential material basis for the connection between the pathogenesis of depression and the signaling of ferroptosis.
The iron in brain tissue mainly comes from the serum iron transported in the cerebral microvasculature, and the Fe3+ entering brain tissue can be absorbed and utilized by neurons and gliocyte.86 Iron in neurons and gliocyte should be maintained at a certain level, and excessive accumulation of iron will lead to a rise in the labile iron pool (LIP) and an increase of ROS,87 which will cause neuroinflammation and neuronal damage through a series of signal transduction pathways, and participate in the development and deterioration of nervous system diseases.88 Similar to previous results,89,90 the expression of GFAP was down-regulated and IBA1 was up-regulated in the hippocampus of mice with stress-induced depression, which confirmed that neuroinflammation and gliocyte damage were indeed involved in the mechanism of depression, which may be related to the inflammatory response caused by ROS release during ferroptosis.
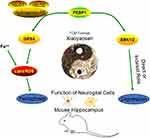
In this experiment, treatment with Xiaoyaosan and fluoxetine not only ameliorated the depressive behavior of CUMS model mice but also regulated related ferroptosis signaling, and significantly improved the expressions of GFAP and IBA1, which had the effect of improving the function of the hippocampal nerve. Fluoxetine, as a selective serotonin reuptake inhibitor (SSRIs), plays an antidepressant role by selectively inhibiting the reuptake of 5-HT released from the presynaptic membrane of the central nervous system. At present, it has been confirmed that fluoxetine can significantly inhibit LPS-induced microglial activation and reduce the release of inflammatory factors,91 alleviate depression by preventing oxidative stress and mitochondrial dysfunction,92,93 and play a role in nerve protection by regulating PI3K/AKT and MAPK/ERK signaling pathways.94 Therefore, the modulatory effect of fluoxetine on ferroptosis-related substances may be related to its neuroprotective and anti-inflammatory effects. This therapeutic Chinese herbal compound has multi-component and multi-target principles and characteristics, and there is a self-assembly phenomenon among the effective components in the preparation process that has a unique compatibility and synergistic effect.95 Xiaoyaosan, as a Chinese herbal compound formula, has a good therapeutic effect on depression. From the experimental results, we could see that its antidepressant mechanism might be achieved by regulating the expression of PEBP1, affecting the expression of ERK1/2 and its phosphorylation level, intervening in the process of ferroptosis involved in GPX4, and improving the central gliocyte function (Figure 10).
Conclusion
In conclusion, the expressions of ferroptosis-related substances were changed in the hippocampi of CUMS depressed model mice, and PEBP1 might be a bridge between the pathogenesis of depression and ferroptosis signaling. Moreover, this study further expanded the antidepressant mechanism of Xiaoyaosan, and found that Xiaoyaosan could play a therapeutic role by improving the expression of GPX4 and other ferroptosis-related substances in the hippocampi of depressed mice. However, this experiment only detected the activation of ferroptosis signaling in depression, preliminarily verified the possibility of PEBP1 as the common material basis of depression and ferroptosis, and observed the possible regulatory effect of Xiaoyaosan on PEBP1-GPX4-mediated ferroptosis. The specific mechanism of ferroptosis in the pathogenesis of depression and how Xiaoyaosan regulates ferroptosis and exerts an antidepressant effect need further investigation.
Acknowledgments
This research was supported by grants from the National Natural Science Foundation of China (No. 81630104, 81973748) and the Huang Zhendong Research Fund for Traditional Chinese Medicine of Jinan University (No. 201911).
Disclosure
The authors declare no conflicts of interest in connection with this work.
