Back to Journals » OncoTargets and Therapy » Volume 13
The Long Non-Coding RNA IDH1-AS1 Promotes Prostate Cancer Progression by Enhancing IDH1 Enzyme Activity
Authors Wu S, Ding L, Xu H, Gao J, Shao Y, Zhang S, Wei Z
Received 29 February 2020
Accepted for publication 25 June 2020
Published 7 August 2020 Volume 2020:13 Pages 7897—7906
DOI https://doi.org/10.2147/OTT.S251915
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Nicola Silvestris
Shuo Wu, Liucheng Ding, Hewei Xu, Jie Gao, Yunpeng Shao, Sicong Zhang, Zhongqing Wei
Department of Urology, The Second Affiliated Hospital of Nanjing Medical University, Nanjing, Jiangsu, People’s Republic of China
Correspondence: Zhongqing Wei
Department of Urology The Second Affiliated Hospital of Nanjing Medical University, Nanjing, Jiangsu, People’s Republic of China
Email [email protected]
Purpose: Long non-coding RNAs (lncRNAs) are involved in the development of various tumors including prostate cancer. The purpose of this study was to explore the function of a natural antisense RNA, IDH1-AS1, exerting potential carcinogenic effects in prostate cancer through a novel molecular mechanism.
Materials and Methods: GEPIA and CCLE databases were searched to identify alterations in the expression of IDH1-AS1, which were then verified by RT-qPCR in 20 pairs of matched tumor and normal tissue samples. Subsequently, CCK-8, EdU, and transwell assays were conducted to investigate the carcinogenic effect of IDH1-AS1. RT-qPCR, Western blot, and isocitrate dehydrogenase 1 (IDH1) enzyme activity assays were used to explore the functional relationship between IDH1-AS1 and its sense gene IDH1.
Results: IDH1-AS1 expression was found to be significantly increased in prostate cancer tissues and cell lines. IDH1-AS1 knockdown significantly inhibited the proliferation and migration of prostate cancer cells. Interestingly, RT-qPCR and Western blot analyses revealed that IDH1-AS1 did not significantly affect the expression of IDH1 mRNA or protein but was involved in the regulation of IDH1 enzyme activity in prostate cancer cells.
Conclusion: Our experiments revealed that the carcinogenic effects of IDH1-AS1 in prostate cancer may depend on a new molecular mechanism, which directly alters IDH1 enzyme activity. Our findings indicate that IDH1-AS1 is a novel candidate target for prostate cancer treatment.
Keywords: antisense RNA, divergent transcription, bioinformatics analysis, enzyme activity
Introduction
Worldwide, prostate cancer (PCa) is the most diagnosed malignant tumor in men and the leading cause of mortality among tumors of the urinary tract.1 In the past decade, PCa exhibited the highest incidence rate in China, causing an increase in overall tumor-related mortality, and gradually becoming a serious social problem.2 The main therapeutic strategies for PCa at initial diagnosis include radical surgery and radiotherapy, but high incidence of metastasis and recurrence are still major clinical issues. Moreover, PCa patients undergoing conservative androgen deprivation therapy (ADT) eventually progress to castration-resistant prostate cancer (CRPC).3 PCa progression to end-stage CRPC greatly increases the risk of invasion and metastasis, and contributes to poor patient survival.4 Currently, prostate-specific antigen (PSA, also known as KLK4) is widely used for clinical screening. However, its low specificity may result in overdiagnosis and overtreatment.5 In-depth understanding of PCa pathogenesis may help predict PCa and develop more effective treatment strategies.
