Back to Journals » Research and Reports in Neonatology » Volume 10
Time to Death and its Predictors among Neonates Admitted in the Intensive Care Unit of the University of Gondar Comprehensive Specialized Hospital, Northwest Ethiopia
Authors Gudayu TW , Zeleke EG, Lakew AM
Received 9 October 2019
Accepted for publication 8 January 2020
Published 29 January 2020 Volume 2020:10 Pages 1—10
DOI https://doi.org/10.2147/RRN.S233828
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Robert Schelonka
Temesgen Worku Gudayu,1 Ejigu Gebeye Zeleke,2 Ayenew Molla Lakew2
1Department of Clinical Midwifery, School of Midwifery, College of Medicine and Health Sciences, University of Gondar, Gondar, Ethiopia; 2Department of Epidemiology and Biostatistics, Institute of Public Health, College of Medicine and Health Sciences, University of Gondar, Gondar, Ethiopia
Correspondence: Temesgen Worku Gudayu
College of Medicine and Health Sciences, School of Midwifery, University of Gondar, PO.Box 196, Gondar, Ethiopia
Tel +2510911755087
Email [email protected]
Introduction: Neonatal mortality is among the leading concerns globally and its burden is highest in sub-Saharan Africa. Despite all the efforts made, neonatal mortality remains high in Ethiopia in general and in the Amhara region in particular. In this study, we assessed the time to death and its predictors among neonates admitted to the intensive care unit of the University of Gondar Comprehensive Specialized Hospital.
Patients and Methods: A retrospective follow-up study was conducted among 504 randomly selected neonates admitted to the neonatal intensive care unit between January 1 and December 31, 2017. The extracted data were entered into epi-info version 7.2, exported to and analyzed using STATA version 14. Failure probability table and Kaplan–Meier failure function were computed. Gompertz proportional hazard regression model was used to identify predictors of time to neonatal death. The hazard ratio with a 95% confidence interval was reported and statistical significance was declared at a p-value of ≤ 0.05.
Results: The proportion of neonatal mortality in this study was 17.3 (95% CI: 14.2, 20.9). Out of 504 studied neonates, 87 (173 per 1000 live births) died. Neonates who did not cry at birth and those who were born before 34 weeks had a higher risk of death in both births-to-death and admission-to-death analysis. Whereas, in admission-to-death analysis, neonates who were born at home had a higher risk of death.
Conclusion: In this study, neonatal mortality is far higher than the national and sustainable development goal targets. Strengthening access to health facilities, improving the community’s awareness of giving birth at health facilities and strengthening facilities and providers’ capacity for caring for high-risk neonates prior to the referral is recommended.
Keywords: neonate, neonatal mortality, neonatal death, neonatal intensive care unit, Ethiopia
Introduction
Neonates are young infants in the age category of birth to 28 days of life. Most fetuses who completed normal gestational age adapt to extrauterine life in the early neonatal period. However, some premature neonates, those having respiratory distress and other malformations, need some assistance for adjustment.1 Though adaptation is physiological, all newborns are vulnerable and the period from birth to the following few days of life hold the highest concentrated risk of death in a human lifetime.2
A 2013 estimate on neonatal mortality demonstrated that about 2.8 million neonatal deaths occurred worldwide. More than half of the world’s neonatal death in the same year were contributed by five countries, namely: India, Nigeria, Pakistan, the Democratic Republic of Congo and China.3 In the 14 years from 2000 to 2013, a quicker decline of mortality was observed among children aged 1–59 months than neonates, and under-5 death during the neonatal period increased from about 37% in 2000 to nearly 42% in 2013.3,4
Ethiopia is among the highest neonatal death contributing countries placed at the 6th position globally, the 2nd in the Sub-Saharan Africa next to Nigeria and the leading in east Africa. In Ethiopia, neonatal death per 1000 live births was reported as 30.5 at 1st day, 61.7 at 1st week and 22.7 at 2nd week of life.5
Recent evidence has shown a slight drop in neonatal mortality rate in Ethiopia between 2000 and 2016 from 49 to 29 per 1000 live births.6 The distribution of neonatal mortality throughout the country ranged from 18 in Addis Ababa, 34–38 in other regions, and 41 in the Somali region. Amhara is the leading region for neonatal death in the country with 47 neonatal deaths per 1000 live births.6
Different studies of neonatal-period mortality in Ethiopia report 35.5 in Jimma Zone,7 62.5 in Tigray region,8 68 in Dabat Health and Demographic Surveillance site (found in the Amhara region)9 and 23.15 in neonatal care unit of St. Paul’s Hospital10 per 1000 live births.