References
1. Smith K. Mental health: a world of depression. Nature. 2014;515(7526):181. doi:10.1038/515180a
2. Vos T, Allen C, Arora M, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet. 2016;388(10053):1545–1602.
3. Barnett R. Depression. Lancet. 2019;393(10186):2113. doi:10.1016/S0140-6736(19)31151-1
4. Wu J, Minikes AM, Gao M, et al. Intercellular interaction dictates cancer cell ferroptosis via NF2-YAP signalling. Nature. 2019;572(7769):402–406. doi:10.1038/s41586-019-1426-6
5. Dixon SJ, Lemberg KM, Lamprecht MR, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149(5):1060–1072. doi:10.1016/j.cell.2012.03.042
6. Yu H, Guo P, Xie X, Wang Y, Chen G. Ferroptosis, a new form of cell death, and its relationships with tumourous diseases. J Cell Mol Med. 2017;21(4):648–657. doi:10.1111/jcmm.13008
7. Wang LF, Yokoyama KK, Chen TY, et al. Male-specific alleviation of iron-induced striatal injury by inhibition of autophagy. PLoS One. 2015;10(7):e0131224. doi:10.1371/journal.pone.0131224
8. Do Van B, Gouel F, Jonneaux A, et al. Ferroptosis, a newly characterized form of cell death in Parkinson’s disease that is regulated by PKC. Neurobiol Dis. 2016;94:169–178. doi:10.1016/j.nbd.2016.05.011
9. Galluzzi L, Vitale I, Aaronson SA, et al. Molecular mechanisms of cell death: recommendations of the nomenclature committee on cell death 2018. Cell Death Differ. 2018;25(3):486–541.
10. Chen L, Hambright WS, Na R, Ran Q. Ablation of the ferroptosis inhibitor glutathione peroxidase 4 in neurons results in rapid motor neuron degeneration and paralysis. J Biol Chem. 2015;290(47):28097–28106. doi:10.1074/jbc.M115.680090
11. Bernier I, Jollès P. Purification and characterization of a basic 23 kDa cytosolic protein from bovine brain. Biochim Biophys Acta. 1984;790(2):174–181. doi:10.1016/0167-4838(84)90221-8
12. Katada E, Mitake S, Matsukawa N, et al. Distribution of hippocampal cholinergic neurostimulating peptide (HCNP)-like immunoreactivity in organs and tissues of young Wistar rats. Histochem Cell Biol. 1996;105(1):43–51. doi:10.1007/BF01450877
13. Hickox DM, Gibbs G, Morrison JR, et al. Identification of a novel testis-specific member of the phosphatidylethanolamine binding protein family, pebp-2. Biol Reprod. 2002;67(3):917–927. doi:10.1095/biolreprod.101.001446
14. Wang X, Li N, Liu B, et al. A novel human phosphatidylethanolamine-binding protein resists tumor necrosis factor alpha-induced apoptosis by inhibiting mitogen-activated protein kinase pathway activation and phosphatidylethanolamine externalization. J Biol Chem. 2004;279(44):45855–45864. doi:10.1074/jbc.M405147200
15. Rautureau G, Jouvensal L, Vovelle F, Schoentgen F, Locker D, Decoville M. Expression and characterization of the PEBP homolog genes from Drosophila. Arch Insect Biochem Physiol. 2009;71(2):55–69. doi:10.1002/arch.20300
16. Yeung K, Seitz T, Li S, et al. Suppression of Raf-1 kinase activity and MAP kinase signalling by RKIP. Nature. 1999;401(6749):173–177. doi:10.1038/43686
17. Wang JQ, Mao L. The ERK Pathway: molecular Mechanisms and Treatment of Depression. Mol Neurobiol. 2019;56:6197–6205. doi:10.1007/s12035-019-1524-3
18. Dwivedi Y, Rizavi HS, Roberts RC, Conley RC, Tamminga CA, Pandey GN. Reduced activation and expression of ERK1/2 MAP kinase in the post-mortem brain of depressed suicide subjects. J Neurochem. 2001;77(3):916–928. doi:10.1046/j.1471-4159.2001.00300.x
19. Sun L, Zhao J, Wang H, Pan Y, Wang L, Zhang WB. Mechanical stress promotes matrix synthesis of mandibular condylar cartilage via the RKIP-ERK pathway. J Mol Histol. 2017;48(5–6):437–446. doi:10.1007/s10735-017-9741-4
20. Gallimore AR, Kim T, Tanaka-Yamamoto K, De Schutter E. Switching on depression and potentiation in the cerebellum. Cell Rep. 2018;22(3):722–733. doi:10.1016/j.celrep.2017.12.084
21. Hambright WS, Fonseca RS, Chen L, Na R, Ran Q. Ablation of ferroptosis regulator glutathione peroxidase 4 in forebrain neurons promotes cognitive impairment and neurodegeneration. Redox Biol. 2017;12:8–17. doi:10.1016/j.redox.2017.01.021
22. Stoyanovsky DA, Tyurina YY, Shrivastava I, et al. Iron catalysis of lipid peroxidation in ferroptosis: regulated enzymatic or random free radical reaction? Free Radic Biol Med. 2019;133:153–161.
