Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 13
Tiotropium/olodaterol versus tiotropium in Japanese patients with COPD: results from the DYNAGITO study
Authors Ichinose M, Nishimura M , Akimoto M, Kurotori Y, Zhao Y, de la Hoz A, Mishima M
Received 2 April 2018
Accepted for publication 21 May 2018
Published 13 July 2018 Volume 2018:13 Pages 2147—2156
DOI https://doi.org/10.2147/COPD.S169941
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Masakazu Ichinose,1 Masaharu Nishimura,2 Manabu Akimoto,3 Yasuhiro Kurotori,3 Yihua Zhao,4 Alberto de la Hoz,5 Michiaki Mishima6
1Department of Respiratory Medicine, Tohoku University Graduate School of Medicine, Sendai, Japan; 2Department of Pulmonary Medicine, Faculty School of Medicine, Hokkaido University, Sapporo, Japan; 3Nippon Boehringer Ingelheim Co. Ltd, Tokyo, Japan; 4Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield, CT, USA; 5Boehringer Ingelheim International GmbH, Ingelheim, Germany; 6Osaka Saiseikai Noe Hospital, Osaka, Japan
Background: The DYNAGITO study was a Phase IIIb, randomized, double-blind, multicenter, active-controlled, parallel-group, 52-week study designed to determine the efficacy and safety of tiotropium and olodaterol combination therapy (TIO+OLO 5/5 μg) versus tiotropium monotherapy (TIO 5 μg) for reducing moderate-to-severe exacerbations of COPD. This is a prespecified analysis of the DYNAGITO data in Japanese patients.
Patients and methods: Enrolled patients had a diagnosis of COPD with at least one moderate-to-severe exacerbation in the previous 12 months. Of the total 7,880 treated patients in the DYNAGITO study, 461 (TIO+OLO 5/5 μg: n=226, TIO 5 μg: n=235) were Japanese. The primary endpoint was the annualized rate of moderate-to-severe COPD exacerbations. The key secondary endpoint was the time to first moderate-to-severe COPD exacerbation, and other secondary endpoints included the annualized rate of exacerbations leading to hospitalization, time to first COPD exacerbation leading to hospitalization, and all-cause mortality. Safety data were analyzed descriptively.
Results: Combination therapy with TIO+OLO resulted in a 29% lower rate of moderate-to-severe COPD exacerbations relative to TIO monotherapy (rate ratio 0.71; 99% CI: 0.46, 1.10; p=0.0434). The risk of a first moderate-to-severe COPD exacerbation was 19% lower with TIO+OLO combination therapy than with TIO monotherapy (HR 0.81; 99% CI: 0.57, 1.17; p=0.1379), although this difference was not statistically significant. The annualized rate of COPD exacerbations requiring hospitalization was 14% lower in the TIO+OLO arm than in the TIO arm (rate ratio 0.86; 95% CI: 0.52, 1.42; p=0.5654). The adverse event incidence was balanced between treatment arms.
Conclusion: In a prespecified subgroup analysis of Japanese patients in the DYNAGITO study, combination therapy with TIO+OLO was more effective than TIO in reducing exacerbations. Both treatments were well tolerated.
