Back to Journals » International Journal of Nanomedicine » Volume 15
The Influence of Nanoparticle Properties on Oral Bioavailability of Drugs
Authors Wang Y, Pi C, Feng X, Hou Y, Zhao L, Wei Y
Received 7 April 2020
Accepted for publication 13 July 2020
Published 24 August 2020 Volume 2020:15 Pages 6295—6310
DOI https://doi.org/10.2147/IJN.S257269
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Mian Wang
Yuanyuan Wang,* Chao Pi,* Xianhu Feng, Yi Hou, Ling Zhao, Yumeng Wei
Central Nervous System Drug Key Laboratory of Sichuan Province, School of Pharmacy, Southwest Medical University, Luzhou, Sichuan 646000, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Ling Zhao; Yumeng Wei Tel/ Fax +86 830 3162292
; +86 830 3162291
Email [email protected]; [email protected]
Abstract: Oral administration has been the most common therapeutic regimen in various diseases because of its high safety, convenience, lower costs, and high compliance of patients. However, susceptible in hostile gastrointestinal (GI) environment, many drugs show poor permeability across GI tract mucus and intestinal epithelium with poor oral absorption and limited therapeutic efficacy. In recent years, nanoparticulate drug delivery systems (NDDS) have become a hot research spot because of their unique advantages including protecting drug from premature degrading and interacting with the physiological environment, increasing intracellular penetration, and enhancing drug absorption. However, a slight change in physicochemistry of nanoparticles can significantly impact their interaction with biological pathways and alter the oral bioavailability of drugs. Hence, this review focuses on the factors affecting oral bioavailability from two aspects. On the one hand, the factors are the biochemical and physiological barriers in oral drugs delivery. On the other hand, the factors are the nanoparticle properties including size, surface properties, and shape of nanoparticles.
Keywords: oral bioavailability, nanoparticle properties, size, shape, surface properties
Introduction
Oral administration has been the most common manner of drug delivery for thousands of years, with some advantages including the safety, it being well-tolerated, with low treatment costs, good compliance, and convenience.1,2 Despite its outstanding advantages, the traditionally oral administered drugs are faced with the daunting challenges of poor and highly variable bioavailability, which can be frequently caused by inherent instability and low solubility in variable conditions of the gastrointestinal (GI) tract, poor permeability through GI barriers, and, in some cases, extensive pre-systemic metabolism in the GI tract and liver (eg, cytochrome P-450).2,5
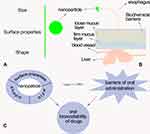
To surmount these barriers in the GI tract and significantly improve oral bioavailability, designing and developing some effective oral drug delivery systems is quite urgent.6 Thus, a large variety of new techniques and dosages of drugs have been developed such as protein or polymer conjugates,7 solid drug dispersions,8 nanoparticle technologies,9 and macroscopic systems such as capsules, gels, and films.10 Among these strategies, nanoparticulate drug delivery systems (NDDS) have been attracting more attention since they can protect the drug from premature degradation and interaction with the physiological environment, increase intracellular penetration, and enhance drug absorption (Figure 1).11,14 By far, numerous NDDS including albumin nanoparticles,15 chitosan (CS) nanoparticles,16 liposomes,17 polymeric micelles,18 etc. have been developed for the oral delivery of different drugs.
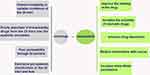 |
Figure 1 The benefits of using nanoparticles.Abbreviation: GI, gastrointestinal. |
However, previous studies have reported that a slight change in physicochemistry of nanoparticles can significantly impact on their interaction with biological pathways and alter the oral bioavailability of drugs.6,11,19 For example, the control of dimension under 200 nm was more absorbed by the GI tract.4,20,21 And other reports found that polystyrene (PS) nanoparticles decorated with vitamin B12 (VitB12) were more easily avoiding lysosomal digestion as well as entering epithelial cells, compared with both soluble VitB12 ligand and unmodified nanoparticles.22 In addition to the influence of size and surface properties of nanoparticles, another paper reported that oral bioavailability of drugs was also significantly impacted by nanoparticle geometry,23–25 such as the fact that rod-nanoparticles had better oral bioavailability compared with sphere-nanoparticles.26 Therefore, it is necessary to summarize the role of the size, surface properties, and shape of nanoparticle in oral drug delivery. We believe that understanding the role of nanoparticle properties in GI tract transport as well as cellular uptake can bring forth a paradigm shift in nanoparticle engineering for oral delivery. Hence, this review focuses on the factors affecting oral bioavailability, in which these factors are divided into two categories: the biochemical and physiological barriers in oral drugs delivery; and the nanoparticle properties including size, surface properties, and shape of nanoparticles (Figure 2).
Physiological and Biochemical Disorders in Oral Delivery
Although oral drug delivery has been widely used in clinical practice, oral bioavailability has always been limited by the disorders of oral administration, including the variation in PH conditions, surfactants, and rich enzyme content in the GI tract, loose and firm mucus, and transporters in vivo. Thus, it is vital to give an insight into the obstacles for the development of oral drugs and the boosting of oral bioavailability of drugs.27
Biochemical Barriers
After oral administration, drugs would confront a harsh biochemical environment,28 which comprises of PH variation in the GI tract, metabolizing enzymes, surfactants like bile salt, and the liver itself.4 The drugs can be destroyed by acidic environment, especially materials that are sensitive to acidic conditions.28 Furthermore, the variation in PH conditions is from acidic to alkaline along the GI tract, which imposes a hurdle to the stability of drugs and sometimes their carriers.29 In addition, surfactants in the GI tract also have a substantial impact on the structural integrity of drugs.29 Beyond these, the drugs after oral administration still have to face a particularly rich enzyme content, composed of proteases, lipases, or amylases.30 These enzymes exist in the digestive tract, especially in the lower one, which are responsible for the metabolism.4 After entering intestinal epithelial cells, the drugs will be threatened by extra-hepatic microsomal enzymes on the endoplasmic reticulum inside the cytoplasm liver (eg, the cytochrome P450 3A family of phaseⅠ metabolic enzymes).31 In conclusion, the bioavailability of drugs can be limited by the biochemical barriers after oral administration. Hence, the numerous drugs are encapsulated to nanoparticles. Furthermore, the materials used as the nanocarrier should be carefully chosen or chemically modified adequately.28
Physiological Barriers
Mucus Barriers
Some reports mentioned that drugs must be capable of penetrating and crossing mucus barriers in order to be absorbed and gain access to the circulatory system and target tissue.32,33 The mucus layer is ubiquitous in the GI tract composed of water, proteins, lipids, electrolytes, antimicrobial peptides, sloughed epithelial cells, bile salts, and other components available in the GI tract.34,35 Mucus can be divided into two types: loose and firm mucus (Figure 3B).36 Loose mucus is easy to be removed by suction as well as shear and high thickness of the layer.37,38 However, firm mucus adheres firmly or anchors to the epithelium surface and is resistant to the removal by suction and shear.37 The main structural constituent of the mucus layer is mucin (Figure 3A) that can be defined as glycoproteins containing heavily O-glycosylated serine/threonine-rich tandem repeat domains.33,39 The oligosaccharide side chains including a terminal carboxyl group or ester sulfate groups give mucus its negative charge.33,40,41 Some positively charged drugs and their carriers have been shown to have electrostatic interactions with the glycoproteins from mucus, limiting the drugs absorption rate and decreasing the oral bioavailability of drugs.34 Meanwhile, hydrophobic bare globular regions (non-glycosylated regions) with high density located on mucin chains generate multiple low-affinity adhesion interactions with hydrophobic regions of foreign particles.42 The large number of hydrophobic drugs through the mucus can be rapidly removed, impairing drug effective therapy.42 In addition, mucin molecules can form a network via disulfide bonds. The non-glycosylated regions of the mucin molecule are the site of interchain disulfide bridge that connects the glycoprotein subunits.43 The mesh space and the brush-like structure of mucus can act as a size exclusion filter, reducing the mobility of large molecules, thereby increasing their clearance rate.40,44,46 Another intriguing finding is that the thickness of the mucosal barrier varies with the location of the GI region (Table 1).35,36 Crossing the mucus layer implies a particle size below 200 nm.28 Indeed, nanocarriers should be small enough to avoid being blocked by the mucin mesh. Moreover, interactions between the surface of nanocarriers and mucus must be kept at a minimum.
