Back to Archived Journals » Cell Health and Cytoskeleton » Volume 7
The kinesin–tubulin complex: considerations in structural and functional complexity
Authors Olmsted Z, Colliver A, Paluh J
Received 2 October 2014
Accepted for publication 2 December 2014
Published 26 February 2015 Volume 2015:7 Pages 83—97
DOI https://doi.org/10.2147/CHC.S75310
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Denis Wirtz
Video abstract presented by Janet L Paluh
Views: 754
Zachary T Olmsted, Andrew G Colliver, Janet L Paluh
State University of New York Polytechnic Institute, Colleges of Nanoscale Science and Engineering, College of Nanoscale Science, Nanobioscience Constellation, Albany, NY, USA
Abstract: The ability of cells to respond to external cues by appropriately manipulating their internal environment requires a dynamic microtubule cytoskeleton that is facilitated by associated kinesin motor interactions. The evolutionary adaptations of kinesins and tubulins when merged generate a highly adaptable communication and infrastructure cellular network that is important to understanding specialized cell functions, human disease, and disease therapies. Here, we review the state of the field in the complex relationship of kinesin–tubulin interactions. We propose 12 mechanistic specializations of kinesins. In one category, referred to as sortability, we describe how kinesin interactions with tubulin isoforms, isotypes, or posttranslationally modified tubulins contribute to diverse cellular roles. Fourteen kinesin families have previously been described. Here, we illustrate the great depth of functional complexity that is possible in members within a single kinesin family by mechanistic specialization through discussion of the well-studied Kinesin-14 family. This includes new roles of Kinesin-14 in regulating supramolecular structures such as the microtubule-organizing center γ-tubulin ring complex of centrosomes. We next explore the value of an improved mechanistic understanding of kinesin–tubulin interactions in regard to human development, disease mechanisms, and improving treatments that target kinesin–tubulin complexes. The ability to combine the current kinesin nomenclature along with a more precisely defined kinesin and tubulin molecular toolbox is needed to support more detailed exploration of kinesin–tubulin interaction mechanisms including functional uniqueness, redundancy, or adaptations to new roles upon cell specialization, and to thereby accelerate applications in human health.
Keywords: microtubule, cellular network, cell functions, sortability, transport, disease, signaling
Introduction
Three decades ago when “conventional” kinesin was discovered, the incredible diversity of kinesin–tubulin interactions along with their fundamental roles in a range of cellular processes and multicellular development was not foreseen. The molecular nature of the kinesin–tubulin interface is highly evolved and includes combined kinesin and tubulin sequence specializations that enable unique functional capabilities. Fourteen kinesin families have been identified.1 In addition, a variety of microtubule networks exist that are made of multiple tubulin family isoforms and isotypes. Posttranslational modifications of kinesins and tubulins provide additional spatial and temporal control.2,3 This review evaluates progress in understanding the underlying molecular mechanisms of kinesin–tubulin interactions. Specifically, we focus on those mechanistic features of kinesins and tubulins that enable their contribution to diverse cellular processes such as mitosis, neurophysiology, immunology, and intracellular transport, which impact cell–cell communication networks and human pathologies when disrupted. Here, we use the general term kinesin to refer to both conventional kinesin (Kinesin-1 family) and kinesin-like proteins (Kinesin-2 through Kinesin-14 families) or otherwise provide specific kinesin family or member names. While 14 kinesin families have been proposed,1 the functional diversity that is achieved within families through sequence and domain specializations extends further. We illustrate this point through discussion of a subset of well-studied Kinesin-14 family members with emphasis on key roles in mitosis. We discuss recently identified kinesin–tubulin interactions that direct the architecture and function of the supramolecular centrosome, cilia, and flagella structures, and how incorporating the knowledge of kinesin–tubulin interactions will improve biomedical understanding, bioinformatic modeling, and biomimetic design.
Deciphering kinesin–tubulin functions by evaluation of their molecular toolbox
Kinesins are most often thought of in terms of motility. After all, it is this function that led to the initial discovery of conventional kinesin as a novel protein translocator of organelles on microtubules of the squid giant axon.4 Indeed, motility and transport are of high importance in the functional repertoire of kinesin–tubulin interactions; however, an exclusive focus on this function would neglect the extensive capabilities designed into this complex. Modeling kinesins into phylogenetic trees5,6 and families1 paired with experimental research identifies some general functional patterns. Briefly, kinesins perform the following functions: mediate the transport of numerous cellular components that include organelles (Kinesin-1 through Kinesin-37), RNA (Kinesin-18), and chromosomes (Kinesin-4 and Kinesin-109) along microtubule tracks; provide control of microtubule organization by cross-linking or sliding parallel or antiparallel microtubules (Kinesin-5,10 Kinesin-6,11 and Kinesin-1412); and regulate microtubule depolymerization (Kinesin-8 and Kinesin-1313), microtubule growth (Kinesin-7 and Kinesin-1014), and microtubule flux (Kinesin-5 and Kinesin-1315–19). Newly recognized functions include control of microtubule nucleation from the γ-tubulin ring complex (γ-TuRC) of human centrosomes and yeast spindle pole bodies (Kinesin-14 and Kinesin-520) and regulation of mother centriole stability that impacts ciliogenesis (Kinesin-1321). Kinesins can also cross-bridge microtubules with other cytoskeleton networks that are intermediate filaments22 and actin.23 The overall complexity of kinesin–tubulin interactions currently prevents the ability to rapidly predict all functional roles. An advanced understanding of underlying molecular mechanisms will assist in addressing this challenge.
For a comprehensive view of kinesin–tubulin functions, we must discuss how mechanistic specialization of kinesins and tubulins is achieved, as outlined in Figure 1 and Figure 2. This includes the following: monomeric or oligomeric state; force generation; kinesin step size; directional motility, processivity, and speed; tethered diffusion; microtubule dynamics; cross-linking and sliding of microtubules; cooperative kinesin–tubulin interactions; cross-cytoskeleton bridging; sortability; and cargo, transport, and signaling. This further illustrates why a highly detailed mechanistic analysis of each kinesin is indispensible to create a map of functional kinesin–tubulin interactions that reflects their scope and incredible diversity.
Kinesin monomeric or oligomeric state
Kinesins typically bind microtubule polymers as monomeric, dimeric, or tetrameric forms by utilizing an adenosine triphosphate (ATP)-dependent motor domain. The oligomeric state of partnering kinesins may be as homodimers or homotetramers or instead be heteromeric, and it is enabled by the electrostatic binding of amino acids in a coiled coil “stalk” domain. The stalk includes a “breathable” terminal neck linker region that connects to the motor domain and interacts with this domain during ATP hydrolysis. Dimeric or tetrameric kinesins have different capabilities to organize microtubules. For example, a tetrameric kinesin with motor domains positioned at each end in the complex can both cross-link and slide parallel or antiparallel microtubule arrays. Oligomeric kinesins participate in microtubule organization for such functions as bipolar spindle assembly, axon transport, immunological synapse formation, and other specialized cellular needs.24,25 Homodimer forms of Kinesin-2, Kinesin-3, Kinesin-4, Kinesin-6, Kinesin-7, Kinesin-8, Kinesin-10, Kinesin-12, Kinesin-13, and Kinesin-14 members exist. However, alternate oligomeric states are also observed for kinesins in several of these families,26 including a heterodimeric Kinesin-14 in which one partner has an ATP-independent motor domain. The ability of some kinesins to move between oligomeric states in vivo has also been reported, such as Caenorhabditis elegans Kinesin-3 Unc104 in which the transition between monomeric to dimeric state regulates its potential for motility.27
Force generation
The ~360-amino acid globular motor domain of kinesin is its defining feature that contains conserved HYPIR and LAGSE sequence elements along with an ATP-binding catalytic site.28 In the kinesin–tubulin ATPase cycle, the binding of the motor domain to microtubules stimulates the ATP turnover rate by 1,000-fold.29 The affinity of the motor domain to the microtubule is altered upon ATP hydrolysis and release of Mg-ADP. This action generates a molecular switch that can couple ATP hydrolysis with neck linker rotation and affect speed and motility. The rate of ATP hydrolysis is determined by ATPase activity and ADP plus Pi release, and affects the force generated and speed of translocation.30 The force can be applied toward directed motility, to generate a ratcheting motion for depolymerizing microtubule ends, or allow release and recycling of kinesins from α/β-tubulin heterodimers.
