Back to Journals » International Journal of Nanomedicine » Volume 9 » Issue 1
Tissue-specific direct microtransfer of nanomaterials into Drosophila embryos as a versatile in vivo test bed for nanomaterial toxicity assessment
Authors Vega-Alvarez S, Herrera A, Rinaldi C , Carrero-Martínez F
Received 23 October 2013
Accepted for publication 10 January 2014
Published 22 April 2014 Volume 2014:9(1) Pages 2031—2041
DOI https://doi.org/10.2147/IJN.S56459
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Sasha Vega-Alvarez,1 Adriana Herrera,2 Carlos Rinaldi,2–4 Franklin A Carrero-Martínez1,5
1Department of Biology, 2Department of Chemical Engineering, University of Puerto Rico-Mayagüez, Mayagüez, Puerto Rico; 3J Crayton Pruitt Family Department of Biomedical Engineering, 4Department of Chemical Engineering, University of Florida, Gainesville, FL, USA; 5Department of Anatomy and Neuroscience, University of Puerto Rico, Medical Sciences Campus, San Juan, Puerto Rico
Abstract: Nanomaterials are the subject of intense research, focused on their synthesis, modification, and biomedical applications. Increased nanomaterial production and their wide range of applications imply a higher risk of human and environmental exposure. Unfortunately, neither environmental effects nor toxicity of nanomaterials to organisms are fully understood. Cost-effective, rapid toxicity assays requiring minimal amounts of materials are needed to establish both their biomedical potential and environmental safety standards. Drosophila exemplifies an efficient and cost-effective model organism with a vast repertoire of in vivo tools and techniques, all with high-throughput scalability and screening feasibility throughout its life cycle. Here we report tissue specific nanomaterial assessment through direct microtransfer into target tissues. We tested several nanomaterials with potential biomedical applications such as single-wall carbon nanotubes, multiwall carbon nanotubes, silver, gold, titanium dioxide, and iron oxide nanoparticles. Assessment of nanomaterial toxicity was conducted by evaluating progression through developmental morphological milestones in Drosophila. This cost-effective assessment method is amenable to high-throughput screening.
Keywords: nanotoxicity, Drosophila, microtransfer, nanoparticle, iron oxide, silver, gold, titanium dioxide, carbon nanotube
Introduction
Nanomaterials have been the subject of intense research focused on their synthesis, modification, and applications.1–3 During the last few years, nanoparticles have become important tools, with an expectation that they will have a considerable effect in the biomedical sciences, and are attractive for applications including imaging agents, gene and drug delivery vehicles, in vivo and in vitro biosensors, and nanoscale thermal therapies.4–9 Nanoparticles can also be found in some everyday use products and in agricultural applications.10,11 Among the wide variety of nanomaterials possible, some of the most commonly used are single (SWCNTs) and multiwalled carbon nanotubes (MWCNTs), metallic silver and gold nanoparticles, oxides such as titanium dioxide and iron oxide (IOs), and semiconductor quantum dots.
It stands to reason that the increase in nanomaterial production associated with their wide range of applications implies a higher risk for human and environmental exposure.12 To date, neither environmental effects nor toxicity of nanomaterials to organisms are fully understood. Toxicity assessments can provide the necessary information to establish adverse effects a substance may have in an organism at the cellular, tissue, and organ levels. Unfortunately, cost-effective toxicity assessments have not been developed in tandem with the fast-growing field of nanomaterials synthesis. To reach a consensus and establish clear and specific regulations for human and environmental safety, one needs to follow a product-focused, science-based approach.13,14 Product-specific assessments will yield conclusions based on the specific properties of the nanomaterial (size, surface modification, concentration, and exposure route), instead of broad generalizations.
The primary advantage of in vitro assessments is their reductionist approach. Unfortunately, this limits the scope of the research to specific homogeneous cell types and may not be relevant to biological events in an otherwise more complex functional organism.15–18 Although cell cultures can include multiple cell types mimicking in vivo situations,19,20 in vitro assessments are not able to simulate the microenvironment present in a complex organism, including, but not limited to, three-dimensional gradients of molecular cues, exposure routes, dosing, and lack of physical barriers intrinsic to tissues and organs.