 |
Table 1 Thickness of Mucus in Different Parts of the Human Gastrointestinal Tract |
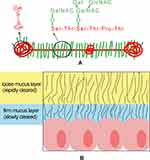 |
Figure 3 (A) Highly O-glycosylated mucin domains. Red, protein core; Green, oligosaccharides. (B) Schematic representation illustrating two types of mucus. |
Absorption Barriers of the Intestinal Epithelium
Intestinal epithelium with dense and orderly brush border on the top surface is an intrinsic physical barrier that consists of a single layer of columnar epithelial cells comprised of absorptive (enterocytes) and secretory cells (goblet cells and paneth cells).47,49 Epithelium permeation of drugs can occur via transcellular route, paracellular avenues, and the densely clustered M cells located on the surface of Payer’s patches.47,50 The transcellular pathway mainly absorbs particles through the cell membrane of enterocytes relying on their size and hydrophobicity.50 In the absence of aggregation, the particles of smaller size are more easily absorbed by intestinal epithelial cells. It has been reported that a particle size smaller than 300 nm could be untaken by enterocytes, while a size larger than 500 nm was more likely to be absorbed in jejuna Payer’s patches.50,52 The paracellular pathway is another limiting step in transepithelial transport, mediated through tight junctions (TJs), which is the way for the hydrophilic molecules uptake.49,50,53 Moreover, most macromolecular drugs do not readily enter systemic circulation through intercellular connections. It is reported that Ca2+ chelating agents could increase the paracellular permeability by reversible opening of the tight junctions.54,55 Payer’s patches are actually the submucosal mass lymph node of the small intestine, which are the main site of intestinal mucosal immune induction. M cells located on the surface of Payer’s patches act as an entrance for drugs to the lymphatic vessels, especially lipid soluble drugs, from the intestinal cavity to lymphoid cells, which shall reduce the primary clearance of the liver.50 However, the lymphatic pathway shows only a high affinity for lipophilic compounds. Ling et al56 increased the bioavailability of cefotaxime via liposome preparation compared with aqueous solution and the physical mixture.
Transporters in vivo
In addition, there are a variety of transporters in vivo, such as P-glycoprotein (P-gp), cytoplasmic transporter, breast cancer drug resistant proteins, and multidrug resistance associated protein, which release drugs from the intracellular environment to outside of the cell.4 P-gp encoded by multidrug resistance-1 gene is the major drug efflux transporter system.4 A lot of drugs (eg, paclitaxel (PTX), docetaxel, etoposide, vinblastine, vincristine, and doxorubicin) are known as potential substrates of efflux transporters. Various efflux transporters in vivo infinitely restrict oral absorption of drugs.4 Therefore, active modification of drug carriers is essential.
The Effect of Size for Oral Bioavailability
It has been reported that the bioadhesion of solid lipid nanoparticles (SLNs) in the GI tract can be improved via smaller size to prolong their residence time and thus play a contributing role in improving oral bioavailability.4,57,58 Retinoic acid-SLNs showed an increase of 3-fold oral bioavailability of retinoic acid when particle size decreases from 328.8 nm to 89.3 nm.59 The fact proved that particle size plays a crucial role in oral drug delivery.60
It has been concluded previously that particle size has a great impact on the movement of particles in the mucus. For instance, Maisel et al61 found that particles of 40 and 100 nm in size spread over the mucosa reaching to the deep, folded surfaces of the colorectal epithelium, whereas particles 200 and 500 nm in size just spread over the surface. The data indicate that particles with smaller size are easily reaching to the firm mucus that is resistant to the removal by suction and shear, thereby prolonging their residence time. In addition, the cellular uptake and uptake pathway of particles are also affected by the size of nanoparticles. The smaller nanostructured lipid carriers (NLC-100 nm) show higher uptake efficiency in the Caco-2 cell (P<0.05) as well as higher permeation ability in Caco-2 cell monolayer (P<0.01) compared with NLC-200 nm and NLC-300 nm.62 Moreover, pharmacokinetic studies shown that NLC-100 nm exhibited highest Cmax and AUC compared with the others.62 Zhao et al63 also mentioned that the 100 nm nanovaccine exhibited better pharmacokinetic efficacy than the 500 nm nanovaccine in the presence of alum adjuvant. Li et al62 concluded from their data that the size of 100 nm might be the most suitable size for oral delivery of nanoparticles. However, the data of Banerjee et al6 indicated that particle uptake by Caco-2 and Caco-2/HT-29 cells was inversely related to their size, with uptake of 50 nm > 200 nm > 500 nm > 1000 nm.6 Thus, the hypothesis can be mentioned that nanoparticle size has a negative correlation influence on oral bioavailability of drugs during the certain range. However, the optimal particle size still needs to be actively explored by researchers in oral drug delivery.
The Effects of Surface Properties for Oral Bioavailability
NDDS have many desirable characteristics for drug administration such as improving solubility and stability of drugs.64 However, even the most potent extended release nanoparticle would be rapidly cleared from the body if it adheres to the superficial layers of mucus, reducing delivery of the drug to the target tissues.65 The report demonstrated that the surface chemistry of nanoparticles can greatly impact their interaction with biological pathways and alter efficacy (Table 2).11 Efficient oral delivery of drugs based on NDDS requires simultaneously overcoming physiological and biochemical disorders of the body, which needs very different or even contradictory surface properties of nanocarriers.66 In general, it is a necessity to achieve a variety of treatment goals with different materials or different modifications to nanoparticles. Thus, definitely learning the role of surface properties of nanoparticles about oral delivery is important.
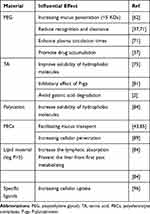 |
Table 2 The Effect of Various Surface Modification Materials of Nanoparticles on the Oral Administration |
The Particle Containing a Hydrophilic Group
As previously discussed, after entering the GI tract through the esophagus, the oral drugs can be removed by non-covalent bonds (such as hydrophobic bonds, hydrogen bonds, and electrostatic forces) with GI mucus, especially the superficial GI mucus, to prevent it entering the systemic circulation, thereby reducing the effective dosage of drugs. Neutral hydrophilic materials (Figure 4) get more and more attention because of their difficulty in hydrophobic and electrostatic interaction with GI mucus. Such substances that are easy to penetrate the mucus barrier are called mucopenetrating particles (MPP), which can improve epithelial distribution and retention time by penetrating the outer, rapidly clear mucus layers to reach the more slowly cleared layers and increase the effective amount of drugs into the systemic circulation.67
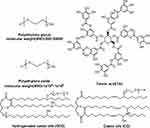 |
Figure 4 Structural formula of partial materials containing hydrophilic groups. |
PEGylated Drug Delivery Vehicles
The poly(ethylene glycol) (PEG), a passive mucopenetrating system, has been widely used in surface modification of nanoparticles due to the property of reducing interactions with both luminal components and mucus in the gut.36 Maisel et al61 used PEG-modified poly(lactic-co-glycolic acid) (PLGA) nanoparticles to study the mucus-penetrating of particles under different conditions (eg, providing normal and colitis mice model, or increasing the volume of gavage), and concluded that the particles were uniformly distributed on the mucosa of epithelial cells. In the PEGylated doxorubicin (DOX) loaded PLGA-Nanoparticles (PEGylated-DOX-PLGA-NPs) observed a 6.8-fold oral bioavailability as compared to DOX-S in pharmacokinetics studies.68 In addition, Feeney et al69 modified lipid nanoparticles (LN) by using carrier materials of hydrogenated castor oil (HCO) and castor oil (CO) (containing PEG groups). They found that the relative bioavailability (defined as the ratio of AUC 0–∞ for the modified LN and its structurally analogous LN) of the formulations was 120% and 182% for HCO and CO formulations, respectively. However, some articles also mentioned PEG had a strong viscosity, which speculated hydrogen bonding between ether oxygen atoms in PEG and sugars on glycosylated mucins. Wang et al70 explored the physicochemical properties of PEG-coated nanoparticles, specifically PEG molecular weight and degree of surface coverage, to reconcile the paradoxical reports of PEGs interactions with mucus. They explored the physicochemical properties of PEG-coated nanoparticles which were formulated from PS modified PEGs of various molecular weights (2, 5, 10 kDa) to reconcile the paradoxical reports of PEGs interactions with mucus. They found that PS-PEG2K(High) nanoparticles penetrated mucus with effective speed only 7-fold reduced compared with in water. And they concluded that low molecular weight and high (dense) PEG surface coverage was required for rapid mucus penetration of coated particles, and that dense PEG coatings transferred from being mucoinert to mucoadhesive when a critical molecular weight existed between 5 and 10 kDa.
