Back to Journals » Diabetes, Metabolic Syndrome and Obesity » Volume 9
The relationship between the presence of lower urinary tract symptoms and waist circumference
Authors Oliveira M, Varella L , Angelo P, Micussi M
Received 11 February 2016
Accepted for publication 16 March 2016
Published 11 July 2016 Volume 2016:9 Pages 207—211
DOI https://doi.org/10.2147/DMSO.S106221
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Ming-Hui Zou
Maria Clara Eugênia de Oliveira, Larissa Ramalho Dantas Varella, Priscylla Helouyse melo Angelo, Maria Thereza Albuquerque Barbosa Cabral Micussi
Physical Therapy Department, Federal University of Rio Grande do Norte, Natal, Rio Grande do Norte, Brazil
Purpose: The aim of the study is to evaluate the pressure of the pelvic floor muscles (PFM) according to waist circumference (WC) and correlate the presence of urinary tract symptoms (UTS) with WC.
Patients and methods: The study was observational and cross-sectional. One-hundred and sixty-four females between 45 and 65 years of age were evaluated. The sample was divided into two groups, according to WC: Group ≤80 (G≤80) was composed of females in whom WC was up to 80 cm; and Group >80 (G>80) was composed of females with WC above 80 cm. The subjects were assessed in terms of sociodemographic data, pre-existing conditions, urogynecological and obstetric history, and the presence of lower UTS (LUTS), as well as physical examination, measurement of WC, height, and weight. The PFM assessment was made by perineometry. To compare the mean between groups, the independent samples t-test was applied, and to correlate the WC with perineometry and LUTS, the Pearson’s correlation test was used.
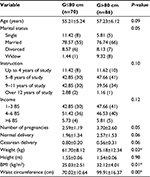
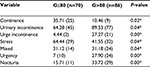
Results: The final sample was composed of 156 patients. The average age of participants was 55.21 (±24.5) years in G≤80 and 57.23 (±6.12) years in G>80. There were significant differences regarding the presence of LUTS between the groups (P<0.05); as to the perineometry, there was a significant difference (P=0.03) between the groups: 38.68±13.63 cmH2O for G≤80 and 30.11±11.20 cmH2O for G>80. There was a correlation between the presence of urinary urgency (r=0.7; P=0.00), nocturia (r=0.7; P=0.00), and urinary incontinence (r=0.9; P=0.00) with WC.
Conclusion: Females with larger abdominal diameter have a higher prevalence of LUTS such as urinary incontinence, nocturia, and urinary urgency, as well as a lower PFM pressure. Furthermore, a relationship between LUTS with WC was also observed.
Keywords: abdominal obesity, pelvic floor, urinary incontinence, females
Introduction
Lower urinary tract symptoms (LUTS) are subjective indicators of a disease or change in condition perceived by the patient and are generally qualitative indicators.1 Among the most common symptoms are urinary frequency and urgency, nocturia, and urinary incontinence (UI).2 Among the symptoms, UI has a greater prevalence and is defined, according to International Continence Society,1 as all involuntary loss of urine originating from different processes that alter its storage in the bladder. It can be classified as urinary urgency; UI accompanied by strong urge to urinate; stress UI, when the loss occurs after exercise, coughing, or sneezing; and mixed, when there are simultaneously stress and urge UI.3
It is known that the presence of LUTS may be associated with the absence or reduction of the strength of the pelvic floor muscles (PFM). These muscles have the role of supporting the pelvic organs, assisting in the sexual function, and maintaining urinary and fecal continence through the ability to contract strongly, fast, and in a reflex manner.4,5 The rapid and strong ability to contract those muscles, for example, generates an adequate closing pressure on the urethra, maintaining its pressure higher than that inside the bladder, thus preventing urine leakage.6
Literature shows that several risk factors are associated with the presence of LUTS, such as age, parity, mode of delivery, birth weight, gestational age, menopause, and obesity.7,8 Particularly in cases of UI, some studies have shown significant tendencies in females with high body mass index (BMI) for developing UI.9,10 This has been attributed to excess weight in the abdominal area, which increases the abdominal pressure, and, as a consequence, increases the pressure of the bladder and causes mobility of the urethra.11,12
It is important to note that although some females have high BMI, central adiposity is not necessarily observed. Central obesity can be defined as the accumulation of fat in the upper body, especially the abdomen, unlike femoral gluteal obesity, in which there is as much fat deposit at the lower body as in the gluteal region. Therefore, it is necessary to measure the waist circumference (WC) to identify the increase in abdominal fat.
Although there have been studies in the field related to the influence of BMI on PFM and UI, data for LUTS addressing WC as the central adiposity identification factor is lacking. Therefore, this study aims to evaluate and correlate the presence of urinary symptoms with WC and PFM pressure.