Keywords: all-cause mortality, COPD, fixed-dose combination therapy, hospitalization, moderate-to-severe exacerbations, monotherapy
Corrigendum for this paper has been published
Introduction
COPD is characterized by respiratory symptoms and airflow limitations1 and is the fourth leading cause of death globally.2 Approximately 10% of the Japanese population aged ≥40 years is estimated to have airflow limitation, according to the Nippon COPD epidemiology study.3 Among these individuals, only 9.4% are diagnosed with COPD, and 44% of these individuals have moderate-to-very severe airflow limitation.3
The Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2017 guidance document and Japanese Respiratory Society guidelines recommend long-acting bronchodilators (eg, long-acting muscarinic antagonists [LAMAs] and long-acting beta agonists [LABAs]) for COPD. In several studies, these agents have been shown to improve lung function, symptoms, and quality of life, and to prevent COPD exacerbations.4–7 GOLD 2017 guidelines suggest that a patient’s frequency of exacerbations within the previous 12 months and the degree of symptoms should determine treatment selection, rather than the severity of airflow limitation.1
Therapeutic strategies for COPD management focus on improvement of symptoms, reduction of the risk of exacerbations, and improved prognosis. Based on the benefits demonstrated, LAMA/LABA combinations play a crucial role in COPD maintenance treatment. Long-acting inhaled bronchodilators have been investigated for their potential to reduce COPD exacerbations in several studies.4–6,8,9 A review of these studies shows that the LAMA tiotropium (TIO) has greater efficacy against exacerbations than LABAs (POET-COPD and INVIGORATE studies5,6). In addition, the efficacy of TIO in reducing exacerbations was shown to be non-inferior to that of fixed-dose combination therapy with inhaled corticosteroids (ICS) and LABA salmeterol (INSPIRE study4). In the SPARK study, fixed-dose combination therapy with LABA indacaterol and LAMA glycopyrronium was not superior to TIO monotherapy in reducing moderate and severe exacebrations.7
Olodaterol (OLO) is a new once-daily LABA bronchodilator that effectively improves lung function.10–13 The combination of TIO and OLO provides additional improvements in lung function and improves health-related quality of life.14–16 The DYNAGITO study was performed to compare the safety and efficacy of TIO and OLO combination therapy versus TIO monotherapy in reducing exacerbations in COPD patients with a history of at least one exacerbation in the previous 12 months that required treatment with systemic corticosteroids, antibiotics, and/or COPD-related hospitalization.17
The characteristics of COPD patients in the Japanese population, including age, body mass index, proportions of men and women, previous smoking history, and pulmonary medication at baseline, differ from other populations.18 Therefore, we performed a prespecified subgroup analysis for Japanese patients included in the DYNAGITO study.17
Patients and methods
Study participants
Patients with moderate-to-very severe COPD who had one or more moderate-to-severe COPD exacerbations within the past 12 months were recruited.
Key inclusion criteria were as follows: age ≥40 years, with diagnosed COPD; post-bronchodilator forced expiratory volume in 1 second (FEV1) <60% of predicted normal; history of one or more moderate-to-severe exacerbations in the previous 12 months that required treatment with systemic corticosteroids and/or antibiotics and/or hospitalization; and current or former smokers with a smoking history of more than 10 pack-years. The exclusion criteria were significant comorbidity, or a current diagnosis of asthma, thyrotoxicosis, or active tuberculosis. The full inclusion and exclusion criteria have been previously reported.17
The trial was registered at ClinicalTrials.gov (NCT02296138) and was conducted in accordance with ethical principles of the Declaration of Helsinki, Good Clinical Practice guidelines, all applicable regulatory requirements, and the approval of all relevant national and local ethics review boards (details of all institutional review boards are provided in Supplemental List 1). All patients provided written informed consent to participate in the study.
Study design
DYNAGITO was a Phase IIIb, randomized, double-blind, multicenter, active-controlled, parallel-group, 52-week exacerbation study conducted between January 2015 and March 2017 in 51 countries. The Japanese subgroup analysis was prespecified in the statistical analysis plan.
Patients were randomly assigned to receive once daily TIO+OLO 5/5 μg or TIO 5 μg (both administered orally via the Respimat® inhaler; Boehringher Ingelheim, Ingelheim am Rhein, Germany) for 52 weeks. The details of the study have been provided elsewhere.17
Efficacy and safety analyses
The primary endpoint was the annualized rate of moderate-to-severe COPD exacerbations during the actual treatment period. The key secondary endpoint was time to first moderate-to-severe COPD exacerbation during the actual treatment period. The annualized rate of exacerbations leading to hospitalization, time to first COPD exacerbation leading to hospitalization, and all-cause mortality were other secondary endpoints. All adverse events (AEs) were collected during the on-treatment period, defined as from the drug start date to 21 days after the last dose.
Statistical methods
The annualized rate of events was analyzed with a negative binomial model including the fixed, categorical effect of treatment and the logarithm of the treatment duration as an offset, without adjustment for covariates. For all time-to-first events analyses, a Cox’s proportional hazard model was used to estimate the HR and corresponding CI. All efficacy analyses included all randomized patients who received the study medication (ie, the treated set) and considered the actual treatment period (from drug start until 1 day after the last drug administration date). SAS version 9.4 (SAS Institute Inc., Cary, NC, USA) was used in all statistical analyses. In the DYNAGITO study, the target significance level for the primary and key secondary endpoints was set to 1%. To maintain consistency with the DYNAGITO study, 99% CIs are presented for the primary and key secondary endpoint analyses for the Japanese subgroup. For analyses of other endpoints, 95% CIs are presented as in the DYNAGITO study. Because the prespecified significance level for the primary endpoint in the DYNAGITO study was not met, all p-values presented should be considered nominal.