Coating Particles with Tannic Acid (TA)
TA is a type of the natural polyphenols which is constituted plentiful catechol and pyrogallol moieties and generally recognized as safe by the United States Food and Drug Administration (FDA).71,72 In recent years, it has been reported that TA can improve solubility of hydrophobic molecules by hydrogen bonding.73 Shen et al72 proved that TA anchored onto the surface of hydrophobic PTX nanocore via multiple hydrogen bonding between pyrogallol or catechol groups of TA and hydroxyl groups or oxygen atoms of PTX molecules. The X-ray diffraction (XRD) pattern of the pure PTX powder showed a number of sharp Bragg peaks, corresponding to highly crystalline PTX.74 No discernible peaks appeared for the TA-PTX complex, indicating the amorphous nature of the complex coating.72 The data of release kinetics indicated that TA-PTX complex nanoparticles displayed a gradual release profile without burst release at an initial stage and finally reached 90% of drug release after 36 hours.72 The good dissolution behavior can be attributed to their amorphous nature of PTX in the complex nanoparticles, which was helpful to improve bioavailability.75 In addition, TA has been verified to exhibit high biological activities,76,77 such as inhibited effect of P-gp. It is all known that P-gp-mediated efflux is one of the major absorptive barriers in vivo.78 Kitagawa et al79 demonstrated that TA displayed the P-gp inhibitory function by ATPase inhibition. Some scholars believed that nanoparticles conjugated TA can avoid gastric acid degradation, achieve PH dependent intestinal site release, and increase oral absorption of drugs.2 They rationally developed a hydrogen-bonded TA acid-based nanoparticle, TA-PTX nanoparticles, via a flash nanoprecipitation process, and explored PH-sensitive behaviors of TA-PTX nanoparticles at different pH conditions. They found that the TA-PTX nanoparticles solution exhibited strong opaque property at acidic conditions and then recovered to 85% optical transmittance after adjusting to pH 7.4. The particles’ diameter immediately increased from 54 nm to about 2 µm after dispersing in pH 2, while reduced to 70 nm in pH 7.4. In addition, it was worth mentioning that microaggregates can be found in pH 2, and the approximate recovery of particle size to original state of TA-PTX nanoparticles when pH was adjusted to 6.8 or 7.4. These results suggested that TA-PTX nanoparticles can reduce the burst release of drugs, possibly prevent the acidic degradation of PTX in the stomach, and achieve intestinal site-specific drug release through physiological pH stimulation.
Coating Particles with Other Hydrophilic Substance
A great number of other hydrophilic substances are abounding in nature, such as polyethylene oxide (PEO), poloxamer 188, and pluronic F-127 (PF-127). To enhance mucus permeation and promote cellular uptake compared with cabazitaxel (CTX), Ren et al80 formulated CTX polymer–lipid hybrid nanoparticles (CTX-PMONP). The nanocarriers were comprised of a poly(ε-caprolactone) (PCL) and chain triglyceride hybrid core for drug loading, and encased in PEO shell by insertion of poloxamer 188.80 The oral bioavailability of CTX was elevated from 7.7% CTX solution (CTX-Sol) to 56.6% after oral administration of CTX-PMONPs, approximately 7.3-times higher than that of CTX-Sol.80 In addition, Li et al81 found that when the mucoinert polymer PF-127 was incorporated, the mucus penetrating properties of liposomes were improved significantly. PF127-inlaid liposomes and PF127-adsorbed liposomes were prepared by a thin-film hydration method followed by extrusion, in which coumarin 6 was loaded as a fluorescence marker.81 A modified Franz diffusion cell mounted with the intestinal mucus of rats was used to study the diffusion characteristics of the two types of PF127 liposomes.81 The diffusion efficiency of the two types of PF127-modified liposomes through intestinal rat mucus was 5-7-fold higher than that of unmodified liposomes. In general, hydrophilic materials are easy to penetrate the mucus barrier because of their difficulty in hydrophobic and electrostatic interaction with GI mucus, thereby increasing the effective amount of drugs into the systemic circulation.
The Particle Containing Polycationic Group
Formulation drugs with polycationic and surface-modified polycation particles have been shown to increase solubility, protect labile compounds from pH changes, and digestive enzymes.82 And some studies also illustrated that apart from uncharged nanoparticles with hydrophilic and a low hydrogen bonding capability, a densely charged nanoparticle yet net neutral surface can be also considered as MPP. These particles with neutral surface, high surface charge density of polyelectrolyte complexes (PECs), can create a hydrophilic surface that decreases its hydrophobic interactions with mucus and facilitate mucus transport.42,83 CS, an attractive polymer, has been extensively used in oral delivery. The combination of CS and chondroitin sulfate was used to prepare PECs that could penetrate mucus to the higher extent compared with reference nanoparticles.84 In addition, interferon alpha (IFNα)-loaded CS-nanoparticles were prepared by the ionotropic gelation method between the polycationic CS and sodium tripolyphosphate pentabasic anions, which were a potential delivery system for the oral administration compared with IFNα.85 Dyawanapelly et al86 developed PLGA nanoparticles, surface-modified chitosan oligosaccharide (COS)-PLGA nanoparticles, and CS-PLGA nanoparticles and compared mucoadhesion of CS and COS surface-modified polymer nanoparticles for mucosal delivery of proteins. Their data indicated that positively charged COS-PLGA nanoparticles and CS-PLGA nanoparticles exhibited higher mucoadhesion than negatively charged PLGA nanoparticles. Wang et al87 also prepared PLGA nanoparticle coating with CS and performed cellular uptake mechanisms as well as transmembrane permeability in Madin-Darby canine kidney-cell monolayers. They found that, under all conditions, CS-PLGA nanoparticles showed a greater potential to be transported into cells. Furthermore, pharmacokinetic studies demonstrated marked improvement of 3.53-fold and 8.03-fold in Wistar rat’s plasma as well as brain higher oral bioavailability through CS-coated-Irinotecan (IRN)-loaded-PLGA nanoparticles when compared with IRN solution.88
The Particle Containing Lipid Material Group
The majority of orally administered drugs are absorbed into the portal blood to reach the systemic circulation. However, the lipophilic substances (log P>5) usually reach the systemic circulation through the lymphatic pathway.60 In addition, some papers have mentioned that lipophilic substances and macromolecular substances with large molecular weights can increase the lymphatic absorption of drugs.89 Moreover, the permeability of lymphatic vessels to nanoparticles is significantly higher than that of capillaries.90 In addition, absorption of drugs through lymphatic vessels, due to its unique physiological and anatomical structure, can prevent the first pass metabolizing of liver.60,91 Therefore, through lymphatic absorption, the bioavailability of oral drugs can be significantly increased.92 However, different lengths of fatty acid chains have different absorption effects. The fatty acid chains from c-14 to c-18 promote greater lymphatic absorption.93 Khoo et al94 also found that long-chain triglycerides promoted absorption more effectively than medium-chain triglycerides. Moreover, certain classes of phospholipids and surfactants used in the formulations of liposomes can suppress the P-gp efflux system, thereby increased absorption of drugs.4
The Particle Containing Specific Ligands
Nanoparticle coated specific ligand has better intracellular uptake trend. For instance, in order to study the effect of folate functionalization on the extracellular transport of PTX, a chemotherapy drug with poor oral bioavailability, Roger et al95 modified PTX-PLGA nanoparticles with folate. Using the Caco-2 monolayer as an in vitro model, confocal microscopy showed that PTX loaded in PLGA nanoparticles increased the apparent permeability in Caco-2 cells by 5-times compared with free PTX. Functionalization of nanoparticles with folic acid further increased the transport capacity (8-fold compared with free PTX).
As we all know, the pharmacokinetic process of cargos in vivo is very complicated, and it is difficult to achieve effective drug delivery via modifying nanoparticles with only one kind of material. Therefore, nowadays, many studies modify or prepare nanoparticles with different or even opposite materials. Netsomboon et al36 mentioned that negatively charged particles being capable of changing zeta-potential to a positive value once having permeated the mucus and having reached the epithelium might be a promising strategy. Negatively charged and uncharged particles can easily move through the mucus,32,96 whereas positively charged particles show a comparatively much higher cell uptake via endocytosis than negatively charged particles.97 A typical example was that, after incubation with intestinal alkaline phosphatase, zeta potential of nanoparticles exhibiting phosphotyrosine substructures on their surface changed from negative to positive due to cleavage of negative charges in the form of phosphate residues.98 In addition, Shan et al66 reported a simple zwitterions-based nanoparticle delivery platform, the dense and hydrophilic coating of zwitterions endowed the nanoparticles with excellent mucus penetrating ability and affinity with epithelial cells, which significantly improved (4.5-fold) the cellular uptake of nanoparticles, compared with PEGlated nanoparticles. Wang et al11 developed a bilayer modification on the surface of mesoporous silica nanoparticles (MSNs) consisting of polyethylenimine-coated carbon dots (PCD) for effective transepithelial absorption and PEG polymers for improved mucus permeability, which enhanced the stability in the physiological environment, altered cell uptake mechanisms, and increased distribution in various intestinal sections.