In vivo assessments are performed using whole organisms in which spatial organization is unaltered. The most common in vivo nanotoxicity assessments use rodents as model organisms, but other model organisms such as Caenorhabditis elegans, Danio reiro, and Drosophila are gaining popularity.21–26 Rodents, being mammals, can be used to study complicated processes underlying normal human development, diseases, and behavior. Using rats or mice as model organisms allows scientists to mimic possible exposure routes that occur in humans, such as inhalation,27 dermal exposure,28 and injections.29 In addition, organ biodistribution and dose equivalencies such as minimum lethal dose and median lethal dose can be directly extrapolated.29 Rats have recently lost favor as animal models in some fields. One reason is that genetic manipulations are limited in this model organism because its genome does not tolerate the insertion of foreign DNA to the extent of other organisms like the mouse, C. elegans, or Drosophila.30
C. elegans and Drosophila possess many of the same advantages but differ mostly on the degree of tractability and accessibility that can be used in experimental manipulation. Drosophila presents the possibility of assessing the six principal exposure routes: intravenous, dermal, subcutaneous, inhalation, intraperitoneal, and oral.31 However, these different exposure routes have not been fully assessed, and oral ingestion is the most widely employed exposure route in nanotoxicity research using Drosophila.26,32–34
Drosophila exemplifies an efficient and cost-effective model organism with a vast repertoire of tools and techniques, all with high-throughput scalability and screening feasibility,35–37 throughout its life cycle.26 A female can lay as many as 3,000 eggs in her lifetime,38 providing a constant supply of individuals in every stage of development. Furthermore, as a result of Drosophila’s small size, the amount of nanomaterial required for in vivo testing is in the nanogram range.26,32 This is orders of magnitude smaller than what is required for testing in other model systems, such as C. elegans21,22 and zebrafish.23,24 Drosophila’s single-cell resolution, together with its neuromuscular system consisting of a series of segmental repeats in a well-known pattern,39 allows for accessible, simple, and precise identification of developmental stages from morphological and molecular perspectives. In addition, single identifiable cells can be tracked throughout the entire embryonic development, thanks to the existence of a clear cuticle during the embryonic and larval stages. This in turn allows the study of developmental effects of nanomaterials in a specific area or system of interest.40
Methods
Drosophila embryos
Canton S wild-type Drosophila melanogaster embryos kept at 25°C and 60% relative humidity were dechorionated using a 50% hypochlorite wash solution followed by staging according to Campos-Ortega and Hartenstein.41 Briefly, stage 15 embryos were selected and placed with their dorsoventral axis parallel to the 0.5 cm2 coverslip in which they were fixed and then covered with a drop of halocarbon oil series 700 (Halocarbon Products Corp, River Edge, NJ, USA). These steps were performed using an Olympus MVX10 MacroView (Center Valley, PA, USA).
Nanomaterials
Silver, titanium dioxide (TiO2), and gold nanoparticles and carbon nanotubes were obtained commercially. The silver (Ag) nanoparticles (MKnano, Mississauga, ON, Canada) had a diameter smaller than 90 nm and purity of 99.9%. Gold (Au) nanoparticles (Sigma-Aldrich, St Louis, MO, USA) had a diameter smaller than 150 nm and purity of 99.9%. The TiO2 nanoparticles (Degussa P25; Evonik Industries, Piscataway Township, NJ, USA) had a diameter smaller than 20 nm and purity of 99.9%. The SWCNT (Cheap Tubes, Inc., Brattleboro, VT, USA) had an outer diameter of 1 to 2 nm, an inner diameter of 0.8 to 1.6 nm, length of 5 to 30 μm, a surface area (SA) of 407 m2/g, and purity higher than 90 wt%. The MWCNT (Cheap Tubes, Inc.) had an outer diameter smaller than 8 nm, an inner diameter of 2 to 5 nm, length of 10 to 30 μm, an SA of 500 m2/g, and purity higher than 95 wt%.
IO nanoparticles were synthesized by the coprecipitation42 or thermal decomposition method43 and coated with carboxymethyl dextran (CMDx), using an amine silane as a grafting agent.44,45 The methods for obtaining these nanoparticles have been published by our group, along with detailed characterization of their colloidal properties.44–46 We refer to CMDx-coated IO obtained by coprecipitation as Cop-IO. The primary particle diameter determined by transmission electron microscopy (JEOL 1200 EX; Jeol Ltd, Tokyo, Japan) was 12±2 nm, and the hydrodynamic diameter, determined by dynamic light scattering (BI-90Plus; Brookhaven Instruments, Holtsville, NY, USA), was 77±5 nm. We have previously shown that particles obtained by these methods consist of small aggregates of primary nanoparticles coated with a CMDx shell.45 We refer to CMDx-coated IO obtained by thermal decomposition as thermo-IO. The primary particle diameter, determined by transmission electron microscopy, was 12±1 nm, and the hydrodynamic diameter, determined by dynamic light scattering, was 38±5 nm. We have previously shown that particles obtained by these methods consist of single IO primary particles coated with a CMDx shell.44
Tissue-specific nanomaterial microtransfer in Drosophila
We used the Sutter’s Xenoworks micromanipulator and digital microinjector System (Sutter Instruments, Novato, CA, USA) in conjunction with TransferTip-R microcapillary needles (Eppendorf, Hamburg, Germany) mounted on a fully motorized Olympus IX81 inverted microscope (Olympus) to deliver nanomaterials. We used a modified pulsed-flow approach with regulated injection pressures, allowing for greater control and consistency of delivered sample. An essential aspect of our approach is the application of the smallest amount of pressure possible once inside the living embryo to ensure delivery of nanomaterials with minimal disruption of cell membranes. We refer to this approach as microtransferring of nanomaterials, and it results in the presentation of selected nanomaterials at cell/tissue interfaces. Taking advantage of the multisegmented body plan in Drosophila, we consistently delivered nanomaterials in the abdominal segments 5/6 intersegmental boundary by following an anterior trajectory after entry through the posterior embryonic axis.
As bare nanomaterials, such as Ag, Au, and TiO2 nanoparticles, and single-walled SWCNT and MWCNT have tendencies to sediment in water, and the colloidal stability of TiO2 nanoparticles has been shown to improve when suspended in a solution of 10% fetal bovine serum or 1% human serum albumin,47 we decided to suspend our nanomaterials in 10% bovine serum albumin solution (BSA 10%). This resulted in nanomaterial suspensions with minimized tendencies to clog fine needle tips. Our control groups consisted of embryos subject to ultrapure H2O and BSA 10% microtransfers. IO nanoparticles were suspended in ultrapure H2O; all other nanomaterials were suspended in BSA 10%. After microtransfer procedures, each embryo was allowed to recover at 25°C and 60% relative humidity (RH). Mortality assessments were recorded 48 hours after the procedure. Mortality determinations took into consideration any embryonic development past stage 15 (Figure 1).