Neonatal, infant and under-5 death are inversely related to the gross domestic product per capita purchasing power of, particularly, developing countries.11 In an effort to tackle this problem, the world paid attention and developed a strategy, the millennium development goal (MDG). Though huge resources were invested to implement the goals, health-related goals are unfinished globally12 in general, and Ethiopia’s13 agenda in particular.
In an effor to reduce neonatal mortality, Ethiopia is working to increase skilled birth assistance through the facility and health care provider expansion, family planning service utilization, antenatal care utilization, and postnatal care utilization.6 Despite all these efforts, neonatal mortality remains high in Ethiopia, predominantly in the Amhara region.6 This study, therefore, was intended to assess time-to-death and its predictors among neonates admitted to the Neonatal Intensive Care Unit (NICU) of the University of Gondar Comprehensive Specialized Hospital (UoGCSH).
Materials and Methods
Study Design and Participants
A retrospective follow-up study was conducted among neonates admitted to the NICU from January 1 to December 31, 2017 at the University of Gondar Comprehensive Specialized Hospital (UoGCSH).
Data were extracted from April 16 to May 15, 2018, at the UGCSH NICU. The Hospital is found in Gondar town and serves more than 7 million people residing in northwest Ethiopia. The NICU is a unit under the pediatrics and child health department and it provides an inpatient medical service for neonates. It has a caring capacity of about 30 beds at a time. About 2500 neonates were admitted to the intensive care unit between January 1 and December 31, 2017.
Neonates admitted to the NICU of UoGCSH from January 1 to December 31, 2017 were included in this study. Neonates whose charts had an incomplete observation on major variables (for example, date of admission and date of discharge/death and treatment outcomes such as discharge or death) were excluded.
Sample Size and Sampling Technique
The sample size was determined using STATA statistical package version 14 considering the following assumptions. A 0.37 hazard-ratio of associated maternal complication,8 variability of 0.5, a 0.0625 probability of neonatal death in Northern Ethiopia,8 5% marginal error, 80% power, and 5% contingency. The final estimated sample size was 535 neonatal charts.
The sample size was allocated proportionally to each month based on an estimated caseload. Then, a sequential number, starting from one, was assigned for each neonate according to the admission order for each month. Thereafter, a simple random sampling technique was used to select neonates and neonatal charts were identified by the medical registration numbers.
Measurements
Time-to-death was defined as a time in days from both birth and admission to death/transfer/discharge of neonates in the NICU. Death/event was considered as any form of neonatal death in the NICU except due to congenital malformations incompatible with life, such as anencephaly. Neonates who were discharged or transferred to other hospitals or self-discharged were considered as censored.
In this study, antenatal care utilization during index pregnancy was considered as utilization when a mother received at least one antenatal visit. The gestational age of the neonate at birth was measured from the last normal menstrual period of the mother (if available), first-trimester ultrasound estimates (if available), or clinical estimation of the Ballard score of neonates.
Weight for gestational age was determined based on sex-specific World Health Organization charts and categorized as large for those greater than 90th percentile, appropriate between 10th and 90th percentile, and small if less than 10th percentile.
Other variables such as perinatal asphyxia (PNA), hyaline membrane disease (HMD), sepsis, meconium aspiration syndrome (MAS) and hyperbilirubinemia, bilirubin encephalopathy, and hypothermia measured as yes or no if the diagnosis was made at the admission of neonates to NICU.
Data Extraction and Quality Control Procedures
Semi-structured data extraction checklist was used and the checklist was developed based on literature and reviewing patient charts. It was prepared in English and data extraction was done by trained individuals. The data extraction process was evaluated on a daily basis and necessary adjustment was made.
During the data extraction period, about 120 randomly selected charts were incomplete or major variables were missing and the charts were replaced with new, randomly selected, ones. Finally, out of a planned 535 charts, a total of 504 charts were included in the analysis.
Data Management and Analysis
Data were checked manually for completeness and entered into Epi-info software version 7.2. Then, the data were transported to STATA version 14 and cleaned before analysis. Descriptive findings were presented in tables, figures, and in text form. Failure probability table and Kaplan–Meier failure function were computed.