All treated patients were included in the safety analysis (safety set), and all analyses of the Japanese subgroup were descriptive. Because patients were followed up for exacerbations and vital status, missing data for these endpoints were not imputed.
Results
Patient demographics and clinical characteristics
A total of 7,880 treated patients, of whom 461 (5.85%) were Japanese, were randomly assigned to study medications in the DYNAGITO study. Characteristics of the Japanese subgroup were as follows: mean ± SD age was 71.6±7.2 years; mean body mass index was 21.4±3.5 kg/m2 (Table 1); and more than 90% of patients were men. With regard to smoking history, 83.3% of patients were ex-smokers, and there were no never-smokers.
Mean percent predicted FEV1 after bronchodilator use was ~43%, and many patients had severe airflow limitation (GOLD stages III and IV). Almost 50% of patients had had at least two moderate COPD exacerbations or one severe COPD exacerbation in the previous year. Approximately 41% of patients were receiving LAMA+LABA+ICS therapy at baseline. The mean ± SD of total score on the St George’s Respiratory Questionnaire at baseline was 40.4±17.1 (Table 1).
TIO monotherapy was administered to 235 patients, with 185 completing the study (Figure 1). A total of 226 patients received TIO+OLO combination therapy, with 203 completing the study. A greater proportion of patients in the TIO than TIO+OLO arm discontinued earlier from treatment (21.3% vs 10.2%; HR 0.45; 95% CI: 0.27, 0.73; p=0.001; Figures 1 and 2). Most discontinuations were due to AEs including worsening COPD. Baseline demographics and clinical characteristics were similar between the two treatment arms (Table 1; Table S1).
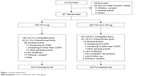  | Figure 1 Patient disposition. |
  | Figure 2 Probability of treatment discontinuation by treatment group. |
Efficacy
The annualized rate of moderate-to-severe COPD exacerbations was 29% lower in the TIO+OLO arm than the TIO arm (0.94 vs 1.32; rate ratio [RR] 0.71; 99% CI: 0.46, 1.10; p=0.0434; Figure 3 and Table S2). Similarly, the annualized rate of severe exacerbations was 19% lower in the TIO+OLO arm than TIO arm (0.35 vs 0.43; RR 0.81; 95% CI: 0.51, 1.31; p=0.3924; Figure 3 and Table S2). Subgroup analyses of the primary endpoint according to baseline characteristics and respiratory medicines at baseline are presented in Figure 4; the tendency of TIO+OLO to reduce COPD exacerbations beyond that of TIO monotherapy was consistent across relevant subgroups based on demographics and respiratory medications at baseline. The time to first moderate-to-severe COPD exacerbation was longer in the TIO+OLO arm than the TIO arm (HR 0.81; 99% CI: 0.57, 1.17; p=0.1379; Figure 5).
  | Figure 5 Cumulative risk of first moderate-to-severe COPD exacerbation by treatment group. |
The annualized rate of moderate-to-severe COPD exacerbations treated with antibiotics only did not differ between the TIO+OLO and TIO arms (0.38 vs 0.32; RR 1.19; 95% CI: 0.76, 1.85; p=0.4504; Figure 6 and Table S2), while the annualized rate of exacerbations treated with systemic corticosteroids only was lower in the TIO+OLO arm than the TIO arm (0.12 vs 0.32; RR 0.38; 95% CI: 0.17, 0.89; p=0.0252; Figure 6 and Table S2); the annualized rate of exacerbations treated with antibiotics and systemic corticosteroids was also lower in the TIO+OLO arm than the TIO arm (0.41 vs 0.67; RR 0.61; 95% CI: 0.39, 0.98; p=0.0393; Figure 6 and Table S2). The annualized rate of COPD exacerbations requiring hospitalization was numerically lower in the TIO+OLO arm than the TIO arm (0.31 vs 0.36; RR 0.86; 95% CI: 0.52, 1.42; p=0.5654; Figure 6 and Table S2).
