Back to Journals » OncoTargets and Therapy » Volume 13
The Roles of Integrin α5β1 in Human Cancer
Authors Hou J, Yan D, Liu Y, Huang P, Cui H
Received 24 July 2020
Accepted for publication 21 October 2020
Published 31 December 2020 Volume 2020:13 Pages 13329—13344
DOI https://doi.org/10.2147/OTT.S273803
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Yong Teng
Jianbing Hou,1– 3 Du Yan,4 Yudong Liu,1– 3 Pan Huang,1– 3 Hongjuan Cui1– 3
1State Key Laboratory of Silkworm Genome Biology, Key Laboratory for Sericulture Biology and Genetic Breeding, Ministry of Agriculture and Rural Affairs, Southwest University, Chongqing 400716, People’s Republic of China; 2Cancer Center, Medical Research Institute, Southwest University, Chongqing 400716, People’s Republic of China; 3Chongqing Engineering and Technology Research Centre for Silk Biomaterials and Regenerative Medicine, Chongqing 400716, People’s Republic of China; 4Chongqing University Central Hospital, Chongqing Emergency Medical Center, Chongqing 400716, People’s Republic of China
Correspondence: Hongjuan Cui
State Key Laboratory of Silkworm Genome Biology, Key Laboratory for Sericulture Biology and Genetic Breeding, Ministry of Agriculture and Rural Affairs, Southwest University, Chongqing 400716, People’s Republic of China
Tel +86-23-68251713
Fax +86-23-68251128
Email [email protected]
Abstract: Cell adhesion to the extracellular matrix has important roles in tissue integrity and human health. Integrins are heterodimeric cell surface receptors that are composed by two non-covalently linked alpha and beta subunits that mainly participate in the interaction of cell-cell adhesion and cell-extracellular matrix and regulate cell motility, adhesion, differentiation, migration, proliferation, etc. In mammals, there have been eighteen α subunits and 8 β subunits and so far 24 distinct types of αβ integrin heterodimers have been identified in humans. Integrin α 5β 1, also known as the fibronectin receptor, is a heterodimer with α 5 and β 1 subunits and has emerged as an essential mediator in many human carcinomas. Integrin α 5β 1 alteration is closely linked to the progression of several types of human cancers, including cell proliferation, angiogenesis, tumor metastasis, and cancerogenesis. In this review, we will introduce the functions of integrin α 5β 1 in cancer progression and also explore its regulatory mechanisms. Additionally, the potential clinical applications as a target for cancer imaging and therapy are discussed. Collectively, the information reviewed here may increase the understanding of integrin α 5β 1 as a potential therapeutic target for cancer.
Keywords: integrin α 5β 1, prognostic indicator, tumorigenesis, molecular target
Introduction
Integrins, the family of heterodimeric cell surface receptors that are expressed in most cells including pericytes, endothelial cells, fibroblasts, and tumor cells, which have emerged as important regulators for providing both mechanical engagement of cell to extracellular matrix, and generation of signals that are implicated in various diseases such as autoimmune diseases, deleterious embryonic development, cardiovascular diseases and cancer malignancies.1,2 In mammals, eighteen α subunits and 8 β subunits form at least 24 distinct types of αβ integrin heterodimers, which play as true receptors of tissue and organ-specific ligands.3 Both α and β subunits possess a large extracellular domain, a small cytoplasmic tail, and a transmembrane domain.4 The extracellular domains act as the cells sense and respond to the microenvironment cues such as adhesion proteins and growth factors.3 The cytoplasmic tail is linked to the actin cytoskeleton and intracellular signaling pathways such as Src family kinase, focal adhesion kinase (FAK), and mitogen-activated protein kinase (MAPK), as well as protein kinase B (AKT).5 Notably, integrins have been received attention as important regulators in mediating the hallmarks that characterize human cancers, including cell proliferation, metastasis, immune evasion, tumor angiogenesis, and resistance to chemotherapy and radiotherapy.
Integrin α5β1was firstly reported in the 1992s and was the only known α5 integrin.6 Upon binding to the ligand, the cytoplasmic tails of integrin α5β1 bind to cytoskeleton and then drive reorganization of the cytoskeleton through the intracellular signaling pathway, that is, the “outside-in” signaling pathway.7,8 Integrin α5β1-mediated intracellular signals can in turn activate extracellular regions and assist ECM assembly, that is, the “inside-out” signaling pathway.9,10 This two-way signaling pathway contributes to various biological activities, such as cell adhesion, migration, and survival.11 And these behaviors can be mediated by arginine-glycine-aspartate (RGD) peptides, specific antibodies, as well as the surface glycosylation.12 Integrin α5β1 has been defined as a proangiogenic factor involve in regulating tumor angiogenesis by interacting with the Vascular Endothelial Growth Factor Receptor (VEGFR) and angiopoietin-Tie systems.13 Moreover, the important roles in tumorigenesis, tumor metastasis, and resistance to chemotherapy and radiotherapy have been highlighted for integrin α5β1.14 In this review, we focus on the recent findings and important progress to summarize the roles and related mechanisms of integrin α5β1, and discuss the potential strategies targeting integrin α5β1 for improving cancer patient’s outcomes.
Structure, Regulation, Ligands and Functions of Integrin α5β1
Structural Domains of Integrin α5β1
Integrin α5β1, as a member of the integrin family, is a heterodimer composed by two subunits, α5 and β1, and both are necessary for complete biological functions.15 The human integrin alpha 5 gene (ITGA5) encodes the α5 subunit and is localized at 12q11. The extracellular domain of α5 subunit has a thigh domain and a β-propeller domain, which is responsible for the recognition of the RGD motifs on the fibronectin and fibrinogen.16 The integrin beta 1 gene (ITGB1) has been proved to reside in chromosome 10p11.2, and the extracellular part of this subunit is made up of a plexin/semaphorin/integrin (PSI) domain, a hybrid domain, a βI domain (with a metal ion-dependent adhesion site [MIDAS] structure), and four EGF like domains.17 The interactions of integrin α5β1 and its extracellular ligands are dependent on the MIDAS structure and divalent cations.18 A recent crystal structure of α5β1 integrin has demonstrated that the specific residue (Asp154) could be used to distinguish α5 from other α subunits as its strong preference for fibronectin over other RGD ligands, and also indicated that Ca2+ is an important cation for ligand-binding of α5β1 integrin.14,16
Regulation of Integrin α5β1
miRNA Pathways Contribute to Post-Transcriptional Regulation of Integrin α5β1
miRNAs are a class of small endogenous noncoding RNAs and have emerged as important molecules that post-transcriptionally regulate gene expression.19 The expression of α5β1integrin is determined by the transcriptional activity of ITGA5 and ITGB1 genes. The 3ʹ-untranslated region of the ITGA5 and ITGB1mRNA possess several miRNA target sequences. Recently, some miRNAs were reported to regulate the expression of integrin α5β1 under various pathological conditions. For example, both integrin α5 and β1 were directly targeted by miR-17 in ovarian cancer cell lines, and forced expression of miR-17 significantly blocked adhesion and invasion of ovarian cancer cells by inhibiting the expression of integrin α5 and β1.20 In addition, miR-23a directly targeted the 3′ UTR of High Mobility Group Nucleosomal Binding Domain 2 (HMGN2) mRNA, which was involved in integrin α5β1 activation. miR-155 might also regulate integrin α5β1function by control of the expression and chromatin location of the integrin transcription suppressor-Nuclear Factor-I (NFI).21 In breast cancer cells, miR-149 inhibited cancer cell metastasis by directly targeting GIT ArfGAP 1 (GIT1), which was responsible for the lysosome-mediated protein degradation of integrin α5β1.22 Therefore, exploring the network of miRNAs and integrin α5β1 is essential to design strategies for better chemo-therapeutics.
Importance of Post-Translational Modifications (PTMs) in Regulation of Integrin α5β1
PTMs are made up of methylation, acetylation, phosphorylation, ubiquitination, neddylation, sulphation, sumoylation, prenylation, and glycosylation, which are the fundamental process for regulating the function of proteins, such as subcellular location, DNA-binding affinity, molecular half-life, and interactions with other proteins. N-Glycosylation of protein is considered to be as the most abundant PTM, and nearly 50% all known proteins are glycosylated in eukaryotes.23 Integrins as the major glycan-carrying proteins, its complete biological functions rely on the N-Glycosylation modifications. Among the 24 human integrins, the functions of N-Glycosylation on integrin α5β1 have been well characterized.23,24 Gu and colleagues have identified several individual N-glycan sites in both α5 and β1subunits, which are critical for heterodimerization and biological functions of integrin α5β1.25–28 For example, the N-glycan of β1-N343 on the βI domain of β1 subunit is linked to integrin α5β1 activation. Loss of this glycan site led to the persistent activation of integrin α5β1;29 The N-glycan sites on the I-like domain of the β1 subunit (β1S4-6) are important for integrin α5β1-mediated cell spreading and migration;26 The N-glycosylation on the β-propeller domain of the α5 subunit (α5S3-5) are critical for the heterodimerization, and biological functions of integrin α5β1, as well as the formation of α5–syndecan-4 complex.25,27 The site-11 N-glycosylation on calf domain of α5 subunit is also important for the α5-EGFR complex formation and the inhibitory effect on EGFR signaling.28 Beyond glycosylation, ubiquitination of α5 subunit also plays an important role for integrin α5β1-mediated fibroblast migration.30 Therefore, exploring the PTMs of integrin α5β1 is essential to understand the biological function and mechanism of integrin α5β1.