The proportional hazard (PH) assumption was checked graphically and by global and detailed Schoenfeld residuals test and the test result met the proportional hazard assumptions.
After the proportional hazard assumptions were done, Cox proportional hazard regression model and other parametric survival analysis models were fitted by taking the baseline hazard distribution assumptions into account. Then, the final fitted model, Gompertz proportional hazard model, was chosen based on the log-likelihood ratio (LR) test and Akaike Information Criterion (AIC).
Neonatal death was predicted by two models by taking the time variable as birth-to-death, and admission-to-death period. Statistically significant factors for birth-to-death and admission-to-death were identified based on the adjusted hazard ratio and its 95% confidence interval at a p-value ≤ 0.05.
Results
Characteristics of Mothers of Neonates Admitted in NICU
A total of 504 charts of neonates admitted in the NICU were reviewed. The median age of the mothers was 26 (IQR: 9) years and more than three quarters, 396 (78.57%) of them were between the age of 20 and 35 years. The majority, 485 (96.23%), of mothers attended ANC and 444 (88.10%) received tetanus toxoid twice (TT2+) during their index pregnancy. About 157 (31.16%) of the mothers had at least one complication during the index pregnancy.
Approximately 404 (80%) of the mothers had a singleton pregnancy and for about 415 (82%) of the mothers the labor was of spontaneous onset. More than two-thirds, 328 (65.08%), of mothers had a spontaneous vaginal delivery and about 369 (73%) of the births occurred at Hospitals (Table 1).
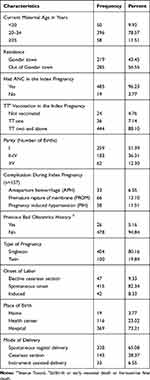 |
Table 1 Socio-demographic and Obstetric Characteristics of Mothers of Neonates Admitted in the NICU of UGCSH, Northwest Ethiopia n=504 |
Characteristics of Neonates Admitted in NICU
Two hundred and ninety-nine (59.44%) neonates were males, and about 209 (41%) were preterm births. Prior to admission to the NICU 129 (25.6%) of the neonates were resuscitated with bag and mask and nearly 237 (47%) were hypothermic at admission. Neonatal sepsis was clinically (based on clinical features) suspected/diagnosed among 321 (63.69%) neonates (Table 2).
 |
Table 2 Characteristics of Neonates Admitted in the NICU of UGCSH, Northwest Ethiopia n=504 |
Neonatal Outcome
Among studied neonates, 87 (17% (95% CI: 14.19, 20.83)) died in the neonatal intensive care unit and only 2 cases were referred to other hospitals. Whereas 80% of neonates improved and were discharged (Figure 1).
 |
Figure 1 Outcome of neonates admitted in the NICU of UoGCSH, Northwest Ethiopia. |
Failure/Death of Neonates
Neonates were followed for 3276 and 2508 neonatal days and neonatal admission days, respectively. Among the neonates followed, 87 (17.3% [95% CI: 14.19, 20.83]) died during the follow-up period and that makes neonatal mortality rate (NMR) 173 per 1000 live births. The overall death incidence rate in this study was 26.56 (95% CI 21.52, 32.77) per 1000 neonatal days for birth-to-death and 34.69 (95% CI: 28.11, 42.80) per 1000 neonatal admission days for admission-to-death.
From all deaths, about 14% of the neonates died in the first 24 hrs, nearly 60% died in the first 3 days and almost 84% of the neonatal deaths occurred in the first 1 week of life. Neonatal death was further analyzed from the time of admission and consequently, about 17% of the deaths occurred in the first 24 hrs, nearly 74% deaths within 3 days and almost 95% deaths occurred in the first week of admission.
At the end of the follow-up, the cumulative failure probability was 26.56 (95% CI: 21.52, 32.77) per 1000 follow-up days for birth-to -death. Similarly, it was 34.69 (95% CI: 28.11, 42.80) per 1000 follow-up days for admission-to-death (Table 3).
 |
Table 3 Failure Probability of Neonates Admitted in the NICU of UGCSH, Northwest Ethiopia n=504 |
The probability of neonatal death during the follow-up time from date of birth and admission was also presented by the failure curve. Accordingly, during the first seven days, the graph went up increasingly showing a higher probability of neonatal death. Also, between days 7 and 14, the probability of death continued and the graph moved upward. In the remaining days of the follow-up period, the graph became straight in the admission-to-death indicating the likelihood of neonatal death remained stable with virtually no deaths occurring. While the probability of death remained slight until 21 days in the birth-to- death graph (Figure 2).