The Effect of Shape for Oral Bioavailability
In addition to surface properties and size of nanoparticles, the shape of nanoparticles has a great influence on their interaction with biological systems, including cellular uptake, plasma circulation and organ distribution and so on. However, understanding of how shape of nanoparticles affects biological systems is far from complete. Nature abounds in viruses and bacteria of various forms.99 It is well known that bacterial forms have an evolutionary advantage due to their interactions with surface mechanisms such as passive diffusion and active transport.100 In Darwinian evolution, changes in bacterial or viral form precede in function due to random mutations. There is a strong case for natural selection of bacteria and viruses with specific forms. In the future of medicine, nanoparticles could also be shaped to suit their function.101
Most current NDDS used in laboratory-scale or clinical trials are spherical due to ease of synthesis. However, nowadays, compounds in large numbers are being presented in different forms of nanoparticles. For instance, Wang et al102 synthesized gold nanoparticles through one pot synthesis with inorganic metal material Au. Different morphologies could be obtained by changing the gold to calcium ion concentration ratios during the synthesis. As the calcium concentration decreased, the morphology changed from hexagons to pentagons and to triangles. Similarly, Huang et al19 fabricated different shaped MSNs (spheres, short rods and long rods), via inorganic nonmetallic material, to study the effect of MSNs on cellular behavior. In addition to inorganic material, Yoo et al103 synthesized PLGA particles with spherical and elliptical disk geometries and investigated the effect of particle shape on rate of particle endocytosis and their intracellular distribution in endothelial cells. This section briefly summarizes and discusses the relationship between different shapes of nanoparticles and common biological endpoints (eg, cell uptake, biological distribution).
Effects of Nanoparticle Morphology on Cellular Uptake in vivo and in vitro
Cell membranes mainly permeate small and non-polar molecules. Most nanoparticles are polar molecules that enter cells via endocytosis rather than cell diffusion.104 Recently, particle shape, an important physicochemical property of nanoparticles, has gained great attention and been demonstrated experimentally and theoretically to exert a great effect on cellular uptake behaviors.105 Different morphological features of nanoparticles affect their endocytosis process. Though some studies have investigated the relationship between shape of nanoparticles and uptake pathways, the revealed results have always been inconsistent (Table 3). For instance, it is well documented that rod-shaped nanoparticles have a lower capacity of entering the cells in comparison with their spherical counterparts.106,107 The data of Wang et al107 also showed the same results that sphere-shaped nanoparticles could enter mesenchymal stem cells at a faster rate in the initial 4 hours than rod-shaped nanoparticles. A possible explanation for their results is that nanorods would seriously destroy the cytoskeleton to cause the reorganization of the cytoskeleton, thereby prolonging the rate of cell internalization and resulting in slow absorption. In contrast, nanospheres have less impact on the cytoskeleton and eventually get into cells faster.107 However, Gratton et al108 found that rod-shaped nanoparticles had the highest internalization rates compared with spheres, cylinders, and cubes. The results of Banerjee et al6 demonstrated that the rod and disc-shaped nanoparticles were internalized 2-fold higher than spherical nanoparticles. The possible reasons for the uptake behaviors are governed by cell types, size, surface chemistry, and particle composition, which play a dominant role.
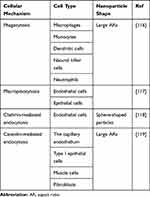 |
Table 3 The Different Internalization Pathways Corresponding to Different Cells and the Main Shapes Associated with Them |
Endocytosis has two stages: adhesion and internalization.109 The shape of nanoparticles has a significant impact on the cell adhesion of a wide variety of nanoparticles. Salatin et al110 mentioned that the elongated nanoparticles showed higher efficiency in adhering to the cells rather than the spherical nanoparticles. They explained that the curve shape of spherical particles allowed a limited number of their binding sites to interact with target cell receptors while the elongated nanoparticles had higher surface area, which facilitated the multivalent interaction of these particles with cell surface. Similarly, Zhang et al109 mentioned that the prickly nanoparticles had a higher anchoring amount compared with the round nanoparticles. The results could be understood as originating from the contact surface area difference of the two types of nanoparticles with the plasma membrane. The paper of He and Park111 suggested that a smaller diameter allowed the microsphere to adhere to cells faster and stronger. Therefore, we can see that the larger surface area is likely to leadd to a stronger adhesion to the cell. Since strong adhesion between the nanoparticles and the cells are required for cells to internalize the nanoparticles, a larger the surface area implies a higher chance internalized by cells.112
NDDS can be capable to enter live cells, often through several endocytic pathways, namely phagocytosis, macropinocytosis, clathrin-mediated endocytosis, and caveolin-mediated endocytosis.113 Phagocytosis only occurs in specialized cells such as macrophages, monocytes, dendritic cells, natural killer cells, and neutrophils, which in turn form intracellular phagosomes.114 Macropinocytosis is a process by which extracellular fluid and its debris are internalized into a nonspecific manner within large, heterogeneous vesicles, named macropinosomes.115 Clathrin-mediated endocytosis is initiated by the interaction between ligands and specific receptors. Following entry into the cell, a fraction of internalized guest matters would be recycled back to the cell exterior, while the remaining fractions are transferred into lysosomes, resulting in the degradation of the sequestered cargo material by the lysosomal enzymes.116 The caveolae-mediated endocytosis pathway is characteristic by the evolution of caveolae-derivatives of the subdomains of sphingolipid and cholesterol-rich cell membrane fractions, which mediates the translocation to the Golgi apparatus, to endoplasmic reticulum or entered into the endosomal pathway.117
It has been revealed that the pathway of nanoparticles into cells can be regulated by shape. For instance, Hao et al105 used sphere-nanoparticles (NS), short-rod nanoparticles (NSR), and long-rod nanoparticles (NLR) to incubate with Hela cells. Their results preliminarily demonstrated that NS particles preferred to be internalized into cells via the clathrin-mediated pathway, whereas the uptake is shifted to the caveolae-mediated pathway for the particles with large aspect ratios (ARs). In addition, Agarwal et al118 mentioned that, although macropinocytosis was used by both epithelial and endothelial cells, epithelial cells uniquely internalized these nanoparticles using the caveolae-mediated pathway. Human umbilical vein endothelial cells, on the other hand, used clathrin-mediated uptake for all shapes and show a significantly higher uptake efficiency compared with epithelial cells. Although less significantly, it further confirmed that the effect of particle geometry is cell type-specific. This shall be one of the reasons for the inconsistency between different cells and dependence on shape. Agarwal et al118 also proposed that when nanoparticle surface properties and composition were kept constant, each cell type could “sense” the nanoscale geometry (both shape and size) and trigger unique uptake pathways and thus have different shape-dependent internalization efficiencies. Therefore, it is paramount to consider the main intercellular pathways of different cells in exploring the effects of nanoparticle morphology on cellular uptake.
Sharma et al119 stretched three different volumes of spherical PS particle, ranging from 0.5–3.6 µm in diameter to form prolate or oblate ellipsoids which utilized the film-stretching method and studied their attachment and internalization in RAW 264.7 murine macrophages. Regardless of the volume, the order of particle adhesion to the membrane surface was the oblong>oblate>sphere. However, the order of internalization was the oblate>sphere>oblong. Prior to this, Champion and Mitragotri120 also explored the effect of nanoparticle morphology on macrophage uptake in a similar approach. They found that regions of higher curvature were associated with a larger degree of wrapping or phagocytosis. As we all know, nanoparticles enter in the system circulation after oral administration where nanoparticles could be absorbed by immune cells in the blood and various phagocytes in tissues. To avoid being devoured by various immune cells or macrophages before reaching the target tissue, this effect could be minimized by controlling the local shape of the nanoparticles, thereby increasing the bioavailability.103 In addition to macrophages, particle geometry has also been shown to have a significant impact on the context of epithelial drug delivery. Banerjee et al6 investigated the uptake and transport of smaller stretch nanoparticles in a triple-cell co-culture model of the intestine. They found that rod-shaped and disc-shaped particles, stretched from 200 nm sphere-shaped nanoparticles, were more readily absorbed and transported by epithelial cells than spheres. Also, Huang et al19 fabricated and functionalized different shaped MSNs (spheres, short rods, and long rods) with fluorescein isothiocyanate (FITC) and rhodamine B isothiocyanate (RITC) for imaging and quantification of MSNs uptake. Then the ability of different shaped MSNs to be transported into model A375 cells was compared. They found that the number of intracellular particles varied with particle shape and exhibited a strong dependence. Compared with spherical and short rod-shaped nanoparticles, long rod-shaped nanoparticles were easier to internalize. The authors suggested that one possible explanation was the curvature of different shapes. The area of contact between rod nanoparticles and the cell membrane was larger than that of spherical nanoparticles because the longitudinal axis of rod nanoparticles interacted with the cell membrane.
Although the main internalization pathways of nanoparticles change with different cells, it could be speculated from the above studies that the curvature of nanoparticles and the area of contact with cells play important roles.