Potential Trafficking Machinery of Integrin α5β1
As transmembrane proteins, the transport of integrins to the cell surface is determined by the integrin trafficking machinery including exocytosis of integrins by vesicles and endocytosis of integrins at the plasma membrane. Integrin trafficking is considered to be an important regulator of cell adhesion and migration. The trafficking of α5β1 integrin is affected by several proteins such as CD151, Ras Homolog Family Member C (RhoC), Cytoskeleton-Associated Protein 4 (CKAP4), PTPRF Interacting Protein Alpha 1 (PPFIA1), Ankyrin-B, protein kinase B, syntaxins 3 and 4, Vesicle-Associated Membrane Protein 2 (VAMP2), Adaptor Protein, phosphotyrosine interacting with ph domain and leucine zipper 1 (APPL1), TGF-β type III receptor (TβRIII), and Neuropilin-2 (NRP-2).31–41 For example, CD151 was functionally linked to integrin-mediated cell migration by control of the endocytosis and/or vesicular trafficking of α3β1, α5β1, and α6β1 integrins.31 And mutation of the YXXφ endocytosis/sorting motif on the C-terminal cytoplasmic domain of CD151 significantly disrupted CD151-mediated cell migration.31 In pancreatic carcinoma cells, RhoC over-expression enhanced integrin α5β1 internalization and trafficking, increasing the levels of α5β1 integrin at the cell surface and promoting cell metastasis.32 TβRIII, a ubiquitous co-receptor for TGF-β, inhibited cell motility by control of β-arrestin2 dependent α5β1 internalization and recycling. In addition, TβRIII expression was significantly associated with α5 localization and overall survival in breast cancer patients.40
Extracellular Molecules Modulate Integrin α5β1
Extracellular molecules such as growth factor and receptors, cytokine and cytokine receptors, as well as extracellular matrix proteins are crucial regulatory factors affecting the biological activity and function of integrin α5β1. In cancer cells, epidermal growth factor (EGF) treatment could promote the p90RSK-dependent phosphorylation of filamin A (FLNa), which was responsible for the inactivation of integrin α5β1.42 In addition, Dudvarski et al reported that epidermal growth factor-like protein 7 (EGFL7) elevated the levels of integrin α5β1 on the cellular surface and then promoted the fibronectin-induced angiogenesis in glioblastoma.43 Interleukin 1β (IL-1β), an inflammatory cytokine, not only induced inflammatory but also increased integrin α5β1-dependent adhesion to fibronectin. Upon IL-1β treatment, the expression of α5 subunit increased and the active β1 subunit were relocated to focal contacts in the transformed human brain microvascular endothelial cells (THBMECs). And using α5-and β1-specific antibodies could remarkably inhibit the transmigration function under IL-1β-induced inflammatory conditions.44 In basal-like breast cancer cells, CD44 elevated the expression and activity of β1 subunit, and also increased the expression of α5 subunit.45 In addition, E-cadherin also associated with the expression and transcription activity of α5β1 in ovarian cancer cells. Sawada et al demonstrated that E-cadherin loss could increase α5β1 expression by regulating the FAK1/ERK1/MAPK signaling pathway.46 Therefore, better understanding of the associations of extracellular molecules and α5β1 is essential for clinical therapy.
Ligands of α5β1
Integrin α5β1 can recognize and adhere to extracellular ligands containing RGD tripeptide motif. Research on the molecular interactions of integrin α5β1 may be essential for interpreting the biological function and underlying mechanisms of α5β1 integrin. Herein, we will discuss the reported ligands and related functions of integrin α5β1, which are summarized in Table 1. Extracellular matrix molecules fibrinogen, fibronectin, and fibrillin-1 could be recognized and bound by integrin α5β1, which have been shown to affect cell adhesion and migration of endothelial and other cells.14,47–50 VEGFR-1 was secreted by endothelial cells and then interacted with integrin α5β1, and this interaction was important for angiogenesis.51 CD97, CD87 and CD154, transmembrane proteins contain RGD peptide, have shown to interact with integrin α5β1 and induce cell adhesion, intracellular signaling, and angiogenesis.52–54 Recently, other ligands of integrin α5β1 have been identified including Porcine hemagglutinating encephalomyelitis virus (PHEV), 25-hydroxycholesterol, Tubulointerstitial nephritis antigen-like 1 (Tinagl1), Pregnancy-Specific Glycoprotein 1 (PSG1), and Neuropilin-2.41,55–58 All of these studies demonstrate that ligand-binding regulation of α5β1 integrin plays an important role for regulating the cellular function, such as cell adhesion, migration, and angiogenesis.
 |
Table 1 Ligands and Related Functions of Integrin α5β1 |
Functions of α5β1
As a transmembrane protein, integrin α5β1possess different domains include extracellular, transmembrane, and cytoplasmic domain that determines the multiple functions of α5β1. The extracellular and transmembrane domains are responsible for binding to ECM proteins, or other extracellular ligands, and contribute to subsequent signaling pathway function, whereas the cytoplasmic domain can interact with cytoskeleton-associated proteins to affect cell migration, invasion, and proliferation.55,59–66 It also reported that integrin α5β1 was involved in anoikis resistance or drug resistance of cancer cells.67–70 Besides, α5β1 integrin was strongly associated with cellular senescence.71 Integrin α5β1 was also linked to the maintenance of bone tissue-forming and the formation of atherogenic inflammation, as well as the function/survival of T cell.72–74 The multiple functions of α5β1 integrin indicated that dysregulation of α5β1 integrin could lead to various diseases, particularly cancer. Indeed, the hyper-expression of α5β1 integrin has been shown to promote tumor metastasis in lung cancer and melanoma. However, α5β1 integrin also act as a tumor-suppressive role in several breast cancer and colon cancer cell lines.14
Implication of α5β1 in Carcinogenesis
It is well known that integrin α5β1 acts an important role in diverse cancer progression and cancerogenesis, and thus the deregulation of α5β1 integrin is highly associated with a series of malignant tumors. Herein, we will discuss the potential roles and related functions of integrin α5β1 on human cancers in this section (Table 2).
 |
Table 2 Functional Roles of Integrin α5β1 Pathway in Different Types of Cancer |
Expression of Integrin α5β1 is Upregulated in Various Types of Cancers
Aberrant upregulation of integrin α5β1 has been implicated in a number of human malignancies and is closely correlated with poor prognosis. Integrin α5β1 expression was slightly expressed in normal brain tissue, but was expressed at significantly high intensity in glioblastoma tissue.75 Research demonstrated that activation of α5β1 integrin was linked to the promotion of cell survival, migration, invasion, angiogenesis, and drug-resistance of glioma cells.43,63,69,76,77 In addition, α5β1 integrin expression was overexpressed in colon cancer cells, and blockade of cell surface α5 integrin by selective antibody significantly suppressed cell adhesion and induced apoptosis.78,79 In MCF-7 human breast carcinoma cells, hyperexpression of integrin α5β1 promoted cell invasion and doxorubicin resistance by enhancing the activity of AKT, mTOR, and ERK1/2 protein kinases.68,80,81 Importantly, blocking α5β1 integrin by PHSCN (Pro-His-Ser-Arg-Asn) peptide significantly prevented cell metastasis in preclinical prostate adenocarcinoma models, and parallel progression Phase I clinical trial.82 Besides, integrin α5β1 was upregulated in some primary and metastatic melanoma cells and positively linked to liver metastasis in melanoma.3,83 In node-negative non-small cell lung cancer (NSCLC), the expression of α5β1 integrin was highly expressed in 50.0% (44/88) node-negative NSCLC patients and significantly associated with the differentiation status and age of the patients.84 Notably, another study demonstrated that α5β1 integrin expression was more frequent in NSCLC with lymph node metastasis.85 Integrin α5β1 was also overexpressed in 84.6% (143/169) cervical cancer samples, and high α5β1 integrin expression was closely linked to poor histologic differentiation, lymph node metastasis, negative chemotherapy response, and recurrence in cervical cancer.86,87 Researchers also showed that the α5 and β1integrin subunits were significantly increased in ovarian cancer compared with the normal tissue, and inhibition of the expression of integrin α5 and β1 may be improved the prognosis of ovarian cancer patients.88,89 Patients with α5β1 integrin hyperexpression tended to have poor overall survival in ewing sarcoma, leukemia, basal cell carcinoma, multiple myeloma, colorectal cancer, osteosarcoma, squamous carcinoma, head and neck squamous cell carcinoma, mesothelioma, pancreatic carcinoma, gastric cancer, cholangiocarcinoma, epidermoid carcinoma, chondrosarcoma, neuroblastoma, epidermoid carcinoma, motile carcinoma, rectal cancer, and transitional carcinoma.32,81,90–107
Integrin α5β1 as a Tumor Suppressor in Several Types of Cancer Cell Lines
Integrin α5β1 as a classic cell surface receptor has been reported as a tumor suppressor due to overexpressing α5β1 integrin in tumor cells are less tumorigenic than its corresponding parent cells. In colon cancer cell line, HT29, α5β1 integrin overexpression showed a strong inhibitory function on lung colonization and metastasis.108 And de novo expression of α5 integrin subunit was linked to suppress cell growth arrest and retard the tumorigenic growth of HT29 cells.109 However, another study demonstrated that upregulation of the α5 integrin subunit suppressed apoptosis triggered by serum deprivation in HT29 cells.110 Then, further research demonstrated that integrin α5β1 level was significantly elevated in the poorly differentiated colon cancer cell lines and was positively associated the tumorigenic capacity. Therefore, the different roles of α5β1 integrin in colon cancer cells might be related to the differentiation status.111 Besides, loss of α5β1 integrin at the cell surface of the uveal melanoma cells was positively associated with the high tumorigenicity and aggressiveness.112–114
Deregulation of Integrin α5β1 Exerts Dramatic Effects on Diverse Cellular Functions
Role of Integrin α5β1 in Angiogenesis
Angiogenesis is a crucial physiological and pathological process for the development of new blood vessels, which was responsible for the tissue repair and fertility, embryonic development, chronic inflammation, tumor growth and metastasis.115 Basic and clinical studies demonstrated that inhibition of angiogenesis could suppress tumor metastasis and progression. Most studies implicate integrins, which are critical modulators of tumor angiogenesis. Among the integrin family, αv, α2, α3, α4, α5, β1 or β2 integrin subunits have been demonstrated to be associated with vasculo- and angiogenesis during development.5,14,65 Research found that loss of fibronectin, the major ligand for α5β1 integrin, led to angiogenesis abnormalities and embryonic death at E9.5 in mice.116 Genetic ablation studies have indicated that β1-integrin-null endothelial cells displayed vascular remodeling effects resulting from adhesion and migration alteration, suggesting the β1 integrin family clearly played an important role in angiogenesis.14,117 Integrins α5 and αv have been considered as key modulators of endothelial cells and vascular smooth muscle cell (vSMC) function. Interestingly, endothelial cell-specific knockout of either integrin α5 and αv do not have obvious angiogenesis defects during embryonic development.118 Researchers found that vSMC-specific knockout of both α5 and αv integrin led to the formation of large aneurysms within the brachiocephalic/carotid arteries and cardiovascular defects, as well as late embryonic lethality.119 These studies indicate that specific integrins are important during the vascular development, and the compensation mechanisms by other integrins are essential for normal angiogenesis.
