Back to Journals » Research and Reports in Urology » Volume 8
The use of urologic investigations among patients with traumatic spinal cord injuries
Authors Welk B , Liu K, Shariff S
Received 4 November 2015
Accepted for publication 18 December 2015
Published 22 February 2016 Volume 2016:8 Pages 27—34
DOI https://doi.org/10.2147/RRU.S99840
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Jan Colli
Blayne Welk,1,2 Kuan Liu,2 Salimah Z Shariff,2
1Department of Surgery and Epidemiology and Biostatistics, Western University, ON, Canada; 2Institute for Clinical Evaluative Sciences – Western (ICES Western), London, ON, Canada
Objective: To assess the use of urologic investigations among traumatic spinal cord injury (TSCI) patients.
Methods: This is a retrospective cohort study from Ontario, Canada. We included all adult TSCI patients injured between 2002 and 2012. The primary outcome was the frequency of urodynamic testing, renal imaging, and cystoscopy. Primary exposure was the year of injury. The impact of age, sex, comorbidity, socioeconomic status, and lesion level was assessed with Cox regression models.
Results: One thousand five hundred and fifty one incident TSCI patients were discharged from a rehabilitation hospital. The median follow-up time of this cohort was 5.0 years (interquartile range =2.9–7.5). At least one urodynamics, renal imaging, or cystoscopy was performed during follow-up for 50%, 80%, and 48% of the cohort, respectively. The overall rate of these tests was 0.22, 0.60, and 0.22 per person-year of follow-up. The proportion of patients who had regular, yearly urodynamics (<2%), renal imaging (6%), or cystoscopy (<2%) was low. There were no significant linear trends in the use of these tests over the 10-year study period. Urodynamics were significantly less likely to be performed in patients over 65 years of age (hazard ratio [HR] =0.63, P<0.01) and those with a higher level of comorbidity (HR =0.72, P<0.01). Patients with quadriplegia were significantly less likely to receive any of the investigations compared to those with paraplegia.
Conclusion: Renal imaging is done at least once for the majority of patients with TSCI; however, only half undergo urodynamics or cystoscopy. Few patients have regular urologic testing. The reality of urologic testing after TSCI is very different from urologist's ideals and practice guidelines.
Keywords: spinal cord injuries, urology, observational study, urodynamics, cystoscopy, renal imaging
Introduction
Traumatic spinal cord injury (TSCI) patients often have significant bladder dysfunction. This dysfunction can lead to urologic complications such as renal failure, autonomic dysreflexia, urinary stone disease, incontinence, urinary infection, and bladder cancer.1
As part of the urologic evaluation of these patients, several investigations are often used to assess for potential risk factors related to the asymptomatic development of the aforementioned complications. A recent systematic review evaluated the evidence for the commonly used screening urologic tests of urodynamics, renal imaging, and cystoscopy.2 Using the available evidence, the authors concluded that routine renal ultrasound was a beneficial screening test and that urodynamics was likely beneficial; there was insufficient evidence to evaluate routine cystoscopy. The optimum frequency of these investigations could not be determined. Despite the limited evidence in the literature, guidelines suggest specific screening regimens for patients with TSCI. For example, the European Association of Urology guidelines on the management of neurogenic bladder recommend twice yearly renal ultrasound, and urodynamic investigation every 1–2 years.3 Guidelines based on expert opinion suggest yearly renal imaging with a baseline urodynamics.4 Other guidelines, such as the Paralyzed Veterans of America bladder management guideline, offer only general directions such as a yearly urologic evaluation, which may include renal imaging, urodynamics, and cystoscopy.5 Surveys of urologists demonstrate that there is variability in their perception of which tests are necessary and how often they are required.6,7
The real-world usage of urologic investigations has not been well studied on a population level. The objective of this study was to characterize the pattern of urodynamics, renal imaging, and cystoscopy among TSCI patients and to assess whether it has changed significantly over time. Our secondary objective was to evaluate the impact of patient characteristics on the likelihood of urologic investigation.
Methods
Study design
This is a retrospective cohort study using administrative data from the province of Ontario, Canada (population of approximately 13 million, all of whom use a single, publicly funded, universal health-care system). The use of administrative data did not require individual patient consent. Institutional ethics approval for this study was provided by Sunnybrook Hospital, Toronto.