Causes of Neonatal Death
Among 87 neonates who were dead during the follow-up period, the three leading causes of death were perinatal asphyxia (PNA), hyaline membrane disease (HMD), and sepsis. Also, few neonatal deaths were reportedly caused by meconium aspiration syndrome (MAS) and bilirubin encephalopathy (Figure 3).
 |
Figure 3 Causes of death among neonates admitted in the NICU of UoGCSH, northwest Ethiopia. |
Factors Associated with the Death of Neonates
In the unadjusted analysis, ten variables showed statistically significant association with the risk of neonatal death at a p-value ≤ 0.2 in birth-to-death and admission-to-death analysis.
However, after controlling confounders in multivariable analysis, not crying at birth (AHR: 3.52 [95% CI: 2.19, 5.64]) and earlier gestational age of the newborn at birth (AHR: 3.25 [95% CI: 1.76, 6.01]), remained statistically significant predictors of birth to death of the neonates at a p-value ≤ 0.05.
Similarly, the risk of death among neonates in the admission to death analysis found to be higher among neonates born at home (AHR: 2.43 [95% CI: 1.03, 5.73])), not crying at birth (AHR: 3.07 [95% CI: 1.92, 4.91]) and born at earlier gestational age (AHR: 2.91 [95% CI: 1.58, 5.35]) at a p-value ≤ 0.05 (Table 4).
 |
Table 4 Gompertz Proportional Hazard Regression of Predictors of Neonatal Mortality Among Neonates Admitted to the NICU of UGSRH, Northwest Ethiopia. N=504 |
Discussion
This study demonstrated a higher NMR (173 per 1000 live births) among neonates admitted in the NICU. This finding is higher than studies conducted in the Tigray region,8 Jimma7 Ethiopia, and EDHS report of the Amhara region and the national findings.6 The study from the Tigray region was focused on the neonates born in the hospital irrespective of their admission to NICU status, whereas the EDHS and Jimma studies were conducted at a community level and in both of the cases, the denominator covers many healthy neonates and thereby the resulting rates could be lower. The higher mortality in our study could be explained by the fact that neonates admitted to the NICU are usually those who need intensive care and held a higher risk of death.
Similar studies conducted in the hospitals showed a higher neonatal mortality rate that ranged from 157 in Cameroon14 and 194 in Nigeria15 to more than 200 in Nepal,16 Mexico17 and Addis Ababa.10 This implies that intensive care unit cohorts have a higher risk of death than general neonates.
In this study, 14% of all neonatal deaths occurred within the first 24 hrs, and the first 3 days hold more than half (60%) of all deaths. Also, a higher proportion (84%) of deaths occurred in the first week of birth. Studies conducted at Addis Ababa10 and Tigray region of Ethiopia8 reported comparable results in the first 24 hrs of life. Conversely, higher mortality in the first 3 days that was reported in the Addis Ababa study might be attributed to the census nature of the study.
In this study, neonates born at home had more than a two-fold risk of death than health institution deliveries in the admission-to-death analysis. A study done in Nigeria18 identified that the risk of death is reduced by 31% among neonates born at health facilities showing that the reverse could have a positive risk of death. Other findings also support a positive association of place of delivery with neonatal mortality.19,20 This might be explained by the fact that home deliveries are assisted by unskilled attendants in unsafe and unequipped settings and hold higher risks of mortality of neonates and mothers.21
In this study, neonates who did not cry at birth had about three times more risk of death compared with those who cried at birth. Healthy neonates are expected to cry at birth and crying vigorously is a measure of breathing effort22 and used as a key component of Apgar score and thereby, to clinically recognize birth asphyxia.23 Neonates who did not cry vigorously at birth could have irregular or slow birthing and cumulative Apgar could be lower. Findings from different studies that also show a higher risk of death among neonates with lower Apgar scores could support this explanation.24–26
The risk of death among neonates delivered before 34 weeks of gestation in this study was found to be nearly three times higher than those born after 37 complete weeks. A study identified that the risk of death was positively associated with hyaline membrane disease cases among premature neonates.27 It is scientifically proven that due to a lack of surfactant production in the fetal lung, hyaline membrane disease occurs among newborns delivered before a gestational age of 34 weeks and eventually will cause respiratory collapse and death.28
Although actionable predictors were identified, this study was generated from a single hospital with a retrospective design. Consequently, its rigorousness and representativeness might be limited. Thus, it is imperative that a large-scale prospective study that further addresses the quality of obstetric and neonatal services be conducted.