23. Tian JS, Peng GJ, Wu YF, et al. A GC-MS urinary quantitative metabolomics analysis in depressed patients treated with TCM formula of Xiaoyaosan. J Chromatogr B Analyt Technol Biomed Life Sci. 2016;1026:227–235. doi:10.1016/j.jchromb.2015.12.026
24. Peng GJ, Tian JS, Gao XX, Zhou YZ, Qin XM. Research on the Pathological Mechanism and Drug Treatment Mechanism of Depression. Curr Neuropharmacol. 2015;13(4):514–523. doi:10.2174/1570159X1304150831120428
25. Xu HY, Zhang YQ, Liu ZM, et al. ETCM: an encyclopaedia of traditional Chinese medicine. Nucleic Acids Res. 2019;47(D1):D976–d982. doi:10.1093/nar/gky987
26. Ding XF, Li YH, Chen JX, et al. Involvement of the glutamate/glutamine cycle and glutamate transporter GLT-1 in antidepressant-like effects of Xiao Yao san on chronically stressed mice. BMC Complement Altern Med. 2017;17(1):326. doi:10.1186/s12906-017-1830-0
27. Liu X, Zheng X, Du G, Li Z, Qin X. Brain metabonomics study of the antidepressant-like effect of Xiaoyaosan on the CUMS-depression rats by (1) H NMR analysis. J Ethnopharmacol. 2019;235:141–154. doi:10.1016/j.jep.2019.01.018
28. Gao X, Liang M, Fang Y, et al. Deciphering the differential effective and toxic responses of bupleuri radix following the induction of chronic unpredictable mild stress and in healthy rats based on serum metabolic profiles. Front Pharmacol. 2017;8:995. doi:10.3389/fphar.2017.00995
29. Banasr M, Dwyer JM, Duman RS. Cell atrophy and loss in depression: reversal by antidepressant treatment. Curr Opin Cell Biol. 2011;23(6):730–737. doi:10.1016/j.ceb.2011.09.002
30. Liu Y, Ding XF, Wang XX, et al. Xiaoyaosan exerts antidepressant-like effects by regulating the functions of astrocytes and EAATs in the prefrontal cortex of mice. BMC Complement Altern Med. 2019;19(1):215. doi:10.1186/s12906-019-2613-6
31. Li XJ, Ma QY, Jiang YM, et al. Xiaoyaosan exerts anxiolytic-like effects by down-regulating the TNF-α/JAK2-STAT3 pathway in the rat hippocampus. Sci Rep. 2017;7(1):353. doi:10.1038/s41598-017-00496-y
32. Zhu X, Xia O, Han W, et al. Xiao yao san improves depressive-like behavior in rats through modulation of β-Arrestin 2-Mediated pathways in hippocampus. Evidence-Based Complementary Altern Med. 2014;2014:902516. doi:10.1155/2014/902516
33. Chen JX, Li W, Zhao X, Yang JX. Effects of the Chinese traditional prescription Xiaoyaosan decoction on chronic immobilization stress-induced changes in behavior and brain BDNF, TrkB, and NT-3 in rats. Cell Mol Neurobiol. 2008;28(5):745–755. doi:10.1007/s10571-007-9169-6
34. Zhu HZ, Liang YD, Ma QY, et al. Xiaoyaosan improves depressive-like behavior in rats with chronic immobilization stress through modulation of the gut microbiota. Biomed Pharmacother. 2019;112:108621. doi:10.1016/j.biopha.2019.108621
35. Vugmeyster Y, Harrold J, Xu X. Absorption, Distribution, Metabolism, and Excretion (ADME) studies of biotherapeutics for autoimmune and inflammatory conditions. AAPS Journal. 2012;14(4):714–727. doi:10.1208/s12248-012-9385-y
36. Ru J, Li P, Wang J, et al. TCMSP: a database of systems pharmacology for drug discovery from herbal medicines. J Cheminform. 2014;6:13. doi:10.1186/1758-2946-6-13
37. Chen CY. TCM Database@Taiwan: the world’s largest traditional Chinese medicine database for drug screening in silico. PLoS One. 2011;6(1):e15939. doi:10.1371/journal.pone.0015939