In most quiescent endothelium, integrin α5β1 was limited to very low levels, but its expression was significantly upregulated in tumor vasculature or neovessels.120,121 Integrin α5β1 participated in regulating angiogenesis by interacting with diverse partners such as CD97, angiopoietin-2 (Ang-2), CD87, VEGFR1, and endostatin.51,54,122–124 Integrin α5β1levels in endothelial cells were induced in response to several angiogenic factor stimuli, such as IL-8, bFGF, EGFL7, Del-1or TNFα, but not by VEGF.43,120,125 Besides, HoxD3 acts as a homeobox gene controlled the expression of integrin α5 by directly binding to the promoter of the α5 subunits.126 Therefore, integrin α5β1 plays an important role in angiogenesis, and blocking α5 and β1 integrin subunits by specific monoclonal antibodies or small peptides has become a potential strategy for anti-angiogenesis therapy.127–130
Integrin α5β1 Regulates the Migration and Invasion of Tumor Cells
The ability of cancer cells to invade locally and further form distant metastasis is partly determined by integrin-mediated attachment to ECM. Integrin α5β1 function as a critical regulator for tumor cell migration and invasion by affecting cytoskeleton rearrangement, cell adhesion, and the production of matrix metalloproteinase (MMP). Some studies have demonstrated that integrin α5β1 enhanced keratinocyte adhesion to fibronectin, and promoted invasion and metastasis via activating various signaling pathways.11,131 Besides, fibronectin binding to integrin α5β1 led to the direct association of α5 integrin with c-Met, which was upstream of Src and FAK. Integrin α5β1 promoted tumor cells invasion and metastasis via activating the c-Met/FAK/Src-dependent signaling pathway.132 Research also found that integrin α5β1 promoted invasiveness and metastasis by regulating the expression and/or activity of MMPs.133 In breast cancer, cells with high integrin α5β1 expression elevated a 3-fold invasive capacity compared with cells exhibiting low α5β1 levels.81 Integrin α5β1 could direct recruit MMP2 collagenase on the surface of breast carcinoma cells, and then regulating cell invasion by control of the levels of MMP2.134 Moreover, MMP-2/α5β1 binding has pivotal role in regulating tumor metastasis by inducing α5β1-mediated IL-6/STAT3 signaling pathway.135 In murine cell line B16F10, integrin α5β1and fibronectin interaction facilitated cell invasion by inducing the activity, mRNA, and protein expression of MMP9. Blocking the α5 integrin receptor by specific antibody remarkably abrogated the fibronectin-induced MMP9 response.136 Moreover, ADAM Metallopeptidase Domain 17 (ADAM17) was reported to directly interacted with integrin α5β1, and this interaction might take place on the same cell or on different cell, with the function to affect cell-cell adhesion and migration.137,138 In addition, Rab-coupling protein (RCP)-driven endocytic recycling of α5β1 integrin also promoted invasion of cancer cells, which was associated with actin cytoskeleton arrangement.7 The effects of integrin α5β1 on the cell adhesion, migration, and invasion of tumor cells indicate that it may be used as the biomarker for the metastasis of tumors.
Integrin α5β1 Mediates the Proliferation of Tumor Cells
Mounting studies have implicated that integrin α5β1 contributed to tumor cell proliferation in vitro and tumor growth in vivo. It has been demonstrated that α5β1 integrin possessed the ability of enhancing cell proliferation depending on the fibroblasts, which are some of the major cells in neoplasm tissues and affect cancer progression. High L1 cell adhesion molecule (L1CAM) levels in fibroblasts promoted cancer cell proliferation by targeting integrin α5β1.139 In addition, interaction of myofibroblasts and soluble fibronectin facilitated the α5β1 integrin-dependent tumor growth in the hepatocellular carcinoma.140
Integrin α5β1 was also considered to be an important regulator for cell cycle-associated proteins. p53 is a universal tumor suppressor implicated in cell cycle arrest, apoptosis, and DNA repair. Research found that the expression of α5 integrin subunit was negatively associated with p53 activity, and depletion of the α5 integrin subunit could increase p53 activity.69 Interestingly, re-activation of p53 by Nutlin-3, a p53-reactivating compound, significantly inhibited the mRNA and protein expression of α5 integrin subunit.141 Thus, the crosstalk between α5β1 integrin and p53 was crucial for tumor growth, and some antagonists of α5β1 integrin have been applied by modulating the integrin α5β1/p53 pathway.77,142 Of note, the integrin α5β1-ERK pathway was also involved in the regulation of cancer cell proliferation.104 Antibodies that block the integrin α5β1 negated the proliferative effect of integrin α5β1 in malignancy cells.143
Role of Integrin α5β1 in Chemoresistance and Radioresistance
Resistance to chemotherapy and radiotherapy is a unique hallmark of neoplasm and is responsible for tumor recurrence and patient relapse.144 Cell adhesion to ECM components is a critical determinant of chemotherapeutic response of human cancers, such as myeloma. For example, Integrin α5β1 promoted K562 chronic myelogenous leukemia (CML) cells bind to fibronectin, and this binding was resistant to apoptosis induced by chemotherapeutic drugs and γ-irradiation.145 Besides, fibronectin/integrin α5β1 binding elevated the efficiency of 2-D colony formation, and provided resistance to paclitaxel-mediated apoptosis.146 Research has demonstrated that integrin α5β1 protected high-grade glioma cells from temozolomide-induced apoptosis by interfering with the p53 pathway in glioma.69 In epithelial ovarian carcinomas, overexpression of integrin α5 was a strong risk factor for drugs resistance.147 Integrin α5β1 also contributed to cell adhesion and drug resistance of multiple myeloma cells through activating the FAK/STAT3/AKT pathways.93 In MCF-7 human breast carcinoma cells, hyperexpression of integrin α5β1 promoted the doxorubicin resistance in an ERK-dependent manner. Besides, silencing of integrin α5β1 significantly inhibited the activity of kinases AKT and ERK in MCF-7 doxorubicin-resistant cells.80
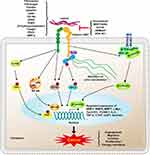
The Signaling Pathways Involved in Integrin α5β1-Mediated Tumor Progression
Integrin α5β1 functions as a cell surface receptor, and ligation of integrin α5β1activates several crucial signaling pathways that are critical in carcinogenesis/tumor progression, such as FAK signaling, Wnt/β-catenin signaling, NF-κB signaling, Yes-associated protein (YAP) signaling, and ERK signaling. Interpreting the molecular mechanism of integrin α5β1 in these pathways may provide a better understanding of carcinogenesis/tumor progression.
Regulation FAK Pathway by Integrin α5β1
Due to lack of intrinsic tyrosine kinase activity, integrins transduce extracellular cues to intracellular signaling pathways require non-receptor tyrosine kinases such as FAK.148,149 Research demonstrated that fibronectin-integrin α5β1 complex facilitated the auto-phosphorylation of the Tyr 379 residue on FAK. Subsequently, the tyrosine kinase Src bound to phosphorylated FAK through its SH2 domain, and induced phosphorylation of FAK at Tyr 925 residue, which then promoted the formation of FAK-Grb2-SOS complex. Ultimately, this complex contributed to cell proliferation, metastasis, and tumorigenesis of cancer cells by activating Ras GTPase and inducing the MAPK/ERK signaling pathway.150,151 Indeed, integrin α5β1-FAK signaling pathway contributed to cell metastasis and cancer progression of several malignant neoplasms, and some specific monoclonal antibody or integrin α5β1 inhibitor could significantly negate these accelerative effects.57,152–154
Regulation Wnt/β-Catenin Pathway
Wnt/β-catenin pathway is critical to facilitate tumor progression, such as cell proliferation, cell cycle, cell metastasis, differentiation, and apoptosis. In pancreatic cancer, integrin α5β1 mediated the adhesion of pancreatic adenocarcinoma cells on fibronectin under serum-free conditions, resulting in the increasing of β-catenin localization throughout the cell.155 In hepatocellular carcinoma, overexpression of CD147 competitively bound to integrin β1that interrupted the fibronectin/integrin β1interaction, which is responsible for E-cadherin degradation and β-catenin nuclear translocation.156 In glioma cells, overexpression and activation of α5β1 integrin by fibronectin facilitated the transactivation of β-catenin gene targets and induced an increase in cell migration.63 In addition, other high-affinity peptide such as cyclized CRRETAWAC also promoted integrin α5β1-mediated Wnt/β-catenin transcriptional activity.157
Regulation of NF-κB Signaling
NF-κB transcription factors and their regulated genes have been recognized as critical mediators involved in tumor initiation, cell proliferation, survival, metastasis, angiogenesis, and resistance to chemotherapy and radiotherapy.158 Fibronectin/integrin α5β1 interaction was responsible for inducing the expression of the p65 component of NF-κB and enhancing the DNA-binding activity of NF-κB in human bronchial epithelial cells.159 Upon fibrinogen binding, integrin α5β1 and αvβ3 promoted the activation of NF-κB and increased the expression of NF-κB-mediated inflammatory chemokines in endothelial cells. And these effects were inhibited by blockage of the integrin α5β1 and αvβ3 with the GRGDS peptide.160 Besides, lunasin, a naturally occurring 43-amino acid peptide isolated from soybean, direct binding with integrin α5β1 and inhibiting the NF-κB signaling in colon cancer cells.152
Activation of YAP by Integrin α5β1
YAP, the crucial transcriptional regulator of the Hippo pathway, is involved in modulating organ size, tissue homeostasis and repair, and tumorigenesis.161 Hyperactivation of YAP is associated with the malignant behavior of neoplasm, such as high proliferation, invasion into the surrounding normal tissue, vascularization, and drug resistance.162 Recent studies demonstrated that activation of integrin α5β1 by the ligand fibronectin significantly increased the phosphorylation of YAP at Tyr357 and induced YAP nuclear translocation via the tyrosine kinase c-Abl in ECs. In contrast, blockage of integrin α5β1 with ATN161or inhibition of c-Abl with bosutinib markedly reduced the levels of integrin α5β1 and p-YAPY357.163 Beyond YAP phosphorylation, the dephosphorylation of YAP (S127) was also regulated by the α5 integrin subunits.164 In Ewing sarcoma cells, integrin α5β1signaling was associated with YAP dephosphorylation and nuclear translocation, and this signaling pathway significantly promoted tumor progression.90
Integrin α5β1 Regulates ERK Signaling Pathway
ERK signaling pathway is hyperactivated in a variety of cancers, which execute programmes related to cell cycle, differentiation, migration and invasion, and apoptosis. Fibronectin and integrin α5β1 binding enhanced Hela cell proliferation by increasing the phosphorylation of ERK at Thr 202 and Tyr 204 residues and then activating the ERK pathway.148 uPAR (CD87), an urokinase receptor frequently upregulated in several types of tumors, bound to integrin α5β1 and then persistently activated the ERK signaling.165 Using site-directed mutagenesis, two single amino acid mutants of the uPAR (S245A and H249A) were respectively identified that fail to facilitate integrin α5β1-mediated ERK signaling.104,166 Moreover, disruption of uPAR/integrin α5β1 interaction by using specific small molecules significantly inhibited ERK activity and tumor progression.104,167
Translational Implications of Integrin α5β1 in Cancer
Integrin α5β1 as a Target for Imaging
Although integrin α5β1 is limited to very low levels in quiescent endothelial cells, it is significantly upregulated in tumor vasculature or neovessels.120,121 And α5β1 integrin is strongly correlated with tumor angiogenesis, suggesting it may be a potential predictive target. So far, several imaging probes for α5β1have been described for tumor molecular imaging. Stefanie et al firstly developed the α5β1-selective antagonists labeled with 68Ga3+ for PET (positron emission tomography) imaging and could verify different patterns of integrin α5β1expression in tumors.168 D’Alessandria et al then successfully developed a 68Ga-labelled α5β1-selective peptidomimetic named FR366, which showed good image quality for PET imaging.169 Through sequential N-methylation analysis, Tobias et al discovered a most potent and selective α5β1-integrin ligand peptide, c(phg-isoDGR-(NMe)k), which was applied for PET imaging by trimerized with the chelator TRAP and labeled with 68Ga.170 In addition, there have been several α5β1-specific probes such as 99m Tc-HisoDGR, 99mTc-AB-3PisoDGR2, and 99mTc-3PisoDGR were developed for SPECT (single-photon emission computed tomography) imaging.171,172 Recently, RNA aptamers have received attention as promising tools for clinical applications due to their smaller size, lack of immunogenicity and toxicity, temperature stability, ease of chemical modification, and lower cost of production.173,174 Fechter and colleagues successfully identified and developed RNA aptamers, aptamer H02, is efficient to distinguish GBM tumor tissues from patient-derived tumor xenografts. This new, original, and powerful aptamer tool may be open roads for α5β1-specific clinical therapy.175
Integrin α5β1 as a Target for Therapy
Integrin α5β1 has become a potential target for cancer therapy, and several specific α5β1 integrin antagonists have been developed and used in preclinical or clinical studies. These antagonists are mainly presented as anti-angiogenic agents due to the pro-angiogenic function of integrin α5β1, and they mainly consisted by specific antibodies and small peptides.