Another study said that the morphology dependence of cell uptake varied with the size of nanoparticles.101 They believed that larger particles had a general pattern of morphological dependence, while smaller nanoparticles did not have morphological dependence in cellular uptake because small particles tended to change the surface properties of nanoparticles. In addition to nanoparticle size, the nanoparticle materials mentioned above also had a significant impact on the shape dependence of cell uptake. Using Jet and Flash Imprint Lithography (J-FIL) for top down fabrication of monodisperse, Agarwal et al118 fabricated PEG-based discoidal and cuboidal rod-shaped nanoparticles. Particles were administered to cells in culture at equal total fluorescence intensity. They found that between discoidal and rod-shaped nanoparticles of similar volume, nanodiscs were more efficiently internalized at any time points. Nevertheless, Barua et al121 reported that for nonspecific hydrophobic PS particles, nanorods and nanodiscs possessed similar uptake in epithelial breast cancer cells. These differences could highlight the effect of nanoparticle size and modification as well as cell types evaluated and emphasize the importance of material and size composition in understanding nanoparticle-mediated intracellular delivery.
Effects of Nanoparticle Morphology on GI Transit
Geometry not only affects cellular uptake, but also influences transport across the physiological barriers. Nanorods and nanospheres were labeled using fluorescence resonance energy transfer (FRET) molecules to track the in vivo fate of intact nanoparticles accurately.122 Li et al122 found that nanorods possessed significantly longer retention time in the GI tract compared with nanospheres. Furthermore, nanorods exhibited a stronger ability of penetration into space of villi than nanospheres, which was the main reason of longer retention time. In addition, mesenteric lymph transported 1.75% nanorods within 10 hours, which was more than nanospheres (0.98%). It indicated the nanoparticles shape could affect the movement route or rate. Yu et al123 found that mesoporous silica rods (80×240 nm) as well as calcium phosphate rods have also been shown to penetrate deeper into the mucosal tissue of the GI tract ex vivo compared with spheres (80 and 140 nm). They guessed that this effect originated from their rotational diffusion combined with shear flows and the mesh-like network of mucus enabling deep penetration of the viscoelastic layer. Combined, these two studies suggested anisotropic and rod-like particles were far superior to targeting the epithelium of the GI tract than their spherical equivalents.
Conclusion and Prospective
The physicochemical properties of nanoparticles can significantly affect oral bioavailability of drugs. However, oral delivery of drugs is a complex process. And variation in one area alone of nanoparticles cannot fully improve the oral bioavailability of drugs. Therefore, it is important to understand the role of nanoparticles in the oral delivery. Finally, efforts should focus not only on the laboratory synthesis/functionalization, but also on the selection of appropriate materials for the preparation of nanoparticles according to clinical needs, so as to increase the oral absorption and improve the oral bioavailability of drugs. Here, we have preliminarily covered the different roles played by different properties of nanoparticles in oral drug delivery to provide corresponding assistance to researchers in this field.
Acknowledgments
This study was supported by the basic research fund of the Science and Technology Department of Sichuan Province (No. 2020YJ0336, 2020YJ0373), the Joint Fund of Luzhou City and Southwest Medical University [No.2017LZXNYD-T02, 2019LZXNYDZ07], the Science and Technology Fund of Luzhou City (No. 2019-SYF-35), and Science and Technology Innovation Team from Jiucheng Science and Technology Talent Cultivation Plan in Luzhou City (2019-1).
Disclosure
Yuanyuan Wang reports grants from the Science and Technology Fund for Distinguished Young Scholars of Sichuan Province and Basic Research, the Joint Fund of Luzhou City and Southwest Medical University, the Science and Technology Fund of Luzhou City, Science and Technology Innovation Team from Jiucheng Science and Technology Talent Cultivation Plan in Luzhou City, the Scientific Research Foundation of the Education Department of Sichuan Province, and a research grant from Traditional Chinese Medicine Administration in Sichuan Province, during the conduct of the study. The authors report no other potential conflicts of interest in this work.
References
1. Munzone E, Colleoni M. Clinical overview of metronomic chemotherapy in breast cancer. Nat Rev Clin Oncol. 2015;12(11):631–644. doi:10.1038/nrclinonc.2015.131
2. Le Z, Chen Y, Han H, et al. Hydrogen-bonded tannic acid-based anticancer nanoparticle for enhancement of oral chemotherapy. ACS Appl Mater Interfaces. 2018;10(49):42186–42197. doi:10.1021/acsami.8b18979
3. Sarparanta MP, Bimbo LM, Makila EM, et al. The mucoadhesive and gastroretentive properties of hydrophobin-coated porous silicon nanoparticle oral drug delivery systems. Biomaterials. 2012;33(11):3353–3362. doi:10.1016/j.biomaterials.2012.01.029
4. Ahmad J, Amin S, Rahman M, et al. Solid matrix based lipidic nanoparticles in oral cancer chemotherapy: applications and pharmacokinetics. Curr Drug Metab. 2015;16(8):633–644. doi:10.2174/1389200216666150812122128
5. van Herwaarden AE, van Waterschoot RA, Schinkel AH. How important is intestinal cytochrome P450 3A metabolism? Trends Pharmacol Sci. 2009;30(5):223–227. doi:10.1016/j.tips.2009.02.003
6. Banerjee A, Qi J, Gogoi R, Wong J, Mitragotri S. Role of nanoparticle size, shape and surface chemistry in oral drug delivery. J Control Release. 2016;238:176–185. doi:10.1016/j.jconrel.2016.07.051
7. Etrych T, Strohalm J, Chytil P, et al. Biodegradable star HPMA polymer conjugates of doxorubicin for passive tumor targeting. Eur J Pharm Sci. 2011;42(5):527–539. doi:10.1016/j.ejps.2011.03.001
8. Chen B, Wang X, Zhang Y, et al. Improved solubility, dissolution rate, and oral bioavailability of main biflavonoids from Selaginella doederleinii extract by amorphous solid dispersion. Drug Deliv. 2020;27(1):309–322. doi:10.1080/10717544.2020.1716876
9. Gong X, Zheng Y, He G, Chen K, Zeng X, Chen Z. Multifunctional nanoplatform based on star-shaped copolymer for liver cancer targeting therapy. Drug Deliv. 2019;26(1):595–603. doi:10.1080/10717544.2019.1625467
10. MacEwan SR, Chilkoti A. From composition to cure: a systems engineering approach to anticancer drug carriers. Angew Chem Int Ed Engl. 2017;56(24):6712–6733. doi:10.1002/anie.201610819
11. Wang Y, Cui Y, Zhao Y, et al. Effects of surface modification and size on oral drug delivery of mesoporous silica formulation. J Colloid Interface Sci. 2018;513:736–747. doi:10.1016/j.jcis.2017.11.065
12. Mazzaferro S, Bouchemal K, Ponchel G. Oral delivery of anticancer drugs III: formulation using drug delivery systems. Drug Discov Today. 2013;18(1–2):99–104. doi:10.1016/j.drudis.2012.08.007
13. Shaikh J, Ankola DD, Beniwal V, Singh D, Kumar MNVR. Nanoparticle encapsulation improves oral bioavailability of curcumin by at least 9-fold when compared to curcumin administered with piperine as absorption enhancer. Eur J Pharm Sci. 2009;37(3–4):223–230. doi:10.1016/j.ejps.2009.02.019