Microtransferred volume quantification
To determine the amount of nanoparticles delivered using the microtransferring technique, we measured volume displacements once we reached the desired delivery location inside the developing embryo. We performed these measurements using our high spatiotemporal resolution imaging and manipulation system and estimated that our system is capable of consistently delivering an average volume of 0.00145 μL per five pulsed microtransfers, and later estimated delivery concentrations and total nanoparticles per embryos. To estimate the volume of the microtransfer, an initial marking was drawn around the outer surface of the needle, and the nanoparticle solution was loaded as close as possible to this marking without surpassing it. Without disconnecting the microneedle from the micropipette holder or pressure tubing of the microinjector, the micropipette holder was placed in a horizontal position over the objective, and an image was acquired before microtransfer (xo) and after every five microtransfers (xn). Using the Volocity 6.3 image acquisition software (PerkinElmer, Waltham, MA, USA), the lengths from the meniscus to the tape were determined. The microtransferred volume was determined using the cylinder volume formula (V=πr2 h), where r is the internal radius of the microneedle and h is the measured length determined from the acquired images. The equation can be written as V=πr2(x1−x2)/n. The concentrations of the microtransfer solutions for each nanoparticle are summarized in Table 1.
We established extrapolation of delivered doses based on body SA from Drosophila embryos to humans. Body SA comparison for dose extrapolation is the method suggested by the US Food and Drug Administration for clinical trials.48 Body SA of a Drosophila embryo was calculated through a simple formula based on a prolate spheroid, a body equivalent ellipsoid by Reading and Freeman49: SA =4πac, c = H/2, H = height =500 μm, a = minor axis =75 um, SA =2.36×10−7 m2. Human SA was calculated by averaging the values obtained from five of the main equations for body SA of a human,50–53 resulting in SA =1.74 m2. A conversion factor between embryo and human SA was calculated (human SA/embryo SA). The amount of nanomaterials per human dosage was calculated by applying the conversion factor to the amount of nanomaterials per embryo dosage. The equivalent microtransferred volume in a human was also established by applying the conversion factor to microinjected volume in an embryo. Dosage (μg/m2) = amount of nanomaterials per microtransfer/SA. Using the conversion factor, the equivalent microtransferred volume was calculated as 10.7 mL, which represents only 0.018% of the volume of an average human (Table 1).
Statistical analysis
Note that n=50 for all conditions tested. The Shapiro–Wilk test showed that our data did not exhibit a normal distribution. Therefore, and to establish the significance of our data set, we performed a nonparametric analysis, using Fisher’s exact test (α=0.05).
Results
Tissue-specific nanotoxicity assessment
Tissue-specific nanomaterial assessment was conducted through direct microtransfer of nanomaterials into target tissues, which yields quantifiable mortality results based on simple developmental morphological milestones in Drosophila. This assessment takes full advantage of the single identifiable cell nature of the Drosophila system, and instead of employing the commonly used microinjection techniques,54 microtransferring resulted in a more gentle and constant release of nanomaterials to the desired location, with no disruption of target tissues. Thus, potential damage to cells caused by accelerated, high-pressure pulsed injections was minimized by direct microtransfer of small amounts of nanomaterials.
We used stage 15 embryos (Figure 2), which have roughly completed 50% of their development, because eggshell membranes are fully developed and dorsal closure is completed.38 These morphological features serve as recognizable landmarks for stage identification and provide important structural integrity.
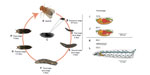  | Figure 2 Drosophila life cycle. |
Developmental effects were assessed 48 hours after microtransfer in terms of overall mortality (OM) and identification of specific developmental stages, in which each embryo was found dead. After multiple preliminary trials, the following trends were chosen as scoring criteria for the quantification of mortality at specific stages of development: number of dead embryos that did not progress past developmental stage 15 (we surmise these embryos died as a result of the delivery procedure), number of dead embryos at late embryogenesis (developmental stages 16 and 17), and number of dead larva (Figure 3). The data obtained through this quantification were analyzed two different ways: by overall mortality, which is the sum of all the scoring criteria, and by scoring criteria with highest mortality. For comparison purposes of the latter, we analyzed the shift in scoring criteria with highest mortality from one concentration to another, as this comparison yields suggestions on stability of the nanomaterial and treatment acuteness.
We tested eight nanomaterials at different concentrations: SWCNTs, MWCNTs, Ag, Au, and TiO2, and IO nanoparticles synthesized by coprecipitation coated with 3-Aminopropyltriethoxysilane (APS) and carboxymethyldextran (Cop-IO) and synthesized by thermo-decomposition coated with CMDx (Thermo-IO).
We employed predicted environmental concentrations (PEC) in water calculated by Muellerand and Nowack12 for TiO2, Ag and SWCNT/MWCNT as our lowest concentrations. PEC values were originally determined by a substance flow analysis from the products to the environment.12 There are no data available on the PEC for Au,55 and in addition, the PEC for iron has not been calculated, as it is such an abundant element in the environment. Therefore, we decided to conduct our trials with the lowest employed concentrations at orders of magnitude similar to those established for TiO2, Ag, and SWCNT/MWCNT.
Effects of PECs
Of the nanomaterials tested at the PEC, only MWCNT treatment showed statistically relevant effects in Drosophila embryo viability compared with the respective control. This suggests that a possible threshold of minimal toxic dose could be established by determining the maximum allowable concentration to be permitted in the environment (Figure 4). None of the IO nanoparticles had statistically relevant effects in Drosophila embryo viability when treated at the lowest concentration, suggesting that if the environmental concentration were to be of a similar order of magnitude as that used for the other nanomaterials, there would not be a statistically relevant mortality effect (Figure 4).