38. Liu Z, Guo F, Wang Y, et al. BATMAN-TCM: a bioinformatics analysis tool for molecular mechanism of traditional Chinese medicine. Sci Rep. 2016;6:21146. doi:10.1038/srep21146
39. Daina A, Michielin O, Zoete V. SwissTargetPrediction: updated data and new features for efficient prediction of protein targets of small molecules. Nucleic Acids Res. 2019;47(W1):W357–w364. doi:10.1093/nar/gkz382
40. Stelzer G, Rosen N, Plaschkes I, et al. The genecards suite: from gene data mining to disease genome sequence analyses. Current Protoc Bioinf. 2016;54(1):
41. Amberger JS, Bocchini CA, Schiettecatte F, Scott AF, Hamosh A. OMIM.org: online Mendelian Inheritance in Man (OMIM®), an online catalog of human genes and genetic disorders. Nucleic Acids Res. 2015;43(Database issue):D789–D798. doi:10.1093/nar/gku1205
42. Wang Y, Zhang S, Li F, et al. Therapeutic target database 2020: enriched resource for facilitating research and early development of targeted therapeutics. Nucleic Acids Res. 2020;48(D1):D1031–d1041. doi:10.1093/nar/gkz981
43. Davis AP, Grondin CJ, Johnson RJ, et al. Comparative Toxicogenomics Database (CTD): update 2021. Nucleic Acids Res. 2020.
44. Zhou N, Bao J. FerrDb: a manually curated resource for regulators and markers of ferroptosis and ferroptosis-disease associations. Database J Biol Databases Curation. 2020;2020. doi:10.1093/database/baaa021
45. UniProt Consortium. UniProt: a worldwide hub of protein knowledge. Nucleic Acids Res. 2019;47(D1):D506–d515. doi:10.1093/nar/gky1049
46. Yan ZY, Li XJ, Ding XF, Liu YY, Chen JX. Evaluating the anti-depression effect of xiaoyaosan on chronically-stressed mice. JoVE. 2019;143. doi:10.3791/58276
47. Liu MY, Yin CY, Zhu LJ, et al. Sucrose preference test for measurement of stress-induced anhedonia in mice. Nat Protoc. 2018;13(7):1686–1698. doi:10.1038/s41596-018-0011-z
48. Morgan JA, Singhal G, Corrigan F, et al. Ceasing exercise induces depression-like, anxiety-like, and impaired cognitive-like behaviours and altered hippocampal gene expression. Brain Res Bull. 2019;148:118–130. doi:10.1016/j.brainresbull.2019.02.014
49. Lian N, Niu Q, Lei Y, Li X, Li Y, Song X. MiR-221 is involved in depression by regulating Wnt2/CREB/BDNF axis in hippocampal neurons. Cell Cycle. 2018;17(24):2745–2755. doi:10.1080/15384101.2018.1556060
50. Alkam T, Kim HC, Hiramatsu M, et al. Evaluation of emotional behaviors in young offspring of C57BL/6J mice after gestational and/or perinatal exposure to nicotine in six different time-windows. Behav Brain Res. 2013;239:80–89. doi:10.1016/j.bbr.2012.10.058
51. Xiao B, Sanders MJ, Carmena D, et al. Structural basis of AMPK regulation by small molecule activators. Nat Commun. 2013;4:3017. doi:10.1038/ncomms4017
52. Hammen C. Stress and depression. Annu Rev Clin Psychol. 2005;1:293–319. doi:10.1146/annurev.clinpsy.1.102803.143938
53. Saito H, Ichikawa K, Nomiyama T, et al. Changes in activities of daily living during treatment of late-life depression. Psychogeriatrics. 2008;8:12–18. doi:10.1111/j.1479-8301.2007.00216.x
54. Huang G-B, Huang GB, Zhao T, et al. Effects of chronic social defeat stress on behaviour, endoplasmic reticulum proteins and choline acetyltransferase in adolescent mice. Int J Neuropsychopharmacol. 2013. doi:10.1017/S1461145713000060