Data sources and validity
Three primary data sources were used for this study. The National Rehabilitation System (NRS) submits data on patients undergoing rehabilitation in Ontario. The Canadian Institutes for Health Information Discharge Abstract Database (CIHI-DAD) records all inpatient encounters for all hospitals in Ontario. Finally, the Ontario Health Insurance Plan (OHIP) database records all fee claims for physicians in the province. Data quality, completeness, and validity are high for all of these sources, and have been previously reported.8
Population, exposure, and outcome definitions
Our patient cohort of TSCI patients was identified using a valid administrative data algorithm based on NRS data (sensitivity =92%, specificity =97%).8 We included all adults with a minimum of 1 year of follow-up after admission to a rehabilitation facility in Ontario for an incident TSCI. We included patients admitted between April 1, 2002 and March 31, 2012. The details of this cohort are outlined in our previous work.8,9
Our primary outcome consisted of the first occurrence of one of three specific procedures: urodynamics (either regular cystometrography or videourodynamics with or without pressure flow studies), renal imaging (either renal/abdominal ultrasound or abdominal CT scan), and cystoscopy. Each of these tests have specific fee codes that are submitted by physicians for compensation for performing or interpreting the relevant procedures (Table S1). As with other physician fee service payments, these codes are expected to have a high sensitivity and positive predictive value.10 Patients were considered to be under observation for these outcomes from the date of their TSCI and were censored at the time of their death, emigration, or on March 31, 2013. Our primary exposure was the year of TSCI. For our secondary analysis, the following covariates were included: age, sex, comorbidity index (using the sum of the Aggregated Diagnostic Groups from the validated Johns Hopkins University Adjusted Case Groups® case-mix system, dichotomized into 0–11 [low comorbidity] versus ≥12 [high comorbidity]11), and socioeconomic status (using the Ontario Marginalization Index scores of residential instability, material deprivation, and ethnic concentration as a proxy for individual marginalization);12 quintiles were ranked from 1 (least marginalization) to 5 (most marginalization). Level of impairment was determined from coding within the NRS, and patients were classified as either quadriplegic, paraplegic, or unknown.
Statistical analysis
Continuous variables are reported as medians with interquartile ranges (IQRs). Categorical variables are reported as percentages (proportion). For our primary objective, the Cochran–Armitage test for linear trend was used. For our secondary analysis, a Cox proportional hazards model was used to assess the impact of primary exposure and covariates on our three primary outcomes. Hazard ratios (HRs), 95% confidence intervals (CIs), and P-values are reported. SAS 9.3 (SAS Institute, Cary, NC, USA) was used.
Results
We identified a cohort of 1,551 TSCI patients. The patients were generally young to middle-aged males (median age =48, IQR =33–63), with a low level of comorbidity prior to their TSCI (Table 1). A median of 161 patients (IQR =140–169) were identified within each fiscal year from 2002 to 2012. This cohort had a median follow-up of 5.0 years (IQR =2.9–7.5). During the study period, 11% (167/1,551) of patients were excluded due to death, and a further 8% (118/1,551) were excluded due to emigration.
Urodynamics were performed at least once on 50% (775/1,551) of TSCI patients after their injury. Only 8% (59/775) of these initial tests were videourodynamic studies. Urodynamic studies were almost always performed by a urologist (89% of cases, 687/775); the remainder were performed by physiatrists or gynecologists. Among the 778 (50%) TSCI patients who had at least 5 years of follow-up, 64% (498/778) had at least one urodynamic study during their first 5 years postinjury. For those who underwent a urodynamic study, the median time to the first study was 0.26 (IQR =0.16–0.61) years and, a median of 2 (IQR =1–3) studies were performed.
Renal imaging was performed at least once on 80% (1,237/1,551) of TSCI patients after their injury. Among the possible imaging modalities, ultrasound accounted for 69% (857/1,237) of the imaging tests, contrast-enhanced abdominal CT (computed tomography) 22% (276/1,237), noncontrast abdominal CT 6% (76/1,237), and a combined contrast- and noncontrast-enhanced CT 2% (27/1,237). Among the 778 TSCI patients with at least 5 years of follow-up, 97% (755/778) had at least one renal imaging test during their first 5 years postinjury. For those who underwent renal imaging, the median time to the first study was 0.21 (0.06–0.91) years, and a median of 3 (IQR =2–5) studies were done. For 20% (249/1,237) of these patients, the imaging test was done without any evidence of urologic consultation during the follow-up period.
Cystoscopy was performed at least once on 48% (751/1,551) of TSCI patients. Among the 778 TSCI patients with at least 5 years of follow-up, 63% (490/778) had at least one cystoscopy during the 5-year period postinjury. For those who underwent cystoscopy, the median time to the first study was 0.38 (0.22–1.29) years, and a median of 2 (IQR =1–3) procedures were repeated among these patients.