The two highest microtransferred amounts of Cop-IO nanoparticles, 1.5 and 2.8 ng, had statistically relevant effects in Drosophila embryo viability with normalized overall mortality percentages of 22% and 42%, respectively (Figure 1). In the case of Cop-IO microtransfer, the shift in scoring criteria with highest mortality from late embryogenesis to immediately after microtransfer occurs from the third to the fourth amount (1.0–1.5 ng). This suggests that the biocompatibility and stabilizing properties of CMDx are having a favorable effect in shifting the toxic effect to higher concentrations.
As with Cop-IO nanoparticles, Thermo-IO treatment presents statistically relevant effects in Drosophila embryo viability only at the second highest microtransferred amounts (2.8 and 5.0 ng), with normalized overall mortality of 38% and 46%, respectively (Figure 1). Even though the two highest concentrations of Thermo-IO-CMDx present higher overall mortality than the two highest concentrations of Cop-IO-APS-CMDx, the shift in highest mortality from late embryogenesis to immediately after microtransfer occurs from the fourth to the fifth microtransferred amount (2.8–5.0 ng). This suggests that nanoparticles synthesized by thermo-decomposition lead to slightly higher overall mortality, but nanoparticles synthesized by coprecipitation present a more acute effect, as the individuals die faster at lower concentrations.
For Ag nanoparticles, all concentrations higher than the PEC (ie, 4.30E−04, 0.04, and 4.3 ng) present statistically relevant effects in Drosophila embryo viability, with normalized overall mortality of 32%, 36%, and 38%, respectively (Figure 1). Furthermore, treatment with Ag nanoparticles shows a shift in scoring criteria, with highest mortality from late embryogenesis to immediately after microtransfer, from the first (PEC) to the second amount (4.30E−08 to 4.30E−04 ng). This suggests that treatment with Ag nanoparticles elicits an acute toxic effect and that Ag nanoparticles have a low effective dose.
For Au nanoparticles, the two highest microtransferred amounts (ie, 0.10 and 10 ng) had statistically relevant effects in Drosophila embryo viability, with normalized overall mortality of 26% and 34%, respectively (Figure 1). Also, treatment with Au nanoparticles shows a shift in scoring criteria, with highest mortality from late embryogenesis to L1 and then to immediately after microtransfer.
For TiO2 nanoparticles, the three highest microtransferred amounts (ie, 0.10, 5.1, and 10 ng) had statistically relevant effects in Drosophila embryo viability, with normalized overall mortality of 22%, 28%, and 32%, respectively (Figure 1). As with treatment with Au nanoparticles, TiO2 nanoparticles show a shift in scoring criteria, with highest mortality from late embryogenesis to L1, and then to immediately after microtransfer. Therefore, as TiO2 and Au nanoparticles were administered at the same concentrations, TiO2 elicits a more acute toxic effect and has a lower effective dose than Au nanoparticles. This is most likely caused by the oxidative stress induced by reactive oxygen species produced by TiO2.56–58
For SWCNTs, the three highest microtransferred amounts (ie, 7.2E−04, 0.07, and 7.2 ng) had statistically relevant effects in Drosophila embryo viability, with normalized overall mortality of 28%, 28%, and 30%, respectively (Figure 1). Furthermore, treatment with SWCNT shows a shift in scoring criteria with highest mortality from late embryogenesis to L1, from the fourth to the fifth microtransferred amount. These results suggest that SWCNTs affect Drosophila embryos similar to Au and TiO2, where embryo mortality is delayed by a shift in scoring criteria with highest mortality from late embryogenesis to L1, and then it shifts back. In contrast, MWCNTs had statistically relevant effects in Drosophila embryo viability only at the lowest (PEC) and the highest microtransferred amounts (7.2E–10 and 7.2 ng), with normalized overall mortality of 22% and 24%, respectively. Contrary to the rest of the nanomaterials, treatment with MWCNTs does not show a clear shift in scoring criteria with higher mortality. MWCNTs only show a slight shift from late embryogenesis to immediately after microtransfer, at the second microtransferred amount, but at the third amount, the shift reverts back to late embryogenesis. Overall mortality results of SWCNT and MWCNT are consistent with what other researchers have found,59–63 and SWCNTs showed higher toxicity than MWCNTs.
The results for MWCNT are puzzling because they show statistically relevant mortality only at the lowest and highest doses. CNTs have a tendency to form agglomerates,64 and there is ongoing debate about whether or not the degree of agglomeration affects CNT toxicity.17,64 With the current methodology, the specific toxic effects cannot be identified, but they can be deduced. Toxicity could be a result of chemical interactions between the biological environment and the nanomaterial or as a result of a physical obstruction. It is possible that as the concentration in the microtransferred solution increases, so does the size of the clusters. An increase in cluster size will diminish the possibility for dispersion, as well as the SA-to-volume ratio, of the nanomaterial. Large enough clusters can be encysted if dispersion is halted and a decrease in SA-to-volume ratio can decrease the amount of free terminals available for interactions with the biological environment. Either case can explain a decrease in mortality after an increase in concentration. Mortality can again increase once a saturation threshold has been surpassed because with an increase in concentration, both the possibilities of agglomeration and the presence of free unclustered nanotubes increase. This could explain not only the effects of CNT but also the effects of Ag, Au, and TiO2 nanoparticle treatment in which the mortality occurs earlier after a first increase in concentration and is delayed after a second increase in concentration.