Conclusion
Neonatal mortality in this study was higher than the national and sustainable development goal targets. Perinatal asphyxia, hyaline membrane disease, and sepsis were identified as the three leading causes of death. The hazard of death found to be higher among neonates who were born at home in admission-to-death analysis. In both admission-to-death and birth-to-death, the hazard of death was higher among neonates born before 34 weeks and those who did not cry at birth. It is important to reinforce giving birth at health facilities by improving access to the service and raising community awareness. Also, to give appropriate care before referral for preterm neonates younger than 34 weeks and neonates who did not cry at birth, it is essential to strengthen the service provision capacity of the health institutions and health service providers.
Abbreviations
ANC, antenatal care; EDHS, Ethiopian Demographic and Health Survey; NICU, neonatal intensive care unit; PH, proportional hazard; UoGCSH, University of Gondar Comprehensive Specialized Hospital.
Ethical Approval and Informed Consent
Ethical clearance was obtained from the Ethical Committee of the Institute of Public Health at the University of Gondar and informed consent was waived as secondary data have been extracted from the chart. Officials from the University of Gondar Comprehensive Specialized Hospital were communicated through a formal letter that was taken from the institute and permission for data collection was granted from the hospital. Confidentiality of information was maintained through not extracting personal identifiers and keeping data in a password secured computer.
Data Sharing Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgments
We would like to express our gratitude to data extractors for their commitment to completing the checklist and identifying incomplete charts for replacement. We would also like to extend our appreciation to all staff of the record office for their commitment to searching the selected charts besides their routine duties.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
The authors declare that they have no competing interests in this work.
References
1. Konar H. DC Dutta’s Textbook of Obstetrics. The Term New Born Infant. New Delhi, London, Philadelphia, Panama: JP Medical Ltd; 2014:447.
2. Lawn JE, Davidge R, Paul VK, et al. Born too soon: care for the preterm baby. Reprod Health. 2013;10(1):S5. doi:10.1186/1742-4755-10-S1-S5
3. Liu L, Oza S, Hogan D, et al. Global, regional, and national causes of child mortality in 2000–13, with projections to inform post-2015 priorities: an updated systematic analysis. Lancet. 2015;385(9966):430–440. doi:10.1016/S0140-6736(14)61698-6
4. Kassebaum NJ, Bertozzi-Villa A, Coggeshall MS, et al. Global, regional, and national levels and causes of maternal mortality during 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2014;384(9947):980–1004. doi:10.1016/S0140-6736(14)60696-6
5. Oza S, Cousens SN, Lawn JE. Estimation of daily risk of neonatal death, including the day of birth, in 186 countries in 2013: a vital-registration and modelling-based study. Lancet Glob Health. 2014;2(11):e635–e644. doi:10.1016/S2214-109X(14)70309-2
6. Central Statistical Agency (CSA) [Ethiopia] and ICF. Ethiopia Demographic and Health Survey 2016. Addis Ababa, Ethiopia, and Rockville, Maryland, USA: CSA and ICF; 2016.
7. Debelew GT, Afework MF, Yalew AW. Determinants and causes of neonatal mortality in Jimma zone, southwest Ethiopia: a multilevel analysis of prospective follow up study. PLoS ONE. 2014;9(9):e107184. doi:10.1371/journal.pone.0107184
8. Mengesha HG, Wuneh AD, Lerebo WT, Tekle TH. Survival of neonates and predictors of their mortality in Tigray region, Northern Ethiopia: prospective cohort study. BMC Pregnancy Childbirth. 2016;16(1):202. doi:10.1186/s12884-016-0994-9
9. Andargie G, Berhane Y, Worku A, Kebede Y. Predictors of perinatal mortality in rural population of Northwest Ethiopia: a prospective longitudinal study. BMC Public Health. 2013;13(1):168. doi:10.1186/1471-2458-13-168
10. Tekleab AM, Amaru GM, Tefera YA. Reasons for admission and neonatal outcome in the neonatal care unit of a tertiary care hospital in Addis Ababa: a prospective study. Res Rep Neonatol. 2016;6:17–23. doi:10.2147/RRN
11. O’Hare B, Makuta I, Chiwaula L, Bar-Zeev N. Income and child mortality in developing countries: a systematic review and meta-analysis. J R Soc Med. 2013;106(10):408–414. doi:10.1177/0141076813489680