Kinesin step size
A microtubule in vivo has a typical diameter of 25 nm that contains 13 protofilaments composed of α/β-tubulin heterodimer building blocks.31,32 The typical kinesin step size on microtubules is 8 nm33 that corresponds to the spacing of β-tubulin proteins within the longitudinal protofilament. Our understanding of the mechanics of the kinesin steps and their size comes largely from work with the dimeric non-mitotic cargo transporter, Kinesin-1.34–36 The asymmetric binding of the dimeric motor heads to microtubules along with asymmetric ATP binding, hydrolysis, and ADP plus Pi release and rotation of the neck linker generates a hand-over-hand stepping motion.37 A recently revised model of the mechanochemical cycle of kinesin on microtubules is shown in Figure 3A, along with the structure of a single ADP-bound kinesin motor head of Kinesin-1 KIF5B docked to an α/β-tubulin heterodimer in Figure 3B. KIF5B binds primarily to 8 nm spaced β-tubulin subunits in the microtubule protofilament, near helices 11 and 12 in the structure.38–40 Kinesins can also bind to γ-tubulin, using residues conserved between β- and γ-tubulin helix 11, indicated in Figure 3C. The kinesin–microtubule ATPase cycle is well studied by many groups and includes more recently a higher resolution reevaluation of structural changes upon stepping39 and a better understanding of molecular adaptations within motor domains that affect ATPase activity and directional motility.41 For monomeric kinesins, a short charged sequence element referred to as the K-loop is present. The K-loop binds to the C-terminal tails of microtubule tubulins at the interface of the α/β-tubulin heterodimers. This interaction provides a second microtubule-binding site during stepping that prevents complete dissociation of the monomeric kinesin from the microtubule upon ATP hydrolysis.42,43 Nonequivalent stepping of dimeric heads by Kinesin-14 Kar3/Vik1 heterodimer has also been reported44 as well as the ability of Kinesin-8 Kip3 to take a larger lateral step onto side-by-side protofilaments.45 The latter would permit the ability to bypass obstacles, such as other motor proteins, avoiding traffic on microtubules. Advances in imaging technology26 continue to reveal new findings in regard to step size and trafficking.
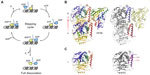  | Figure 3 The kinesin–tubulin complex. |
Directional motility, processivity, and speed
Not all kinesins are motile. Among motile kinesins, the retention time on microtubules before dissociation, referred to as processivity, and the rate of motion may differ. Processivity can be understood in terms of the number of ATP hydrolyses (number of steps) per initial microtubule-binding event. Highly processive kinesins, such as Kinesin-1, may take over 100 steps per encounter with a microtubule, hydrolyzing a single ATP per step.33,46 For non-processive kinesins, such as Drosophila Kinesin-14 Ncd and human Kinesin-14 KIFC1, only a single step or ATP hydrolysis occurs before the kinesin dissociates from the microtubule.47 The degree of processivity of a kinesin on a microtubule is not yet fully predictable. It is influenced by sequence elements in both the neck linker and motor domain that interact upon microtubule binding and ATP hydrolysis. Removal of the neck linker region diminishes processivity,48 while the addition of positive or negative charges to the neck linker region increases or decreases processivity, respectively.49 Directional motility is primarily associated with the kinesin neck domain.50 In fact, altering conserved elements in the neck sequence of Drosophila Kinesin-14 Ncd allowed directional reversal of motility from minus-end to plus-end directed.51 Within a kinesin polypeptide that has an N-terminal motor domain, the neck sequence follows, whereas the neck sequence precedes a C-terminal-positioned motor domain (Figure 2). Neck linker sequences differ between kinesins in regard to N-terminal or C-terminal motor domain position and along with the motor confer plus- or minus-end directionality, respectively. Motile members of the Kinesin-14 family are minus-end directed. This paradigm helps to classify motor proteins into kinesin families but is not predictive of processivity or rates of motility that can be very different.52 Some tubulin modifications can selectively affect kinesin rate of motility. The polyglutamylation of the C-terminal tails of α/β-tubulins has been shown to enhance the motility of members of Kinesin-1 and Kinesin-2 but not Kinesin-13.53
Tethered diffusion
Nonmotile or low-processivity kinesins have the capability to track along microtubules by tethered diffusion in a one-dimensional random walk.30 That is, the initial three-dimensional diffusion to find microtubules is reduced in dimensionality to one dimension, following the initial contact with microtubules. In the case of the central motor domain kinesins, Kinesin-13 XKCM1 (MCAK) and XKIF2,13,54 one-dimensional tethered diffusion directs these kinesins to microtubule ends where the ATPase cycle is used in depolymerization of microtubules.13 In single-molecule microscopy assays, MCAK on average diffused 0.83 seconds with a diffusion coefficient of 0.38 μm2/s.55 Strong binding to the microtubule occurs in the ATP-bound state, whereas diffusion occurs during the weakly bound ADP state. For MCAK, tethered diffusion increased the concentration of kinesin at microtubule plus- or minus-ends over 100-fold compared to free diffusion in three dimensions.55 Two Kinesin-14 dimeric family members also exhibit tethered diffusion, that for Drosophila Ncd is unbiased56 and for Schizosaccharomyces pombe Pkl1 is biased57 diffusion along the microtubule.
Microtubule dynamics
Microtubules exhibit dynamic instability58 that is an inherent ability to transition between periods of stable growth or rapid disassembly of subunits, referred to as polymerization and depolymerization, respectively.31 Microtubules also undergo a period of attenuated dynamics or a pause state during which they neither grow nor shorten appreciably.59,60 Heterodimeric α/β-tubulin subunits assemble as polarized longitudinal protofilaments that become linked laterally as a sheet that closes to form a tube.60,61 The polar microtubule lattice terminates in α-tubulin at minus-ends and is stabilized by a GTP-β-tubulin cap. The hydrolysis of this cap to GDP-tubulin triggers rapid depolymerization31 that can be regulated by kinesins and other microtubule-associated proteins. Depolymerization mechanisms are not fully understood and may generate either small or large changes to microtubule length. Kinesin-13 mediates bending of individual protofilaments away from the longitudinal face of the microtubule and generates large changes in microtubule length, while Kinesin-8 either suppresses dynamics or removes just one or two heterodimers and may not deform protofilaments.13 Certain members of the Kinesin-4 family also regulate dynamics. Kinesin-4 Kif7 regulates microtubule length in the cilium by depolymerization that is also linked to Hedgehog signaling.62 The Sonic Hedgehog pathway is critical for vertebrate organogenesis in the developing embryo, including brain development. Microtubule plus-end proteins Kinesin-10 Nod and Kinesin-7 CENPE both promote microtubule plus-end elongation14 but either do so by an ATP hydrolysis-independent pathway, as for Nod,63 or require an ATP turnover for polymerizations, as for CENPE.64 Multicellular eukaryotes encode genes for Kinesin-4, Kinesin-7, Kinesin-8, Kinesin-10, and Kinesin-13 families, while only Kinesin-8 is present in fission and budding yeasts.13,65 The translocation of α/β-tubulin heterodimers within microtubule protofilaments during mitosis is referred to as flux.66 Coupling plus-end addition of α/β-tubulin subunits with minus-end loss of subunits during flux can generate minus-end-directed transport of microtubule-bound proteins. The flux mechanism is unknown but requires Kinesin-5 in Xenopus, although not in human cells.67,68 Microtubule dynamics facilitate rapid organization and reorganization of microtubule architectures, regulate multiple microtubule-dependent signaling pathways, and provide forces to push and pull as needed for chromosome capture and alignment during mitosis.
Cross-linking and sliding of microtubules
The ability to cross-link and/or slide parallel or antiparallel microtubules is a classical role of dimeric or tetrameric kinesins that drives the assembly of larger microtubule arrays. These actions are integral to ubiquitous processes such as spindle assembly and anaphase B elongation in dividing cells as well as cell specialization such as transport in neuronal axons and dendrites that contain microtubule bundles of opposite orientation. Tetrameric Kinesin-5 members help to drive bipolar mitotic spindle assembly and anaphase B spindle elongation by cross-linking and sliding antiparallel microtubules extending from opposing spindle poles.69 In fission yeast, the Kinesin-6 Klp9 is the anaphase B motor and is regulated by the mitotic Cdc2/cyclin B kinase.70 Cross-linking without sliding can be used to generate a molecular brake to maintain integrity of bipolar microtubule arrays. For example, C. elegans Kinesin-5 BMK-1 resists outward sliding during anaphase B to prevent spindle collapse.71 A similar braking mechanism is proposed to regulate neuronal process outgrowth by Kinesin-5 and Kinesin-12 members.72,73 The homodimer Drosophila Kinesin-14 Ncd cross-links microtubules via its motor domain and tail region that contains ATP-independent tubulin-binding elements and allows Ncd to span two adjacent microtubules.74,75 The positively charged tubulin binding elements in the Ncd tail bind to negatively charged residues in the C-terminus of α/β-tubulins, which extend from the microtubule surface. As with tetrameric kinesins, Ncd can either drive antiparallel microtubule sliding or act as a brake on cross-linked microtubule arrays.12 Kinesin-5 and Kinesin-14 families in Drosophila provide balanced antagonistic forces that enable spatial and temporal control of mitotic spindle assembly.76 This antagonistic relationship is now considered ubiquitous in all eukaryotes. It also likely extends to nondividing cells, such as neurons for the organization of axon and dendrite microtubule arrays, in which Kinesin-5 is already known to play a role.72
Cooperative kinesin–tubulin interactions
The ability of multiple copies of a single kinesin protein bound on microtubules to uniformly coordinate their actions requires cooperation. It is of particular importance to such processes as antiparallel microtubule sliding in which coordinated effort may be needed for large force-requiring events, such as anaphase B, and where uncoordinated efforts can result in braking and stuttered movements. A model of cooperative action of Drosophila Kinesin-14 Ncd proteins on microtubules is proposed based on differing electrostatic interactions in its coiled coil stalk, a structural domain that participates in dimerization.77 The Ncd stalk contains differing coiled coil regions. Weak electrostatic interactions are adjacent to its tail domain, followed by a strongly dimerizing region.78,79 In the Ncd cooperativity model, the weak coiled coil segment will collapse under force when non-synchronized with force-generating Ncd proteins. In addition, for both Drosophila Ncd and S. pombe Pkl1, the diffusive component of movement is reduced with increasing motor numbers that may facilitate cooperation.57,77 Continued studies are needed to determine detailed mechanisms through which collective motor coordination contributes to microtubule organization and other processes such as intracellular transport.