Discussion
Interaction of nanoparticles with living organisms to determine toxicity effects and safety considerations must be understood. Drosophila is emerging as a suitable organism for the study of toxicity of several nanomaterials. Nanotoxicity assessment studies have been previously conducted. Most of these studies use oral ingestion routes during third instar larval32,65–68 and adult stages33,67,69–74 to assess nanotoxicologic effects of several nanomaterials. Unfortunately, and because of the relatively small amounts of food intake during these stages, it is very difficult to accurately estimate actual amounts of ingested food. In addition, it is possible that nanomaterials in Drosophila food may change its composition. Food composition plays an important part in Drosophila’s feeding behaviors,75–79 and thus, is an important factor to consider when using oral administration routes. In addition, several recent studies addressed the effect of silver nanoparticle toxicity, using oral ingestion as their administration routes, during third instar larva32,68 and adult stages.33,69–71,73,74 If we consider that nanosilver has strong antimicrobial and antifungal properties,80,81 together with the fact that Drosophila feeds mostly on microorganisms, particularly yeast, it is then possible to speculate that we may have confounding mortality effects arising from the unfavorable feeding conditions. Ingestion represents an important administration route, but more accurate screening tools are required.67 To avoid the potential pitfalls of this and other indirect methods, we chose a direct microtransfer approach. This ensures accurate exposure to the nanomaterials under consideration in specific tissues and at known concentrations in the nanogram range, thus allowing for more accurate assessment of toxicity, which is of utmost importance when determining safety exposure margins.
Our assay consists of a uniform methodology that allows for overall mortality quantification, which can be normalized against a control trial of the solution in which the nanomaterials were suspended. This assessment also includes a novel and simple methodology for volume quantification that allows for dosage extrapolation. The controls also account for the mortality caused by the mechanical damage of needle puncturing that precedes microtransfer, leading to results that are independent of human manipulation and that are, consequently, more reproducible. Because of the small amounts of nanomaterials required and the relatively short life cycle of Drosophila, we were able to use a high number of replicates (n=50) for each condition tested. This high-resolution assessment allows not only for a general evaluation of embryonic viability but also for the identification of specific stage of mortality. In turn, this can render information in terms of minimal toxic dose, acuteness of toxic effect, maximum allowable concentration in the environment, and stability of surface modification as a function of how delayed the toxic effects elicited by nanomaterials are.
The toxicity assessment of IO, Ag, Au, and TiO2 nanoparticles, SWCNTs, and MWCNTs yielded important information on their intrinsic and relative toxicity. The results on mortality at predicted environmental concentrations can help establish future safety regulations in terms of maximum allowable concentrations in the environment, particularly for MWCNTs. Methods such as those described here can be applied to systematic studies aiming to modify nanomaterial physicochemical properties to minimize their adverse effect on organisms in the environment. Furthermore, our assessment can be further developed to establish more specific molecular interactions linked to the toxicity of specific tissues or organs.
Drosophila allows us to register morphological changes throughout development, and as future work, this methodology could be adapted to other stages of development. The nanomaterials could be traced across the life cycle in the surviving embryos, especially if fluorescently tagged nanomaterials are employed. Other tools such as transgenic flies with fluorescent markers against caspase 3; lactate dehydrogenase, to identify necrotic tissue; detection of intact lysosomes, and detection of reactive oxygen species, to assess stress response, can be integrated as mortality markers. This way, more-specific conclusions could be reached and specific organ and/or system toxic effects could be assessed (ie, neurotoxicity). As a validated model for human diseases, Drosophila also presents the possibility of simultaneously assessing effects on viability and nanomaterial applications in the treatment or understanding of human diseases. Finally, Drosophila’s cost-effectiveness, requiring nanomaterial amounts in the nanogram ranges, increases the possibility of this assessment being conducted as a high-throughput assay.
Conclusion
The current rate at which new nanomaterial compositions, morphologies, and synthesis routes are developed far outpaces the rate at which their in vivo toxicity can be tested using traditional mammalian animal models. We have developed a cost-effective, tissue-specific nanomaterial toxicity assay using direct microtransfer of nanomaterials to embryos of Drosophila melanogaster. Monitoring progression through simple development morphological milestones allows for overall mortality quantification and identification of specific stages of mortality in only 48 hours. The described methods are systematic and general enough to be employed in the assessment of other nanomaterials. Because of the small amounts of nanomaterials needed per embryo, and because of the short life cycle of Drosophila, the reported method lends itself for large numbers of replicates. Furthermore, given the wide array of molecular tools available for manipulation of Drosophila and its widespread use in a variety of disease models, the direct microtransfer technique described here could also enable application of Drosophila for in vivo testing of nanomaterial efficacy in a variety of biomedical applications.
Acknowledgments
This material is based on work supported by, or in part by, the US Army Research Laboratory and the US Army Research Office under contract/grant number W911NF-09-1-0219 to FAC-M and CR, which supported AH and SV-A, as well as US National Science Foundation grant NSF-IOS-0818243, which supported SV-A. Carlos Marti, Rosa Martínez, and Geidy Acevedo-Méndez helped in embryo collection and preparation techniques. Finally, the National Institute of Health’s RISE-2-BEST Program (grant 1R25GM088023) also supported the work of SV-A.
Author contributions
FAC-M conceived and designed all Drosophila experiments. CR designed and supervised nanoparticle synthesis and characterization. SV-A performed the Drosophila experiments, including microtransfer, micromanipulation, and microscopy. AH performed synthesis and characterization of magnetic nanoparticles. All authors participated in data analysis and result discussions, and contributed to manuscript writing and critique.