55. Jin HM, Muna SS, Bagalkot TR, Cui Y, Chung YC. The effects of social defeat on behavior and dopaminergic markers in mice. Neuroscience. 2015;288:167–177. doi:10.1016/j.neuroscience.2014.12.043
56. Yoshino K, Oda Y, Kimura M, et al. The alterations of glutamate transporter 1 and glutamine synthetase in the rat brain of a learned helplessness model of depression. Psychopharmacology. 2020;237(8):2547–2553. doi:10.1007/s00213-020-05555-3
57. Ribeiro DE, Casarotto PC, Staquini L, Silva MAPE, Joca S. Reduced P2X receptor levels are associated with antidepressant effect in the learned helplessness model. PeerJ. 2019;7(3):e7834. doi:10.7717/peerj.7834
58. Willner P. The chronic mild stress (CMS) model of depression: history, evaluation and usage. Neurobiol Stress. 2017;6:78–93. doi:10.1016/j.ynstr.2016.08.002
59. Calabrese F, Brivio P, Sbrini G, et al. Effect of lurasidone treatment on chronic mild stress-induced behavioural deficits in male rats: the potential role for glucocorticoid receptor signalling. J Psychopharmacol. 2020;34(4):420–428. doi:10.1177/0269881119895547
60. Dai Y, Li Z, Xue L, et al. Metabolomics study on the anti-depression effect of xiaoyaosan on rat model of chronic unpredictable mild stress. J Ethnopharmacol. 2010;128(2):482–489. doi:10.1016/j.jep.2010.01.016
61. Yan HC, Cao X, Das M, Zhu XH, Gao TM. Behavioral animal models of depression. Neurosci Bull. 2010;26(4):327–337. doi:10.1007/s12264-010-0323-7
62. Papp M, Willner P, Muscat R. An animal model of anhedonia: attenuation of sucrose consumption and place preference conditioning by chronic unpredictable mild stress. Psychopharmacology. 1991;104(2):255–259. doi:10.1007/BF02244188
63. Willner P, Towell A, Sampson D, Sophokleous S, Muscat R. Reduction of sucrose preference by chronic unpredictable mild stress, and its restoration by a tricyclic antidepressant. Psychopharmacology. 1987;93(3):358–364. doi:10.1007/BF00187257
64. Stunkard AJ, Fernstrom MH, Price RA, Buss E, Frank E, Kupfer DJ. Weight change in depression: influence of “disinhibition” is mediated by body mass and other variables. Psychiatry Res. 1991;38(2):197–200. doi:10.1016/0165-1781(91)90044-P
65. Pothion S, Bizot JC, Trovero F, Belzung C. Strain differences in sucrose preference and in the consequences of unpredictable chronic mild stress. Behav Brain Res. 2004;155(1):135–146. doi:10.1016/j.bbr.2004.04.008
66. Vucković MG, Wood RI, Holschneider DP, et al. Memory, mood, dopamine, and serotonin in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-lesioned mouse model of basal ganglia injury. Neurobiol Dis. 2008;32(2):319–327. doi:10.1016/j.nbd.2008.07.015
67. Francis-Oliveira J, Ponte B, Barbosa AP, et al. Fluoxetine exposure during pregnancy and lactation: effects on acute stress response and behavior in the novelty-suppressed feeding are age and gender-dependent in rats. Behav Brain Res. 2013;252:195–203. doi:10.1016/j.bbr.2013.05.064
68. Zhao X, Cao F, Liu Q, et al. Behavioral, inflammatory and neurochemical disturbances in LPS and UCMS-induced mouse models of depression. Behav Brain Res. 2019;364:494–502. doi:10.1016/j.bbr.2017.05.064
69. Gao XX, Cui J, Zheng XY, et al. An investigation of the antidepressant action of xiaoyaosan in rats using ultra performance liquid chromatography-mass spectrometry combined with metabonomics. PTR. 2013;27(7):1074–1085. doi:10.1002/ptr.4805