Long non-coding RNAs (lncRNAs), longer than 200 bp, have been recently discovered owing to the advancements in sequencing technologies.6,7 LncRNAs have vital biological functions, as they participate in the regulation of diverse biological processes.8 Emerging evidence shows that lncRNAs are involved in tumorigenesis and cancer progression by regulating the proliferation, metastasis, and invasion of cancer cells.9 For example, in glioma, the lncRNA, MALAT1, serves as a competing endogenous RNA of zinc fingers and homeoboxes 1 (ZHX1), and MALAT1 knockdown attenuates the viability of glioblastoma cells.10 Furthermore, DLGP1-AS1 is significantly upregulated in hepatocellular carcinoma and promotes tumor progression by regulating the miR-486-5p/H3F3B axis.11 Natural antisense RNAs, a type of lncRNAs, are characterized by a complementary or overlapping position with its homologous RNA in gene location.12 Natural antisense RNAs are classified as trans-natural and cis-natural depending on their location with respect to their sense RNAs.12 Natural antisense RNAs control their sense RNAs by a specialized divergent transcription, which is recently attracting increasing attention.13 In particular, FGFR3-AS1 upregulates the transcription of its sense gene, FGFR3, thus maintaining gene stability in osteosarcoma.14 Previous studies have reported that IDH1-AS1 is aberrantly upregulated in prostate cancer and is considered as an oncogene.15 The gene for isocitrate dehydrogenase-1 (IDH1) is located on the sense strand with respect to IDH1-AS1. However, the interaction between IDH1-AS1 and IDH1 and its role in PCa remains unclear. We hypothesized that IDH1-AS1 could induce PCa progression by regulating its sense gene.
The purpose of this study was to characterize the IDH1-AS1/IDH1 interaction and elucidate its role in PCa. First, we employed bioinformatics tools and RT-qPCR to explore IDH1-AS1 expression in PCa. Subsequently, the impact of IDH1-AS1 on the proliferation and migration of PCa cell lines was addressed. Finally, IDH1-AS1-induced regulation of IDH1 expression and activity was examined by RT-qPCR, Western blot, and enzyme activity assays. Our study provides new insights into the function of IDH1-AS1 and identifies this molecule as a promising therapeutic target in PCa.
Materials and Methods
GEPIA and CCLE Databases
Gene Expression Profiling Interactive Analysis (GEPIA, http://gepia.cancer-pku.cn)16 is a public database containing information on 9736 tumor samples and 857 normal tissue samples, and is normally used to identify genes with differential expression in human tumors. The Cancer Cell Line Encyclopedia (CCLE, https://portals.broadinstitute.org/ccle)17 is an ongoing project allowing for free integrated computational analyses on human tumors, and provides detailed molecular information on more than 1100 cell lines.
Tissue Collection
Our twenty pairs of tumor tissues and adjacent normal prostate epithelial tissues were obtained from PCa patients of the Second Affiliated Hospital of Nanjing Medical University who underwent radical resection without neoadjuvant chemotherapy or endocrine therapy prior to surgery. All samples were collected within 30 min after isolation, and stored in the refrigerator at −80°C. Liquid nitrogen was used to transport samples to the laboratory for subsequent experiments. All samples were confirmed to be tumor or normal tissue samples by two independent pathologists. Patients were informed about their participation in the controlled clinical trial, and signed an informed consent document before the trial. This study was approved by Ethics Committee of the Second Affiliated Hospital of Nanjing Medical University.
Cell Culture and Transfection
PCa (PC3, DU145, LNCaP, 22RV1) and normal control cells (WPMY-1) were purchased from Zhong Qiao Xin Zhou Biotechnology (Shanghai, China). PC3 and DU145 cells were cultured in F-12K medium, LNCaP and 22RV1 cells in 1640 medium, and WPMY1 cells in DMEM medium. Complete medium was obtained by adding 10% fetal bovine serum (FBS, ScienCell, USA) and 1% penicillin and streptomycin. The cell incubator was set at 37°C and 5% CO. The small interfering RNAs (si-RNAs) for cell transfection and the transfection kit were purchased from RiboBio company (Guangzhou, China). The transfection procedure was conducted according to the manufacturer’s instructions.
RT-qPCR
For RT-qPCR, total RNA was isolated from cells or tissues using TRIzol Reagent (TaKaRa, Dalian, China). A PrimerScript RT-PCR kit (TaKaRa, Dalian, China) was used for reverse transcribe 1 µg of total RNA into cDNA according to the manufacturer’s protocol. Then, cDNA, gene-specific primers, and SYBR Green PCR kit (TaKaRa, Dalian, China) reagents were mixed. Real-time PCR was performed by using a Roche LightCycler 480II machine (Roche, Switzerland), and the procedure was performed according to the product instructions. The relative expression of RNAs was calculated by the 2−ΔΔCt method, with GAPDH as an internal control. The primers were as follows: IDH1-AS1-forward: GTCATGGAGGTGTCTGTGTTAG.