Disclosure
FAC-M is currently the AAAS Roger Revelle Fellow in Global Stewardship. The other authors have no conflicts of interest to disclose in respect of this work.
References
Gupta AK, Gupta M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials. 2005;26(18):3995–4021. | |
Karousis N, Tagmatarchis N, Tasis D. Current progress on the chemical modification of carbon nanotubes. Chem Rev. 2010;110(9):5366–5397. | |
Wang Z, Ma L. Gold nanoparticle probes. Coordination Chem Rev. 2009;253(11–12):1607–1618. | |
Bonini M, Berti D, Baglioni P. Nanostructures for magnetically triggered release of drugs and biomolecules. Curr Opinion Colloid Interface Sci. 2013;18(5):459–467. | |
Cho K, Wang X, Nie S, Chen ZG, Shin DM. Therapeutic nanoparticles for drug delivery in cancer. Clin Cancer Res. 2008;14(5):1310–1316. | |
De M, Ghosh PS, Rotello VM. Applications of nanoparticles in biology. Adv Mater. 2008;20(22):4225–4241. | |
Martin CR, Kohli P. The emerging field of nanotube biotechnology. Nat Rev Drug Discov. 2003;2(1):29–37. | |
Murphy CJ, Gole AM, Stone JW, et al. Gold nanoparticles in biology: beyond toxicity to cellular imaging. Acc Chem Res. 2008;41(12):1721–1730. | |
Pankhurst QA, Thanh NTK, Jones SK, Dobson J. Progress in applications of magnetic nanoparticles in biomedicine. J Physics D: Appl Physics. 2009;42(22):224001. | |
Kalpana Sastry R, Anshul S, Rao NH. Nanotechnology in food processing sector-An assessment of emerging trends. J Food Sci Technol. 2013;50(5):831–841. | |
Khot LR, Sankaran S, Maja JM, Ehsani R, Schuster EW. Applications of nanomaterials in agricultural production and crop protection: A review. Crop Protection. 2012;35(C):64–70. | |
Mueller NC, Nowack B. Exposure modeling of engineered nanoparticles in the environment. Environ Sci Technol. 2008;42(12):4447–4453. | |
Hamburg MA. Science and regulation. FDA’s approach to regulation of products of nanotechnology. Science. 2012;336(6079):299–300. | |
Holden JP, Sunstein CR, Siddiqui IA. Policy principles for the US decision-making concerning regulation and oversight of applications of nanotechnology and nanomaterials. Memorandum for the Heads of Executive Departments and Agencies. Washington, DC: Executive Office of the President; 2011. | |
Kroll A, Pillukat MH, Hahn D, Schnekenburger J. Current in vitro methods in nanoparticle risk assessment: limitations and challenges. Eur J Pharm Biopharm. 2009;72(2):370–377. | |
Stone V, Johnston H, Schins RP. Development of in vitro systems for nanotoxicology: methodological considerations. Crit Rev Toxicol. 20039(7):613–626. | |
Lewinski N, Colvin V, Drezek R. Cytotoxicity of nanoparticles. Small. 2008;4(1):26–49. | |
Rogers EJ, Hsieh SF, Organti N, Schmidt D, Bello D. A high throughput in vitro analytical approach to screen for oxidative stress potential exerted by nanomaterials using a biologically relevant matrix: human blood serum. Toxicol In Vitro. 2008;22(6):1639–1647. | |
Rothen-Rutishauser BM, Kiama SG, Gehr P. A three-dimensional cellular model of the human respiratory tract to study the interaction with particles. Am J Respir Cell Mol Biol. 2005;32(4):281–289. | |
Jepson MA, Clark MA. Studying M cells and their role in infection. Trends Microbiol. 1998;6(9):359–365. | |
Pluskota A, Horzowski E, Bossinger O, von Mikecz A. In Caenorhabditis elegans nanoparticle-bio-interactions become transparent: silica-nanoparticles induce reproductive senescence. PLoS One. 2009;4(8):e6622. | |
Mohan N, Chen CS, Hsieh HH, Wu YC, Chang HC. In vivo imaging and toxicity assessments of fluorescent nanodiamonds in Caenorhabditis elegans. Nano Lett. 2010;10(9):3692–3699. | |
Usenko CY, Harper SL, Tanguay RL. In vivo evaluation of carbon fullerene toxicity using embryonic zebrafish. Carbon N Y. 2007;45(9):1891–1898. | |
Asharani PV, Lian Wu Y, Gong Z, Valiyaveettil S. Toxicity of silver nanoparticles in zebrafish models. Nanotechnology. 2008;19(25):255102. | |
Fako VE, Furgeson DY. Zebrafish as a correlative and predictive model for assessing biomaterial nanotoxicity. Adv Drug Deliv Rev. 2009;61(6):478–486. | |
Liu X, Vinson D, Abt D, Hurt RH, Rand DM. Differential toxicity of carbon nanomaterials in Drosophila: larval dietary uptake is benign, but adult exposure causes locomotor impairment and mortality. Environ Sci Technol. 2009;43(16):6357–6363. | |
Sung JH, Ji JH, Park JD, et al. Subchronic inhalation toxicity of silver nanoparticles. Toxicol Sci. 2009;108(2):452–461. | |
Wu J, Liu W, Xue C, et al. Toxicity and penetration of TiO2 nanoparticles in hairless mice and porcine skin after subchronic dermal exposure. Toxicol Lett. 2009;191(1):1–8. | |
