Back to Journals » Clinical Ophthalmology » Volume 12
The ophthalmic fallout in Utah after the Great American Solar Eclipse of 2017
Authors Ricks C , Montoya A, Pettey J
Received 6 June 2018
Accepted for publication 1 August 2018
Published 21 September 2018 Volume 2018:12 Pages 1853—1857
DOI https://doi.org/10.2147/OPTH.S174808
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Christopher Ricks,1 Alexandrea Montoya,2 Jeff Pettey1
1John A. Moran Eye Center, Salt Lake City, UT, USA; 2Department of Ophthalmology, University of Utah School of Medicine, Salt Lake City, UT, USA
Background: Solar retinopathy is a rare ophthalmic disorder resulting from sustained viewing of the sun without protective eyewear. Incidence of solar retinopathy typically increases following a solar eclipse due to attempted visualization of the sun without appropriate eye protection. This paper serves as a case series of all available reported cases of solar retinopathy present in Utah resulting from the August 21st, 2017 solar eclipse.
Methods: Twenty-seven patients had presented with concern for vision changes after the solar eclipse and six patients had exam findings consistent with solar retinopathy. Of these six cases, charts were available for three.
Results: The common finding in all cases was a central scotoma with a correlating change to the inner segment/outer segment junction on optical coherence tomography. Demographically, all three patients were young males.
Conclusion: This data provides insights on populations in Utah at risk for ophthalmic damage and can aid in targeting education programs in the future.
Keywords: OCT, ophthalmology, retina, solar retinopathy, solar eclipse
Introduction
Solar retinopathy is a rare ophthalmic disorder resulting from sustained view of the sun without protective eyewear. Symptoms are typically bilateral and are characterized by blurred vision, various patterns of scotoma, chromatopsia, headache, and photosensitivity.1,2 Though rare, numbers of patients presenting with solar retinopathy typically increase following a solar eclipse.2–7 Retinal injury as a result of unprotected sungazing occurs in the photoreceptor cells of the fovea and parafovea. It has been suggested that damage occurs secondarily to the disruption of photoreceptor outer segment membranes and mitochondria by oxidative damage as a result of electromagnetic radiation.3,8 High definition spectral domain optical coherence tomography (OCT), fluorescein angiography (FA), fundoscopy, and various visual acuity and contrast sensitivity tests are used to determine the severity of retinal damage.1–7,9 The vast majority of symptoms resolve within 1–3 months, but there are several documented cases of achromatopsia, central scotoma, and decreased visual acuity remaining 7 months to 1 year after exposure.7,10
On August 21st, 2017 a solar eclipse occurred within the view of the entire continental USA. The local academic ophthalmology clinic held several press conferences teaching about eye safety during the solar eclipse. There were also numerous print, radio and news reports that resulted from these press conferences. There was a high concern for counterfeit safety glasses and Amazon.com initiated a recall of many suspect glasses.11 Data regarding cases of solar retinopathy as a result of this eclipse is still limited. The aim of our case study is to explore all cases of solar retinopathy that presented after the 2017 solar eclipse in Utah. This data will be used to develop a better understanding of at-risk populations, develop improved educational materials of the risks of unprotected eclipse viewing, and to contribute to data regarding solar retinopathy patient outcomes.
Materials and methods
After approval was obtained from the University of Utah Institutional Review Board (IRB) Committee, patients with solar retinopathy secondary to sun gazing during the solar eclipse in August 2017 were found for this report through a short survey sent to 92% of ophthalmologists in Utah; there were approximately 135 surveys sent. The survey asked how many patients were seen with complaints of vision changes after the solar eclipse, how many patients had exam findings consistent with solar retinopathy, and if we could contact the provider to request the patient’s chart. If a provider did not respond, it was assumed that he or she did not have any cases of solar retinopathy. Not all charts were available for review. Once available charts were obtained, they were evaluated for the following criteria where applicable and available: age, gender, chief complaint, visual acuity, exam findings, OCT findings (CIRRUS™, Carl Zeiss Meditec, Dublin, CA, USA; SPECTRALIS®, Heidelberg Engineering, Franklin, MA, USA), FA findings, if eye protection was worn, if the eye protection worn was properly certified, time spent looking at the sun without eye protection, if pain was associated with the symptoms, if the symptoms resolved, and if there were any other eye problems.
Ethics approval
This study was done under IRB# 105,746 through the University of Utah and meets standards set by the Declaration of Helsinki. The University of Utah IRB did not feel that patient consent was required due to the fact that this study was done with de-identified information as a retrospective chart review and only one patient was available for follow up. Appropriate measures were taken to ensure patient data confidentiality.
Results
Of the 135 surveys sent out, 38 physicians responded. These 38 physicians reported 27 patients that had presented with concern for vision changes after the solar eclipse. Of these 27, six patients had exam findings consistent with solar retinopathy. Of these six cases, charts were available for three cases. These three cases are presented below.
Case 1
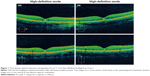
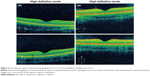
A 17-year-old male presented 24 hours after the eclipse with complaints of bilateral central scotoma after direct visualization of the eclipse for 15 seconds, three separate times. The patient demonstrated a visual acuity of 20/25 OU without correction. Funduscopic exam showed bilateral yellow foveal spots. Time-domain OCT (Cirrus™, Carl Zeiss Meditec) showed bilateral sub-foveal nodules (Figure 1) suggestive of possible changes to the ellipsoid zone. The patient presented at 2-month follow up with visual acuity of 20/25+2 OD and 20/20 OS without correction, and with a self-perceived vision improvement of 20% with persisting scotoma. Funduscopic exam was significant for a small yellow spot persisting within the fovea of the left eye but was otherwise normal. OCT (Cirrus™, Carl Zeiss Meditec) findings demonstrated resolution of previous findings (Figure 2).