14. Swarnakar NK, Jain AK, Singh RP, Godugu C, Das M, Jain S. Oral bioavailability, therapeutic efficacy and reactive oxygen species scavenging properties of coenzyme Q10-loaded polymeric nanoparticles. Biomaterials. 2011;32(28):6860–6874. doi:10.1016/j.biomaterials.2011.05.079
15. Zhao Y, Chen G, Meng Z, et al. A novel nanoparticle drug delivery system based on PEGylated hemoglobin for cancer therapy. Drug Deliv. 2019;26(1):717–723. doi:10.1080/10717544.2019.1639846
16. Alam S, Khan ZI, Mustafa G, et al. Development and evaluation of thymoquinone-encapsulated chitosan nanoparticles for nose-to-brain targeting: a pharmacoscintigraphic study. Int J Nanomedicine. 2012;7:5705–5718. doi:10.2147/IJN.S35329
17. Ansari MJ, Anwer MK, Jamil S, et al. Enhanced oral bioavailability of insulin-loaded solid lipid nanoparticles: pharmacokinetic bioavailability of insulin-loaded solid lipid nanoparticles in diabetic rats. Drug Deliv. 2016;23(6):1972–1979. doi:10.3109/10717544.2015.1039666
18. Ganea GM, Fakayode SO, Losso JN, van Nostrum CF, Sabliov CM, Warner IM. Delivery of phytochemical thymoquinone using molecular micelle modified poly(D, L lactide-co-glycolide) (PLGA) nanoparticles. Nanotechnology. 2010;21(28):285104. doi:10.1088/0957-4484/21/28/285104
19. Huang X, Teng X, Chen D, Tang F, He J. The effect of the shape of mesoporous silica nanoparticles on cellular uptake and cell function. Biomaterials. 2010;31(3):438–448. doi:10.1016/j.biomaterials.2009.09.060
20. Torchilin V. Tumor delivery of macromolecular drugs based on the EPR effect. Adv Drug Deliv Rev. 2011;63(3):131–135. doi:10.1016/j.addr.2010.03.011
21. Nakamura H, Fang J, Maeda H. Development of next-generation macromolecular drugs based on the EPR effect: challenges and pitfalls. Expert Opin Drug Deliv. 2015;12(1):53–64. doi:10.1517/17425247.2014.955011
22. Fowler R, Vllasaliu D, Trillo FF, et al. Nanoparticle transport in epithelial cells: pathway switching through bioconjugation. Small. 2013;9(19):3282–3294. doi:10.1002/smll.201202623
23. Champion JA, Mitragotri S. Shape induced inhibition of phagocytosis of polymer particles. Pharm Res. 2009;26(1):244–249. doi:10.1007/s11095-008-9626-z
24. Caldorera-Moore M, Guimard N, Shi L, Roy K. Designer nanoparticles: incorporating size, shape and triggered release into nanoscale drug carriers. Expert Opin Drug Deliv. 2010;7(4):479–495. doi:10.1517/17425240903579971
25. Li Y, Yue T, Yang K, Zhang X. Molecular modeling of the relationship between nanoparticle shape anisotropy and endocytosis kinetics. Biomaterials. 2012;33(19):4965–4973. doi:10.1016/j.biomaterials.2012.03.044
26. Cong VT, Gaus K, Tilley RD, Gooding JJ. Rod-shaped mesoporous silica nanoparticles for nanomedicine: recent progress and perspectives. Expert Opin Drug Deliv. 2018;15(9):881–892. doi:10.1080/17425247.2018.1517748
27. He B, Lin P, Jia Z, et al. The transport mechanisms of polymer nanoparticles in Caco-2 epithelial cells. Biomaterials. 2013;34(25):6082–6098. doi:10.1016/j.biomaterials.2013.04.053
28. Florek J, Caillard R, Kleitz F. Evaluation of mesoporous silica nanoparticles for oral drug delivery - current status and perspective of MSNs drug carriers. Nanoscale. 2017;9(40):15252–15277. doi:10.1039/C7NR05762H
29. Agoram B, Woltosz WS, Bolger MB. Predicting the impact of physiological and biochemical processes on oral drug bioavailability. Adv Drug Deliv Rev. 2001;50(Suppl 1):S41–S67. doi:10.1016/S0169-409X(01)00179-X
30. Yun Y, Cho YW, Park K. Nanoparticles for oral delivery: targeted nanoparticles with peptidic ligands for oral protein delivery. Adv Drug Deliv Rev. 2013;65(6):822–832. doi:10.1016/j.addr.2012.10.007
31. Ye W, Chen R, Chen X, et al. AhR regulates the expression of human cytochrome P450 1A1 (CYP1A1) by recruiting Sp1. FEBS J. 2019;286(21):4215–4231. doi:10.1111/febs.14956
32. Crater JS, Carrier RL. Barrier properties of gastrointestinal mucus to nanoparticle transport. Macromol Biosci. 2010;10(12):1473–1483. doi:10.1002/mabi.201000137
33. Sigurdsson HH, Kirch J, Lehr CM. Mucus as a barrier to lipophilic drugs. Int J Pharm. 2013;453(1):56–64. doi:10.1016/j.ijpharm.2013.05.040
34. Varum FJ, Veiga F, Sousa JS, Basit AW. Mucus thickness in the gastrointestinal tract of laboratory animals. J Pharm Pharmacol. 2012;64(2):218–227. doi:10.1111/j.2042-7158.2011.01399.x
35. Juge N. Microbial adhesins to gastrointestinal mucus. Trends Microbiol. 2012;20(1):30–39. doi:10.1016/j.tim.2011.10.001
36. Netsomboon K, Bernkop-Schnurch A. Mucoadhesive vs. mucopenetrating particulate drug delivery. Eur J Pharm Biopharm. 2016;98:76–89. doi:10.1016/j.ejpb.2015.11.003
37. Strugala V, Allen A, Dettmar PW, Pearson JP. Colonic mucin: methods of measuring mucus thickness. Proc Nutr Soc. 2003;62(1):237–243. doi:10.1079/PNS2002205
38. Smith DJ, Gaffney EA, Blake JR. Modelling mucociliary clearance. Respir Physiol Neurobiol. 2008;163(1–3):178–188. doi:10.1016/j.resp.2008.03.006
39. Evans CM, Koo JS. Airway mucus: the good, the bad, the sticky. Pharmacol Ther. 2009;121(3):332–348. doi:10.1016/j.pharmthera.2008.11.001
40. Boegh M, García-Díaz M, Müllertz A, Nielsen HM. Steric and interactive barrier properties of intestinal mucus elucidated by particle diffusion and peptide permeation. Eur J Pharm Biopharm. 2015;95:136–143. doi:10.1016/j.ejpb.2015.01.014
41. Lock JY, Carlson TL, Carrier RL. Mucus models to evaluate the diffusion of drugs and particles. Adv Drug Deliv Rev. 2018;124:34–49. doi:10.1016/j.addr.2017.11.001
42. Lai SK, Wang YY, Hanes J. Mucus-penetrating nanoparticles for drug and gene delivery to mucosal tissues. Adv Drug Deliv Rev. 2009;61(2):158–171. doi:10.1016/j.addr.2008.11.002
43. Svensson O, Arnebrant T. Mucin layers and multilayers-physicochemical properties and applications. Curr Opin Colloid Interface Sci. 2010;15(6):395–405. doi:10.1016/j.cocis.2010.05.015
44. Ensign LM, Cone R, Hanes J. Oral drug delivery with polymeric nanoparticles: the gastrointestinal mucus barriers. Adv Drug Deliv Rev. 2012;64(6):557–570. doi:10.1016/j.addr.2011.12.009
45. Nordgard CT, Draget KI. Oligosaccharides as modulators of rheology in complex mucous systems. Biomacromolecules. 2011;12(8):3084–3090. doi:10.1021/bm200727c
46. Lieleg O, Ribbeck K. Biological hydrogels as selective diffusion barriers. Trends Cell Biol. 2011;21(9):543–551. doi:10.1016/j.tcb.2011.06.002
47. Chen MC, Sonaje K, Chen KJ, Sung HW. A review of the prospects for polymeric nanoparticle platforms in oral insulin delivery. Biomaterials. 2011;32(36):9826–9838. doi:10.1016/j.biomaterials.2011.08.087
48. Renukuntla J, Vadlapudi AD, Patel A, Boddu SH, Mitra AK. Approaches for enhancing oral bioavailability of peptides and proteins. Int J Pharm. 2013;447(1–2):75–93. doi:10.1016/j.ijpharm.2013.02.030
49. Bruno BJ, Miller GD, Lim CS. Basics and recent advances in peptide and protein drug delivery. Ther Deliv. 2013;4(11):1443–1467. doi:10.4155/tde.13.104
50. Luo YY, Xiong XY, Tian Y, Li ZL, Gong YC, Li YP. A review of biodegradable polymeric systems for oral insulin delivery. Drug Deliv. 2016;23(6):1882–1891. doi:10.3109/10717544.2015.1052863
51. Bakhru SH, Furtado S, Morello AP, Mathiowitz E. Oral delivery of proteins by biodegradable nanoparticles. Adv Drug Deliv Rev. 2013;65(6):811–821. doi:10.1016/j.addr.2013.04.006
52. He C, Yin L, Tang C, Yin C. Size-dependent absorption mechanism of polymeric nanoparticles for oral delivery of protein drugs. Biomaterials. 2012;33(33):8569–8578. doi:10.1016/j.biomaterials.2012.07.063
53. Turner JR. Intestinal mucosal barrier function in health and disease. Nat Rev Immunol. 2009;9(11):799–809. doi:10.1038/nri2653