IDH1-AS1-reverse: GTCACTCTGCGGATGTTTCT; IDH1-forward: GGCTGAAGAAGCAGAGGATAG, IDH1-reverse: TTCACCACTCTGTCGGTTTAG; U6-forward: CTCGCTTCGGCAGCACA, U6-reverse: AACGCTTCACGAATTTGCGT; GAPDH-forward: GGTGTGAACCATGAGAAGTATGA, GAPDH-reverse: GAGTCCTTCCACGATACCAAAG.
CCK-8 Assay
PCa cell proliferation capacity was measured using a CCK-8 kit (Beyotime, Shanghai, China) according to the manufacturer’s instruction. After cell counting, 100 µL of cell suspension containing 4×103 cells was added to each well of a 96-well plate. Ten microliters of CCK-8 reagent were added at each time point. At 2 h after the addition of CCK-8 reagent, a Microplate Reader (Thermo, USA) was used to measure the absorbance at 450 nm, reflecting the rate of cell proliferation.
EdU Assay
The EdU assay was conducted to determine the proliferative activity of PCa cells. According to the protocol of the Cell-light™ EdU Kit (Ribobio, China), PC3 and LNCaP cells were harvested and seeded into 96‐well plates (1×105 cells/well). After labeling with EdU reagent (50 μM) for 2 h and fixation with glycine and 4% paraformaldehyde for 30 min, the cells were stained with DAPI solution for 30 min. EdU-positive cells were observed and counted by using a fluorescence microscope (Olympus, Japan).
Transwell Assay
A transwell assay was used to determine the migration ability of PCa cells. After cell counting, about 1.5×104 cells per well were seeded into the upper chamber of the transwell compartment with 100 µL of serum-free medium, and 600 µL of 20% FBS-containing medium was added to the lower chamber. The cells were cultured under standard conditions for 24 h, and the cells that had migrated to the lower chamber were collected, fixed with 4% formaldehyde, and stained with 0.5% crystal violet dye for 10 min. Finally, the migrated cells were washed with PBS and placed under a microscope for observation and counting.
Subcellular Fractionation
To separate nuclear and cytosolic fractions of PCa cells, subcellular fractionation was conducted by using a PARIS Kit (Life Technologies, USA) according to the manufacturer’s instructions. Specifically, the Cell Disruption Buffer was used to cells lysis on ice for 5mins. The Cell Fractionation Buffer was added into the obtained cell lysate, then nuclear and cytoplasmic fractions were separated by centrifugation at 4°C. The subsequent experiments including RNA isolation and protein analysis were then performed.
Western Blot
PCa cell proteins were extracted with RIPA buffer (Beyotime, Shanghai, China) and boiled for denaturation. Subsequently, the protein samples were separated by 10% SDS-PAGE and transferred to PVDF membranes (Millipore Corporation). The membranes were blocked with non-fat milk and incubated overnight at 4°C with primary antibodies against IDH1 or GAPDH. Finally, the membranes were incubated with horseradish peroxidase (HRP)-conjugated secondary antibodies (Abcam, UK), and protein bands visualized by enhanced chemiluminescence.
IDH1 Enzyme Activity Assay
As already mentioned, the IDH1 enzyme mainly functions in the cytoplasm.18 After protein extraction, IDH1 was purified by affinity chromatography. Enzyme activity was measured according to the instructions of the IDH Activity Assay Kit (Sigma, Shanghai, China). In brief, 38 µL of IDH assay buffer, 8 µL of developer, 2 µL of IDH substrate, 2 µL of NADP+, and 2 µL of NAD+ were mixed at 37°C. Then, IDH1 enzyme activity was calculated by measuring the OD 450 nm. The mean values of triplicate experiments were reported.
Statistical Analyses
All data were obtained from three independent experiments. The mean ± standard deviation was used for statistical analysis. Unpaired sample t-test was applied for two-group comparisons, while Pearson correlation analysis was used for the correlation of gene expression values. The threshold of statistical significance was set at P<0.05. The data were analyzed by using GraphPad7.0 software.