Case 2
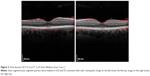
A 36-year-old-male presented 8 days after the eclipse with bilateral central scotoma with no eye pain. He stated that onset of symptoms began the day of the eclipse and were accompanied by a headache that had since subsided. The patient described direct visualization of the eclipse for 5–10 seconds followed by the use of solar eclipse viewing glasses for another 20 seconds. It is unknown if the glasses he used were properly certified. The patient demonstrated a visual acuity of 20/20+1 OD, and 20/15–3 OS. Funduscopic exam showed small central foveal lesions. Time-domain OCT (Cirrus™, Carl Zeiss Meditec) showed abnormal inner segment/outer segment (IS/OS) junctions bilaterally as well as foveal outer retinal abnormalities consistent with solar maculopathy (Figure 3). There was no data on follow up.
Case 3
A 21-year-old-male presented 36 days after the eclipse with vision changes in his right eye. He stated that he directly visualized the eclipse for 1 second. The patient demonstrated a visual acuity of 20/50 OD, and 20/20 OS. Funduscopic exam showed a red lesion in the macula surrounded by gray pigment OD, and no positive findings OS. Time-domain OCT (SPECTRALIS®, Heidelberg Engineering) showed a macular scar with disruption of rods and cones. FA was performed and showed window defects in the early and late macula. There was no data on follow up, and no figures are available.
Discussion
Despite millions of people across Utah viewing the solar eclipse on August 21, 2017, very few people were found to have ophthalmic damage from the solar eclipse. This can be attributed to effective education about the risks and ready access to eye protection. As far as the authors of this paper know, no one in Utah had eye damage from faulty solar eclipse viewing glasses. The one exception being the man in Case 2, as the certification status of his solar eclipse viewing glasses was unknown. All of the patients in the cases mentioned previously did not use safety glasses when they viewed the eclipse, which led directly to the development of solar retinopathy. The common finding in all cases was a central scotoma with a correlating change to the IS/OS junction on OCT. Changes in IS/OS junctions are typically caused by disruption of photoreceptor cells. This corresponds well with data suggesting that electromagnetic radiation contributes to photoreceptor damage through inducing oxidative damage.3,8 Demographically, all cases available and presented in this series were young males. The weaknesses of this case series include the lack of follow up information and the unavailability of patient records on three of the six confirmed cases of solar retinopathy. Our data possibly underestimates the incidence of solar retinopathy resulting from unprotected eclipse viewing as we cannot account for those patients with symptoms that chose to forego medical attention. Physicians that did not respond to the survey may have also provided more patient data. It is unknown what the prevalence of solar retinopathy is during a non-solar eclipse year. The next total solar eclipse will be July 2, 2019, visible from the South Pacific and South America. It will be important to continue the public education efforts that were seen in preparing for the Great American Solar Eclipse.
Conclusion
Although rare, solar retinopathy is highly preventable with education efforts. This case series provides insight into the rise in prevalence of cases following a solar eclipse, and populations at the greatest risk. Data can be used to tailor education programs.
Acknowledgments
Unrestricted grants from Research to Prevent Blindness and the Utah chapter of the Achievement Rewards for College Scientists Foundation.
Disclosure
The authors report no conflicts of interest in this work.
References
Bruè C, Mariotti C, de Franco E, Fisher Y, Guidotti JM, Giovannini A. Solar retinopathy: a multimodal analysis. Case Rep Ophthalmol Med. 2013;2013:1–5. | ||
Codenotti M, Patelli F, Brancato R. OCT findings in patients with retinopathy after watching a solar eclipse. Ophthalmologica. 2002;216(6):463–466. | ||
Chen KC, Jung JJ, Aizman A. High definition spectral domain optical coherence tomography findings in three patients with solar retinopathy and review of the literature. Open Ophthalmol J. 2012;6:29–35. | ||
Dhir SP, Gupta A, Jain IS. Eclipse retinopathy. Br J Ophthalmol. 1981;65(1):42–45. | ||
Khatib N, Knyazer B, Lifshitz T, Levy J. Acute eclipse retinopathy: a small case series. J Optom. 2014;7(4):225–228. | ||
Michaelides M, Rajendram R, Marshall J, Keightley S. Eclipse retinopathy. Eye. 2001;15(Pt 2):148–151. | ||
Wong SC, Eke T, Ziakas NG. Eclipse burns: a prospective study of solar retinopathy following the 1999 solar eclipse. Lancet. 2001;357(9251):199–200. | ||
Hope-Ross MW, Mahon GJ, Gardiner TA, Archer DB. Ultrastructural findings in solar retinopathy. Eye. 1993;7 (Pt 1):29–33. | ||
Jain A, Desai RU, Charalel RA, Quiram P, Yannuzzi L, Sarraf D. Solar retinopathy: comparison of optical coherence tomography (OCT) and fluorescein angiography (FA). Retina. 2009;29(9):1340–1345. | ||
Moran S, O’Donoghue E. Solar retinopathy secondary to sungazing. BMJ Case Rep. 2013;2013:bcr2012008402bcr2012008402. | ||
KSL.com. Amazon, John A. Moran Eye Center recall unverified eclipse glasses. Salt Lake City, UT: KSL Broadcasting Salt Lake City UT; 2017 [August 14]. Available from: https://www.ksl.com/?nid=148&sid=45418100. Accessed September 12, 2018. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.