Safety
The incidences of AEs in the two treatment arms were similar: AEs occurred in 196 patients (83.4%) in the TIO arm and 194 patients (85.8%) in the TIO+OLO arm (Table 2). The most frequently reported AE in both treatment arms was COPD (including exacerbations and worsening), followed by viral upper respiratory tract infection (Table 2). Drug-related AEs occurred in 10 patients (4.3%) and 13 patients (5.8%) in the TIO and TIO+OLO arms, respectively. Serious AEs occurred in 76 patients (32.3%) in the TIO arm and in 76 patients (33.6%) in the TIO+OLO arm. The most frequently reported serious AE was COPD in both treatment arms, followed by pneumonia. AEs resulting in study discontinuation occurred in 27 patients (11.5%) in the TIO arm and 14 patients (6.2%) in the TIO+OLO arm. AEs leading to death occurred in six patients (2.6%) in the TIO arm and in one patient (0.4%) in the TIO+OLO arm during the planned study period, although none of these AEs were considered related to treatment.
  | Table 2 Summary of AEs by preferred terms (safety set) |
Discussion
The objective of this prespecified subgroup analysis was to descriptively compare the efficacy and safety of TIO+OLO combination therapy with TIO monotherapy in the reduction of moderate-to-severe exacerbations in the Japanese subgroup of the DYNAGITO study. The prespecified level of significance of 1% for the primary endpoint was not met in the DYNAGITO study. The data show, within the limitations of this exploratory analysis, that TIO+OLO combination therapy had better efficacy at reducing the annualized rate of moderate-to-severe COPD exacerbations than TIO monotherapy. The effect of TIO+OLO combination therapy in the Japanese subgroup was numerically larger on moderate-to-severe exacerbations compared with those seen in the overall study population (Table S2). Although the rationale for the numerical difference between overall study patients and Japanese patients is unclear, this difference may be due to differences in clinical characteristics between the overall study population and Japanese patients.
Compared with the overall study population, the Japanese patients were older, with longer smoking histories; in addition, the proportions of men and ex-smokers were higher, body mass index was lower, LAMA use at baseline was more widespread, and ICS use at baseline was less common (Table S1).
The proportion of patients using triple ICS/LAMA/LABA combination therapy at baseline was ~40% for both treatment arms, which is similar to the overall study population; however, more patients in the Japanese subgroup received LAMA/LABA (24.3% vs 11.9%) and fewer received ICS/LABA combination therapy (5.2% vs 25.8%) than in the overall population (Table S1).
The annualized rates of moderate-to-severe COPD exacerbations treated with antibiotics only did not differ between the TIO+OLO and TIO arms. However, the annualized rates of moderate-to-severe exacerbations treated with systemic corticosteroids only and treated with systemic corticosteroids in combination with antibiotics were reduced in the TIO+OLO arm relative to the TIO arm. It is difficult to identify factors that might explain differences in rates of exacerbations requiring systemic corticosteroids or antibiotics. This may be related to the OLO-mediated anti-inflammatory effect, as previously described by Wex et al.19 It could also be because the decision to select between antibiotic and corticosteroid treatment, or a combination of the two, is made by the physician and is not based on formal standards set for the different types of exacerbation. However, systemic corticosteroid-treated events may represent more severe events than events treated with antibiotics alone, because systemic corticosteroid-treated events are associated with worse future outcomes.20 According to Japanese Respiratory Society guidelines,21 bronchodilators (mainly short-acting β2-agonists) are used as first-line therapy for exacerbations, with the addition of antibiotics if purulent sputum or infection arises. Systemic corticosteroid administration is recommended to COPD patients with airflow limitation rated worse than “severe”, or if hospitalization is necessary.
The incidence of AEs reported in the Japanese subgroup did not differ between the TIO and TIO+OLO arms (83.4% and 85.8%, respectively). However, AE incidences were slightly higher than those reported in the overall study population (TIO arm 74.5%; TIO+OLO arm 74.1%; Table S3). These slightly greater rates in the Japanese subgroup were consistent for most AEs, except for the AEs of COPD, dyspnea, and cough (Table S3). They may be due to the higher average age in the Japanese subgroup (71.6 years vs 66.4 years; Table S1). Although the incidences of viral upper respiratory tract infection (24.3% vs 7.3%), pneumonia (14.5% vs 4.9%), and bronchitis (10.6% vs 3.1%) were greater in the Japanese subgroup than in the overall study population, the incidence of COPD (including exacerbations) was lower (31.5% vs 43.9%; Table S3). These findings were evident, even though fewer patients were using ICS monotherapy (0.2% vs 2.5%) or an ICS plus LABA combination (5.2% vs 25.8%) at baseline (Table S1).