Results
Bioinformatics Analysis Revealed Aberrant Expression of IDH1-AS1 and IDH1 in PCa Tissues and Cells
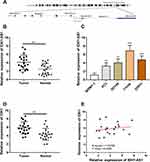
GEPIA and CCLE databases were employed to investigate the mRNA levels of IDH1-AS1 and IDH1 in PCa tissues and cells. A total of 492 tumor cases and 152 normal controls were examined. We found that the expression of both IDH1-AS1 and IDH1 was markedly upregulated in PCa tissues compared to normal tissues (Figure 1A and B). Then, a correlation analysis was performed by using the GEPIA plug-in, which showed the absence of correlation between IDH1-AS1 and IDH1 expression (Figure 1C). However, data visualization from GEPIA database showed that IDH1-AS1 expression in prostate cancer was outstanding among various cancers (Figure 1D). To confirm the aberrant expression of IDH1-AS1 in cancer cells, we used the CCLE database to analyze the level of IDH1 in multiple tumor cell lines. Human prostate cancer cell lines exhibited the highest level of IDH1-AS1 expression (Figure 1E). The expression of IDH1-AS1 was consistently high in different PCa cell lines (Figure 1F). In conclusion, the combined analysis of GEPIA and CCLE databases revealed that IDH1-AS1 was highly expressed in PCa.
IDH1-AS1 and IDH1 are Complementary Strand Genes Highly Expressed in Clinical PCa Samples
According to the University of California, Santa Cruz (UCSC, http://genome.ucsc.edu) website, IDH1-AS1 and its sense mRNA, IDH1, were located head-to-head with a short gap on chromosome 2 (Figure 2A). PCa samples and cells were analyzed by RT-qPCR. IDH1-AS1 was expressed at a significantly higher level in PCa tissues compared to adjacent normal prostate epithelial tissues (Figure 2B). Consistently, higher levels of IDH1-AS1 were detected in PCa cells than in normal prostatic epithelial cells (Figure 2C). We further examined IDH1 mRNA level in clinical samples, and found it upregulated in tumors compared to normal tissues (Figure 2D). Next, we performed a correlation analysis in PCa samples, which clearly showed the absence of unequivocal correlation between IDH1-AS1 and IDH1 expression (Pearson’s correlation, Figure 2E). On the other hand, previous studies suggested that both IDH1-AS1 and IDH1 play pivotal roles in tumorigenesis.18 Therefore, the functional relationship between IDH1-AS1 and IDH1 in PCa might not be based on a mechanism distinct from AS1-IDH1-mediated regulation of IDH1 transcription.
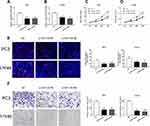
IDH1-AS1 Promotes the Proliferation and Migration of PCa Cells
In order to clarify the exact role of IDH1-AS1 in PCa progression, we designed loss-of-function experiments, based on the use of a specific small interfering RNA (siRNA) in two selected prostate cancer cell lines, PC3 and LNCaP. RT-qPCR was used to verify the efficiency of IDH1-AS1 silencing (Figure 3A and B). IDH1-AS1 knockdown inhibited the proliferation of PCa cells, as determined by CCK-8 assays (Figure 3C and D). Consistently, EdU assays showed a decline in the proliferation of both PC3 and LNCaP cells under the same conditions (Figure 3E). Finally, transwell experiments revealed that IDH1-AS1 knockdown reduced the migration ability of PCa cells (Figure 3F). Collectively, these data indicated that IDH1-AS1 played an important role in PCa progression and tumor cell migration.