A series of blocking antibodies was developed to target the interaction between integrin α5β1 and fibronectin. IIA1, an integrin α5β1 function-blocking murine antibody, was generated and used to inhibit in vitro angiogenesis, cell adhesion, invasion, and survival of tumor cells.46,130,176 Notably, Ramakrishnan and colleagues firstly developed a chimeric human IgG4 version of IIA1 antibody, volociximab, with similar affinity for α5β1 integrin and similar activity by inhibition of fibronectin binding than IIA1.130 Volociximab as a potential anti-antigenic drug and has been shown to be effective, safe, and tolerable in phase I b studies in patients with non-small-cell lung cancer, and in Phase II studies in patients with epithelial ovarian or primary peritoneal cancer.177–179 The MINT1526A is a function-blocking anti-α5β1 monoclonal antibody, has been used in anti-angiogenic therapy combining α5β1 and VEGF inhibition, and has been shown to be well tolerated and safe in phase I study.180 Recently, a bispecific antibody (BsAbα5β1/αv) simultaneously targeting the degradation of αv and α5β1 integrins. And this combinatorial strategy was superior to monospecific antibodies in abrogating cell adhesion, migration, survival in prostate cancer cells.181
Integrins α5β1can recognize the RGD motif of fibronectin and then directly bind to it. Recent years, many studies focus on the design antagonists with enhanced selectivity of α5β1 integrin. Some antagonists have been developed and used in preclinical or clinical studies, such as SJ749 and JSM6427, and ATN-161.120,142,182,183 Among them, ATN-161 (Ac-PHSCN-NH2), a competitive inhibitor of the FN-α5β1 interactions firstly developed by Attenuon LLC (San Diego, CA, USA), have moved to Phase II clinical trails.14
Although many preclinical studies supported the anti-angiogenic therapies of blocking antibody and small molecule, early clinical responses have been disappointing. Most α5β1 integrin inhibitors and antibodies were clinical investigations in phases 1 or 2 that have not progressed through phases 3, revealed no treatment benefit.184 Murphy and colleagues revealed that FN and the FN receptors, α5 and αv, were dispensable for tumor angiogenesis through a series of genetic tools and pre-clinical models (transplant models and RIP1-Tag2 model of pancreatic cancer), suggesting that the antagonism of antibodies or small molecules on tumor angiogenesis may occur through a dominant-negative effect, rather than a simple block of the FN-integrin α5β1binding.184 And they also found that tumor growth was not affected by the absence of FN and its integrin receptors.184 In addition, they revealed the potential compensatory mechanism that several RGD-containing extracellular matrix proteins, such as fibrillins, collagens, and nidogens, might be important in compensating for the loss of FN.184 Therefore, further in vivo genetic studies were necessary to resolve the targeting difficulties.
Conclusion
In this review, we briefly illustrate our understanding on the structure, regulation, ligands and biological functions of integrin α5β1, and reveal the roles of integrin α5β1in various tumors. The dysregulation of α5β1 integrin significantly relates to the development and progression of many neoplasms and can be used as a valuable indicator of poor prognosis. Functionally, integrin α5β1 can recognize and adhere to extracellular ligands containing RGD tripeptide motif, and this integrin α5β1/ligand binding modulates diverse cellular progression by activation of several classic oncogenic signaling pathways, such as FAK signaling, Wnt/β-catenin signaling, NF-κB signaling, YAP signaling, and ERK signaling (Figure 1). The important role of integrin α5β1 in the tumor angiogenesis is that provides the potential predictive possibility for tumor molecular imaging, such as PET, SPECT, and RNA aptamers. Moreover, several specific α5β1 integrin antagonists have been developed and/or used in preclinical or clinical studies. Considering the pivotal cellular role of the integrin α5β1, it is reasonable to assume that advances in integrin α5β1 research will facilitate the development of molecular diagnosing and therapy of tumors in the future.
Acknowledgments
This work was supported by the Natural Science Foundation of Chongqing (No. cstc2019jcyj-zdxmX0033 and cstc2020jcyj-msxm2003), the National Natural Science Foundation of China (81872071, 81672502, and 81972357) and the Fundamental Research Funds for the Central Universities (SWU118097).
Author Contributions
All authors made substantial contributions to conception and design; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
The authors declare no conflicts of interest.
References
1. Desgrosellier JS, Cheresh DA. Integrins in cancer: biological implications and therapeutic opportunities. Nat Rev Cancer. 2010;10:9–22. doi:10.1038/nrc2748
2. Schwartz MA, Ginsberg MH. Networks and crosstalk: integrin signalling spreads. Nat Cell Biol. 2002;4:E65–68. doi:10.1038/ncb0402-e65
3. Huang R, Rofstad EK. Integrins as therapeutic targets in the organ-specific metastasis of human malignant melanoma. J Exp Clin Cancer Res. 2018;37:92. doi:10.1186/s13046-018-0763-x
4. Campbell ID, Humphries MJ. Integrin structure, activation, and interactions. Cold Spring Harb Perspect Biol. 2011;3:a004994–a004994. doi:10.1101/cshperspect.a004994
5. Naci D, Vuori K, Aoudjit F. Alpha2beta1 integrin in cancer development and chemoresistance. Semin Cancer Biol. 2015;35:145–153. doi:10.1016/j.semcancer.2015.08.004
6. Pacifici R, Basilico C, Roman J, Zutter MM, Santoro SA, McCracken R. Collagen-induced release of interleukin 1 from human blood mononuclear cells. Potentiation by fibronectin binding to the alpha 5 beta 1 integrin. J Clin Invest. 1992;89:61–67. doi:10.1172/JCI115586
7. Paul NR, Allen JL, Chapman A, et al. alpha5beta1 integrin recycling promotes Arp2/3-independent cancer cell invasion via the formin FHOD3. J Cell Biol. 2015;210:1013–1031. doi:10.1083/jcb.201502040
8. Delon I, Brown NH. Integrins and the actin cytoskeleton. Curr Opin Cell Biol. 2007;19:43–50. doi:10.1016/j.ceb.2006.12.013
9. Hynes RO. Integrins: bidirectional, allosteric signaling machines. Cell. 2002;110:673–687. doi:10.1016/S0092-8674(02)00971-6
10. Ridley AJ, Schwartz MA, Burridge K, et al. Cell migration: integrating signals from front to back. Science. 2003;302:1704–1709. doi:10.1126/science.1092053
11. Nieberler M, Reuning U, Reichart F, et al. Exploring the role of RGD-recognizing integrins in cancer. Cancers (Basel). 2017;9:116. doi:10.3390/cancers9090116
12. Rubtsov MA, Syrkina MS, Aliev G. RGD-based therapy: principles of selectivity. Curr Pharm Des. 2016;22:932–952. doi:10.2174/1381612822666151209153636
13. Li L, Welser-Alves J, van der Flier A, Boroujerdi A, Hynes RO, Milner R. An angiogenic role for the alpha5beta1 integrin in promoting endothelial cell proliferation during cerebral hypoxia. Exp Neurol. 2012;237:46–54. doi:10.1016/j.expneurol.2012.06.005
14. Schaffner F, Ray AM, Dontenwill M. Integrin alpha5beta1, the fibronectin receptor, as a pertinent therapeutic target in solid tumors. Cancers (Basel). 2013;5:27–47. doi:10.3390/cancers5010027
15. Rocha LA, Learmonth DA, Sousa RA, Salgado AJ. alphavbeta3 and alpha5beta1 integrin-specific ligands: from tumor angiogenesis inhibitors to vascularization promoters in regenerative medicine? Biotechnol Adv. 2018;36:208–227. doi:10.1016/j.biotechadv.2017.11.004
16. Nagae M, Re S, Mihara E, Nogi T, Sugita Y, Takagi J. Crystal structure of alpha5beta1 integrin ectodomain: atomic details of the fibronectin receptor. J Cell Biol. 2012;197:131–140. doi:10.1083/jcb.201111077
17. Barczyk M, Carracedo S, Gullberg D. Integrins. Cell Tissue Res. 2010;339:269–280.
18. Carman CV, Springer TA. Integrin avidity regulation: are changes in affinity and conformation underemphasized? Curr Opin Cell Biol. 2003;15:547–556. doi:10.1016/j.ceb.2003.08.003