Cross-cytoskeleton bridging
In addition to classical microtubule cross-linking, kinesins can also bridge microtubules to additional cytoskeleton networks, which is often referred to as cytoskeletal intersections.80 Two studies using the squid conventional kinesin, Kinesin-1, indicate an ability to link microtubules to intermediate filaments via light chains that are a feature of this kinesin family.81–83 The kinesin attachment to the intermediate filament is static, but motile on the microtubule, and contributes to the assembly of large intermediate filament networks that are important for directional protrusion of lamella and cell migration.84 Kinesins can also cross-bridge microtubules to actin. In the plant Nicotiana tabacum, cell division requires Kinesin-14 cross-bridging of actin to microtubules that uses an internal kinesin calponin homology domain (KCH).23 In budding yeast Saccharomyces cerevisiae, the Kinesin-1-like Smy1 colocalizes with myosin V motor Myo2, binds the Myo2 tail, and is able to suppress myosin defects.85–87 Smy1 has also been shown to have a role in actin cable assembly by binding to the FH2 domain of the formin Bnr1 to decrease the rate of actin filament elongation and enable proper actin architecture and dynamic function.88 Although a distant kinesin relative, any microtubule-based roles are yet to be defined. No kinesin-facilitated tubulin–actin interactions in metazoans have been identified, although non-motor adaptor proteins like the actin–microtubule linker protein ACF7 that is found primarily in neural network, muscle, and lung could possibly assist in cell-specific interactions.80
Sortability
Here, we define the ability to direct kinesin–tubulin interactions to specific cellular locations or to promote or prevent interactions. Three primary strategies enable separation of kinesin–tubulin interactions that are kinesin adaptor proteins, kinesin specialization or tubulin diversity, and modifications. Kinesin adaptor proteins and specialized intrinsic kinesin targeting domains both frequently involve the tail regions and are exemplified in the Kinesin-14 family described in a later section. The restriction of kinesin–tubulin interactions by specific tubulin isoforms, isotypes, or posttranslational modifications (PTMs) is sometimes referred to as the tubulin code.89 These strategies are expanded in multicellular eukaryotes through additional kinesin families and members and an extended tubulin superfamily and PTMs.53,90 In yeasts, the limited tubulin diversity includes only three tubulin isoforms that are α, β, and γ and two α-tubulin isotypes. In contrast, the human genome has seven genes that code for α-tubulin, eight for β-tubulin,91 two genes for γ-tubulin,92 as well as genes encoding other tubulin members.90,93,94 Tubulin PTMs further contribute to sortability and compartmentalized functions as exemplified in a recent review in regard to neural physiology3 as well as a comprehensive bioinformatic analysis of the C-terminal tail of tubulins. The tubulin tail is where most PTMs occur,94 except for acetylation.95 The other PTMs found in tubulins include phosphorylation, tyrosination, polyglutamylation, and polyglycylation that can selectively serve as a code for specific cell functions.89 Examples of use of the tubulin code include Kinesin-1 KIF5 that navigates to axonal but not dendritic microtubules by differences in tyrosination96 and inability of Kinesin-13 MCAK and Kif2A to depolymerize neuronal microtubules that are detyrosinated at the α-tubulin C-terminal tail and stabilized.13
Cargo, transport, and signaling
Kinesin-mediated transport along microtubules has been studied extensively due to the wide scope of cargo. This includes RNA, proteins, chromosomes, vesicles, organelles, small multiprotein complexes, and other microtubules.97 Multiple Kinesin families participate in intracellular transport with cargo-specific domains and include Kinesin-1, Kinesin-2, Kinesin-3, Kinesin-4, Kinesin-7, and Kinesin-10 members.25,98 Tight spatiotemporal control of transport by kinesins is achieved by both auto-inhibition and phosphorylation-dependent mechanisms acting on kinesins as molecular switches.26 Numerous kinesin–microtubule transport functions contribute to such critical processes as assembly and functional dynamics of the mitotic spindle,99 establishment of correct microtubule length and architecture of cilia and flagella via intraflagellar transport,100 and neuronal function.101 Detailed mechanisms for cargo release and handoff at a given destination are largely unknown. Thus, it is unclear whether the kinesin is recycled by active retrograde transport with dynein, simple diffusion, or used singly and then eliminated through proteasome degradation pathways. For example, experimental analysis of human Kinesin-1 Kif5 coupled with mathematical modeling supports diffusional recycling after cargo transport and release at neurite tips.102 In mitosis, Kinesin-7 CENPE transports misaligned chromosomes to plus-ends at the metaphase plate, switching from microtubule lateral association to plus-end tracking that couples dynamic microtubule ends to kinetochores.103–105 This action also links CENPE to spindle assembly checkpoint signaling that monitors chromosome bipolar attachment and tension.106 Other examples linking cargo, transport, and signaling include Drosophila Kinesin-4 Cos2 that transports the transcription factor Ci required for Hedgehog signaling107,108 and Kinesin-1 that transports SMAD2 in the TGF-β signaling pathway.109 Members of a single kinesin family may regulate the same signaling pathway in different ways between eukaryotes. Mammalian Kinesin-4 KIF7/KIF27 regulates the Hedgehog signaling pathway in cilia by controlling cilia length through microtubule depolymerization at plus-ends and not by participation in microtubule-based transport as does Drosophila Kinesin-4 Cos2.62
Kinesin-14-tubulin paradigm for functional specialization
Only one kinesin family, Kinesin-14, is capable of minus-end-directed motility. This may explain why its diverse functions include all mechanistic specializations reviewed so far as well as incorporation into supramolecular machines discussed in the following section. This detailed appreciation of Kinesin-14 member capabilities comes from their analysis by site-directed mutations, partial deletions, chimeric kinesins, cross-species functional studies, single-molecule and motility assays, and structural and bioinformatic modeling. Specialization of tail domain sequences differs between S. pombe Pkl1 and Drosophila Ncd allowing these proteins to sort to and function at γ-TuRC or on microtubules, respectively.12,74,110 Pkl1 is nonmotile but actively diffuses along microtubules to spindle poles.57 The Ncd tail has two specialized elements for ATP-independent microtubule binding, including a lysine-rich so-called E-Hook-binding sequence that binds negatively charged residues in the tubulin C-terminal tails referred to as the E-Hook.74 With microtubule-binding capabilities at both ends, Ncd can cross-link parallel or antiparallel microtubule arrays and slide microtubules. As mentioned previously, specialization of the Ncd stalk is proposed to coordinate the action of multiple Ncd proteins on microtubules.79 The budding yeast S. cerevisiae Kinesin-14 Kar3 is best studied in regard to its heterodimeric partnering with Cik1 and Vik1 proteins.111–115 The ATP-independent motor domains of both Cik1 and Vik1 generate an alternate stepping mechanism with the Kar3 ATP-regulated motor head. The Cik1 and Vik1 partners also provide sortability by targeting Kar3/Vik1 to the spindle pole body protein Spc72 during meiosis or Kar3/Cik1 to microtubules during mitosis.113,116 Typically, eukaryotes have two to three Kinesin-14 members, except for the single Ncd in Drosophila.1 Multifunctional properties often exist in Kinesin-14 members that are not fully understood mechanistically. Pkl1 participates in spindle assembly checkpoint signaling.117,118 Ncd has additional described roles in meiosis with γ-tubulin.119,120 Human Kinesin-14 HSET cross-links microtubules in human cells121,122 but binds to γ-TuRC in cross-species studies in S. pombe.110 Kinesin-14 members currently share no sequence similarities outside of conserved neck linker and kinesin motor elements. Thus, understanding separation of function either among multiple members between eukaryotes or within a single eukaryote continues to require detailed functional knowledge aided by understanding of specialized domains.