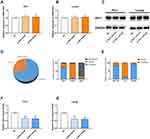
IDH1-AS1 is Involved in the Regulation of IDH1 Enzyme Activity in PCa Cells
To further explore the involvement of IDH1-AS1 in transcriptional or post-transcriptional IDH1 regulation, RT-qPCR, Western blot, and IDH1 enzyme activity assays were performed in PC3 and LNCAP cells knocked down for IDH1-AS1. IDH1-AS1 knockdown did not affect the level of IDH1 mRNA (Figure 4A and B) or IDH1 protein (Figure 4C), as determined by RT-qPCR and Western blot, respectively. Then, the intracellular distribution of IDH1 was examined after subcellular fractionation. Both IDH1 (Figure 4D) and IDH1-AS1 (Figure 4E) were predominantly localized in the cytoplasm. So that we can purify IDH1 enzyme from its abundant family enzyme proteins. IDH1 is known to play a key role in tumor progression, by controlling the oxidative decarboxylation of isocitrate to 2-oxopentaric acid and participating in important aspects of mitochondrial metabolism.20 The impact of IDH1-AS1 knockdown on the activity of cytoplasmic IDH1 was determined. IDH1 activity was reduced in both PC3 and LNCaP cells following IDH1-AS1 suppression (Figure 4F and G). In conclusion, IDH1-AS1 appeared to function by a mechanism distinct from that of a variety of endogenous antisense RNAs. Specifically, it affected IDH1 enzyme activity without significantly altering the expression of the corresponding mRNA or protein.
Discussion
Prostate cancer is a common urologic tumor, but the mechanism of tumorigenesis is still unclear.19 A high proportion of patients with prostate cancer suffer from metastasis and tumor recurrence due to ineffective treatment strategies.20 Therefore, improved diagnosis and treatment of PCa are urgently needed to control this growing clinical and social issue. A large number of lncRNAs have been found dysregulated in tumors, and these changes are correlated with tumorigenesis and tumor progression.21,22 Recent studies revealed that the level of prostate cancer-associated lncRNAs reflects the stage of tumor development. These lncRNAs appear to have crucial functions in various signaling pathways, functioning as transcriptional or post-transcriptional regulators, and acting as oncogenes or tumor suppressors.23 In previous studies, it was found that the lncRNA, IDH1-AS1, is an oncogene involved in PCa.15 A critical finding of the latter study was that IDH1-AS1 is involved in autophagy. In particular, IDH1-AS1 participates in a novel molecular pathway, acting as a competing endogenous RNA (ceRNA) that regulates ATG5 mRNA in PCa.15 Nevertheless, to date, the possible role of IDH1-AS1 as a natural antisense RNA and its functional relationship with the corresponding sense RNA has been largely neglected. On the other hand, evidence for a role of the IDH1-AS1-IDH1 axis in PCa growth was previously reported.24
It is well known that, in addition to regulating the stability of specific genes. 25, 26 similarly to most other lncRNAs, natural antisense RNAs regulate the transcription and function of their sense genes involved in tumor progression.27–29 IDH1-AS1 is located on chromosome 2, almost head-to-head, with a short gap, against the IDH1 gene, which encodes an enzyme catalyzing the oxidative decarboxylation of isocitric acid, resulting in the production of alpha-ketoglutarate.30 IDH1 proteins are key enzymes regulating citric acid metabolism, and their expression and activity are closely related to PCa progression.31 In PCa, reduced IDH1 activity leads to abnormal isocitrate oxidation and production of oxalosuccinic acid, which induces a series of epigenetic changes.32 These reports indicate that IDH1 plays an important role in the development of PCa. According to some studies, IDH1 is a potential target of therapies aimed at interfering with energy metabolism in cancer.33 Notably, a new way of IDH1-AS1-mediated regulation of IDH1 enzyme activity, independent of IDH1 transcription, has been previously demonstrated in Hela and HCT116 cells.25 However, to date, no evidence for this mechanism has been reported in PCa. We hypothesized that the IDH1-AS1 gene could regulate the expression or activity of the enzyme encoded by its sense gene, IDH1, thus contributing to continuous prostate tumor progression. To verify this hypothesis, the effect of IDH1-AS1 knockdown on IDH1 gene transcription, protein translation, and IDH1 enzyme activity was investigated. Small fluctuations in IDH1 enzyme activity were observed within 1–2 days of transfection. In line with a previous study, our results suggested that IDH1-AS1 altered IDH1 enzyme activity without affecting IDH1 mRNA or protein level. However, the details of this regulatory mechanism are still unknown. Notably, IDH1 functions as a homodimer.34 Therefore, it is reasonable to hypothesize that IDH1-AS1 may alter the stability of IDH1 heterodimers. This hypothesis needs to be verified by further experiments.