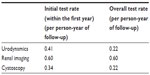
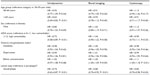
The cumulative frequency of urodynamics, renal imaging, and cystoscopy after 1–5 years of follow-up is shown in Table 2, and the median time between successive tests is shown in Table 3. The 1-year and overall investigation rate per person-year of follow-up for the patient cohort is shown in Table 4.
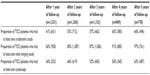
The proportion of TSCI patients that would fit into one of two routine investigative patterns (at least once every year, or at least once every 2 years) was determined for each urologic investigation. This pattern of usage of these urologic investigations is shown in Figure 1. After 5 years of follow-up, consistent yearly urodynamics (<2%), renal imaging (6%), or cystoscopy (<2%) was carried out in very few patients. Similarly, with a less stringent pattern of investigation of at least once every 2 years, consistent use of urodynamics (10%), renal imaging (30%), and cystoscopy (12%) was rarely observed after 6 years of follow-up.
The primary exposure of year of injury was assessed to see if there was a significant change in the usage of any of these investigations. Urodynamics (P=0.18), renal imaging (P=0.47), and cystoscopy (P=0.59) did not demonstrate a significant linear trend in their usage within the first year after TSCI. Our secondary analysis evaluated the effect of age, sex, comorbidity, socioeconomic status, and lesion level on the likelihood of receiving one of these investigations (Table 5). Urodynamics were significantly less likely to be performed in patients over 65 years of age (HR =0.63, 95% CI =0.49–0.82) and those with a higher level of comorbidity (HR =0.72 [95% CI =0.58–0.89]). Patients with quadriplegia were significantly less likely to receive any of the investigations compared to those with paraplegia.
Discussion
The urinary morbidity associated with a TSCI is significant. Overall, these patients require three times as many health-care resources as age-matched cohorts13,14 and are often rehospitalized after their injury.15 Despite significant improvements in genitourinary-specific mortality rates, many long-term urologic health complications have not decreased significantly.16 It is important to try and prevent these urologic complications not only because of their direct morbidity but also because they lead to increased rehospitalization17 and a significantly higher rate of physician usage.14
To try and optimize urologic health for TSCI, urologists are often involved in the long-term management of these patients.9 In addition to a clinical assessment, patients often will have urodynamics, renal imaging, and cystoscopy tests performed.
Urodynamics are considered an essential part of understanding the bladder function of a TSCI patient,3 and we demonstrated that the majority of these tests are done by urologists. Certain urodynamic features, such as detrusor sphincter dyssynergia, poor compliance, or high-pressure detrusor overactivity, are usually treated to minimize renal risk.18 Previous studies have shown that urodynamics are valuable: almost half of TSCI patients require a management change based on the test results in order to prevent renal deterioration, worsening bladder symptoms, or increased autonomic dysreflexia.19 Once the initial urodynamic studies are complete, follow-up testing is necessary as parameters change within TSCI patients over time.20
Despite the obvious importance of urodynamics, we found that only about two-thirds of TSCI patients in Ontario actually received them in the first 5 years after their injury. Further, if we consider a fairly liberal definition of urodynamic follow-up (once every 2 years), only 10% of TSCI patients meet this definition. This is a significant variation from established practice guidelines suggesting urodynamics at least once every 2 years3 and surveys of urology management practices that suggest that 65%–80% of urologists believe that SCI patients should get yearly urodynamics.6,7 Among the population of patients who do receive repeat urodynamics, the time interval between successive studies seems to be approximately yearly. Patients with quadriplegia, those over 65 years of age, and those with increased comorbidities were significantly less likely to receive urodynamics, probably as a result of chronic indwelling catheter usage in this population.
Routine renal imaging for TSCI patients is suggested in order to assess for the asymptomatic development of hydronephrosis, renal stone disease, or renal scarring.21 A previous case series has suggested that these complications can occur in up to a third of patients over time.22 This screening test does not require the expertise or involvement of a urologist, and instead may be ordered by any physician; however, only 20% are occurring in patients with no urologic visits during the study period. We demonstrated in this study that while the vast majority (97%) of patients do have at least one imaging test within the first 5 years, once again a minority actually have repeat studies performed at either 1- (6%) or 2 (30%)-year intervals. Furthermore, a large proportion of these imaging tests are carried out in the immediate post-TSCI period and are not repeated in the future. These early imaging tests are likely related to the initial trauma or hospital stay and have little bearing on the long-term urologic health of TSCI patients. Surveys of urologists again consistently suggest that these tests should be performed annually,6,7 similar to the previous referenced guidelines. Given the minimal morbidity associated with renal ultrasound, and its apparent benefit,2 this low utilization was unexpected.