Kinesin–tubulin integration into supramolecular machines and cell specialization
Kinesin–tubulin interactions have important conserved roles in multiprotein complexes to coordinate function with signaling. Required mitotic complexes include spindle kinetochores for chromosome attachment,123 γ-TuRC for microtubule nucleation from spindle poles,20,124 and passenger proteins that transfer from kinetochores to a spindle midzone for temporal control of anaphase.105,125 Orientation of the spindle axis with the cell cortex by astral microtubules also uses cytoplasmic microtubule plus-end complexes.105 The following are well studied in cell specialization: intraflagellar trafficking to control microtubule length, and architecture and function in cilia and flagella;100,126,127 neurophysiology of axon architecture and regeneration as well as neuronal growth cone formation and turning;101 cell–cell interfacial communication through adherens junctions;128 and developmental roles such as cell migration84 and regulation of nodal flow.126 Here, we discuss briefly the newly emerging roles in centrosomal complexes as well as summarize the now extensive studies on cilia and flagella, also reviewed in detail elsewhere.98,100,126,129
The centrosome is a massive microtubule-generating and microtubule-organizing center and hub for intracellular signaling.130 A pair of centrioles within a pericentriolar material helps to organize this complex matrix that includes numerous γ-TuRC sites that nucleate microtubules. The microtubule polarity enabled by centrosomal γ-TuRC contributes to classical roles such as mitosis and meiosis, as well as specialized functions in neurophysiology and immune cell targeting via the immunologic synapse. Centriolar biogenesis is a semiconservative process in which a daughter centriole is generated from a mother template. The Kinesin-13 Kif24 has been shown to asymmetrically bind to mother centrioles of the centrosome in S/G2 phases of the cell cycle to assist in remodeling of centriolar microtubules.21 Kif24 does not alter cytoplasmic microtubules or arrays. Kinesin-14 and Kinesin-5 were recently shown to antagonistically bind to γ-TuRC to regulate microtubule nucleation.20,124 The γ-TuRC provides a 13-protofilament ring template for microtubule growth and minus-end attachment.131 The kinesin motor domains bind γ-TuRC through γ-tubulin helix 11 (Figure 3C), predicted to be similar to kinesin–β-tubulin microtubule interactions.132 Additional targeting to γ-TuRC, blocking of microtubule nucleation, and antagonism of Kinesin-14 by Kinesin-5 require specialized tail domain elements in Kinesin-14 Pkl1110,124 and the BimC domain in Kinesin-5 Cut7.20 Pkl1 tail elements alone are both necessary and sufficient to block microtubule nucleation by γ-TuRC in human breast cancer cells.20,110,124 Incorporation of kinesin–tubulin roles at centrosome centriolar and nucleation sites may underlie needs for precise coupling to spindle mechanisms and non-mitotic centrosome functions.
Eukaryotic cilia and flagella microtubule-based organelles serve multiple roles in chemo-, thermo-, and mechano-sensation, cell motility, and fluid flow rate. Immotile primary cilia are found in eye rod photoreceptor cells and olfactory receptors, while motile cilia line fluid compartments. The latter includes lining of the trachea that protects the lungs from fluid accumulation and the fallopian tubes to facilitate transport of the ovum to the uterus. An axoneme structure in primary cilia has a ring of nine microtubule doublets (9+0), while motile cilia and flagella have two additional microtubule singlets within the ring for a 9+2 arrangement. In Caenorhabditis reinhardtii, Kinesin-13 is positioned at flagellar microtubule ends where it controls microtubule assembly and disassembly in this organelle.133 Kinesin mechanisms for microtubule depolymerization in ciliary, flagellar, and centriole length control are reviewed elsewhere.13 A heterotrimeric Kinesin-II complex that is composed of a heterodimeric KIF3A/KIF3B and KAP protein regulates intraflagellar cargo transport in multiple species. Polarized cargo transport from the cell body to the developing cilium assists ciliogenesis by participating in assembly and regeneration.127 Patterning in mammalian and fly embryos uses kinesin–tubulin regulation of Hedgehog pathway signaling, as described earlier, in transport and depolymerization roles affecting cilia architecture and function.62,107,134–134 These roles are needed during mesodermal induction and neurogenesis.126,137
The kinesin–tubulin complex in development, disease, and human health
Mutations in kinesins and tubulins as well as inappropriate up- or downregulation of these proteins in specific cell or tissue types have been linked to cancer, transport-based neuropathologies, plasticity-related neuropathologies, memory, liposomal diseases, and developmental defects (Table 1). Deciphering the cause of disease requires an understanding of kinesin–tubulin mechanistic functions. Although a cellular process such as transport may be affected, the precise point where the mechanism fails is not always evident. Transport-based failures that lead to disease may occur via a variety of onslaughts to kinesin or tubulin proteins that alter any of the mechanistic functions described in this review as well as protein stability and concentration. Here, we highlight both mechanistic functions and additional considerations in the kinesin–tubulin complex as it relates to development, disease, and potential therapies to improve human health.
  | Table 1 Kinesin–tubulin interactions in human diseases |
Although mechanisms underlying kinesin–tubulin interactions are often studied in regard to a particular cell process, functional diversification can exist. We define that as the ability of a kinesin to participate in multiple processes in different cell types using all or a subset of its mechanisms. Functional diversification often exists in multicellular eukaryotes that must be considered in human disease treatment strategies. In cancers, the important roles that mitotic kinesins play in spindle function and genomic stability are apparent when disrupted due to their impact on tumorigenesis and cancer progression.138–140 In this regard, further disrupting mitotic kinesins or spindle microtubule dynamics are effective anticancer therapies139,141 by promoting apoptotic cell death.142,143 However, current microtubule poisons such as paclitaxel in clinical use that bind tubulins and alter microtubule dynamics,141,144,145 while effective, can also interfere with microtubule function of nondividing differentiated cells such as neurons.146 Similarly, Kinesin-5 Eg5 drugs that block bipolar spindle assembly147 that are in Phase I clinical development must contend with non-mitotic roles of this kinesin. The ability of Kinesin-5 to slide microtubules is utilized in neurons to promote axonal outgrowth by focusing of microtubule arrays.72 The impact of altered function of Kinesin-5 on neural health may be significant as revealed by Ostergaard et al.148 In this study, 14 mutations in the KIF11 gene encoding human Kinesin-5 Eg5 were identified that map to the stalk region and occur in 15 patients with autosomal-dominant microcephaly accompanied by lymphedema and/or chorioretinopathy. Stalk mutations including the neck linker may alter the kinesin–tubulin interface by affecting the kinesin oligomeric state, step size, or directional motility parameters. The ability to target a specific cell process by affecting kinesin–tubulin interactions, without also affecting other processes, remains challenging and requires knowledge on the scope of functional diversification.
During human development, kinesin–tubulin interactions participate in both more confined short-range processes such as mitosis and cell–cell communication as well as in extended and complex systemic architectures possible with the human neural network. For proper neurophysiological functions, microtubule networks in neuronal axons that transport organelles and synaptic vesicles can extend to a meter long and must be maintained and repaired to prevent neuronal death and impaired neural function.149 Accumulation of materials in neuronal compartments is a characteristic of Alzheimer’s and Huntington’s disease.150,151 Mutations in Kinesin-1 KIF5A, Kinesin-3 KIF1B, and Kinesin-4 KIF21A, as well as in tubulin isotypes, have been reviewed in regard to neuronal disease contributions3,149 along with detailed studies of the KIF5 family in transport-based accumulation diseases like Alzheimer’s150,152 and KIF13B in cholesterol homeostasis.8 Mutations to the KIF5A motor or stalk that impair axonal transport are linked to Charcot–Marie–Tooth disease 2 (CMT2)153 as well as hereditary spastic paraplegia in humans.153,154 CMT2 also results from mutations in KIF1B that reduce transport of vesicle proteins.155,156 Mutations in KIF21A underlie atrophy of ocular motor muscles and the onset of congenital fibrosis of the extraocular muscle (referred to by the acronyms CFEOM1 and CFEOM3).157–159 In mice, the absence of Kif5A leads to development of epilepsy due to the failed transport of the GABAA receptor protein.160 Mutations in tubulin proteins that inhibit neuronal migration have been linked to brain development. This includes insufficient brain folds and grooves seen in lissencephaly, as well as microcephaly that is characterized by a small brain size.3
Changes to higher order functions such as memory are also linked to kinesin–tubulin interactions due to effects on transport for synaptic function, neural architecture, and neuroplasticity. A recent patient study by Willemsen et al161 found that a disruptive mutation in Kinesin-4 KIF4A (exon skipping) was linked to mild intellectual disability (ID) and epilepsy, whereas a patient with a missense mutation in Kinesin-1 KIF5C (Glu-237-Lys) exhibited severe ID, epilepsy, microcephaly, and cortical malformation. A truncating mutation in the synaptic Kinesin-2 KIF17 was identified in one patient with Schizophrenia during a bioinformatic screen.162 Altered kinesin functions in these patients may interrupt the balance between excitatory and inhibitory synaptic excitability required for normal brain function and normal development.