19. Anastasiadou E, Jacob LS, Slack FJ. Non-coding RNA networks in cancer. Nat Rev Cancer. 2018;18:5–18.
20. Gong C, Yang Z, Wu F, Han L, Liu Y, Gong W. miR-17 inhibits ovarian cancer cell peritoneal metastasis by targeting ITGA5 and ITGB1. Oncol Rep. 2016;36:2177–2183. doi:10.3892/or.2016.4985
21. Teng Y, Miao J, Shen X, et al. The modulation of MiR-155 and MiR-23a manipulates Klebsiella pneumoniae adhesion on human pulmonary epithelial cells via integrin alpha5beta1 signaling. Sci Rep. 2016;6:31918. doi:10.1038/srep31918
22. Chan SH, Huang WC, Chang JW, et al. MicroRNA-149 targets GIT1 to suppress integrin signaling and breast cancer metastasis. Oncogene. 2014;33:4496–4507. doi:10.1038/onc.2014.10
23. Gu J, Isaji T, Sato Y, Kariya Y, Fukuda T. Importance of N-glycosylation on alpha5beta1 integrin for its biological functions. Biol Pharm Bull. 2009;32:780–785. doi:10.1248/bpb.32.780
24. Gu J, Taniguchi N. Regulation of integrin functions by N-glycans. Glycoconj J. 2004;21:9–15. doi:10.1023/B:GLYC.0000043741.47559.30
25. Isaji T, Sato Y, Zhao Y, et al. N-glycosylation of the beta-propeller domain of the integrin alpha5 subunit is essential for alpha5beta1 heterodimerization, expression on the cell surface, and its biological function. J Biol Chem. 2006;281:33258–33267. doi:10.1074/jbc.M607771200
26. Isaji T, Sato Y, Fukuda T, Gu J. N-glycosylation of the I-like domain of beta1 integrin is essential for beta1 integrin expression and biological function: identification of the minimal N-glycosylation requirement for alpha5beta1. J Biol Chem. 2009;284:12207–12216. doi:10.1074/jbc.M807920200
27. Hang Q, Isaji T, Hou S, et al. Regulator of cell adhesion: identification and characterization of important N-glycosylation sites on integrin alpha5 for cell migration. Mol Cell Biol. 2017;37. doi:10.1128/MCB.00558-16
28. Hang Q, Isaji T, Hou S, Zhou Y, Fukuda T, Gu J. N-Glycosylation of integrin alpha5 acts as a switch for EGFR-mediated complex formation of integrin alpha5beta1 to alpha6beta4. Sci Rep. 2016;6:33507. doi:10.1038/srep33507
29. Cai X, Thinn AMM, Wang Z, Shan H, Zhu J. The importance of N-glycosylation on beta3 integrin ligand binding and conformational regulation. Sci Rep. 2017;7:4656. doi:10.1038/s41598-017-04844-w
30. Lobert VH, Brech A, Pedersen NM, et al. Ubiquitination of alpha 5 beta 1 integrin controls fibroblast migration through lysosomal degradation of fibronectin-integrin complexes. Dev Cell. 2010;19:148–159. doi:10.1016/j.devcel.2010.06.010
31. Liu L, He B, Liu WM, Zhou D, Cox JV, Zhang XA. Tetraspanin CD151 promotes cell migration by regulating integrin trafficking. J Biol Chem. 2007;282:31631–31642. doi:10.1074/jbc.M701165200
32. Li NF, Gemenetzidis E, Marshall FJ, et al. RhoC interacts with integrin alpha5beta1 and enhances its trafficking in migrating pancreatic carcinoma cells. PLoS One. 2013;8:e81575. doi:10.1371/journal.pone.0081575
33. Osugi Y, Fumoto K, Kikuchi A. CKAP4 regulates cell migration via the interaction with and recycling of integrin. Mol Cell Biol. 2019;39. doi:10.1128/MCB.00073-19
34. Mana G, Clapero F, Panieri E, et al. PPFIA1 drives active alpha5beta1 integrin recycling and controls fibronectin fibrillogenesis and vascular morphogenesis. Nat Commun. 2016;7:13546. doi:10.1038/ncomms13546
35. Qu F, Lorenzo DN, King SJ, Brooks R, Bear JE, Bennett V. Ankyrin-B is a PI3P effector that promotes polarized alpha5beta1-integrin recycling via recruiting RabGAP1L to early endosomes. Elife. 2016;5.
36. Roberts MS, Woods AJ, Dale TC, Van Der Sluijs P, Norman JC. Protein kinase B/Akt acts via glycogen synthase kinase 3 to regulate recycling of alpha v beta 3 and alpha 5 beta 1 integrins. Mol Cell Biol. 2004;24:1505–1515. doi:10.1128/MCB.24.4.1505-1515.2004
37. Day P, Riggs KA, Hasan N, Corbin D, Humphrey D, Hu C. Syntaxins 3 and 4 mediate vesicular trafficking of alpha5beta1 and alpha3beta1 integrins and cancer cell migration. Int J Oncol. 2011;39:863–871.
38. Hasan N, Hu C. Vesicle-associated membrane protein 2 mediates trafficking of alpha5beta1 integrin to the plasma membrane. Exp Cell Res. 2010;316:12–23. doi:10.1016/j.yexcr.2009.10.007
39. Diggins NL, Kang H, Weaver A, Webb DJ. alpha5beta1 integrin trafficking and Rac activation are regulated by APPL1 in a Rab5-dependent manner to inhibit cell migration. J Cell Sci. 2018;131:jcs207019. doi:10.1242/jcs.207019
40. Mythreye K, Knelson EH, Gatza CE, Gatza ML, Blobe GC. TbetaRIII/beta-arrestin2 regulates integrin alpha5beta1 trafficking, function, and localization in epithelial cells. Oncogene. 2013;32:1416–1427. doi:10.1038/onc.2012.157
41. Cao Y, Hoeppner LH, Bach S. Neuropilin-2 promotes extravasation and metastasis by interacting with endothelial alpha5 integrin. Cancer Res. 2013;73:4579–4590. doi:10.1158/0008-5472.CAN-13-0529
42. Vial D, McKeown-Longo PJ. Epidermal growth factor (EGF) regulates alpha5beta1 integrin activation state in human cancer cell lines through the p90RSK-dependent phosphorylation of filamin A. J Biol Chem. 2012;287:40371–40380. doi:10.1074/jbc.M112.389577
43. Dudvarski Stankovic N, Bicker F, Keller S, et al. EGFL7 enhances surface expression of integrin alpha5beta1 to promote angiogenesis in malignant brain tumors. EMBO Mol Med. 2018;10. doi:10.15252/emmm.201708420
44. Labus J, Woltje K, Stolte KN, et al. IL-1beta promotes transendothelial migration of PBMCs by upregulation of the FN/alpha5beta1 signalling pathway in immortalised human brain microvascular endothelial cells. Exp Cell Res. 2018;373:99–111. doi:10.1016/j.yexcr.2018.10.002
45. McFarlane S, McFarlane C, Montgomery N, Hill A, Waugh DJ. CD44-mediated activation of alpha5beta1-integrin, cortactin and paxillin signaling underpins adhesion of basal-like breast cancer cells to endothelium and fibronectin-enriched matrices. Oncotarget. 2015;6:36762–36773.
46. Sawada K, Mitra AK, Radjabi AR, et al. Loss of E-cadherin promotes ovarian cancer metastasis via alpha 5-integrin, which is a therapeutic target. Cancer Res. 2008;68:2329–2339. doi:10.1158/0008-5472.CAN-07-5167
47. Mould AP, Akiyama SK, Humphries MJ. Regulation of integrin alpha 5 beta 1-fibronectin interactions by divalent cations. Evidence for distinct classes of binding sites for Mn2+, Mg2+, and Ca2+. J Biol Chem. 1995;270:26270–26277. doi:10.1074/jbc.270.44.26270
48. Suehiro K, Gailit J, Plow EF. Fibrinogen is a ligand for integrin alpha5beta1 on endothelial cells. J Biol Chem. 1997;272:5360–5366. doi:10.1074/jbc.272.8.5360
49. Tian J, Zhang FJ, Lei GH. Role of integrins and their ligands in osteoarthritic cartilage. Rheumatol Int. 2015;35:787–798.
50. Bax DV, Bernard SE, Lomas A, et al. Cell adhesion to fibrillin-1 molecules and microfibrils is mediated by alpha 5 beta 1 and alpha v beta 3 integrins. J Biol Chem. 2003;278:34605–34616. doi:10.1074/jbc.M303159200
51. Orecchia A, Lacal PM, Schietroma C, Morea V, Zambruno G, Failla CM. Vascular endothelial growth factor receptor-1 is deposited in the extracellular matrix by endothelial cells and is a ligand for the alpha 5 beta 1 integrin. J Cell Sci. 2003;116:3479–3489. doi:10.1242/jcs.00673
52. Wang T, Ward Y, Tian L, et al. CD97, an adhesion receptor on inflammatory cells, stimulates angiogenesis through binding integrin counterreceptors on endothelial cells. Blood. 2005;105:2836–2844. doi:10.1182/blood-2004-07-2878
53. Leveille C, Bouillon M, Guo W, et al. CD40 ligand binds to alpha5beta1 integrin and triggers cell signaling. J Biol Chem. 2007;282:5143–5151. doi:10.1074/jbc.M608342200
54. Tarui T, Andronicos N, Czekay RP, et al. Critical role of integrin alpha 5 beta 1 in urokinase (uPA)/urokinase receptor (uPAR, CD87) signaling. J Biol Chem. 2003;278:29863–29872. doi:10.1074/jbc.M304694200
55. Lv X, Li Z, Guan J, et al. Porcine hemagglutinating encephalomyelitis virus activation of the integrin alpha5beta1-FAK-cofilin pathway causes cytoskeletal rearrangement to promote its invasion of N2a cells. J Virol. 2019;93.
56. Pokharel SM, Shil NK, Gc JB, et al. Integrin activation by the lipid molecule 25-hydroxycholesterol induces a proinflammatory response. Nat Commun. 2019;10:1482. doi:10.1038/s41467-019-09453-x
57. Shen M, Jiang YZ, Wei Y, et al. Tinagl1 suppresses triple-negative breast cancer progression and metastasis by simultaneously inhibiting integrin/FAK and EGFR signaling. Cancer Cell. 2019;35:64–80 e67. doi:10.1016/j.ccell.2018.11.016
58. Rattila S, Dunk CEE, Im M, et al. Interaction of pregnancy-specific glycoprotein 1 with integrin alpha5beta1 is a modulator of extravillous trophoblast functions. Cells. 2019;8:1369. doi:10.3390/cells8111369
59. Cosset EC, Godet J, Entz-Werle N, et al. Involvement of the TGFbeta pathway in the regulation of alpha5 beta1 integrins by caveolin-1 in human glioblastoma. Int J Cancer. 2012;131:601–611. doi:10.1002/ijc.26415
60. De Toni-costes F, Despeaux M, Bertrand J, et al. A new alpha5beta1 integrin-dependent survival pathway through GSK3beta activation in leukemic cells. PLoS One. 2010;5:e9807. doi:10.1371/journal.pone.0009807
61. Kim S, Kang HY, Nam EH, et al. TMPRSS4 induces invasion and epithelial-mesenchymal transition through upregulation of integrin alpha5 and its signaling pathways. Carcinogenesis. 2010;31:597–606. doi:10.1093/carcin/bgq024
62. Zhang W, Ju J, Rigney T, Tribble G. Integrin alpha5beta1-fimbriae binding and actin rearrangement are essential for Porphyromonas gingivalis invasion of osteoblasts and subsequent activation of the JNK pathway. BMC Microbiol. 2013;13:5.