70. Gao M, Monian P, Pan Q, Zhang W, Xiang J, Jiang X. Ferroptosis is an autophagic cell death process. Cell Res. 2016;26(9):1021–1032. doi:10.1038/cr.2016.95
71. Cheng Z, Li Y. What is responsible for the initiating chemistry of iron-mediated lipid peroxidation: an update. Chem Rev. 2007;107(3):748–766. doi:10.1021/cr040077w
72. Lachaier E, Louandre C, Godin C, et al. Sorafenib induces ferroptosis in human cancer cell lines originating from different solid tumors. Anticancer Res. 2014;34(11):6417–6422.
73. Latunde-Dada GO. Ferroptosis: role of lipid peroxidation, iron and ferritinophagy. Biochimica Et Biophysica Acta Gen Subj. 2017;1861(8):1893–1900. doi:10.1016/j.bbagen.2017.05.019
74. Yang WS, Kim KJ, Gaschler MM, Patel M, Shchepinov MS, Stockwell BR. Peroxidation of polyunsaturated fatty acids by lipoxygenases drives ferroptosis. Proc Natl Acad Sci U S A. 2016;113(34):E4966–E4975. doi:10.1073/pnas.1603244113
75. Kagan VE, Mao G, Qu F, et al. Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis. Nat Chem Biol. 2017;13(1):81–90. doi:10.1038/nchembio.2238
76. Shimada K, Skouta R, Kaplan A, et al. Global survey of cell death mechanisms reveals metabolic regulation of ferroptosis. Nat Chem Biol. 2016;12(7):497–503. doi:10.1038/nchembio.2079
77. Seiler A, Schneider M, Förster H, et al. Glutathione peroxidase 4 senses and translates oxidative stress into 12/15-lipoxygenase dependent- and AIF-mediated cell death. Cell Metab. 2008;8(3):237–248. doi:10.1016/j.cmet.2008.07.005
78. Yang WS, SriRamaratnam R, Welsch ME, et al. Regulation of ferroptotic cancer cell death by GPX4. Cell. 2014;156(1–2):317–331. doi:10.1016/j.cell.2013.12.010
79. Ratan RR. The chemical biology of ferroptosis in the central nervous system. Cell Chem Biol. 2020;27(5):479–498. doi:10.1016/j.chembiol.2020.03.007
80. Oh DR, Yoo JS, Kim Y, et al. Vaccinium bracteatum leaf extract reverses chronic restraint stress-induced depression-like behavior in mice: regulation of hypothalamic-pituitary-adrenal axis, serotonin turnover systems, and ERK/Akt phosphorylation. Front Pharmacol. 2018;9:604. doi:10.3389/fphar.2018.00604
81. Trakul N, Menard RE, Schade GR, Qian Z, Rosner MR. Raf kinase inhibitory protein regulates Raf-1 but not B-Raf kinase activation. J Biol Chem. 2005;280(26):24931. doi:10.1074/jbc.M413929200
82. Khawaja X, Xu J, Liang JJ, Barrett JE. Proteomic analysis of protein changes developing in rat hippocampus after chronic antidepressant treatment: implications for depressive disorders and future therapies. J Neurosci Res. 2004;75(4):451–460. doi:10.1002/jnr.10869
83. Andreasen J, Olsen G, Wiborg O, Redrobe J. Antidepressant-like effects of nicotinic acetylcholine receptor antagonists, but not agonists, in the mouse forced swim and mouse tail suspension tests. J Psychopharmacol. 2009;23(7):797–804. doi:10.1177/0269881108091587
84. Sun Y, Luo ZM, Zheng MJ, Liu X. Phosphatidylethanolamine-binding protein 1 (PEBP1) as a potential target for the treatment for depression. CNS Neurosci Ther. 2013;19(12):982–983. doi:10.1111/cns.12185
85. Wenzel SE, Tyurina YY, Zhao J, et al. PEBP1 wardens ferroptosis by enabling lipoxygenase generation of lipid death signals. Cell. 2017;171(3):628–641.e626. doi:10.1016/j.cell.2017.09.044
86. McCarthy RC, Kosman DJ. Iron transport across the blood-brain barrier: development, neurovascular regulation and cerebral amyloid angiopathy. CMLS. 2015;72(4):709–727.