Animal studies of kinesin–tubulin interactions and functions extend studies in model eukaryotes and have focused primarily on the brain. These studies include generation of knockout genes in mice, analysis of brain sections, and establishment of primary cultures and explants. During development, mitotic-cycling cells stop dividing, exit the cell cycle, and differentiate. In this transition, kinesins typically thought to be mitotic specific may be repurposed into non-mitotic roles, or alternate family members may perform these roles. The story is expected to be complex, since in murine brain development, as many as 38 kinesin superfamily proteins (KIFs) may be required.163,164 Analysis of murine KIFs may offer solutions for human diseases that build off of kinesin–tubulin knowledge from model eukaryotes. For example, strategies for enhancing regeneration of adult axons via inhibition of Kinesin-5.165
Future directions
Nature has coevolved both kinesins and tubulins to generate extensive functional capabilities. The complex nature of kinesin–tubulin interactions includes not only mechanistic specializations but also functional diversification. The latter is particularly evident in multicellular eukaryotes in which cells exit the cell cycle and differentiate to accommodate new cellular and systemic roles, yet may take advantage of kinesin–tubulin interactions originally only needed for mitotic cell division. The ability of kinesin–tubulin interactions to contribute to human development and higher order functions such as memory, and impact these through diseases can be used as a powerful tool to help identify the molecular basis of these processes. Structural studies continue to provide an insider’s view at the molecular scale but currently remain incomplete in resolution detail and provide static time points. Computational bioinformatics will help to bridge the gap between experiment, structure, and in vitro models as has been done to understand the role of tubulin C-terminal tails,94 microtubule dynamics of tubulin polymerization/depolymerization,60 mesoscale communication networks,166 and in vitro models to test kinesin–microtubule functional and organizational plasticity.52,167 As well, biomimicry is important to test and apply our understanding of kinesin–tubulin interactions. Biomimicry takes its template from biology to incorporate functionality or design from nature,168 including copying, repurposing, or developing novel emerging functions. The ability to copy nature is not trivial, and so hybrid biosynthetic systems are likely to emerge first. Manipulating the kinesin–tubulin interaction has been attempted in vitro and informs on nature’s design but still pales in contrast to the dynamic adaptability of the natural system.52 While we have only begun to scratch the surface of kinesin–tubulin complexity and its impact on cellular functions, multicellular development, eukaryotic diversity, and human diseases, a new excitement exists in the cytoskeleton field to unlock these secrets.
Disclosure
The authors report no conflicts of interest in this work.
References
Lawrence CJ, Dawe RK, Christie KR, et al. A standardized kinesin nomenclature. J Cell Biol. 2004;167(1):19–22. | |
Daire V, Poüs C. Kinesins and protein kinases: key players in the regulation of microtubule dynamics and organization. Arch Biochem Biophys. 2011;510(2):83–92. | |
Breuss M, Keays DA. Microtubules and neurodevelopmental disease: the movers and the makers. Adv Exp Med Biol. 2014;800:75–96. | |
Vale R, Reese T, Sheetz M. Identification of a novel force-generating protein, kinesin, involved in microtubule-based motility. Cell. 1985;42(1):39–50. | |
Dagenbach EM, Endow SA. A new kinesin tree. J Cell Sci. 2004;117: 3–7. | |
Lawrence CJ, Malmberg RL, Muszynski MG, Dawe RK. Maximum likelihood methods reveal conservation of function among closely related kinesin families. J Mol Evol. 2002;54:42–53. | |
Verhey KJ, Kaul N, Soppina V. Kinesin assembly and movement in cells. Annu Rev Biophys. 2011;40:267–288. | |
Kanai Y, Dohmae N, Hirokawa N. Kinesin transports RNA: isolation and characterization of an RNA-transporting granule. Neuron. 2004;43: 513–525. | |
Mazumdar M, Misteli T. Chromokinesins: multitalented players in mitosis. Trends Cell Biol. 2005;15(7):349–355. | |
Kapitein LC, Peterman EJG, Kwok BH, Kim JH, Kapoor TM, Schmidt CF. The bipolar mitotic kinesin Eg5 moves on both microtubules that it crosslinks. Nature. 2005;435:114–118. | |
Braun M, Lansky Z, Fink G, Ruhnow F, Diez S, Janson ME. Adaptive braking by Ase1 prevents overlapping microtubules from sliding completely apart. Nat Cell Biol. 2011;13(10):1259–1264. | |
Fink G, Hajdo L, Skowronek KJ, Reuther C, Kasprzak A, Diez S. The mitotic kinesin-14 Ncd drives directional microtubule-microtubule sliding. Nat Cell Biol. 2009;11(6):717–723. | |
Walczak CE, Gayek S, Ohi R. Microtubule-depolymerizing kinesins. Annu Rev Cell Dev Biol. 2013;29:417–441. | |
Drummond DR. Regulation of microtubule dynamics by kinesins. Semin Cell Dev Biol. 2011;22(9):927–934. | |
Brust-Mascher I, Scholey JM. Mitotic motors and chromosome segregation: the mechanism of anaphase B. Biochem Soc Trans. 2011;39(5):1149–1153. | |
Kwok BH, Kapoor TM. Microtubule flux: drivers wanted. Curr Opin Cell Biol. 2007;19(1):36–42. | |
Mitchison TJ. Polewards microtubule flux in the mitotic spindle: evidence from photoactivation of fluorescence. J Cell Biol. 1989;109(2):637–652. | |
Rogers GC, Rogers SL, Sharp DJ. Spindle microtubules in flux. J Cell Sci. 2005;118(6):1105–1116. | |
Sawin KE, Mitchison TJ. Poleward microtubule flux mitotic spindles assembled in vitro. J Cell Biol. 1991;112(5):941–954. | |
Olmsted ZT, Colliver AG, Riehlman TD, Paluh JL. Kinesin-14 and kinesin-5 antagonistically regulate microtubule nucleation by γ-TuRC in yeast and human cells. Nat Commun. 2014;5:5339. | |
Kobayashi T, Tsang WY, Li J, Lane W, Dynlacht BD. Centriolar kinesin Kif24 interacts with CP110 to remodel microtubules and regulate ciliogenesis. Cell. 2011;145(6):914–925. | |
Kreitzer G, Liao G, Gundersen GG. Detyrosination of tubulin regulates the interaction of intermediate filaments with microtubules in vivo via a kinesin-dependent mechanism. Mol Biol Cell. 1999;10:1105–1118. | |
Klotz J, Nick P. A novel actin-microtubule cross-linking kinesin, NtKCH, functions in cell expansion and division. New Phytol. 2012;193(3):576–589. | |
Cross RA, McAinsh A. Prime movers: the mechanochemistry of mitotic kinesins. Nat Rev Mol Cell Biol. 2014;15(4):257–271. | |
Hirokawa N, Noda Y, Tanaka Y, Niwa S. Kinesin superfamily motor proteins and intracellular transport. Nat Rev Mol Cell Biol. 2009;10(10):682–696. | |
Verhey KJ, Hammond JW. Traffic control: regulation of kinesin motors. Nat Rev Mol Cell Biol. 2009;10(11):765–777. | |
Al-Bassam J, Cui Y, Klopfenstein D, Carragher BO, Vale RD, Milligan RA. Distinct conformations of the kinesin Unc104 neck regulate a monomer to dimer motor transition. J Cell Biol. 2003;163(4):743–753. | |
Vale R, Fletterick R. The design plan of kinesin motors. Annu Rev Cell Dev Biol. 1997;13:745–777. | |
Hackney DD. Kinesin ATPase: rate-limiting ADP release. Proc Natl Acad Sci U S A. 1988;85:6314–6318. | |
Friel CT, Howard J. Coupling of kinesin ATP turnover to translocation and microtubule regulation: one engine, many machines. J Muscle Res Cell Motil. 2012;33(6):377–383. | |
Desai A, Mitchison TJ. Microtubule polymerization dynamics. Annu Rev Cell Dev Biol. 1997;13:83–117. | |
Downing KH, Nogales E. Tubulin and microtubule structure. Curr Opin Cell Biol. 1998;10(1):16–22. | |
Schnitzer MJ, Block SM. Kinesin hydrolyses one ATP per 8-nm step. Nature. 1997;388:386–390. | |
Vale R, Funatsu T, Pierce D, Romberg L. Direct observation of single kinesin molecules moving along microtubules. Nature. 1996;380(6573):451–453. | |