63. Renner G, Noulet F, Mercier MC, et al. Expression/activation of alpha5beta1 integrin is linked to the beta-catenin signaling pathway to drive migration in glioma cells. Oncotarget. 2016;7:62194–62207. doi:10.18632/oncotarget.11552
64. Oh SH, Kim JW, Kim Y, et al. The extracellular matrix protein Edil3 stimulates osteoblast differentiation through the integrin alpha5beta1/ERK/Runx2 pathway. PLoS One. 2017;12:e0188749. doi:10.1371/journal.pone.0188749
65. Subbaram S, Dipersio CM. Integrin alpha3beta1 as a breast cancer target. Expert Opin Ther Targets. 2011;15:1197–1210. doi:10.1517/14728222.2011.609557
66. Wang W, Tang M, Zhang L, et al. Clinical implications of CSN6 protein expression and correlation with mutant-type P53 protein in breast cancer. Jpn J Clin Oncol. 2013;43:1170–1176. doi:10.1093/jjco/hyt148
67. Guha D, Saha T, Bose S, et al. Integrin-EGFR interaction regulates anoikis resistance in colon cancer cells. Apoptosis. 2019;24:958–971. doi:10.1007/s10495-019-01573-5
68. Morozevich GE, Kozlova NI, Susova OY, Lupatov AY, Berman AE. Hyperexpression of integrin alpha5beta1 promotes resistance of MCF-7 human breast carcinoma cells to doxorubicin via ERK protein kinase down-regulation. Biochemistry (Mosc). 2017;82:1017–1024. doi:10.1134/S0006297917090048
69. Janouskova H, Maglott A, Leger DY, et al. Integrin alpha5beta1 plays a critical role in resistance to temozolomide by interfering with the p53 pathway in high-grade glioma. Cancer Res. 2012;72:3463–3470.
70. Nakahara S, Miyoshi E, Noda K, et al. Involvement of oligosaccharide changes in alpha5beta1 integrin in a cisplatin-resistant human squamous cell carcinoma cell line. Mol Cancer Ther. 2003;2:1207–1214.
71. Lopez-Luppo M, Catita J, Ramos D, et al. Cellular senescence is associated with human retinal microaneurysm formation during aging. Invest Ophthalmol Vis Sci. 2017;58:2832–2842. doi:10.1167/iovs.16-20312
72. Di Maggio N, Martella E, Frismantiene A, et al. Extracellular matrix and alpha5beta1 integrin signaling control the maintenance of bone formation capacity by human adipose-derived stromal cells. Sci Rep. 2017;7:44398. doi:10.1038/srep44398
73. Al-Yafeai Z, Yurdagul A, Peretik JM, Alfaidi M, Murphy PA, Orr AW. Endothelial FN (Fibronectin) deposition by alpha5beta1 integrins drives atherogenic inflammation. Arterioscler Thromb Vasc Biol. 2018;38:2601–2614. doi:10.1161/ATVBAHA.118.311705
74. Bachsais M, Naddaf N, Yacoub D, et al. The interaction of CD154 with the alpha5beta1 integrin inhibits fas-induced T cell death. PLoS One. 2016;11:e0158987.
75. Gingras MC, Roussel E, Bruner JM, Branch CD, Moser RP. Comparison of cell adhesion molecule expression between glioblastoma multiforme and autologous normal brain tissue. J Neuroimmunol. 1995;57:143–153. doi:10.1016/0165-5728(94)00178-Q
76. Fujita M, Yamamoto T, Iyoda T, et al. Autocrine production of PDGF stimulated by the tenascin-C-derived peptide TNIIIA2 induces hyper-proliferation in glioblastoma cells. Int J Mol Sci. 2019;20:3183. doi:10.3390/ijms20133183
77. Renner G, Janouskova H, Noulet F, et al. Integrin alpha5beta1 and p53 convergent pathways in the control of anti-apoptotic proteins PEA-15 and survivin in high-grade glioma. Cell Death Differ. 2016;23:640–653. doi:10.1038/cdd.2015.131
78. Murillo CA, Rychahou PG, Evers BM. Inhibition of alpha5 integrin decreases PI3K activation and cell adhesion of human colon cancers. Surgery. 2004;136:143–149. doi:10.1016/j.surg.2004.04.006
79. Toquet C, Colson A, Jarry A, et al. ADAM15 to alpha5beta1 integrin switch in colon carcinoma cells: a late event in cancer progression associated with tumor dedifferentiation and poor prognosis. Int J Cancer. 2012;130:278–287. doi:10.1002/ijc.25891
80. Morozevich GE, Kozlova NI, Ushakova NA, Preobrazhenskaia ME, Berman AE. [Implication of integrin alpha5beta1 in human breast carcinoma apoptosis and drug resistance]. Biomed Khim. 2011;57:77–84. Russian. doi:10.18097/pbmc20115701077
81. Mierke CT, Frey B, Fellner M, Herrmann M, Fabry B. Integrin alpha5beta1 facilitates cancer cell invasion through enhanced contractile forces. J Cell Sci. 2011;124:369–383. doi:10.1242/jcs.071985
82. Veine DM, Yao H, Stafford DR, Fay KS, Livant DL. A D-amino acid containing peptide as a potent, noncovalent inhibitor of alpha5beta1 integrin in human prostate cancer invasion and lung colonization. Clin Exp Metastasis. 2014;31:379–393. doi:10.1007/s10585-013-9634-1
83. Ryu SH, Heo SH, Park EY, et al. Selumetinib inhibits melanoma metastasis to mouse liver via suppression of EMT-targeted genes. Anticancer Res. 2017;37:607–614. doi:10.21873/anticanres.11354
84. Adachi M, Taki T, Higashiyama M, Kohno N, Inufusa H, Miyake M. Significance of integrin alpha5 gene expression as a prognostic factor in node-negative non-small cell lung cancer. Clin Cancer Res. 2000;6:96–101.
85. Han JY, Kim HS, Lee SH, Park WS, Lee JY, Yoo NJ. Immunohistochemical expression of integrins and extracellular matrix proteins in non-small cell lung cancer: correlation with lymph node metastasis. Lung Cancer. 2003;41:65–70. doi:10.1016/S0169-5002(03)00146-6
86. Wang HY, Chen Z, Wang ZH, Wang H, Huang LM. Prognostic significance of alpha5beta1-integrin expression in cervical cancer. Asian Pac J Cancer Prev. 2013;14:3891–3895. doi:10.7314/APJCP.2013.14.6.3891
87. Zhu H, Chen A, Li S, et al. Predictive role of galectin-1 and integrin alpha5beta1 in cisplatin-based neoadjuvant chemotherapy of bulky squamous cervical cancer. Biosci Rep. 2017;37. doi:10.1042/BSR20170958
88. Villegas-Pineda JC, Garibay-Cerdenares OL, Hernandez-Ramirez VI, et al. Integrins and haptoglobin: molecules overexpressed in ovarian cancer. Pathol Res Pract. 2015;211:973–981. doi:10.1016/j.prp.2015.10.002
89. Xie X, Long L, Wang H, Zheng Y, Liu S. The specifical inhibition of the expression of integrin alpha5/beta1 probably enhances the treatment effects and improves the prognosis of epithelial ovarian cancer. Med Hypotheses. 2015;84:68–71. doi:10.1016/j.mehy.2014.11.018
90. He S, Huang Q, Hu J, et al. EWS-FLI1-mediated tenascin-C expression promotes tumour progression by targeting MALAT1 through integrin alpha5beta1-mediated YAP activation in Ewing sarcoma. Br J Cancer. 2019;121:922–933. doi:10.1038/s41416-019-0608-1
91. Yi L, Hu Q, Zhou J, Liu Z, Li H. Alternative splicing of Ikaros regulates the FUT4/Le(X)-alpha5beta1 integrin-FAK axis in acute lymphoblastic leukemia. Biochem Biophys Res Commun. 2019;510:128–134. doi:10.1016/j.bbrc.2019.01.064
92. Kuonen F, Surbeck I, Sarin KY, et al. TGFbeta, fibronectin and integrin alpha5beta1 promote invasion in basal cell carcinoma. J Invest Dermatol. 2018;138:2432–2442. doi:10.1016/j.jid.2018.04.029
93. Lin L, Yan F, Zhao D, et al. Reelin promotes the adhesion and drug resistance of multiple myeloma cells via integrin beta1 signaling and STAT3. Oncotarget. 2016;7:9844–9858. doi:10.18632/oncotarget.7151
94. Adil MM, Levine RM, Kokkoli E. Increasing cancer-specific gene expression by targeting overexpressed alpha5beta1 integrin and upregulated transcriptional activity of NF-kappaB. Mol Pharm. 2014;11:849–858. doi:10.1021/mp400535v
95. Odagiri H, Kadomatsu T, Endo M, et al. The secreted protein ANGPTL2 promotes metastasis of osteosarcoma cells through integrin alpha5beta1, p38 MAPK, and matrix metalloproteinases. Sci Signal. 2014;7:ra7. doi:10.1126/scisignal.2004612
96. Amaral Pereira AL, Lopes FF, da Cruz MC, et al. Role of integrins in the carcinogenesis of squamous cell carcinoma of the tongue and lower lip. Appl Immunohistochem Mol Morphol. 2013;21:154–158.
97. Jung AC, Ray AM, Ramolu L, et al. Caveolin-1-negative head and neck squamous cell carcinoma primary tumors display increased epithelial to mesenchymal transition and prometastatic properties. Oncotarget. 2015;6:41884–41901.