87. Mills E, Dong XP, Wang F, Xu H. Mechanisms of brain iron transport: insight into neurodegeneration and CNS disorders. Future Med Chem. 2010;2(1):51–64. doi:10.4155/fmc.09.140
88. Sabens Liedhegner EA, Gao XH, Mieyal JJ. Mechanisms of altered redox regulation in neurodegenerative diseases–focus on S–glutathionylation. Antioxid Redox Signal. 2012;16(6):543–566. doi:10.1089/ars.2011.4119
89. Araya-Callís C, Hiemke C, Abumaria N, Flugge G. Chronic psychosocial stress and citalopram modulate the expression of the glial proteins GFAP and NDRG2 in the hippocampus. Psychopharmacology. 2012;224(1):209–222. doi:10.1007/s00213-012-2741-x
90. Yamawaki Y, Yoshioka N, Nozaki K, et al. Sodium butyrate abolishes lipopolysaccharide-induced depression-like behaviors and hippocampal microglial activation in mice. Brain Res. 2018;1680:13–38. doi:10.1016/j.brainres.2017.12.004
91. Zhang F, Zhou H, Wilson BC, Shi JS, Hong JS, Gao HM. Fluoxetine protects neurons against microglial activation-mediated neurotoxicity. Parkinsonism Relat Disord. 2012;18(Suppl 1(0 1)):S213–S217. doi:10.1016/S1353-8020(11)70066-9
92. Da silva Souza SV, da Rosa PB, Neis VB, Moreira JD, Rodrigues ALS, Moretti M. Effects of cholecalciferol on behavior and production of reactive oxygen species in female mice subjected to corticosterone-induced model of depression. Naunyn-Schmiedeberg’s arch pharmacol. 2020;393(1):111–120. doi:10.1007/s00210-019-01714-2
93. Ludka FK, Dal-Cim T, Binder LB, Constantino LC, Massari C, Tasca CI. Atorvastatin and Fluoxetine Prevent Oxidative Stress and Mitochondrial Dysfunction Evoked by Glutamate Toxicity in Hippocampal Slices. Mol Neurobiol. 2017;54(5):3149–3161. doi:10.1007/s12035-016-9882-6
94. Huang W, Zhao Y, Zhu X, et al. Fluoxetine upregulates phosphorylated-AKT and phosphorylated-ERK1/2 proteins in neural stem cells: evidence for a crosstalk between AKT and ERK1/2 pathways. J Mol Neurosci. 2013;49(2):244–249. doi:10.1007/s12031-012-9822-5
95. Wang L, Zhou GB, Liu P, et al. Dissection of mechanisms of Chinese medicinal formula Realgar-Indigo naturalis as an effective treatment for promyelocytic leukemia. Proc Natl Acad Sci U S A. 2008;105(12):4826–4831. doi:10.1073/pnas.0712365105
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.