It has been previously reported that Japanese COPD patients may experience fewer exacerbations than Caucasian patients.22 Accordingly, Japanese patients in this study may not have experienced COPD exacerbations, even when they developed an infection, and their conditions may have stabilized, even in the presence of viral upper respiratory tract infections. Conversely, the higher incidence of pneumonia in the Japanese subgroup may have been due to higher average age, the treatment setting in Japan and, in particular, diagnostic facilitation by a general practitioner with access to radiographic imaging.23,24 Fewer deaths occurred in the Japanese subgroup versus the overall study population (1.5% vs 3.0%; Table S3) during the planned study period, regardless of the higher mean age (71.6 years vs 66.4 years), longer smoking history (62.5 pack-years vs 44.8 pack-years), and higher proportion of men (94.1% vs 71.4%; Table S1); this may be one of the clinical features of this Japanese subgroup.
In conclusion, TIO+OLO combination therapy provided a numerically greater reduction in moderate-to-severe exacerbations than TIO monotherapy in this Japanese subgroup analysis. In comparison to the overall study population, TIO+OLO showed a slightly greater reduction in COPD exacerbations in Japanese COPD patients. The safety profiles of TIO+OLO and TIO were comparable in the Japanese subgroup and the overall study population. Based on previously demonstrated benefits on lung function, symptoms, and quality of life,18,25 and findings from this analysis, TIO+OLO combination therapy is a viable treatment option for moderate-to-very severe COPD in Japanese patients.
Acknowledgments
The authors would like to thank all investigators and patients involved in this study, and Lourdes Briz and David Murdoch of Edanz Medical Writing for providing medical writing assistance, which was funded by Boehringer Ingelheim.
This study was funded by Nippon Boehringer Ingelheim Nippon Boehringer Ingelheim contributed to the design of the study, data collection, analysis, and interpretation of the study results. Nippon Boehringer Ingelheim also funded the medical writing support, publication charges, and open access fee for publication of this manuscript.
Author contributions
MI, MN, MA, YK, YZ, AD, and MM contributed to the manuscript; YZ analyzed the data and all the authors contributed to the interpretation of the data. MI drafted the manuscript, and all the authors contributed to critically revising the manuscript, read and approved the final manuscript.
Disclosure
MI has received lecture honoraria from AstraZeneca, Nippon Boehringer Ingelheim, and Novartis Pharma. MN has received research funding from AstraZeneca K.K. and Novartis Pharma, and lecture honoraria from Nippon Boehringer Ingelheim. MA and YK are employees of Nippon Boehringer Ingelheim. YZ is an employee of Boehringer Ingelheim Pharmaceuticals, Inc. AD is an employee of Boehringer Ingelheim International GmbH. MM received honoraria from Nippon Boehringer Ingelheim and AstraZeneca K.K. The authors report no other conflicts of interest in this work.
References
Vogelmeier CF, Criner GJ, Martinez FJ, et al. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Lung Disease 2017 report. GOLD executive summary. Am J Respir Crit Care Med. 2017;195(5):557–582. | ||
World Health Organisation. Mortality and global health estimates. 2015 [cited 2018 Jan 12]. Available from: http://www.who.int/gho/mortality_burden_disease/en/. Accessed February 12, 2018. | ||
Fukuchi Y, Nishimura M, Ichinose M, et al. COPD in Japan: the Nippon COPD Epidemiology study. Respirology. 2004;9(4):458–465. | ||
Wedzicha JA, Calverley PM, Seemungal TA, et al. The prevention of chronic obstructive pulmonary disease exacerbations by salmeterol/fluticasone propionate or tiotropium bromide. Am J Respir Crit Care Med. 2008;177(1):19–26. | ||
Vogelmeier C, Hederer B, Glaab T, et al. Tiotropium versus salmeterol for the prevention of exacerbations of COPD. N Engl J Med. 2011;364(12):1093–1103. | ||
Decramer ML, Chapman KR, Dahl R, et al. Once-daily indacaterol versus tiotropium for patients with severe chronic obstructive pulmonary disease (INVIGORATE): a randomised, blinded, parallel-group study. Lancet Respir Med. 2013;1(7):524–533. | ||