Conclusions
The present study confirmed that IDH1-AS1 was upregulated in PCa and functioned as an oncogene. In addition, we demonstrated a novel IDH1-AS1-mediated regulatory mechanism involved in PCa progression, based on post-translational regulation. We found that both IDH1-AS1 and IDH1 were highly expressed in prostate cancer tissues, and implicated in the proliferation and invasion of PCa cells. Our experiments discovered a new underlying mechanism of IDH1-AS1, and it may become a new target for prostate cancer therapy.
Ethical Approval
This study was approved by the Ethics Committee of the Second Affiliated Hospital of Nanjing Medical University.
Acknowledgments
We sincerely thank all individuals for supporting and pushing on our work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Ferlay J, Colombet M, Soerjomataram I, et al. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int J Cancer. 2019;144(8):1941–1953. doi:10.1002/ijc.31937
2. Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66(2):115–132. doi:10.3322/caac.21338
3. Cattrini C, Castro C, Lozano L, et al. Current treatment options for metastatic hormone-sensitive prostate cancer. Cancers. 2019;11(9):E1355. doi:10.3390/cancers11091355
4. Lowrance WT, Murad MH, Oh WK, et al. Castration-resistant prostate cancer: AUA guideline amendment 2018. J Urol. 2018;200(6):1264–1272. doi:10.1016/j.juro.2018.07.090
5. Pezaro C, Woo HH, Davis ID. Prostate cancer: measuring PSA. Int Med J. 2014;44(5):433–440. doi:10.1111/imj.12407
6. Tehrani SS, Karimian A, Parsian H, et al. Multiple functions of long non-coding RNAs in oxidative stress, DNA damage response and cancer progression. J Cell Biochem. 2018;119:223–236. doi:10.1002/jcb.26217
7. Ruan X. Long non-coding RNA central of glucose homeostasis. J Cell Biochem. 2016;117:1061–1605. doi:10.1002/jcb.25427
8. Quinn JJ, Chang HY. Unique features of long non-coding RNA biogenesis and function. Nat Rev Genet. 2016;17(1):47–62. doi:10.1038/nrg.2015.10
9. Huarte M. The emerging role of lncRNAs in cancer. Nat Med. 2015;21(11):1253–1261. doi:10.1038/nm.3981
10. Liao K, Lin Y, Gao W, et al. Blocking lncRNA MALAT1/miR-199a/ZHX1 axis inhibits glioblastoma proliferation and progression. Mol Ther Nucleic Acids. 2019;18:388–399. doi:10.1016/j.omtn.2019.09.005
11. Peng X, Wei F, Hu X. Long noncoding RNA DLGAP1-AS1 promotes cell proliferation in hepatocellular carcinoma via sequestering miR-486-5p. J Cell Biochem. 2020;121(2):1953–1962. doi:10.1002/jcb.29430
12. Balbin OA, Malik R, Dhanasekaran SM, et al. The landscape of antisense gene expression in human cancers. Genome Res. 2015;25(7):1068–1079. doi:10.1101/gr.180596.114
13. Lepoivre C, Belhocine M, Bergon A, et al. Divergent transcription is associated with promoters of transcriptional regulators. BMC Genomics. 2013;14(1):914. doi:10.1186/1471-2164-14-914
14. Sun J, Wang X, Fu C, et al. Long noncoding RNA FGFR3-AS1 promotes osteosarcoma growth through regulating its natural antisense transcript FGFR3. Mol Biol Rep. 2016;43(5):427–436. doi:10.1007/s11033-016-3975-1
15. Zhang N, Li Z, Bai F, et al. PAX5-induced upregulation of IDH1-AS1 promotes tumor growth in prostate cancer by regulating ATG5-mediated autophagy. Cell Death Dis. 2019;10(10):734. doi:10.1038/s41419-019-1932-3
16. Tang Z, Li C, Kang B, et al. GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res. 2017;45(W1):W98–W102. doi:10.1093/nar/gkx247
17. Ghandi M, Huang FW, Jané-Valbuena J, et al. Next-generation characterization of the cancer cell line encyclopedia. Nature. 2019;569(7757):503–508. doi:10.1038/s41586-019-1186-3