54. Su FY, Lin KJ, Sonaje K, et al. Protease inhibition and absorption enhancement by functional nanoparticles for effective oral insulin delivery. Biomaterials. 2012;33(9):2801–2811. doi:10.1016/j.biomaterials.2011.12.038
55. Chuang EY, Lin KJ, Su FY, et al. Calcium depletion-mediated protease inhibition and apical-junctional-complex disassembly via an EGTA-conjugated carrier for oral insulin delivery. J Control Release. 2013;169(3):296–305. doi:10.1016/j.jconrel.2012.11.011
56. Ling SS, Magosso E, Khan NA, Yuen KH, Barker SA. Enhanced oral bioavailability and intestinal lymphatic transport of a hydrophilic drug using liposomes. Drug Dev Ind Pharm. 2006;32(3):335–345. doi:10.1080/03639040500519102
57. Hanafy A, Spahn-Langguth H, Vergnault G, et al. Pharmacokinetic evaluation of oral fenofibrate nanosuspensions and SLN in comparison to conventional suspensions of micronized drug. Adv Drug Deliv Rev. 2007;59(6):419–426. doi:10.1016/j.addr.2007.04.005
58. Zhuang CY, Li N, Wang M, et al. Preparation and characterization of vinpocetine loaded nanostructured lipid carriers (NLC) for improved oral bioavailability. Int J Pharm. 2010;394(1–2):179–185. doi:10.1016/j.ijpharm.2010.05.005
59. Hu L, Tang X, Cui F. Solid lipid nanoparticles (SLNs) to improve oral bioavailability of poorly soluble drugs. J Pharm Pharmacol. 2004;56(12):1527–1535. doi:10.1211/0022357044959
60. Das S, Chaudhury A. Recent advances in lipid nanoparticle formulations with solid matrix for oral drug delivery. AAPS PharmSciTech. 2011;12(1):62–76. doi:10.1208/s12249-010-9563-0
61. Maisel K, Ensign L, Reddy M, Cone R, Hanes J. Effect of surface chemistry on nanoparticle interaction with gastrointestinal mucus and distribution in the gastrointestinal tract following oral and rectal administration in the mouse. J Control Release. 2015;197:48–57. doi:10.1016/j.jconrel.2014.10.026
62. Li H, Chen M, Su Z, Sun M, Ping Q. Size-exclusive effect of nanostructured lipid carriers on oral drug delivery. Int J Pharm. 2016;511(1):524–537. doi:10.1016/j.ijpharm.2016.07.049
63. Zhao Z, Hu Y, Hoerle R, et al. A nanoparticle-based nicotine vaccine and the influence of particle size on its immunogenicity and efficacy. Nanomedicine. 2017;13(2):443–454. doi:10.1016/j.nano.2016.07.015
64. de la Fuente M, Csaba N, Garcia-Fuentes M, Alonso MJ. Nanoparticles as protein and gene carriers to mucosal surfaces. Nanomedicine (Lond). 2008;3(6):845–857. doi:10.2217/17435889.3.6.845
65. Ensign LM, Tang BC, Wang YY, et al. Mucus-penetrating nanoparticles for vaginal drug delivery protect against herpes simplex virus. Sci Transl Med. 2012;4(138):138ra179. doi:10.1126/scitranslmed.3003453
66. Shan W, Zhu X, Tao W, et al. Enhanced oral delivery of protein drugs using zwitterion-functionalized nanoparticles to overcome both the diffusion and absorption barriers. ACS Appl Mater Interfaces. 2016;8(38):25444–25453. doi:10.1021/acsami.6b08183
67. Ensign LM, Schneider C, Suk JS, Cone R, Hanes J. Mucus penetrating nanoparticles: biophysical tool and method of drug and gene delivery. Adv Mater. 2012;24(28):3887–3894. doi:10.1002/adma.201201800
68. Ahmad N, Ahmad R, Alam MA, Ahmad FJ. Enhancement of oral bioavailability of doxorubicin through surface modified biodegradable polymeric nanoparticles. Chem Cent J. 2018;12(1):65. doi:10.1186/s13065-018-0434-1
69. Feeney OM, Williams HD, Pouton CW, Porter CJ. ‘Stealth’ lipid-based formulations: poly(ethylene glycol)-mediated digestion inhibition improves oral bioavailability of a model poorly water soluble drug. J Control Release. 2014;192:219–227. doi:10.1016/j.jconrel.2014.07.037
70. Wang YY, Lai SK, Suk JS, Pace A, Cone R, Hanes J. Addressing the PEG mucoadhesivity paradox to engineer nanoparticles that “slip” through the human mucus barrier. Angew Chem Int Ed Engl. 2008;47(50):9726–9729. doi:10.1002/anie.200803526
71. Ejima H, Richardson JJ, Liang K, et al. One-step assembly of coordination complexes for versatile film and particle engineering. Science. 2013;341(6142):154–157. doi:10.1126/science.1237265
72. Shen G, Xing R, Zhang N, Chen C, Ma G, Yan X. Interfacial cohesion and assembly of bioadhesive molecules for design of long-term stable hydrophobic nanodrugs toward effective anticancer therapy. ACS Nano. 2016;10(6):5720–5729. doi:10.1021/acsnano.5b07276
73. Jackson JK, Letchford K. The effective solubilization of hydrophobic drugs using epigallocatechin gallate or tannic acid-based formulations. J Pharm Sci. 2016;105(10):3143–3152. doi:10.1016/j.xphs.2016.06.027
74. Bentley WE, Payne GF. Nature’s other self-assemblers. Science. 2013;341(6142):136–137. doi:10.1126/science.1241562
75. Hancock BC, Parks M. What is the true solubility advantage for amorphous pharmaceuticals? Pharm Res. 2000;17(4):397–404. doi:10.1023/A:1007516718048
76. Shutava TG, Balkundi SS, Vangala P, et al. Layer-by-layer-coated gelatin nanoparticles as a vehicle for delivery of natural polyphenols. ACS Nano. 2009;3(7):1877–1885. doi:10.1021/nn900451a
77. Xiang S, Yang P, Guo H, et al. Green tea makes polyphenol nanoparticles with radical-scavenging activities. Macromol Rapid Commun. 2017;38(23):1700446. doi:10.1002/marc.201700446
78. Wang X, Chen Y, Dahmani FZ, Yin L, Zhou J, Yao J. Amphiphilic carboxymethyl chitosan-quercetin conjugate with P-gp inhibitory properties for oral delivery of paclitaxel. Biomaterials. 2014;35(26):7654–7665. doi:10.1016/j.biomaterials.2014.05.053
79. Kitagawa S, Nabekura T, Nakamura Y, Takahashi T, Kashiwada Y. Inhibition of P-glycoprotein function by tannic acid and pentagalloylglucose. J Pharm Pharmacol. 2007;59(7):965–969. doi:10.1211/jpp.59.7.0008
80. Ren T, Wang Q, Xu Y, et al. Enhanced oral absorption and anticancer efficacy of cabazitaxel by overcoming intestinal mucus and epithelium barriers using surface polyethylene oxide (PEO) decorated positively charged polymer-lipid hybrid nanoparticles. J Control Release. 2018;269:423–438. doi:10.1016/j.jconrel.2017.11.015
81. Li X, Chen D, Le C, et al. Novel mucus-penetrating liposomes as a potential oral drug delivery system: preparation, in vitro characterization, and enhanced cellular uptake. Int J Nanomedicine. 2011;6:3151–3162. doi:10.2147/IJN.S25741
82. Schulz JD, Gauthier MA, Leroux JC. Improving oral drug bioavailability with polycations? Eur J Pharm Biopharm. 2015;97(Pt B):427–437. doi:10.1016/j.ejpb.2015.04.025
83. Bourganis V, Karamanidou T, Samaridou E, Karidi K, Kammona O, Kiparissides C. On the synthesis of mucus permeating nanocarriers. Eur J Pharm Biopharm. 2015;97(Pt A):239–249. doi:10.1016/j.ejpb.2015.01.021
84. Pereira de Sousa I, Steiner C, Schmutzler M, et al. Mucus permeating carriers: formulation and characterization of highly densely charged nanoparticles. Eur J Pharm Biopharm. 2015;97(Pt A):273–279. doi:10.1016/j.ejpb.2014.12.024
85. Cánepa C, Imperiale JC, Berini CA, Lewicki M, Sosnik A, Biglione MM. Development of a drug delivery system based on chitosan nanoparticles for oral administration of interferon-α. Biomacromolecules. 2017;18(10):3302–3309. doi:10.1021/acs.biomac.7b00959
86. Dyawanapelly S, Koli U, Dharamdasani V, Jain R, Dandekar P. Improved mucoadhesion and cell uptake of chitosan and chitosan oligosaccharide surface-modified polymer nanoparticles for mucosal delivery of proteins. Drug Deliv Transl Res. 2016;6(4):365–379. doi:10.1007/s13346-016-0295-x