Wedzicha JA, Decramer M, Ficker JH, et al. Analysis of chronic obstructive pulmonary disease exacerbations with the dual bronchodilator QVA149 compared with glycopyrronium and tiotropium (SPARK): a randomised, double-blind, parallel-group study. Lancet Respir Med. 2013;1(3):199–209. | ||
Koch A, Watz H, Maleki-Yazdi MR, et al. Comprehensive assessment of the safety of olodaterol 5 μg in the Respimat® device for maintenance treatment of COPD: comparison with the long-acting beta2-agonist formoterol. NPJ Prim Care Respir Med. 2017;27(1):60. | ||
Joos GF, Aumann JL, Coeck C, et al. A randomised, double-blind, four-way, crossover trial comparing the 24-h FEV1 profile for once-daily versus twice-daily treatment with olodaterol, a novel long-acting beta2-agonist, in patients with chronic obstructive pulmonary disease. Respir Med. 2015;109(5):606–615. | ||
Feldman GJ, Bernstein JA, Hamilton A, Nivens MC, Korducki L, LaForce C. The 24-h FEV1 time profile of olodaterol once daily via Respimat® and formoterol twice daily via Aerolizer® in patients with GOLD 2-4 COPD: results from two 6-week crossover studies. Springerplus. 2014;3:419. | ||
Ferguson GT, Feldman GJ, Hofbauer P, et al. Efficacy and safety of olodaterol once daily delivered via Respimat® in patients with GOLD 2–4 COPD: results from two replicate 48-week studies. Int J Chron Obstruct Pulmon Dis. 2014;9:629–645. | ||
Koch A, Pizzichini E, Hamilton A, et al. Lung function efficacy and symptomatic benefit of olodaterol once daily delivered via Respimat® versus placebo and formoterol twice daily in patients with GOLD 2–4 COPD: results from two replicate 48-week studies. Int J Chron Obstruct Pulmon Dis. 2014;9:697–714. | ||
Lange P, Hamilton J-L, Tetzlaff K, Ting N, Derom E. The 24 hour lung function time profile of olodaterol once daily versus placebo and tiotropium in patients with moderate to very severe chronic obstructive pulmonary disease. J Pulm Resp Med. 2014;4:196. | ||
Buhl R, Maltais F, Abrahams R, et al. Tiotropium and olodaterol fixed-dose combination versus mono-components in COPD (GOLD 2–4). Eur Respir J. 2015;45(4):969–979. | ||
Beeh KM, Westerman J, Kirsten AM, et al. The 24-h lung-function profile of once-daily tiotropium and olodaterol fixed-dose combination in chronic obstructive pulmonary disease. Pulm Pharmacol Ther. 2015;32:53–59. | ||
Singh D, Ferguson GT, Bolitschek J, et al. Tiotropium + olodaterol shows clinically meaningful improvements in quality of life. Respir Med. 2015;109(10):1312–1319. | ||
Calverley PMA, Anzueto AR, Carter K, et al. Tiotropium and olodaterol in the prevention of chronic obstructive pulmonary disease exacerbations: a double-blind, randomised, parallel-group, active-controlled, trial. Lancet Respir Med. 2018;6(5):337–344. | ||
Ichinose M, Taniguchi H, Takizawa A, et al. The efficacy and safety of combined tiotropium and olodaterol via the Respimat® inhaler in patients with COPD: results from the Japanese sub-population of the Tonado® studies. Int J Chron Obstruct Pulmon Dis. 2016;11:2017–2027. | ||
Wex E, Kollak I, Duechs MJ, Naline E, Wollin L, Devillier P. The long-acting beta2-adrenoceptor agonist olodaterol attenuates pulmonary inflammation. Br J Pharmacol. 2015;172(14):3537–3547. | ||
Calverley PMA, Anzueto AR, Dusser D, et al. Treatment of exacerbations as a predictor of subsequent outcomes in patients with COPD. Int J Chron Obstruct Pulmon Dis. 2018;3:1297–1308. | ||
Japanese Respiratory Society. COPD Guidelines for Diagnosis and Treatment. 4th ed. Japanese Respiratory Society; Tokyo; 2013. | ||
Suzuki M, Makita H, Ito YM, et al. Clinical features and determinants of COPD exacerbation in the Hokkaido COPD cohort study. Eur Respir J. 2014;43(5):1289–1297. | ||
Berrington de González A, Darby S. Risk of cancer from diagnostic X-rays: estimates for the UK and 14 other countries. Lancet. 2004;363:345–351. | ||
Kumamaru KK, Machitori A, Koba R, et al. Global and Japanese regional variations in radiologist potential workload for computed tomography and magnetic resonance imaging examinations. Jpn J Radiol. 2018;36(4):273–281. | ||
Ichinose M, Kato M, Takizawa A, et al. Long-term safety and efficacy of combined tiotropium and olodaterol in Japanese patients with chronic obstructive pulmonary disease. Respir Investig. 2017;55(2):121–129. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.