18. Zhao S, Lin Y, Xu W, et al. Glioma-derived mutations in IDH1 dominantly inhibit IDH1 catalytic activity and induce HIF-1alpha. Science. 2009;324(5924):261–265. doi:10.1126/science.1170944
19. Spratt DE, Zumsteg ZS, Feng FY, et al. Translational and clinical implications of the genetic landscape of prostate cancer. Nat Rev Clin Oncol. 2016;13(10):597–610. doi:10.1038/nrclinonc.2016.76
20. Resnick MJ, Koyama T, Fan K-H, et al. Long-term functional outcomes after treatment for localized prostate cancer. N Engl J Med. 2013;368(5):436–445. doi:10.1056/NEJMoa1209978
21. Kondo Y, Shinjo K, Katsushima K. Long non-coding RNA s as an epigenetic regulator in human cancers. Cancer Sci. 2017;108(10):1927–1933. doi:10.1111/cas.13342
22. Chen L, Dzakah EE, Shan G. Targetable long non-coding RNAs in cancer treatments. Cancer Lett. 2018;418:119–124. doi:10.1016/j.canlet.2018.01.042
23. Xu Y-H, Deng J-L, Wang G, et al. Long non-coding RNAs in prostate cancer: functional roles and clinical implications. Cancer Lett. 2019;464:37–55. doi:10.1016/j.canlet.2019.08.010
24. Xiang S, Gu H, Jin L, et al. LncRNA IDH1-AS1 links the functions of c-Myc and HIF1α via IDH1 to regulate the warburg effect. Proc Natl Acad Sci U S A. 2018;115(7):E1465–E1474. doi:10.1073/pnas.1711257115
25. Bo C, Li X, He L, et al. A novel long noncoding RNA HHIP-AS1 suppresses hepatocellular carcinoma progression through stabilizing HHIP mRNA. Biochem Biophys Res Commun. 2019;520(2):333–340. doi:10.1016/j.bbrc.2019.09.137
26. Diao P, Ge H, Song Y, et al. Overexpression of ZEB2-AS1 promotes epithelial-to-mesenchymal transition and metastasis by stabilizing ZEB2 mRNA in head neck squamous cell carcinoma. J Cell Mol Med. 2019;23(6):4269–4280. doi:10.1111/jcmm.14318
27. Shan H, Yang Y, Zhu X, et al. FAM83H-AS1 is associated with clinical progression and modulates cell proliferation, migration, and invasion in bladder cancer. J Cell Biochem. 2019;120(3):4687–4693. doi:10.1002/jcb.27758
28. Xu M, Xu X, Pan B, et al. LncRNA SATB2-AS1 inhibits tumor metastasis and affects the tumor immune cell microenvironment in colorectal cancer by regulating SATB2. Mol Cancer. 2019;18(1):135. doi:10.1186/s12943-019-1063-6
29. Zhao Y, Wang N, Zhang X, et al. LncRNA ZEB1-AS1 down-regulation suppresses the proliferation and invasion by inhibiting ZEB1 expression in oesophageal squamous cell carcinoma. J Cell Mol Med. 2019;23(12):8206–8218. doi:10.1111/jcmm.14692
30. Parker SJ, Metallo CM. Metabolic consequences of oncogenic IDH mutations. Pharmacol Ther. 2015;152:54–62. doi:10.1016/j.pharmthera.2015.05.003
31. Gonthier K, Poluri RTK, Weidmann C, et al. Reprogramming of isocitrate dehydrogenases expression and activity by the androgen receptor in prostate cancer. Mol Cancer Res. 2019;17:1699–1709. doi:10.1158/1541-7786.MCR-19-0020
32. Molenaar RJ, Maciejewski JP, Wilmink JW, et al. Wild-type and mutated IDH1/2 enzymes and therapy responses. Oncogene. 2018;37(15):1949–1960. doi:10.1038/s41388-017-0077-z
33. Caino MC, Altieri DC. Molecular pathways: mitochondrial reprogramming in tumor progression and therapy. Clin Cancer Res. 2016;22(3):540–545. doi:10.1158/1078-0432.CCR-15-0460
34. Xu X, Zhao J, Xu Z, et al. Structures of human cytosolic NADP-dependent isocitrate dehydrogenase reveal a novel self-regulatory mechanism of activity. J Biol Chem. 2004;279(32):33946–33957. doi:10.1074/jbc.M404298200
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.