Liu T, Li L, Teng X, et al. Single and repeated dose toxicity of mesoporous hollow silica nanoparticles in intravenously exposed mice. Biomaterials. 2011;32(6):1657–1668. | |
Hunter P. The paradox of model organisms. The use of model organisms in research will continue despite their shortcomings. EMBO Rep. 2008;9(8):717–720. | |
Fischer HC, Chan WC. Nanotoxicity: the growing need for in vivo study. Curr Opin Biotechnol. 2007;18(6):565–571. | |
Ahamed M, Posgai R, Gorey TJ, Nielsen M, Hussain SM, Rowe JJ. Silver nanoparticles induced heat shock protein 70, oxidative stress and apoptosis in Drosophila melanogaster. Toxicol Appl Pharmacol. 2010;242(3):263–269. | |
Posgai R, Cipolla-McCulloch CB, Murphy KR, Hussain SM, Rowe JJ, Nielsen MG. Differential toxicity of silver and titanium dioxide nanoparticles on Drosophila melanogaster development, reproductive effort, and viability: size, coatings and antioxidants matter. Chemosphere. 2011;85(1):34–42. | |
Barandeh F, Nguyen PL, Kumar R, et al. Organically modified silica nanoparticles are biocompatible and can be targeted to neurons in vivo. PLoS One. 2017;(1):e29424. | |
Carrero-Martínez F, Chiba A. Cell adhesion molecules at the Drosophila neuromuscular junction. In: Umemori H, Hortsch M, editors. The Sticky Synapse. New York: Springer; 2009:11–38. | |
Tweedie S, Ashburner M, Falls K, et al; FlyBase Consortium. FlyBase: enhancing Drosophila Gene Ontology annotations. Nucleic Acids Res. 2009;37(Database issue):D555–D559. | |
Das T, Cagan R. Drosophila as a novel therapeutic discovery tool for thyroid cancer. Thyroid. 2010;20(7):689–695. | |
Ashburner M, Golic K, Hawley RS, editors. Drosophila: A Laboratory Handbook. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press; 2004. | |
Hoang B, Chiba A. Single-cell analysis of Drosophila larval neuromuscular synapses. Dev Biol. 2001;229(1):55–70. | |
Margaritis LH, Kafatos FC, Petri WH. The eggshell of Drosophila melanogaster. I. Fine structure of the layers and regions of the wild-type eggshell. J Cell Sci. 1980;43:1–35. | |
Campos-Ortega JA, Hartenstein V. The Embryonic Development of Drosophila melanogaster. Berlin: Springer-Verlag; 1985. | |
Bee A, Massart R, Neveu S. Synthesis of very fine maghemite particles. J Magnetism Magnetic Mater. 1995;149(1):6–9. | |
Park J, An K, Hwang Y, et al. Ultra-large-scale syntheses of monodisperse nanocrystals. Nat Mater. 2004;3(12):891–895. | |
Barrera C, Herrera A, Zayas Y, Rinaldi C. Surface modification of magnetite nanoparticles for biomedical applications. J Magnetism Magnetic Mater. 2009;321(10):1397–1399. | |
Herrera AP, Barrera C, Rinaldi C. Synthesis and functionalization of magnetite nanoparticles with aminopropylsilane and carboxymethyldextran. J Mater Chem. 2008;18:3650–3654. | |
Creixell M, Herrera AP, Latorre-Esteves M, et al. The effect of grafting method on the colloidal stability and in vitro cytotoxicity of carboxymethyl dextran coated magnetic nanoparticles. J Mater Chem. 2010;20:8539–8547. | |
Allouni ZE, Cimpan MR, Høl PJ, Skodvin T, Gjerdet NR. Agglomeration and sedimentation of TiO2 nanoparticles in cell culture medium. Colloids Surf B Biointerfaces. 2009;68(1):83–87. | |
Guidance for Industry. Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers. 2005; http://www.fda.gov/downloads/Drugs/GuidanceCompliance RegulatoryInformation/Guidances/UCM078932.pdf. Accessed August 21, 2011. | |
Reading BD, Freeman B. Simple formula for the surface area of the body and a simple model for anthropometry. Clin Anat. 2005;18(2):126–130. | |
Du Bois D, Du Bois EF. A formula to estimate the approximate surface area if height and weight be known. 1916. Nutrition. 1989;5(5):303–311. | |
Gehan EA, George SL. Estimation of human body surface area from height and weight. Cancer Chemother Rep. 1970;54(4):225–235. | |
Haycock GB, Schwartz GJ, Wisotsky DH. Geometric method for measuring body surface area: a height-weight formula validated in infants, children, and adults. J Pediatr. 1978;93(1):62–66. | |
Mosteller RD. Simplified calculation of body-surface area. N Engl J Med. 1987;317(17):1098. | |
Kiehart DP, Crawford JM, Montague RA. Collection, dechorionation, and preparation of Drosophila embryos for quantitative microinjection. CSH Protoc. 2007;2007:pdb.prot4717. | |
Joner EJ, Hartnik T, Amundsen CE, editors. Nanoparticles and the Environmen. Environmental Fate and Ecotoxicity of Engineered Nanoparticles. ås, Norway: Bioforsk, Norwegian Pollution Control Authority; 2008. | |
Hussain SM, Hess KL, Gearhart JM, Geiss KT, Schlager JJ. In vitro toxicity of nanoparticles in BRL 3A rat liver cells. Toxicol In Vitro. 2005;19(7):975–983. | |