In TSCI patients, cystoscopy is used to assess for bladder calculi, urethral pathology, and bladder cancer. It has been advocated as a screening tool for SCI patients due to an increased risk of bladder cancer; however, there is no evidence that cystoscopy fulfills the requirements of a screening test, and most patients develop bladder cancer many years after their initial injury.23 There is no high-quality evidence to suggest that routine cystoscopy in an asymptomatic neurogenic bladder patient is warranted. Given the practice patterns observed in this study, cystoscopy is likely being used to evaluate specific symptoms.
The results of this study demonstrate that in a real-world setting, most TSCI patients are being managed with a more conservative approach to urologic screening, and many patients are not necessarily receiving even an initial assessment. This may be due to multiple factors, including patient preference and compliance, limited access to physicians/investigations, and a more individualized approach to TSCI patient assessment and follow-up. While some authors have reported a poor concordance between symptoms and urodynamic abnormalities,24 others have successfully managed patients using clinical assessments as a trigger for further investigation. Edokpolo and Foster25 reviewed 48 TSCI patients who had initial urodynamics to define bladder function, and then yearly renal ultrasound to follow for hydronephrosis. They found no significant preventable complications during a mean follow-up of 7 years. Vaidyanathan et al26 demonstrated that ultrasound abnormalities were primarily related to symptoms and that ultrasound findings among asymptomatic patients did not result in any management changes. Other studies have also demonstrated a poor adherence among other populations to urologic screening: among 7,162 patients with SCI in the USA, only 25% of patients received a urologic clinical assessment, serum creatinine analysis, and renal imaging study over a 2-year period; cystoscopy and urodynamics were only performed among 12% and 7%, respectively.27
It is important to note the limitations of this study: we used administrative data, which provide a large and comprehensive patient sample, but have limited clinical details (such as the lesion level or method of bladder management). We are unable to determine the reason the urologic investigations were performed: some may have been for screening purposes, and some for symptoms or signs. In addition, it is likely that some physicians involved in the care of TSCI patients do perform an assessment of urologic health, and that these clinical assessments play a role in determining if a patient goes on having renal imaging or urodynamics. The safety and cost-effectiveness of this approach, however, are largely untested. Finally, we are not able to determine if obtaining these urologic investigations actually improves care and outcomes for these patients.
Conclusion
We found that at least one urodynamics or renal imaging test was performed after TSCI for 50% and 80% of patients, respectively. This is significantly different from urologists’ perceptions and guideline recommendations. This has not changed significantly over the last decade. The “real-world” management of these patients needs to be taken into account during the development of future guidelines in this area. Future research should include the prospective study of patients undergoing different follow-up regimens and the ability of urodynamic and renal imaging frequency to act as quality-of-care indicators.
Acknowledgment
Funding provided by the Ontario Neurotrauma Foundation, in partnership with the Rick Hansen Institute. This study was supported by the Institute for Clinical Evaluative Sciences (ICES), which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC). The study was conducted through ICES Western, which is funded by an operating grant from the Academic Medical Organization of Southwestern Ontario (AMOSO). The opinions, results, and conclusions reported in this paper are those of the authors and are independent from the funding sources. No endorsement by ICES, the Ontario MOHLTC, or AMOSO is intended or should be inferred.
Disclosure
The authors report no conflicts of interest in this work.