Carter NJ, Cross RA. Mechanics of the kinesin step. Nature. 2005;435(7040):308–312. | |
Clancy B. A universal pathway for kinesin stepping. Nat Struct Mol Biol. 2011;18(9):1020–1027. | |
Asbury CL, Fehr AN, Block SM. Kinesin moves by an asymmetric hand-over-hand mechanism. Science. 2003;302(5653):2130–2134. | |
Nogales E, Wolf SG, Downing KH. Structure of the alpha beta tubulin dimer by electron crystallography. Nature. 1998;391(6663):199–203. | |
Amos LA, Hirose K. A cool look at the structural changes in kinesin motor domains. J Cell Sci. 2007;120(22):3919–3927. | |
Hoenger A, Thormählen M, Diaz-Avalos R, et al. A new look at the microtubule binding patterns of dimeric kinesins. J Mol Biol. 2000;297(5):1087–1103. | |
Goulet A, Major J, Jun Y, Gross SP, Rosenfeld SS, Moores CA. Comprehensive structural model of the mechanochemical cycle of a mitotic motor highlights molecular adaptations in the kinesin family. Proc Natl Acad Sci U S A. 2014;111(5):1837–1842. | |
Okada Y, Hirokawa N. Mechanism of the single-headed processivity: diffusional anchoring between the K-loop of kinesin and the C terminus of tubulin. Proc Natl Acad Sci U S A. 2000;97(2):640–645. | |
Skiniotis G, Cochran JC, Müller J, Mandelkow E, Gilbert SP, Hoenger A. Modulation of kinesin binding by the C-termini of tubulin. EMBO J. 2004;23(5):989–999. | |
Rank KC, Chen CJ, Cope J, et al. Kar3Vik1, a member of the kinesin-14 superfamily, shows a novel kinesin microtubule binding pattern. J Cell Biol. 2012;197(7):957–970. | |
Bormuth V, Nitzsche B, Ruhnow F, et al. The highly processive kinesin-8, Kip3, switches microtubule protofilaments with a bias toward the left. Biophys J. 2012;103(1):L4–L6. | |
Coy DL, Wagenbach M, Howard J. Kinesin takes one 8-nm step for each ATP that it hydrolyzes. J Biol Chem. 1999;274(6):3667–3671. | |
Endow SA, Barker DS. Processive and nonprocessive models of kinesin movement. Annu Rev Physiol. 2003;65(9):161–175. | |
Romberg L, Pierce DW, Vale RD. Role of the kinesin neck region in processive microtubule-based motility. J Cell Biol. 1998;140(6):1407–1416. | |
Thorn KS, Ubersax JA, Vale RD. Engineering the processive run length of the kinesin motor. J Cell Biol. 2000;151(5):1093–1100. | |
Rice S, Lin AW, Safer D, et al. A structural change in the kinesin motor protein that drives motility. Nature. 1999;402:778–784. | |
Sablin EP, Case RB, Dai SC, et al. Direction determination in the minus-end-directed kinesin motor ncd. Nature. 1998;395(6704):813–816. | |
Riehlman TD, Olmsted ZT, Paluh JL. Molecular motors. In: Xie Y, editor. The Nanotechnology Handbook. Boca Raton, FL: CRC Press; 2012:73–112. | |
Sirajuddin M, Rice LM, Vale RD. Regulation of microtubule motors by tubulin isotypes and post-translational modifications. Nat Cell Biol. 2014;16(4):335–344. | |
Desai A, Verma S, Mitchison TJ, Walczak CE. Kin I kinesins are microtubule-destabilizing enzymes. Cell. 1999;96(1):69–78. | |
Helenius J, Brouhard G, Kalaidzidis Y, Diez S, Howard J. The depolymerizing kinesin MCAK uses lattice diffusion to rapidly target microtubule ends. Nature. 2006;441(7089):115–119. | |
Furuta K, Toyoshima YY. Minus-end-directed motor Ncd exhibits processive movement that is enhanced by microtubule bundling in vitro. Curr Biol. 2008;18(2):152–157. | |
Furuta K, Edamatsu M, Maeda Y, Toyoshima YY. Diffusion and directed movement: in vitro motile properties of fission yeast kinesin-14 Pkl1. J Biol Chem. 2008;283(52):36465–36473. | |
Mitchison T, Kirschner M. Dynamic instability of microtubule growth. Nature. 1984;312:237–242. | |
Jordan MA, Wilson L. Microtubules as a target for anticancer drugs. Nat Rev Cancer. 2004;4:253–265. | |
VanBuren V, Cassimeris L, Odde DJ. Mechanochemical model of microtubule structure and self-assembly kinetics. Biophys J. 2005;89(5):2911–2926. | |
Nogales E, Wang H-W. Structural intermediates in microtubule assembly and disassembly: how and why? Curr Opin Cell Biol. 2006;18(2):179–184. | |
He M, Subramanian R, Bangs F, et al. The kinesin-4 protein Kif7 regulates mammalian hedgehog signalling by organizing the cilium tip compartment. Nat Cell Biol. 2014;16(7):663–672. | |
Cui W, Sproul LR, Gustafson SM, Matthies HJG, Gilbert SP, Hawley RS. Drosophila Nod protein binds preferentially to the plus ends of microtubules and promotes microtubule polymerization in vitro. Mol Biol Cell. 2005;16:5400–5409. | |
Sardar HS, Luczak VG, Lopez MM, Lister BC, Gilbert SP. Mitotic kinesin CENP-E promotes microtubule plus-end elongation. Curr Biol. 2010;20:1648–1653. | |
Garcia MA, Koonrugsa N, Toda T. Two kinesin-like Kin I family proteins in fission yeast regulate the establishment of metaphase and the onset of anaphase A. Curr Biol. 2002;12(8):610–621. | |
Ganem N, Compton D. Functional roles of poleward microtubule flux during mitosis. Cell Cycle. 2006;5(5):481–485. | |
Cameron LA, Yang G, Cimini D, et al. Kinesin 5-independent poleward flux of kinetochore microtubules in PtK1 cells. J Cell Biol. 2006;173(2):173–179. | |
Ferenz NP, Gable A, Wadsworth P. Mitotic functions of kinesin-5. Semin Cell Dev Biol. 2010;21(3):255–259. | |
Waitzman JS, Rice SE. Mechanism and regulation of kinesin-5, an essential motor for the mitotic spindle. Biol Cell. 2014;106(1):1–12. | |
Fu C, Ward JJ, Loiodice I, Velve-Casquillas G, Nedelec FJ, Tran PT. Phospho-regulated interaction between kinesin-6 Klp9p and microtubule bundler Ase1p promotes spindle elongation. Dev Cell. 2009;17(2):257–267. | |
Saunders A, Powers J, Strome S, Saxton W. Kinesin-5 acts as a brake in anaphase spindle elongation. Curr Biol. 2007;17(12):1–5. | |
Myers KA, Baas PW. Kinesin-5 regulates the growth of the axon by acting as a brake on its microtubule array. J Cell Biol. 2007;178(6):1081–1091. | |
Liu M, Nadar VC, Kozielski F, Kozlowska M, Yu W, Baas PW. Kinesin-12, a mitotic microtubule-associated motor protein, impacts axonal growth, navigation, and branching. J Neurosci. 2010;30(44):14896–14906. | |
Karabay A, Walker R. Identification of Ncd tail domain-binding sites on the tubulin dimer. Biochem Biophys Res Commun. 2003;305(3):523–528. | |
Oladipo A, Cowan A, Rodionov V. Microtubule motor Ncd induces sliding of microtubules in vivo. Mol Biol Cell. 2007;18:3601–3606. | |
Tao L, Mogilner A, Civelekoglu-Scholey G, et al. A homotetrameric kinesin-5, KLP61F, bundles microtubules and antagonizes Ncd in motility assays. Curr Biol. 2006;16(23):2293–2302. | |
Furuta K, Furuta A, Toyoshima YY, Amino M, Oiwa K, Kojima H. Measuring collective transport by defined numbers of processive and nonprocessive kinesin motors. Proc Natl Acad Sci U S A. 2013;110(2):501–506. | |
Ito M, Morii H, Shimizu T, Tanokura M. Coiled coil in the stalk region of ncd motor protein is nonlocally sustained. Biochemistry. 2006;45(10):3315–3324. | |
Makino T, Morii H, Shimizu T, et al. Reversible and irreversible coiled coils in the stalk domain of ncd motor protein. Biochemistry. 2007;46(33):9523–9532. | |
Fuchs E, Karakesisoglou I. Bridging cytoskeletal intersections. Genes Dev. 2001;15(1):1–14. | |
Liao G, Gundersen G. Kinesin is a candidate for cross-bridging microtubules and intermediate filaments. J Biol Chem. 1998;273(16):9797–9803. | |
Gundersen G, Kreitzer G, Cook T, Liao G. Microtubules as determinants of cellular polarity. Biol Bull. 1998;194:358–360. | |
Prahlad V, Yoon M, Moir RD, Vale RD, Goldman RD. Rapid movements of vimentin on microtubule tracks: kinesin-dependent assembly of intermediate filament networks. J Cell Biol. 1998;143(1):159–170. | |
Etienne-Manneville S. Microtubules in cell migration. Annu Rev Cell Dev Biol. 2013;29:471–499. | |
Lillie SH, Brown SS. Suppression of a myosin defect by a kinesin-related gene. Nature. 1992;356:358–361. | |
Lillie SH, Brown SS. Immunofluorescence localization of the unconventional myosin, Myo2p, and the putative kinesin-related protein, Smy1p, to the same regions of polarized growth in Saccharomyces cerevisiae. J Cell Biol. 1994;125(4):825–842. | |