Jin C, Tang Y, Fan XY, et al. In vivo evaluation of the interaction between titanium dioxide nanoparticle and rat liver DNA. Toxicol Ind Health. 2013;29(3):235–244. | |
Long TC, Saleh N, Tilton RD, Lowry GV, Veronesi B. Titanium dioxide (P25) produces reactive oxygen species in immortalized brain microglia (BV2): implications for nanoparticle neurotoxicity. Environ Sci Technol. 2006;40(14):4346–4352. | |
Jia G, Wang H, Yan L, et al. Cytotoxicity of carbon nanomaterials: single-wall nanotube, multi-wall nanotube, and fullerene. Environ Sci Technol. 2005;39(5):1378–1383. | |
Lam CW, James JT, McCluskey R, Arepalli S, Hunter RL. A review of carbon nanotube toxicity and assessment of potential occupational and environmental health risks. Crit Rev Toxicol. 2006;36(3):189–217. | |
Shvedova AA, Castranova V, Kisin ER, et al. Exposure to carbon nanotube material: assessment of nanotube cytotoxicity using human keratinocyte cells. J Toxicol Environ Health A. 2003;66(20):1909–1926. | |
Smart SK, Cassady AI, Lu GQ, Martin DJ. The biocompatibility of carbon nanotubes. Carbon. 2006;44(6):1034–1047. | |
Warheit DB, Laurence BR, Reed KL, Roach DH, Reynolds GA, Webb TR. Comparative pulmonary toxicity assessment of single-wall carbon nanotubes in rats. Toxicol Sci. 2004;77(1):117–125. | |
Wick P, Manser P, Limbach LK, et al. The degree and kind of agglomeration affect carbon nanotube cytotoxicity. Toxicol Lett. 2007;168(2):121–131. | |
Pandey A, Chandra S, Chauhan LK, Narayan G, Chowdhuri DK. Cellular internalization and stress response of ingested amorphous silica nanoparticles in the midgut of Drosophila melanogaster. Biochim Biophys Acta. 2013;1830(1):2256–2266. | |
Demir E, Turna F, Vales G, Kaya B, Creus A, Marcos R. In vivo genotoxicity assessment of titanium, zirconium and aluminium nanoparticles, and their microparticulated forms, in Drosophila. Chemosphere. 2013;93(10):2304–2310. | |
Vecchio G, Galeone A, Brunetti V, et al. Mutagenic effects of gold nanoparticles induce aberrant phenotypes in Drosophila melanogaster. Nanomedicine. 2012;8(1):1–7. | |
Gorth DJ, Rand DM, Webster TJ. Silver nanoparticle toxicity in Drosophila: size does matter. Int J Nanomedicine. 2016:343–350. | |
Das S, Debnath N, Patra P, Datta A, Goswami A. Nanoparticles influence on expression of cell cycle related genes in Drosophila: a microarray-based toxicogenomics study. Toxicol Environ Chem. 2012;94(5):952–957. | |
Vecchio G, Galeone A, Malvindi MA, Cingolani R, Pompa PP. Ranking the in vivo toxicity of nanomaterials in Drosophila melanogaster. J Nanoparticle Res. 2013;15(9):1936. | |
Philbrook NA, Winn LM, Afrooz AR, Saleh NB, Walker VK. The effect of TiO(2) and Ag nanoparticles on reproduction and development of Drosophila melanogaster and CD-1 mice. Toxicol Appl Pharmacol. 2011;257(3):429–436. | |
Pompa PP, Vecchio G, Galeone A, et al. In Vivo toxicity assessment of gold nanoparticles in Drosophila melanogaster. Nano Research. 2011;4(4):405–412. | |
Key CS, Reaves D, Turner F, Bang JJ. Impacts of silver nanoparticle ingestion on pigmentation and developmental progression in Drosophila. Atlas J Biol. 2011;1:52–61. | |
Panacek A, Prucek R, Safarova D, et al. Acute and chronic toxicity effects of silver nanoparticles (NPs) on Drosophila melanogaster. Environ Sci Technol. 2011;45(11):4974–4979. | |
Wu Q, Wen T, Lee G, Park JH, Cai HN, Shen P. Developmental control of foraging and social behavior by the Drosophila neuropeptide Y-like system. Neuron. 2003;39(1):147–161. | |
Wu Q, Zhang Y, Xu J, Shen P. Regulation of hunger-driven behaviors by neural ribosomal S6 kinase in Drosophila. Proc Natl Acad Sci U S A. 2005;102(37):13289–13294. | |
Wu Q, Zhao Z, Shen P. Regulation of aversion to noxious food by Drosophila neuropeptide Y- and insulin-like systems. Nat Neurosci. 2005;8(10):1350–1355. | |
Kaun KR, Chakaborty-Chatterjee M, Sokolowski MB. Natural variation in plasticity of glucose homeostasis and food intake. J Exp Biol. 2008;211(Pt 19):3160–3166. | |
Lingo PR, Zhao Z, Shen P. Co-regulation of cold-resistant food acquisition by insulin- and neuropeptide Y-like systems in Drosophila melanogaster. Neuroscience. 2007;148(2):371–374. | |
Johnston HJ, Hutchison G, Christensen FM, Peters S, Hankin S, Stone V. A review of the in vivo and in vitro toxicity of silver and gold particulates: particle attributes and biological mechanisms responsible for the observed toxicity. Crit Rev Toxicol. 2010;40(4):328–346. | |
Lubick N. Nanosilver toxicity: ions, nanoparticles – or both? Environ Sci Technol. 2008;42(23):8617. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.