98. Okamoto T, Iwata S, Yamazaki H, et al. CD9 negatively regulates CD26 expression and inhibits CD26-mediated enhancement of invasive potential of malignant mesothelioma cells. PLoS One. 2014;9:e86671. doi:10.1371/journal.pone.0086671
99. Yeh YC, Chang WL, Yang HB, Cheng HC, Wu JJ, Sheu BS. H. pylori cagL amino acid sequence polymorphism Y58E59 induces a corpus shift of gastric integrin alpha5beta1 related with gastric carcinogenesis. Mol Carcinog. 2011;50:751–759. doi:10.1002/mc.20753
100. Utispan K, Sonongbua J, Thuwajit P, et al. Periostin activates integrin alpha5beta1 through a PI3K/Akt-dependent pathway in invasion of cholangiocarcinoma. Int J Oncol. 2012;41:1110–1118. doi:10.3892/ijo.2012.1530
101. Morozevich GE, Kozlova NI, Ushakova NA, Preobrazhenskaya ME, Berman AE. Integrin alpha5beta1 simultaneously controls EGFR-dependent proliferation and Akt-dependent pro-survival signaling in epidermoid carcinoma cells. Aging (Albany NY). 2012;4:368–374. doi:10.18632/aging.100457
102. Tang CH, Keng YT, Liu JF. HMGB-1 induces cell motility and alpha5beta1 integrin expression in human chondrosarcoma cells. Cancer Lett. 2012;322:98–106. doi:10.1016/j.canlet.2012.02.014
103. Wu L, Bernard-Trifilo JA, Lim Y, et al. Distinct FAK-Src activation events promote alpha5beta1 and alpha4beta1 integrin-stimulated neuroblastoma cell motility. Oncogene. 2008;27:1439–1448. doi:10.1038/sj.onc.1210770
104. Chaurasia P, Aguirre-Ghiso JA, Liang OD, Gardsvoll H, Ploug M, Ossowski L. A region in urokinase plasminogen receptor domain III controlling a functional association with alpha5beta1 integrin and tumor growth. J Biol Chem. 2006;281:14852–14863. doi:10.1074/jbc.M512311200
105. Lynch L, Vodyanik PI, Boettiger D, Guvakova MA. Insulin-like growth factor I controls adhesion strength mediated by alpha5beta1 integrins in motile carcinoma cells. Mol Biol Cell. 2005;16:51–63. doi:10.1091/mbc.e04-05-0399
106. Jayne DG, Heath RM, Dewhurst O, Scott N, Guillou PJ. Extracellular matrix proteins and chemoradiotherapy: alpha5beta1 integrin as a predictive marker in rectal cancer. Eur J Surg Oncol. 2002;28:30–36. doi:10.1053/ejso.2001.1182
107. Zhang GJ, Crist SA, McKerrow AK, et al. production by human transitional carcinoma cells upregulates expression of the alpha5beta1 fibronectin receptor. J Urol. 2000;163:1553–1559. doi:10.1016/S0022-5347(05)67678-1
108. Schirner M, Herzberg F, Schmidt R, et al. Integrin alpha5beta1: a potent inhibitor of experimental lung metastasis. Clin Exp Metastasis. 1998;16:427–435. doi:10.1023/A:1006581424490
109. Schmidt R, Streit M, Kaiser R, et al. De novo expression of the alpha5beta1-fibronectin receptor in HT29 colon-cancer cells reduces activity of C-SRC. Increase of C-SRC activity by attachment on fibronectin. Int J Cancer. 1998;76:91–98. doi:10.1002/(SICI)1097-0215(19980330)76:1<91::AID-IJC15>3.0.CO;2-J
110. O’Brien V, Frisch SM, Juliano RL. Expression of the integrin alpha 5 subunit in HT29 colon carcinoma cells suppresses apoptosis triggered by serum deprivation. Exp Cell Res. 1996;224:208–213. doi:10.1006/excr.1996.0130
111. Chantret I, Barbat A, Dussaulx E, Brattain MG, Zweibaum A. Epithelial polarity, villin expression, and enterocytic differentiation of cultured human colon carcinoma cells: a survey of twenty cell lines. Cancer Res. 1988;48:1936–1942.
112. Beliveau A, Berube M, Rousseau A, Pelletier G, Guerin SL. Expression of integrin alpha5beta1 and MMPs associated with epithelioid morphology and malignancy of uveal melanoma. Invest Ophthalmol Vis Sci. 2000;41:2363–2372.
113. Beliveau A, Berube M, Carrier P, Mercier C, Guerin SL. Tumorigenicity of the mixed spindle-epithelioid SP6.5 and epithelioid TP17 uveal melanoma cell lines is differentially related to alpha5beta1 integrin expression. Invest Ophthalmol Vis Sci. 2001;42:3058–3065.
114. Landreville S, Vigneault F, Bergeron MA, et al. Suppression of alpha5 gene expression is closely related to the tumorigenic properties of uveal melanoma cell lines. Pigment Cell Melanoma Res. 2011;24:643–655. doi:10.1111/j.1755-148X.2011.00869.x
115. Carmeliet P. Angiogenesis in life, disease and medicine. Nature. 2005;438:932–936. doi:10.1038/nature04478
116. George EL, Georges-Labouesse EN, Patel-King RS, Rayburn H, Hynes RO. Defects in mesoderm, neural tube and vascular development in mouse embryos lacking fibronectin. Development. 1993;119:1079–1091.
117. Bloch W, Forsberg E, Lentini S, et al. Beta 1 integrin is essential for teratoma growth and angiogenesis. J Cell Biol. 1997;139:265–278. doi:10.1083/jcb.139.1.265
118. van der Flier A, Badu-Nkansah K, Whittaker CA, et al. Endothelial alpha5 and alphav integrins cooperate in remodeling of the vasculature during development. Development. 2010;137:2439–2449. doi:10.1242/dev.049551
119. Turner CJ, Badu-Nkansah K, Crowley D, van der Flier A, Hynes RO. alpha5 and alphav integrins cooperate to regulate vascular smooth muscle and neural crest functions in vivo. Development. 2015;142:797–808. doi:10.1242/dev.117572
120. Kim S, Bell K, Mousa SA, Varner JA. Regulation of angiogenesis in vivo by ligation of integrin alpha5beta1 with the central cell-binding domain of fibronectin. Am J Pathol. 2000;156:1345–1362. doi:10.1016/S0002-9440(10)65005-5
121. Magnussen A, Kasman IM, Norberg S, Baluk P, Murray R, McDonald DM. Rapid access of antibodies to alpha5beta1 integrin overexpressed on the luminal surface of tumor blood vessels. Cancer Res. 2005;65:2712–2721. doi:10.1158/0008-5472.CAN-04-2691
122. Tjong WY, Lin HH. The role of the RGD motif in CD97/ADGRE5-and EMR2/ADGRE2-modulated tumor angiogenesis. Biochem Biophys Res Commun. 2019;520:243–249. doi:10.1016/j.bbrc.2019.09.113
123. Lee HS, Oh SJ, Lee KH, et al. Gln-362 of angiopoietin-2 mediates migration of tumor and endothelial cells through association with alpha5beta1 integrin. J Biol Chem. 2014;289:31330–31340. doi:10.1074/jbc.M114.572594
124. Sudhakar A, Sugimoto H, Yang C, Lively J, Zeisberg M, Kalluri R. Human tumstatin and human endostatin exhibit distinct antiangiogenic activities mediated by alpha v beta 3 and alpha 5 beta 1 integrins. Proc Natl Acad Sci U S A. 2003;100:4766–4771. doi:10.1073/pnas.0730882100
125. Avraamides CJ, Garmy-Susini B, Varner JA. Integrins in angiogenesis and lymphangiogenesis. Nat Rev Cancer. 2008;8:604–617. doi:10.1038/nrc2353
126. Boudreau NJ, Varner JA. The homeobox transcription factor Hox D3 promotes integrin alpha5beta1 expression and function during angiogenesis. J Biol Chem. 2004;279:4862–4868. doi:10.1074/jbc.M305190200
127. Krayem N, Abdelkefi-Koubaa Z, Marrakchi N, Gargouri Y, Luis J. Native and recombinant phospholipases A2 of Scorpio maurus venom glands impair angiogenesis by targeting integrins alpha5beta1 and alphavbeta3. Int J Biol Macromol. 2018;116:305–315. doi:10.1016/j.ijbiomac.2018.04.141
128. Bosnjak M, Dolinsek T, Cemazar M, et al. Gene electrotransfer of plasmid AMEP, an integrin-targeted therapy, has antitumor and antiangiogenic action in murine B16 melanoma. Gene Ther. 2015;22:578–590. doi:10.1038/gt.2015.26
129. Kumar CC, Malkowski M, Yin Z, et al. Inhibition of angiogenesis and tumor growth by SCH221153, a dual alpha(v)beta3 and alpha(v)beta5 integrin receptor antagonist. Cancer Res. 2001;61:2232–2238.
130. Ramakrishnan V, Bhaskar V, Law DA, et al. Preclinical evaluation of an anti-alpha5beta1 integrin antibody as a novel anti-angiogenic agent. J Exp Ther Oncol. 2006;5:273–286.
131. Kiwanuka E, Andersson L, Caterson EJ, Junker JP, Gerdin B, Eriksson E. CCN2 promotes keratinocyte adhesion and migration via integrin alpha5beta1. Exp Cell Res. 2013;319:2938–2946. doi:10.1016/j.yexcr.2013.08.021
132. Mitra AK, Sawada K, Tiwari P, Mui K, Gwin K, Lengyel E. Ligand-independent activation of c-Met by fibronectin and alpha(5)beta(1)-integrin regulates ovarian cancer invasion and metastasis. Oncogene. 2011;30:1566–1576. doi:10.1038/onc.2010.532
133. Pal S, Ganguly KK, Moulik S, Chatterjee A. Modulation of MMPs by cell surface integrin receptor alpha5beta1. Anticancer Agents Med Chem. 2012;12:726–732. doi:10.2174/187152012802650183
134. Morozevich G, Kozlova N, Cheglakov I, Ushakova N, Berman A. Integrin alpha5beta1 controls invasion of human breast carcinoma cells by direct and indirect modulation of MMP-2 collagenase activity. Cell Cycle. 2009;8:2219–2225. doi:10.4161/cc.8.14.8980
135. Kesanakurti D, Chetty C, Dinh DH, Gujrati M, Rao JS. Role of MMP-2 in the regulation of IL-6/Stat3 survival signaling via interaction with alpha5beta1 integrin in glioma. Oncogene. 2013;32:327–340. doi:10.1038/onc.2012.52
136. Sil H, Sen T, Chatterjee A. Fibronectin-integrin (alpha5beta1) modulates migration and invasion of murine melanoma cell line B16F10 by involving MMP-9. Oncol Res. 2011;19:335–348. doi:10.3727/096504011X13079697132925
137. Bax DV, Messent AJ, Tart J, et al. Integrin alpha5beta1 and ADAM-17 interact in vitro and co-localize in migrating HeLa cells. J Biol Chem. 2004;279:22377–22386. doi:10.1074/jbc.M400180200
138. Machado-Pineda Y, Cardenes B, Reyes R, et al. CD9 controls integrin alpha5beta1-mediated cell adhesion by modulating its association with the metalloproteinase ADAM17. Front Immunol. 2018;9:2474. doi:10.3389/fimmu.2018.02474
139. Nakaoka HJ, Tanei Z, Hara T, et al. Mint3-mediated L1CAM expression in fibroblasts promotes cancer cell proliferation via integrin alpha5beta1 and tumour growth. Oncogenesis. 2017;6:e334. doi:10.1038/oncsis.2017.27
140. Yaqoob U, Cao S, Shergill U, et al. Neuropilin-1 stimulates tumor growth by increasing fibronectin fibril assembly in the tumor microenvironment. Cancer Res. 2012;72:4047–4059. doi:10.1158/0008-5472.CAN-11-3907
141. Janouskova H, Ray AM, Noulet F, et al. Activation of p53 pathway by Nutlin-3a inhibits the expression of the therapeutic target alpha5 integrin in colon cancer cells. Cancer Lett. 2013;336:307–318. doi:10.1016/j.canlet.2013.03.018
142. Martinkova E, Maglott A, Leger DY, et al. alpha5beta1 integrin antagonists reduce chemotherapy-induced premature senescence and facilitate apoptosis in human glioblastoma cells. Int J Cancer. 2010;127:1240–1248. doi:10.1002/ijc.25187
143. Aguirre-Ghiso JA, Estrada Y, Liu D, Ossowski L. ERK(MAPK) activity as a determinant of tumor growth and dormancy; regulation by p38(SAPK). Cancer Res. 2003;63:1684–1695.