87. Wang M, Zhang Y, Feng J, et al. Preparation, characterization, and in vitro and in vivo investigation of chitosan-coated poly (d,l-lactide-co-glycolide) nanoparticles for intestinal delivery of exendin-4. Int J Nanomedicine. 2013;8:1141–1154. doi:10.2147/IJN.S41457
88. Ahmad N, Alam MA, Ahmad R, Umar S, Jalees Ahmad F. Improvement of oral efficacy of Irinotecan through biodegradable polymeric nanoparticles through in vitro and in vivo investigations. J Microencapsul. 2018;35(4):327–343. doi:10.1080/02652048.2018.1485755
89. Li H, Lu S, Luo M, Li X, Liu S, Zhang T. A matrix dispersion based on phospholipid complex system: preparation, lymphatic transport, and pharmacokinetics. Drug Dev Ind Pharm. 2020;46(4):557–565. doi:10.1080/03639045.2020.1735408
90. Sanjula B, Shah FM, Javed A, Alka A. Effect of poloxamer 188 on lymphatic uptake of carvedilol-loaded solid lipid nanoparticles for bioavailability enhancement. J Drug Target. 2009;17(3):249–256. doi:10.1080/10611860902718672
91. Chaudhary S, Garg T, Murthy RS, Rath G, Goyal AK. Recent approaches of lipid-based delivery system for lymphatic targeting via oral route. J Drug Target. 2014;22(10):871–882. doi:10.3109/1061186X.2014.950664
92. Trevaskis NL, Charman WN, Porter CJ. Lipid-based delivery systems and intestinal lymphatic drug transport: a mechanistic update. Adv Drug Deliv Rev. 2008;60(6):702–716. doi:10.1016/j.addr.2007.09.007
93. Porter CJ, Charman WN. Intestinal lymphatic drug transport: an update. Adv Drug Deliv Rev. 2001;50(1–2):61–80. doi:10.1016/S0169-409X(01)00151-X
94. Khoo SM, Shackleford DM, Porter CJH, Edwards GA, Charman WN. Intestinal lymphatic transport of halofantrine occurs after oral administration of a unit–dose lipid-based formulation to fasted dogs. Pharm Res. 2003;20(9):1460–1465. doi:10.1023/A:1025718513246
95. Roger E, Kalscheuer S, Kirtane A, et al. Folic acid functionalized nanoparticles for enhanced oral drug delivery. Mol Pharm. 2012;9(7):2103–2110. doi:10.1021/mp2005388
96. Bansil R, Turner BS. Mucin structure, aggregation, physiological functions and biomedical applications. Curr Opin Colloid Interface Sci. 2006;11(2–3):164–170. doi:10.1016/j.cocis.2005.11.001
97. Loretz B, Thaler M, Bernkop-Schnürch A. Role of sulfhydryl groups in transfection? A case study with chitosan-NAC nanoparticles. Bioconjug Chem. 2007;18(4):1028–1035. doi:10.1021/bc0603079
98. Perera G, Zipser M, Bonengel S, Salvenmoser W, Bernkop-Schnurch A. Development of phosphorylated nanoparticles as zeta potential inverting systems. Eur J Pharm Biopharm. 2015;97(Pt A):250–256. doi:10.1016/j.ejpb.2015.01.017
99. Wanger G, Onstott TC, Southam G. Stars of the terrestrial deep subsurface: a novel ‘star-shaped’ bacterial morphotype from a South African platinum mine. Geobiology. 2008;6(3):325–330. doi:10.1111/j.1472-4669.2008.00163.x
100. Young KD. Bacterial morphology: why have different shapes? Curr Opin Microbiol. 2007;10(6):596–600. doi:10.1016/j.mib.2007.09.009
101. Kinnear C, Moore TL, Rodriguez-Lorenzo L, Rothen-Rutishauser B, Petri-Fink A. Form follows function: nanoparticle shape and its implications for nanomedicine. Chem Rev. 2017;117(17):11476–11521. doi:10.1021/acs.chemrev.7b00194
102. Wang C, Ito Y, Pradeep B, Valiyaveettil S. Shape sensitivity on toxicity of gold nanoplates in breast cancer cells. J Nanosci Nanotechnol. 2015;15(12):9520–9530. doi:10.1166/jnn.2015.10780
103. Yoo JW, Doshi N, Mitragotri S. Endocytosis and intracellular distribution of PLGA particles in endothelial cells: effect of Particle Geometry. Macromol Rapid Commun. 2010;31(2):142–148. doi:10.1002/marc.200900592
104. Foroozandeh P, Aziz AA. Insight into cellular uptake and intracellular trafficking of nanoparticles. Nanoscale Res Lett. 2018;13(1):339. doi:10.1186/s11671-018-2728-6
105. Hao N, Li L, Zhang Q, et al. The shape effect of PEGylated mesoporous silica nanoparticles on cellular uptake pathway in Hela cells. Micropor Mesopor Mat. 2012;162:14–23. doi:10.1016/j.micromeso.2012.05.040
106. Chithrani BD, Ghazani AA, Chan WC. Determining the size and shape dependence of gold nanoparticle uptake into mammalian cells. Nano Lett. 2006;6(4):662–668. doi:10.1021/nl052396o
107. Wang J, Yang G, Wang Y, et al. Chimeric protein template-induced shape control of bone mineral nanoparticles and its impact on mesenchymal stem cell fate. Biomacromolecules. 2015;16(7):1987–1996. doi:10.1021/acs.biomac.5b00419
108. Gratton SE, Ropp PA, Pohlhaus PD, et al. The effect of particle design on cellular internalization pathways. Proc Natl Acad Sci U S A. 2008;105(33):11613–11618. doi:10.1073/pnas.0801763105
109. Zhang B, Feng X, Yin H, et al. Anchored but not internalized: shape dependent endocytosis of nanodiamond. Sci Rep. 2017;7:46462. doi:10.1038/srep46462
110. Salatin S, Maleki Dizaj S, Yari Khosroushahi A. Effect of the surface modification, size, and shape on cellular uptake of nanoparticles. Cell Biol Int. 2015;39(8):881–890. doi:10.1002/cbin.10459
111. He Y, Park K. Effects of the microparticle shape on cellular uptake. Mol Pharm. 2016;13(7):2164–2171. doi:10.1021/acs.molpharmaceut.5b00992
112. He Q, Zhang Z, Gao Y, Shi J, Li Y. Intracellular localization and cytotoxicity of spherical mesoporous silica nano- and microparticles. Small. 2009;5(23):2722–2729. doi:10.1002/smll.200900923
113. Salatin S, Yari Khosroushahi A. Overviews on the cellular uptake mechanism of polysaccharide colloidal nanoparticles. J Cell Mol Med. 2017;21(9):1668–1686. doi:10.1111/jcmm.13110
114. Kuhn DA, Vanhecke D, Michen B, et al. Different endocytotic uptake mechanisms for nanoparticles in epithelial cells and macrophages. Beilstein J Nanotechnol. 2014;5:1625–1636. doi:10.3762/bjnano.5.174
115. Commisso C, Davidson SM, Soydaner-Azeloglu RG, et al. Macropinocytosis of protein is an amino acid supply route in Ras-transformed cells. Nature. 2013;497(7451):633–637. doi:10.1038/nature12138
116. Watson P, Jones AT, Stephens DJ. Intracellular trafficking pathways and drug delivery: fluorescence imaging of living and fixed cells. Adv Drug Deliv Rev. 2005;57(1):43–61. doi:10.1016/j.addr.2004.05.003
117. Jiang L, Li X, Liu L, Zhang Q. Cellular uptake mechanism and intracellular fate of hydrophobically modified pullulan nanoparticles. Int J Nanomedicine. 2013;8:1825–1834. doi:10.2147/IJN.S44342
118. Agarwal R, Singh V, Jurney P, Shi L, Sreenivasan SV, Roy K. Mammalian cells preferentially internalize hydrogel nanodiscs over nanorods and use shape-specific uptake mechanisms. Proc Natl Acad Sci U S A. 2013;110(43):17247–17252. doi:10.1073/pnas.1305000110
119. Sharma G, Valenta DT, Altman Y, et al. Polymer particle shape independently influences binding and internalization by macrophages. J Control Release. 2010;147(3):408–412. doi:10.1016/j.jconrel.2010.07.116
120. Champion JA, Mitragotri S. Role of target geometry in phagocytosis. Proc Natl Acad Sci U S A. 2006;103(13):4930–4934. doi:10.1073/pnas.0600997103
121. Barua S, Yoo JW, Kolhar P, Wakankar A, Gokarn YR, Mitragotri S. Particle shape enhances specificity of antibody-displaying nanoparticles. Proc Natl Acad Sci U S A. 2013;110(9):3270–3275. doi:10.1073/pnas.1216893110
122. Li D, Zhuang J, He H, et al. Influence of particle geometry on gastrointestinal transit and absorption following oral administration. ACS Appl Mater Interfaces. 2017;9(49):42492–42502. doi:10.1021/acsami.7b11821
123. Yu M, Wang J, Yang Y, et al. Rotation-facilitated rapid transport of nanorods in mucosal tissues. Nano Lett. 2016;16(11):7176–7182. doi:10.1021/acs.nanolett.6b03515
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.