Patients and methods
The study design was observational and cross-sectional. The sample included 164 females between 45 and 65 years of age, who were assisted at the Maternity School Januário Cicco in Natal, RN, Brazil, divided into two groups according to WC: Group ≤80 (G≤80), was composed of females with WC up to 80 cm (n=70), and Group >80 (G>80) comprised females with WC >80 cm (n=86). Those who satisfied the following criteria were included: not using hormonal medication, not been subjected to gynecological surgery, not had previous physical therapy for pelvic dysfunction, and not doing any specific exercises for the PFM. Excluded were those with genital infection, prolapse grade III or IV identified by Pelvic Organ Prolapse Quantification System (POP-Q),13 anyone who complained about unbearable pain during muscle strength test and perineometry, and anyone who have had trouble contracting. The sample size was defined through the sample calculation for 80% power.
Data collection took place in three stages: 1) application form evaluation, in which sociodemographic data, information about pre-existing diseases, and urological, gynecological, and obstetrical history as well as the presence of LUTS (urgency, nocturia, UI, and type of UI) were collected; 2) anthropometric assessment, which measured the height and weight to calculate BMI. BMI was considered normal for values between 18.5 kg/m2 and 24.99 kg/m2; overweight for values between 25 kg/m2 and 29.99 kg/m2; and obese for values above 30 kg/m2. Also, WC was measured using a metric tape positioned at the midpoint between the last rib and the edge of the iliac crest; and 3) perineometry to assess the pressure exerted by PFM.
For perineometry, we used the PeritronÔ model 9300AV (Cardio Design, Castle Hill, NSW, Australia). The participants adopted the modified gynecological position (supine, knees flexed and hips abducted, with feet flat on the stretcher), and the condom-covered probe was inserted into the vagina to a depth of 9–10 cm. Three maximum voluntary contractions at 30-second intervals were requested.14 During the examination, if the patient did not perform the contraction correctly, the value was discarded. The value considered was the arithmetic average of the three attempts15 and expressed in cmH2O.
Statistical analysis was performed using SPSS for Windows 20.0 (IBM Corporation, Armonk, NY, USA), adopting a 5% significance level for all statistical procedures. To analyze the normality of the data, we used the Kolmogorov–Smirnov normality test. For mean comparisons between groups, we used the independent samples t-test, and to correlate the WC with perineometry and LUTS, we used the Pearson’s correlation test. The Research Ethics Committee of the Federal University of Rio Grande do Norte approved the study (approval no. 1042362). All participants read and signed the informed consent form before being evaluated.
Results
This study evaluated 164 females. However, 4.87% of the subjects (n=8) were excluded because of being unable to perform perineometry, noncoordination during the contraction of PFM, or pain when inserting the probe. During sociodemographic assessment, it was observed that there was no difference between the groups. Regarding physical examination, statistical difference between the weight, BMI, and WC was observed (Table 1).
Significant difference was observed in regard to the presence of LUTS when comparing the two groups (Table 2). Regarding the degree of UI, it was observed that the G>80 group presented it more significantly.
Regarding the pressure of PFM, we observed significant difference (P=0.03) between the groups: for G≤80, it was 38.68±13.63 cmH2O and for G>80 it was 30.11±11.20 cmH2O. There was a correlation between the presence of urinary urgency (r=0.7; P=0.00), nocturia (r=0.7; P=0.00), and UI (r= 0.9; P=0.00) with WC.
Discussion
This study aimed to evaluate the pressure of the PFM according to WC but also to correlate the presence of LUTS with WC. By dividing the subjects according to WC, it was possible to identify those with a larger abdominal diameter, which resulted in higher intra-abdominal pressure.