144. Bonneau C, Rouzier R, Geyl C, et al. Predictive markers of chemoresistance in advanced stages epithelial ovarian carcinoma. Gynecol Oncol. 2015;136:112–120. doi:10.1016/j.ygyno.2014.10.024
145. Damiano JS, Hazlehurst LA, Dalton WS. Cell adhesion-mediated drug resistance (CAM-DR) protects the K562 chronic myelogenous leukemia cell line from apoptosis induced by BCR/ABL inhibition, cytotoxic drugs, and gamma-irradiation. Leukemia. 2001;15:1232–1239. doi:10.1038/sj.leu.2402179
146. Cohen E, Tendler T, Lu H, et al. Collagen I provides a survival advantage to MD-1483 head and neck squamous cell carcinoma cells through phosphoinositol 3-kinase signaling. Anticancer Res. 2013;33:379–386.
147. Hu Z, Gao S, Gao J, et al. Elevated levels of Lewis y and integrin alpha5beta1 correlate with chemotherapeutic drug resistance in epithelial ovarian carcinoma. Int J Mol Sci. 2012;13:15588–15600. doi:10.3390/ijms131215588
148. Ye DJ, Kwon YJ, Shin S, Baek HS, Shin DW, Chun YJ. Induction of integrin signaling by steroid sulfatase in human cervical cancer cells. Biomol Ther (Seoul). 2017;25:321–328. doi:10.4062/biomolther.2016.155
149. Guo W, Giancotti FG. Integrin signalling during tumour progression. Nat Rev Mol Cell Biol. 2004;5:816–826. doi:10.1038/nrm1490
150. Lahlou H, Muller WJ. beta1-integrins signaling and mammary tumor progression in transgenic mouse models: implications for human breast cancer. Breast Cancer Res. 2011;13:229. doi:10.1186/bcr2905
151. Meng XN, Jin Y, Yu Y, et al. Characterisation of fibronectin-mediated FAK signalling pathways in lung cancer cell migration and invasion. Br J Cancer. 2009;101:327–334. doi:10.1038/sj.bjc.6605154
152. Dia VP, Gonzalez de Mejia E. Lunasin potentiates the effect of oxaliplatin preventing outgrowth of colon cancer metastasis, binds to alpha5beta1 integrin and suppresses FAK/ERK/NF-kappaB signaling. Cancer Lett. 2011;313:167–180. doi:10.1016/j.canlet.2011.09.002
153. Zeng F, Luo F, Lv S, et al. A monoclonal antibody targeting neuropilin-1 inhibits adhesion of MCF7 breast cancer cells to fibronectin by suppressing the FAK/p130cas signaling pathway. Anticancer Drugs. 2014;25:663–672.
154. Lv X, Li Z, Guan J, et al. ATN-161 reduces virus proliferation in PHEV-infected mice by inhibiting the integrin alpha5beta1-FAK signaling pathway. Vet Microbiol. 2019;233:147–153. doi:10.1016/j.vetmic.2019.04.029
155. Grzesiak JJ, Smith KC, Burton DW, Deftos LJ, Bouvet M. GSK3 and PKB/Akt are associated with integrin-mediated regulation of PTHrP, IL-6 and IL-8 expression in FG pancreatic cancer cells. Int J Cancer. 2005;114:522–530. doi:10.1002/ijc.20748
156. Lu M, Wu J, Hao ZW, et al. Basolateral CD147 induces hepatocyte polarity loss by E-cadherin ubiquitination and degradation in hepatocellular carcinoma progress. Hepatology. 2018;68:317–332. doi:10.1002/hep.29798
157. Saidak Z, Le Henaff C, Azzi S, et al. Wnt/beta-catenin signaling mediates osteoblast differentiation triggered by peptide-induced alpha5beta1 integrin priming in mesenchymal skeletal cells. J Biol Chem. 2015;290:6903–6912. doi:10.1074/jbc.M114.621219
158. Xia Y, Shen S, Verma IM. NF-kappaB, an active player in human cancers. Cancer Immunol Res. 2014;2:823–830. doi:10.1158/2326-6066.CIR-14-0112
159. Han SW, Roman J. Fibronectin induces cell proliferation and inhibits apoptosis in human bronchial epithelial cells: pro-oncogenic effects mediated by PI3-kinase and NF-kappa B. Oncogene. 2006;25:4341–4349. doi:10.1038/sj.onc.1209460
160. Guo M, Sahni SK, Sahni A, Francis CW. Fibrinogen regulates the expression of inflammatory chemokines through NF-kappaB activation of endothelial cells. Thromb Haemost. 2004;92:858–866. doi:10.1160/TH04-04-0261
161. Moroishi T, Hansen CG, Guan KL. The emerging roles of YAP and TAZ in cancer. Nat Rev Cancer. 2015;15:73–79. doi:10.1038/nrc3876
162. Zanconato F, Battilana G, Cordenonsi M, Piccolo S. YAP/TAZ as therapeutic targets in cancer. Curr Opin Pharmacol. 2016;29:26–33. doi:10.1016/j.coph.2016.05.002
163. Li B, He J, Lv H, et al. c-Abl regulates YAPY357 phosphorylation to activate endothelial atherogenic responses to disturbed flow. J Clin Invest. 2019;129:1167–1179. doi:10.1172/JCI122440
164. Yun S, Hu R, Schwaemmle ME, et al. Integrin alpha5beta1 regulates PP2A complex assembly through PDE4D in atherosclerosis. J Clin Invest. 2019;130:4863–4874. doi:10.1172/JCI127692
165. Hwang JH, Sung JS, Kim JM, et al. Caveolin-1-dependent and -independent uPAR signaling pathways contribute to ganglioside GT1b induced early apoptosis in A549 lung cancer cells. Am J Cancer Res. 2014;4:801–810.
166. Wei Y, Tang CH, Kim Y, et al. Urokinase receptors are required for alpha 5 beta 1 integrin-mediated signaling in tumor cells. J Biol Chem. 2007;282:3929–3939. doi:10.1074/jbc.M607989200
167. Chaurasia P, Mezei M, Zhou MM, Ossowski L. Computer aided identification of small molecules disrupting uPAR/alpha5beta1–integrin interaction: a new paradigm for metastasis prevention. PLoS One. 2009;4:e4617. doi:10.1371/journal.pone.0004617
168. Neubauer S, Rechenmacher F, Beer AJ, et al. Selective imaging of the angiogenic relevant integrins alpha5beta1 and alphavbeta3. Angew Chem Int Ed Engl. 2013;52:11656–11659. doi:10.1002/anie.201306376
169. D’Alessandria C, Pohle K, Rechenmacher F, et al. In vivo biokinetic and metabolic characterization of the (6)(8)Ga-labelled alpha5beta1-selective peptidomimetic FR366. Eur J Nucl Med Mol Imaging. 2016;43:953–963. doi:10.1007/s00259-015-3218-z
170. Kapp TG, Di Leva FS, Notni J, et al. N-methylation of isoDGR peptides: discovery of a selective alpha5beta1-integrin ligand as a potent tumor imaging agent. J Med Chem. 2018;61:2490–2499. doi:10.1021/acs.jmedchem.7b01752
171. Gao H, Luo C, Yang G, et al. Improved in vivo targeting capability and pharmacokinetics of (99m)Tc-labeled isoDGR by dimerization and albumin-binding for glioma imaging. Bioconjug Chem. 2019;30:2038–2048. doi:10.1021/acs.bioconjchem.9b00323
172. Zhao H, Gao H, Zhai L, et al. (99m)Tc-HisoDGR as a potential SPECT probe for orthotopic glioma detection via targeting of integrin alpha5beta1. Bioconjug Chem. 2016;27:1259–1266. doi:10.1021/acs.bioconjchem.6b00098
173. Zhou J, Rossi J. Aptamers as targeted therapeutics: current potential and challenges. Nat Rev Drug Discov. 2017;16:181–202.
174. Bouvier-Muller A, Duconge F. Application of aptamers for in vivo molecular imaging and theranostics. Adv Drug Deliv Rev. 2018;134:94–106. doi:10.1016/j.addr.2018.08.004
175. Fechter P, Cruz Da Silva E, Mercier MC, et al. RNA aptamers targeting integrin alpha5beta1 as probes for cyto- and histofluorescence in glioblastoma. Mol Ther Nucleic Acids. 2019;17:63–77. doi:10.1016/j.omtn.2019.05.006
176. Nam JM, Onodera Y, Bissell MJ, Park CC. Breast cancer cells in three-dimensional culture display an enhanced radioresponse after coordinate targeting of integrin alpha5beta1 and fibronectin. Cancer Res. 2010;70:5238–5248. doi:10.1158/0008-5472.CAN-09-2319
177. Ricart AD, Tolcher AW, Liu G, et al. Volociximab, a chimeric monoclonal antibody that specifically binds alpha5beta1 integrin: a Phase I, pharmacokinetic, and biological correlative study. Clin Cancer Res. 2008;14:7924–7929. doi:10.1158/1078-0432.CCR-08-0378
178. Besse B, Tsao LC, Chao DT, et al. Phase Ib safety and pharmacokinetic study of volociximab, an anti-alpha5beta1 integrin antibody, in combination with carboplatin and paclitaxel in advanced non-small-cell lung cancer. Ann Oncol. 2013;24:90–96. doi:10.1093/annonc/mds281
179. Bell-McGuinn KM, Matthews CM, Ho SN, et al. A Phase II, single-arm study of the anti-alpha5beta1 integrin antibody volociximab as monotherapy in patients with platinum-resistant advanced epithelial ovarian or primary peritoneal cancer. Gynecol Oncol. 2011;121:273–279. doi:10.1016/j.ygyno.2010.12.362
180. Weekes CD, Rosen LS, Capasso A, et al. Phase I study of the anti-alpha5beta1 monoclonal antibody MINT1526A with or without bevacizumab in patients with advanced solid tumors. Cancer Chemother Pharmacol. 2018;82:339–351. doi:10.1007/s00280-018-3622-8
181. Joshi R, Ren W, Mathew P. A bispecific antibody targeting the alphav and alpha5beta1 integrins induces integrin degradation in prostate cancer cells and is superior to monospecific antibodies. Mol Cancer Res. 2019.
182. Martin S, Cosset EC, Terrand J, Maglott A, Takeda K, Dontenwill M. Caveolin-1 regulates glioblastoma aggressiveness through the control of alpha(5)beta(1) integrin expression and modulates glioblastoma responsiveness to SJ749, an alpha(5)beta(1) integrin antagonist. Biochim Biophys Acta. 2009;1793:354–367. doi:10.1016/j.bbamcr.2008.09.019
183. Okazaki T, Ni A, Ayeni OA, et al. alpha5beta1 Integrin blockade inhibits lymphangiogenesis in airway inflammation. Am J Pathol. 2009;174:2378–2387. doi:10.2353/ajpath.2009.080942
184. Murphy PA, Begum S, Hynes RO, Yanagisawa H. Tumor angiogenesis in the absence of fibronectin or its cognate integrin receptors. PLoS One. 2015;10:e0120872. doi:10.1371/journal.pone.0120872
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.