12. Ki-Moon B The Millennium Development Goals Report 2015. United Nations Pubns. 2015.
13. National Planning Commission and the United Nations in Ethiopia. Millennium Development Goals Report 2014 Ethiopia: Assessment of Ethiopia’s Progress Towards the MDGs. United Nations Pubns. Addis Ababa, Ethiopia;2015:40–47
14. Ndombo PK, Ekei QM, Tochie JN, et al. A cohort analysis of neonatal hospital mortality rate and predictors of neonatal mortality in a sub-urban hospital of Cameroon. Ital J Pediatr. 2017;43(1):52. doi:10.1186/s13052-017-0369-5
15. Toma BO, Ige OO, Abok II, Onwuanaku C, Abah RO, Donli A. Pattern of neonatal admissions and outcome in a tertiary institution in north central Nigeria. J Med Tropics. 2013;15(2):121. doi:10.4103/2276-7096.123590
16. Shah GS, Yadav S, Thapa A, Shah L. Clinical profile and outcome of neonates admitted to Neonatal Intensive Care Unit (NICU) at a tertiary care centre in Eastern Nepal. J Nepal Paediatr Soc. 2013;33(3):177–181. doi:10.3126/jnps.v33i3.8447
17. Lona Reyes JC, Perez Ramirez RO, Llamas Ramos L, Gomez Ruiz LM, Benitez Vazquez EA, Rodriguez Patino V. Neonatal mortality and associated factors in newborn infants admitted to a neonatal care unit. Arch Argent Pediatr. 2018;116(1):42–48. doi:10.5546/aap.2018.eng.42
18. Akinyemi JO, Bamgboye EA, Ayeni O. Trends in neonatal mortality in Nigeria and effects of bio-demographic and maternal characteristics. BMC Pediatr. 2015;15(1):36. doi:10.1186/s12887-015-0349-0
19. Adedini SA, Odimegwu C, Imasiku EN, Ononokpono DN, Ibisomi L. Regional variations in infant and child mortality in Nigeria: a multilevel analysis. J Biosoc Sci. 2015;47(2):165–187. doi:10.1017/S0021932013000734
20. Kolola T, Ekubay M, Tesfa E, Morka W. Determinants of neonatal mortality in North Shoa Zone, Amhara Regional State, Ethiopia. PLoS ONE. 2016;11(10):e0164472. doi:10.1371/journal.pone.0164472
21. Berhan Y, Berhan A. Skilled health personnel attended delivery as a proxy indicator for maternal and perinatal mortality: a systematic review. Ethiop J Health Sci. 2014;24:69–80. doi:10.4314/ejhs.v24i0.7S
22. Simon LV, Hashmi MF, Bragg BN. Apgar Score. 2019.
23. Cnattingius S, Norman M, Granath F, Petersson G, Stephansson O, Frisell T. Apgar score components at 5 minutes: risks and prediction of neonatal mortality. Paediatr Perinat Epidemiol. 2017;31(4):328–337. doi:10.1111/ppe.2017.31.issue-4
24. de Castro ECM, Leite ÁJM, de Almeida MFB, Guinsburg R. Perinatal factors associated with early neonatal deaths in very low birth weight preterm infants in Northeast Brazil. BMC Pediatr. 2014;14(1):312. doi:10.1186/s12887-014-0312-5
25. Dzimah DA. Modelling the Risk Factors of Neonatal Mortality Using Survival Analysis. University of Ghana; 2016.
26. Ghorbani F, Heidarzadeh M, Dastgiri S, Ghazi M, Rahkar Farshi M. Survival of premature and low birth weight infants: a multicenter, prospective, cohort study in Iran. Iran J Neonatol. 2017;8(1):16–22.
27. Wesenu M, Kulkarni S, Tilahun T. Modeling determinants of time-to-death in premature infants admitted to neonatal intensive care unit in Jimma University specialized hospital. Ann Data Sci. 2017;4(3):361–381. doi:10.1007/s40745-017-0107-2
28. Purandare C. Fetal lung maturity. J Obstet Gynecol India. 2005;55(3):215–217.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