According to our findings, UI was more prevalent in the G>80 group. Among the forms of urinary loss, stress UI was the most referred to. An earlier study had shown that high levels of body weight with a high level of abdominal obesity were related to UI.16 Furthermore, central fat accumulation lead to a high WC and increased intra-abdominal pressure due to excess body fat.17 Therefore, the PFM can suffer significant influence of chronic stress, stretching and weakening of other structures of the pelvic floor.17
In the literature, it is reported that the severity of UI may be related to the degree of central rather than peripheral obesity.18–21 Therefore, the correlation of the bladder pressure to the circumference or sagittal abdominal diameter is larger than the waist/hip ratio.18 Thus, waist/hip ratio and WC may indicate a correlation with urinary urgency and incontinence,22,23 confirming the data in this work. The results of this study showed that the urinary urgency and urinary urge/incontinence were more frequent in the group with higher WC. It has been shown that adipose tissue produces leptin, which in turn increases the autonomic nervous activity, especially of the noradrenergic sympathetic nerves.24 An increase in perivesical fat, known as lipomatosis, can cause inflammation inside the bladder, leading to urinary frequency, and cause urgency to urinate.24 Moreover, visceral fat produces inflammatory cytokines, which can lead to irritation and, therefore, to the appearance of urinary symptoms.25
We also observed that mixed type of UI was also more frequent in the group with higher WC. Some authors have argued that this form of urinary leakage occurs in people with higher weight only due to mechanical factors;26 that is, increasing BMI and abdominal diameter increases intra-abdominal pressure and bladder pressure proportionally.18,27 However, it is also known that the release of cytokines by adipose tissue can promote the urgency to urinate and thereby contribute to mixed type of UI.25
Our data show that the group with the highest WC showed high frequency of symptoms such as urgency and nocturia. There have been studies showing that irritative bladder symptoms are reported more frequently in the obese population, especially in premenopausal females.28 Tension and weakness in the PFM can occur leading to a deficit in the nerve supply of the support structures which can cause ineffective function.28,29
Previous research evaluating the strength and endurance of PMF in obese and non-obese patients agree with our findings, which concluded that besides a lower muscle response of obese, an increase was observed in symptoms of nocturia associated with increased BMI.19–21,30,31
It is worth emphasizing that patients with a higher WC had a lower PFM pressure compared to the group with WC ≤80. According to the completeness theory proposed by Petros and Ulmsten, in order to have a properly functioning PFM, it is necessary to have completeness of PFM apart from other structures that compose this region such as nerves, fascia, and ligaments.32 In this sense, the overload, that comes from overweight in the waist region, may damage the PFM as well as the other structures that compose it, and so provoke dysfunctions such as UI and other symptoms that routinely coexist, such as urinary frequency (polyuria), nocturia, chronic pelvic pain, and changes in bowel and vesical emptying.10,32
In this context, it is observed in the literature that reducing the WC, resulting from bariatric surgery, resolves LUTS by up to 52%.33 In this context, a question for further research is: What is the effect of strengthening the PFM for the treatment of LUTS with persistence of BMI and high WC?
Unlike other studies that address only the urinary losses and BMI, this study aimed to evaluate WC and its relationship with LUTS, without specifying the types of UI. However, this study was limited to assessing WC only and did not consider the hip circumference or the relationship of WC and hip circumference.
Conclusion
Our results suggest that females with a higher WC suffer more from LUTS, and there is a strong positive relationship between irritative symptoms/UI and larger abdominal diameter. The reasons for these findings are probably not only linked to the direct increase of BMI but also to the accumulation of abdominal fat which can lead to increased intra-abdominal pressure on PFM and also neuroendocrine changes.
Disclosure
The authors report no conflicts of interest in this work.
References
Sand PK, Dmochowski R. Analysis of the standardisation of terminology of lower urinary tract dysfunction: report from the standardisation sub-committee of the International Continence Society. Neurourol Urodyn. 2002;21(1):167–178. | ||
Greer WJ, Richter HE, Bartolucci AA, Burgio KL. Obesity and pelvic floor disorders: a systematic review. Obstet Gynecol. 2008;112(2 pt 1):341–349. | ||
Baracho E. Fisioterapia aplicada à saúde da mulher. 5th ed. Rio de Janeiro: Guanabara Koogan; 2012. | ||
Shishido K, Peng Q, Jones R, Omata S, Constantinou CE. Influence of pelvic floor muscle contraction on the profile of vaginal closure pressure in continente and stress urinary incontinente women. J Urol. 2008;179(5):1917–1922. | ||