Beningo KA, Lillie SH, Brown SS. The yeast kinesin-related protein Smy1p exerts its effects on the class V myosin Myo2p via a physical interaction. Mol Biol Cell. 2000;11(2):691–702. | |
Chesarone-Cataldo M, Guérin C, Yu JH, Wedlich-Soldner R, Blanchoin L, Goode BL. The myosin passenger protein Smy1 controls actin cable structure and dynamics by acting as a formin damper. Dev Cell. 2011;21:217–230. | |
Verhey KJ, Gaertig J. The tubulin code. Cell Cycle. 2007;6: 2152–2160. | |
McKean PG, Vaughan S, Gull K. The extended tubulin superfamily. J Cell Sci. 2001;114(15):2723–2733. | |
Ludueña RF, Banerjee A. The isotypes of tubulin- distribution and functional significance. In: Tito F, editor. Cancer Drug Discovery and Development: The Role of Microtubules in Cell Biology, Neurobiology, and Oncology. Tototwa, NJ: Humana press; 2008:123–175. | |
Wilson PG, Zheng Y, Oakley CE, Oakley BR, Borisy GG, Fuller MT. Differential expression of two gamma-tubulin isoforms during gametogenesis and development in Drosophila. Dev Biol. 1997;184:207–221. | |
Stearns T, Evans L, Kirschner M. Gamma-tubulin is a highly conserved component of the centrosome. Cell. 1991;65:825–836. | |
Tuszynski JA, Carpenter EJ, Huzil JT, Malinski W, Luchko T, Luduena RF. The evolution of the structure of tubulin and its potential consequences for the role and function of microtubules in cells and embryos. Int J Dev Biol. 2006;50:341–358. | |
Janke C, Bulinski JC. Post-translational regulation of the microtubule cytoskeleton: mechanisms and functions. Nat Rev Mol Cell Biol. 2011;12(12):773–786. | |
Konishi Y, Setou M. Tubulin tyrosination navigates the kinesin-1 motor domain to axons. Nat Neurosci. 2009;12(5):559–567. | |
Karcher RL, Deacon SW, Gelfand VI. Motor-cargo interactions: the key to transport specificity. Trends Cell Biol. 2002;12(1):21–27. | |
Hirokawa N, Noda Y. Intracellular transport and kinesin superfamily proteins, KIFs: structure, function, and dynamics. Physiol Rev. 2008;88:1089–1118. | |
Welburn JPI. The molecular basis for kinesin functional specificity during mitosis. Cytoskeleton (Hoboken). 2013;70(9):476–493. | |
Pedersen LB, Rosenbaum JL. Chapter two intraflagellar transport (IFT). Role in ciliary assembly, resorption and signalling. Curr Top Dev Biol. 2008;85:23–61. | |
Conde C, Cáceres A. Microtubule assembly, organization and dynamics in axons and dendrites. Nat Rev Neurosci. 2009;10(5):319–332. | |
Blasius TL, Reed N, Slepchenko BM, Verhey KJ. Recycling of kinesin-1 motors by diffusion after transport. PLoS One. 2013;8(9):e76081. | |
Gudimchuk N, Vitre B, Kim Y. Kinetochore kinesin CENP-E is a processive bi-directional tracker of dynamic microtubule tips. Nat Cell Biol. 2013;15(9):1079–1088. | |
Abrieu A, Kahana JA, Wood KW, Cleveland DW. CENP-E as an essential component of the mitotic checkpoint in vitro. Cell. 2000;102(6):817–826. | |
Tamura N, Draviam VM. Microtubule plus-ends within a mitotic cell are “moving platforms” with anchoring, signalling and force-coupling roles. Open Biol. 2012;2(11):120132. | |
Yao X, Abrieu A, Zheng Y, Sullivan KF, Cleveland DW. CENP-E forms a link between attachment of spindle microtubules to kinetochores and the mitotic checkpoint. Nat Cell Biol. 2000;2:484–491. | |
Farzan SF, Ascano M Jr, Ogden SK, et al. Costal2 functions as a kinesin-like protein in the hedgehog signal transduction pathway. Curr Biol. 2008;18(16):1215–1220. | |
Méthot N, Basler K. An absolute requirement for Cubitus interruptus in hedgehog signaling. Development. 2001;128(5):733–742. | |
Batut J, Howell M, Hill CS. Kinesin-mediated transport of Smad2 is required for signaling in response to TGF-beta ligands. Dev Cell. 2007;12(2):261–274. | |
Simeonov DR, Kenny K, Seo L, Moyer A, Allen J, Paluh JL. Distinct Kinesin-14 mitotic mechanisms in spindle bipolarity. Cell Cycle. 2009;8(21):3563–3575. | |
Allingham JS, Sproul LR, Rayment I, Gilbert SP. Vik1 modulates microtubule-Kar3 interactions through a motor domain that lacks an active site. Cell. 2007;128(6):1161–1172. | |
Chu H, Yun M, Anderson D. Kar3 interaction with Cik1 alters motor structure and function. EMBO J. 2005;24(18):3214–3223. | |
Sproul L, Anderson D, Mackey A. Cik1 targets the minus-end kinesin depolymerase kar3 to microtubule plus ends. Curr Biol. 2005;15(15):1420–1427. | |
Barrett JG, Manning BD, Snyder M. The Kar3p kinesin-related protein forms a novel heterodimeric structure with its associated protein Cik1p. Mol Biol Cell. 2000;11(7):2373–2385. | |
Manning BD, Barrett JG, Wallace JA, Granok H, Snyder M. Differential regulation of the Kar3p kinesin-related protein by two associated proteins, Cik1p and Vik1p. J Cell Biol. 1999;144(6):1219–1233. | |
Gibeaux R, Politi AZ, Nédélec F, Antony C, Knop M. Spindle pole body-anchored Kar3 drives the nucleus along microtubules from another nucleus in preparation for nuclear fusion during yeast karyogamy. Genes Dev. 2013;27(3):335–349. | |
Grishchuk EL, McIntosh JR. Microtubule depolymerization can drive poleward chromosome motion in fission yeast. EMBO J. 2006;25(20):4888–4896. | |
Mayer C, Filopei J, Batac J, Alford L, Paluh JL. An extended anaphase signaling pathway for Mad2p includes microtubule organizing center proteins and multiple motor-dependent transitions. Cell Cycle. 2011;5(13):1456–1463. | |
Endow SA, Hallen MA. Anastral spindle assembly and γ-tubulin in Drosophila oocytes. BMC Cell Biol. 2011;12:1. | |
Hallen MA, Ho J, Yankel CD, Endow SA. Fluorescence recovery kinetic analysis of gamma-tubulin binding to the mitotic spindle. Biophys J. 2008;95(6):3048–3058. | |
Cai S, Weaver L. Kinesin-14 family proteins HSET/XCTK2 control spindle length by cross-linking and sliding microtubules. Mol Biol Cell. 2009;20:1348–1359. | |
Mountain V, Simerly C, Howard L, Ando A, Schatten G, Compton DA. The kinesin-related protein, HSET, opposes the activity of Eg5 and cross-links microtubules in the mammalian mitotic spindle. J Cell Biol. 1999;147:351–365. | |
Foley EA, Kapoor TM. Microtubule attachment and spindle assembly checkpoint signaling at the kinetochore. Nat Rev Mol Cell Biol. 2013;14(1):25–37. | |
Olmsted ZT, Riehlman TD, Branca CN, Colliver AG, Cruz LO, Paluh JL. Kinesin-14 Pkl1 targets γ-tubulin for release from the γ-tubulin ring complex (γ-TuRC). Cell Cycle. 2013;12(5):842–848. | |
Mirchenko L, Uhlmann F. Sli15INCENP dephosphorylation prevents mitotic checkpoint reengagement due to loss of tension at anaphase onset. Curr Biol. 2010;20:1396–1401. | |
Fliegauf M, Benzing T, Omran H. When cilia go bad: cilia defects and ciliopathies. Nat Rev Mol Cell Biol. 2007;8:880–893. | |
Verhey K, Dishinger J, Kee H. Kinesin motors and primary cilia. Biochem Soc Trans. 2011;39(5):1120–1125. | |
Mukhopadhyay B, Nam SC, Choi KW. Kinesin II is required for cell survival and adherens junction positioning in Drosophila photoreceptors. Genesis. 2010;48:522–530. | |
Bhogaraju S, Engel BD, Lorentzen E. Intraflagellar transport complex structure and cargo interactions. Cilia. 2013;2(1):10. | |
Bettencourt-Dias M, Glover DM. Centrosome biogenesis and function: centrosomics brings new understanding. Nat Rev Mol Cell Biol. 2007;8(6):451–463. | |
Moritz M, Braunfeld MB, Guénebaut V, Heuser J, Agard DA. Structure of the gamma-tubulin ring complex: a template for microtubule nucleation. Nat Cell Biol. 2000;2(6):365–370. | |
Rodriguez AS, Batac J, Killilea AN, et al. Protein complexes at the microtubule organizing center regulate bipolar spindle assembly. Cell Cycle. 2008;7(9):1246–1253. | |
Piao T, Luo M, Wang L, et al. A microtubule depolymerizing kinesin functions during both flagellar disassembly and flagellar assembly in Chlamydomonas. Proc Natl Acad Sci U S A. 2009;106(12):4713–4718. | |
Endoh-Yamagami S, Evangelista M, Wilson D, et al. The mammalian Cos2 homolog Kif7 plays an essential role in modulating Hh signal transduction during development. Curr Biol. 2009;19(15):1320–1326. | |