References
Weld KJ, Dmochowski RR. Effect of bladder management on urological complications in spinal cord injured patients. J Urol. 2000;163(3):768–772. | |
Cameron AP, Rodriguez GM, Schomer KG. Systematic review of urological followup after spinal cord injury. J Urol. 2012;187(2):391–397. | |
Stöhrer M, Blok B, Castro-Diaz D, et al. EAU guidelines on neurogenic lower urinary tract dysfunction. Eur Urol. 2009;56(1):81–88. | |
Abrams P, Agarwal M, Drake M, et al. A proposed guideline for the urological management of patients with spinal cord injury. BJU Int. 2008;101(8):989–994. | |
Consortium for Spinal Cord Medicine. Bladder management for adults with spinal cord injury: a clinical practice guideline for health-care providers. J Spinal Cord Med. 2006;29(5):527–573. | |
Razdan S, Leboeuf L, Meinbach DS, Weinstein D, Gousse AE. Current practice patterns in the urologic surveillance and management of patients with spinal cord injury. Urology. 2003;61(5):893–896. | |
Blok BF, Karsenty G, Corcos J. Urological surveillance and management of patients with neurogenic bladder: results of a survey among practicing urologists in Canada. Can J Urol. 2006;13(5):3239–3243. | |
Welk B, Loh E, Shariff SZ, Liu K, Siddiqi F. An administrative data algorithm to identify traumatic spinal cord injured patients: a validation study. Spinal Cord. 2014;52(1):34–38. | |
Welk B, Tran KC, Liu K, Shariff S. The pattern of urologic care among traumatic spinal cord injured patients. Can Urol Assoc J. 2014;8(11–12):E805–E809. | |
Raina P, Torrance-Rynard V, Wong M, Woodward C. Agreement between self-reported and routinely collected health-care utilization data among seniors. Health Serv Res. 2002;37(3):751–774. | |
The Johns Hopkins ACG® Case-Mix System Reference Manual, Version 7. Baltimore, MD: Health Services Research and Development Center; 2007. | |
Matheson FI, Dunn JR, Smith KL, Moineddin R, Glazier RH. Development of the Canadian Marginalization Index: a new tool for the study of inequality. Can J Public Health. 2012;103(8 Suppl 2):S12–S16. | |
Guilcher SJ, Munce SE, Couris CM, et al. Health care utilization in non-traumatic and traumatic spinal cord injury: a population-based study. Spinal Cord. 2010;48(1):45–50. | |
Dryden DM, Saunders LD, Rowe BH, et al. Utilization of health services following spinal cord injury: a 6-year follow-up study. Spinal Cord. 2004;42(9):513–525. | |
Jaglal SB, Munce SE, Guilcher SJ, et al. Health system factors associated with rehospitalizations after traumatic spinal cord injury: a population-based study. Spinal Cord. 2009;47(8):604–609. | |
McKinley WO, Jackson AB, Cardenas DD, DeVivo MJ. Long-term medical complications after traumatic spinal cord injury: a regional model systems analysis. Arch Phys Med Rehabil. 1999;80(11):1402–1410. | |
Jeong SJ, Cho SY, Oh SJ. Spinal cord/brain injury and the neurogenic bladder. Urol Clin North Am. 2010;37(4):537–546. | |
Danforth TL, Ginsberg DA. Neurogenic lower urinary tract dysfunction: how, when, and with which patients do we use urodynamics? Urol Clin North Am. 2014;41(3):445–452. | |
Linsenmeyer TA, Linsenmeyer MA. Impact of annual urodynamic evaluations on guiding bladder management in individuals with spinal cord injuries. J Spinal Cord Med. 2013;36(5):420–426. | |
Shin JC, Lee Y, Yang H, Kim DH. Clinical significance of urodynamic study parameters in maintenance of renal function in spinal cord injury patients. Ann Rehabil Med. 2014;38(3):353–359. | |
Bodley R. Imaging in chronic spinal cord injury – indications and benefits. Eur J Radiol. 2002;42(2):135–153. | |
Ku JH, Choi WJ, Lee KY, et al. Complications of the upper urinary tract in patients with spinal cord injury: a long-term follow-up study. Urol Res. 2005;33(6):435–439. | |
Welk B, McIntyre A, Teasell R, Potter P, Loh E. Bladder cancer in individuals with spinal cord injuries. Spinal Cord. 2013;51(7):516–521. | |
Nosseir M, Hinkel A, Pannek J. Clinical usefulness of urodynamic assessment for maintenance of bladder function in patients with spinal cord injury. Neurourol Urodyn. 2007;26(2):228–233. | |
Edokpolo LU, Foster HE. Renal tract ultrasonography for routine surveillance in spinal cord injury patients. Top Spinal Cord Inj Rehabil. 2013;19(1):54–60. | |
Vaidyanathan S, Hughes PL, Soni BM. A comparative study of ultrasound examination of urinary tract performed on spinal cord injury patients with no urinary symptoms and spinal cord injury patients with symptoms related to urinary tract: do findings of ultrasound examination lead to changes in clinical management? Scientific World Journal. 2006;6:2450–2459. | |
Cameron AP, Lai J, Saigal CS, Clemens JQ, NIDDK Urological Diseases in America Project. Urological surveillance and medical complications after spinal cord injury in the United States. Urology. 2015;86(3):506–510. |
Supplementary material
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.