Deffieux X, Hubeaux K, Porcher R, Ismael SS, Raibaut P, Amarenco G. Abnormal pelvic response to cough in women with stress urinary incontinence. Neurourol Urodyn. 2008;27(4):291–296. | ||
Miller J, Kasper C, Sampselle C. Review of muscle physiology with application to pelvic muscle exercise. Urol Nurs. 1994;14(3):92–97. | ||
Minassian VA, Drutz HP, Al-Badr A. Urinary incontinence as a worldwide problem. Int J Gynaecol Obstet. 2003;82(3):327–338. | ||
Qiu J, Lv L, Lin X, et al. Bodymass index, recreational physical activity and female urinary incontinence in Gansu, China. Eur J Obstet Gynecol Reprod Biol. 2011;159(1):224–229. | ||
Townsend MK, Curhan GC, Resnick NM, Grodstein F. BMI, waist circumference, and incident urinary incontinence in older women. Obesity (Silver Spring). 2008;16(4):881–886. | ||
Jackson RA, Vittinghoff E, Kanaya AM, et al. Urinary incontinence in elderly women: findings from the Health, Aging, and Body Composition Study. Obstet Gynecol. 2004;104(2):301. | ||
Subak LL, Richter HE, Hunskaar S. Obesity and urinary incontinence: epidemiology and clinical research update. J Urol. 2009;182(6 suppl):S2–S7. | ||
Brown JS, Seeley DG, Fong J, Black DM, Ensrud KE, Grady D. Urinary incontinence in older women: who is at risk? Study of Osteoporotic Fractures Research Group. Obstet Gynecol. 1996;87(5 pt 1):715–721. | ||
Bump RC, Mattiasson A, Bo K, et al. The standardization of terminology of female pelvic organ prolapse and pelvic floor dysfunction. Am J Obestet Gynecol. 1996;175(1):10–17. | ||
Ferreira CH, Barbosa PB, de Oliveira Souza F, Antônio FI, Franco MM, Bø K. Inter-rater reliability study of the modified Oxford Grading Scale and the Peritron manometer. Physiotherapy. 2011;97(2):132–138. | ||
Da Roza T, Mascarenhas T, Araujo M, Trindade V, Jorge RN. Oxford Grading Scale vs manometer for assessment of pelvic floor strength in nulliparous sports students. Physiotherapy. 2013;99(3):207–211. | ||
Han MO, Lee NY, Park HS. Abdominal obesity is associated with stress urinary incontinence in Korean women. Int Urogynecol J Pelvic Floor Dysfunct. 2006;17(1):35–39. | ||
Lorenzi DRS, Basso E, Fagundes PO, Saciloto B. Prevalence of overweight and obesity among climacteric women [Prevalência de sobrepeso e obesidade no climatério]. Rev Bras Ginecol Obstet. 2005;27(8):479–484. Portuguese. | ||
Sugerman H, Windsor A, Bessos M, Wolfe L. Intra-abdominal pressure, sagittal abdominal diameter and obesity comorbidity. J Intern Med. 1997;241(1):71–79. | ||
Mommsen S, Foldspang A. Body mass index and adult female urinary incontinence. World J Urol. 1994;12(6):319–322. | ||
Chen CG, Gatmaitan P, Koepp S, et al. Obesity is associated with increased prevalence and severity of pelvic floor disorders in women considering bariatric surgery. Surg Obes Relat Dis. 2009;5(4): | ||
Whitcomb EL, Lukacz ES, Lawrence JM, Nager CW, Luber KM. Prevalence and degree of bother from pelvic floor disorders in obese women. Int Urogynecol J Pelvic Floor Dysfunct. 2009;20(3):289. | ||
Kim I-H, Chun H, Kwon J-W. Gender differences in the effect of obesity on chronic diseases among the elderly Koreans. J Korean Med Sci. 2011;26(2):250–257. | ||
Kuh D, Cardozo L, Hardy R. Urinary incontinence in middle aged women: childhood enuresis and other life time risk factors in a British prospective cohort. J Epidemiol Community Health. 1999;53(8):453–458. | ||
Tong RS, Larner T, Finlay M, Agarwal D, Costello AJ. Pelvic lipomatosis associated with proliferative cystitis occurring in two brothers. Urology. 2002;59(4):602. | ||
Link CL, Steers WD, Kusek JW, McKinlay JB. The association of adiposity and overactive bladder appears to differ by gender: results from the Boston Area Community Health survey. J Urol. 2011;185(3): | ||
Khullar V, Sexton CC, Thompson CL, Milsom I, Bitoun CE, Coyne KS. The relationship between BMI and urinary incontinence subgroups: results from EpiLUTS. Neurourol Urodyn. 2014;33(4):392–399. | ||
Noblett KL, Jensen JK, Ostergard DR. The relationship of body mass index to intra-abdominal pressure as measured by multichannel cystometry. Int Urogynecol J Pelvic Floor Dysfunct. 1997;8(6): | ||
Ramalingam K, Monga A. Obesity and pelvic floor dysfunction. Best Pract Res Clin Obstet Gynaecol. 2015;29(4):541–547. | ||
Alling Moller L, Lose G, Jorgensen TU. Risk factors for lower urinary tract symptoms in women 40 to 60 years of age. Obstet Gynecol. 2000;96(3):446–451. | ||
Higa R, Lopes MH, dos Reis MJ. Risk factors for urinary incontinence in women [Factores de riesgo para incontinencia urinaria em La mujer]. Rev Esc Enferm USP. 2008;42(1):187–192. Spanish. | ||
Silva JC, Prado MC, Fracon e Romão JF, Cestário CE. Pelvic floor muscular force degree in obese and nonobese incontinent women [Grau de força muscular do assoalho pélvico em mulheres incontinentes obesas e não obesas]. Rev Ciência Saúde. 2011;4(2):37–44. Portuguese. | ||
Petros PE, Ulmsten U. An integral theory of female urinary incontinence. Acta Obstet Gynecol Scand Suppl. 1990;153:7–31. | ||
Shimonov M, Groutz A, Schachter P, Gordon D. Is bariatric surgery the answer to urinary incontinence in obese women? Neurourol Urodyn. Epub 2015 Oct 16. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.