Liem KF, He M, Ocbina PJR, Anderson KV. Mouse Kif7/Costal2 is a cilia-associated protein that regulates Sonic hedgehog signaling. Proc Natl Acad Sci U S A. 2009;106:13377–13382. | |
Wilson CW, Nguyen CT, Chen MH, et al. Fused has evolved divergent roles in vertebrate hedgehog signalling and motile ciliogenesis. Nature. 2009;459:98–102. | |
Takeda S, Yonekawa Y, Tanaka Y, Okada Y, Nonaka S, Hirokawa N. Left-right asymmetry and kinesin superfamily protein KIF3A: new insights in determination of laterality and mesoderm induction by kif3A-/- mice analysis. J Cell Biol. 1999;145(4):825–836. | |
Liu X, Gong H, Huang K. Oncogenic role of kinesin proteins and targeting kinesin therapy. Cancer Sci. 2013;104:651–656. | |
Rath O, Kozielski F. Kinesins and cancer. Nat Rev Cancer. 2012;12(8):527–539. | |
Yu Y, Feng YM. The role of kinesin family proteins in tumorigenesis and progression. Cancer. 2010;116:5150–5160. | |
Zhou J, Giannakakou P. Targeting microtubules for cancer chemotherapy. Curr Med Chem Anticancer Agents. 2005;5:65–71. | |
Zhu Y, Zhou Y, Shi J. Post-slippage multinucleation renders cytotoxic variation in anti-mitotic drugs that target the microtubules or mitotic spindle. Cell Cycle. 2014;13(11):1756–1764. | |
Tang Y, Orth J, Xie T, Mitchison T. Rapid induction of apoptosis during Kinesin-5 inhibitor-induced mitotic arrest in HL60 cells. Cancer Lett. 2011;310(1):15–24. | |
Rose WC. Taxol: a review of its preclinical in vivo antitumor activity. Anticancer Drugs. 1992;3:311–321. | |
Pellegrini F, Budman DR. Review: tubulin function, action of antitubulin drugs, and new drug development. Cancer Invest. 2005;23: 264–273. | |
Baas PW, Ahmad FJ. Beyond taxol: microtubule-based treatment of disease and injury of the nervous system. Brain. 2013;136:2937–2951. | |
Mayer TU, Kapoor TM, Haggarty SJ, King RW, Schreiber SL, Mitchison TJ. Small molecule inhibitor of mitotic spindle bipolarity identified in a phenotype-based screen. Science. 1999;286(5441):971–974. | |
Ostergaard P, Simpson MA, Mendola A, et al. Mutations in KIF11 cause autosomal-dominant microcephaly variably associated with congenital lymphedema and chorioretinopathy. Am J Hum Genet. 2012;90(2):356–362. | |
Franker MAM, Hoogenraad CC. Microtubule-based transport – basic mechanisms, traffic rules and role in neurological pathogenesis. J Cell Sci. 2013;126(11):2319–2329. | |
Goldstein LS. Kinesin molecular motors: transport pathways, receptors, and human disease. Proc Natl Acad Sci U S A. 2001;98(13):6999–7003. | |
Millecamps S, Julien J-P. Axonal transport deficits and neurodegenerative diseases. Nat Rev Neurosci. 2013;14:161–176. | |
Mandelkow E, Mandelkow E-M. Kinesin motors and disease. Trends Cell Biol. 2002;12(12):585–591. | |
Crimella C, Baschirotto C, Arnoldi A, et al. Mutations in the motor and stalk domains of KIF5A in spastic paraplegia type 10 and in axonal charcot-Marie-Tooth type 2. Clin Genet. 2012;82:157–164. | |
Füger P, Sreekumar V, Schüle R, et al. Spastic paraplegia mutation N256S in the neuronal microtubule motor KIF5A disrupts axonal transport in a Drosophila HSP model. PLoS Genet. 2012;8(11):e1003066. | |
Gentil BJ, Cooper L. Molecular basis of axonal dysfunction and traffic impairments in CMT. Brain Res Bull. 2012;88(5):444–453. | |
Zhao C, Takita J, Tanaka Y, et al. Charcot-Marie-Tooth disease type 2A caused by mutation in a microtubule motor KIF1B. Cell. 2001;105(5):587–597. | |
Desai J, Velo M, Yamada K. Spatiotemporal expression pattern of KIF21A during normal embryonic development and in congenital fibrosis of the extraocular muscles type 1 (CFEOM1). Gene Expr Patterns. 2012;12:180–188. | |
Lee KH, Lee JS, Lee D, et al. KIF21A-mediated axonal transport and selective endocytosis underlie the polarized targeting of NCKX2. J Neurosci. 2012;32(12):4102–4117. | |
Yamada K. Identification of KIF21A mutations as a rare cause of congenital fibrosis of the extraocular muscles type 3 (CFEOM3). Invest Ophthalmol Vis Sci. 2004;45(7):2218–2223. | |
Nakajima K, Yin X, Takei Y, Seog D-H, Homma N, Hirokawa N. Molecular motor KIF5A is essential for GABA(A) receptor transport, and KIF5A deletion causes epilepsy. Neuron. 2012;76(5):945–961. | |
Willemsen MH, Ba W, Wissink-Lindhout WM, et al. Involvement of the kinesin family members KIF4A and KIF5C in intellectual disability and synaptic function. J Med Genet. 2014;51(7):487–494. | |
Tarabeux J, Champagne N, Brustein E, et al; Synapse toDiseaseTeam. De novo truncating mutation in kinesin 17 associated with schizophrenia. Biol Psychiatry. 2010;68(7):649–656. | |
Goldstein LS, Yang Z. Microtubule-based transport systems in neurons: the roles of kinesins and dyneins. Annu Rev Neurosci. 2000;23:39–71. | |
Miki H, Setou M, Kaneshiro K, Hirokawa N. All kinesin superfamily protein, KIF, genes in mouse and human. Proc Natl Acad Sci U S A. 2001;98(13):7004–7011. | |
Lin S, Liu M, Son YJ, et al. Inhibition of Kinesin-5, a microtubule-based motor protein, as a strategy for enhancing regeneration of adult axons. Traffic. 2011;12:269–286. | |
Bush SF. Toward in vivo nanoscale communication networks: utilizing an active network architecture. Front Comput Sci China. 2011;5(3):316–326. | |
Steuerwald D, Früh S, Griss R, Lovchik R, Vogel V. Nanoshuttles propelled by motor proteins sequentially assemble molecular cargo in a microfluidic device. Lab Chip. 2014;14:30. | |
Mann S. Life as a nanoscale phenomenon. Angew Chem Int Ed Engl. 2008;47:5306–5320. | |
Milic B, Andreasson JOL, Hancock WO, Block SM. Kinesin processivity is gated by phosphate release. Proc Natl Acad Sci U S A. 2014;111(39):14136–14140. | |
Kull F, Sablin E, Lau R, Fletterick R, Vale R. Crystal structure of the kinesin motor domain reveals a structural similarity to myosin. Nature. 1996;380(6574):550–555. | |
Ebbing B, Mann K, Starosta A, et al. Effect of spastic paraplegia mutations in KIF5A kinesin on transport activity. Hum Mol Genet. 2008;17(9):1245–1252. | |
Yamada K, Andrews C, Chan WM, et al. Heterozygous mutations of the kinesin KIF21A in congenital fibrosis of the extraocular muscles type 1 (CFEOM1). Nat Genet. 2003;35:318–321. | |
DeBerg HA, Blehm BH, Sheung J, et al. Motor domain phosphorylation modulates kinesin-1 transport. J Biol Chem. 2013;288: 32612–32621. | |
Morfini GA, You YM, Pollema SL, et al. Pathogenic huntingtin inhibits fast axonal transport by activating JNK3 and phosphorylating kinesin. Nat Neurosci. 2009;12:864–871. | |
Pigino G, Morfini G, Pelsman A, Mattson MP, Brady ST, Busciglio J. Alzheimer’s presenilin 1 mutations impair kinesin-based axonal transport. J Neurosci. 2003;23:4499–4508. | |
Ari C, Borysov SI, Wu J, Padmanabhan J, Potter H. Alzheimer amyloid beta inhibition of Eg5/kinesin 5 reduces neurotrophin and/or transmitter receptor function. Neurobiol Aging. 2014;35: 1839–1849. | |
Lin F, Hiesberger T, Cordes K, et al. Kidney-specific inactivation of the KIF3A subunit of kinesin-II inhibits renal ciliogenesis and produces polycystic kidney disease. Proc Natl Acad Sci U S A. 2003;100: 5286–5291. | |
Hirokawa N, Tanaka Y, Okada Y. Left-right determination: involvement of molecular motor KIF3, cilia, and nodal flow. Cold Spring Harb Perspect Biol. 2009;1:a000802. | |
Waitzman JS, Larson AG, Cochran JC, et al. The loop 5 element structurally and kinetically coordinates dimers of the human kinesin-5, Eg5. Biophys J. 2011;101:2760–2769. | |
Goulet A, Behnke-Parks WM, Sindelar CV, Major J, Rosenfeld SS, Moores CA. The structural basis of force generation by the mitotic motor kinesin-5. J Biol Chem. 2012;287(53):44654–44666. | |
Wood KW, Lad L, Luo L, et al. Antitumor activity of an allosteric inhibitor of centromere-associated protein-E. Proc Natl Acad Sci U S A. 2010;107(13):5839–5844. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.