Back to Journals » Clinical Interventions in Aging » Volume 15
The Extract from Acidosasa longiligula Alleviates in vitro UV-Induced Skin Cell Damage via Positive Regulation of Thioredoxin 1
Authors Huang J, Xu Q , Lin M , Cheng B, Ji C
Received 25 November 2019
Accepted for publication 29 May 2020
Published 17 June 2020 Volume 2020:15 Pages 897—905
DOI https://doi.org/10.2147/CIA.S239920
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Zhi-Ying Wu
Jin-wen Huang, Qiu-yun Xu, Min Lin, Bo Cheng, Chao Ji
Department of Dermatology, The First Affiliated Hospital of Fujian Medical University, Fuzhou, Fujian 350005, People’s Republic of China
Correspondence: Chao Ji; Bo Cheng
Department of Dermatology, The First Affiliated Hospital of Fujian Medical University, Fuzhou, Fujian 350005, People’s Republic of China
Email [email protected]; [email protected]
Introduction: Skin, as the outermost organ, is exposed to a wide range of environmental risk factors including ultraviolet (UV) and all kinds of pollutants. Excessive UV exposure contributes to many disorders, such as photoaging, skin inflammation, and carcinogenesis.
Methods: To determine the effects of bamboo extract (BEX) from our local plant, Acidosasa longiligula, on UV-irritated human skin, we conducted a variety of studies, including Western blot, apoptosis assays, reactive oxygen species (ROS) detection, and thioredoxin (TXN) and thioredoxin reductase (TXNRD) activity assays in primary skin keratinocytes.
Results: We first determined that BEX protects human skin keratinocytes against UV radiation-induced apoptosis and ROS production. UV radiation can robustly impair TXN and TXNRD activity which can, in turn, be significantly rescued by BEX treatment. Moreover, BEX regulates TXN1 levels in primary skin keratinocytes and TXN1 is proved to be required for the protective function of BEX. Last, we found that the NF-κB/p65 pathway mediates the protective function of BEX against UV.
Discussion: Collectively, our work delineates the beneficial role of BEX in UV-induced skin cell damage and provides a novel therapeutic reagent to prevent or alleviate the progress of photoaging and other UV-provoked skin diseases.
Keywords: Acidosasa longiligula, bamboo extract, BEX, ultraviolet radiation, UV radiation, thioredoxin, TXN, NF-κB pathway
Introduction
Skin, the largest organ of human body, has functions of protection, regulation, and sensation.1,2 As the outermost organ, it is exposed to a wide range of environmental risk factors including ultraviolet (UV) and all kinds of pollutants. Cumulative solar exposure, the major risk factor for many skin diseases, is well established to be associated with reactive oxygen species (ROS) production, oxidative stress, and eventually oxidative damage to DNA, lipids, and proteins.3–6 Excessive UV exposure contributes to many disorders, such as photo-aging, skin inflammation, and carcinogenesis.7–9
The TXN system, consisting of TXN, TXNRD, and NADPH, plays critical roles in redox homeostasis and other cellular functions.10–12 It has a highly active redox site (-Cys-X-X-Cys-) and effectively protects organisms from oxidative stress and damages.13 It has been indicated in multiple cellular processes, such as cell metabolism, differentiation, proliferation, and apoptosis.14 Any disturbance of the TXN system may cause health problems, including cancers, neurodegenerative disorders, metabolic syndromes, and cardiovascular diseases.15 Nevertheless, UV-associated effects in the TXN system still warrant further exploration.
Recently, a wide range of natural compounds has become the focus of interest as antioxidants since they are safer than many synthetic compounds. Acidosasa longiligula, one of the most widely distributed bamboo species in central Fujian province (China), grows in mountains at an altitude of 500–1000 meters. It has high protein content, an abundance of calcium phosphorus, and is rich in fiber. Different bamboo products have been reported in treatment of many diseases including cancer, inflammatory disorders, neuropathy, and metabolism disorders.16–19 In addition, recent studies revealed that the traditional Korean food, bamboo salt, can effectively protect mouse skin from 2, 4-dinitrofluorobenzene-induced atopic dermatitis.20 However, the effect of this specific BEX in UV-irradiated skin cells has yet to be determined.
In our study, we investigated the protective effects of BEX on UV-irritated human primary skin keratinocytes. We first determined that BEX protects human skin keratinocytes against UV radiation-induced apoptosis and ROS production. UV radiation can robustly impair TXN and TXNRD activity which can, in turn, be significantly rescued by BEX treatment. Moreover, BEX regulates TXN1 levels in primary skin keratinocytes and TXN1 is proved to be required for the protective function of BEX. Last, we found that the NF-κB/p65 pathway mediates the protective function of BEX against UV. Collectively, our work delineates the beneficial role of BEX in UV-induced skin cell damage and provides a novel therapeutic reagent to prevent or alleviate the progress of photoaging and other UV-provoked skin diseases.
Materials and Methods
Chemicals and Reagents
BEX compound used in the present study was obtained from Xu Hui LLC (Fuzhou, China). The compound powder was freshly made from bamboo leaves and small branches of Acidosasa longiligula by ethanol/water (70/30, v/v) extraction. To prepare the BEX solution, the dry powder (100 mg) was freshly dissolved in 1 mL of distilled water and then filtered with sterile 0.22 µm syringe filter (Millipore).
Cell Culture and UV Radiation
As previously described,21–25 the primary skin keratinocytes from American Type Culture Collection (ATCC) were maintained in Dulbecco’s Modified Eagle Medium (DMEM) (Invitrogen Life Technologies, Carlsbad, CA, USA) with 10% fetal bovine serum (FBS) (Biomeda, Foster city, CA, USA) and 1% penicillin/streptomycin/glutamine (Gibco/Invitrogen, Carlsbad, CA, USA), in a humidified incubator with 5% CO2 at 37°C. UV radiation equipment and procedures have been described previously.21–25
Antibodies and Reagents
The primary antibodies used in our study were rabbit anti-cleaved caspase 3 (#9661, 1:1000), mouse anti-β-actin (#3700, 1:3000), rabbit anti-thioredoxin 1 (#2429, 1:1000), rabbit anti-thioredoxin 2 (#14,907, 1:1000), rabbit anti-thioredoxin reductase 1 (#15,140, 1:1000), mouse anti-thioredoxin reductase 2 (#12,029, 1:1000), rabbit anti-NF-κB p65 (#8242, 1:1000), and rabbit anti-lamin B1 (#13,435,1:1000) from Cell Signaling Technology (Danvers, MA, USA). Species-specific secondary antibodies were purchased from LI-COR Biosciences.
Cell Viability Assays
Methods have been described previously.26,27 Cell viability was examined by Cell Counting Kit-8 (CCK-8) (Dojindo, Japan) according to the manufacturer’s instructions. 2 x 105 cells/well were used in a 96-well plate with 100 µL medium.
Western Blot
This method has been described previously with slight modifications.27 In brief, samples consisting of 50 μg of protein were resolved on a denaturing 8–12% SDS-PAGE gel (Bio-Rad) and then transferred to polyvinylidene fluoride membranes by electroblotting. The membrane was blocked in blocking buffer (LI-COR) at room temperature for 1 hour and incubated with primary antibodies at 4°C overnight. Blots were then incubated with specific secondary antibodies at room temperature for 1 hour the next day. The signals were detected by ECL reagents. β-actin and lamin B1 were used as equivalent loading controls.
Apoptosis Assay
The levels of apoptosis in human skin keratinocytes were examined by Western blot analysis of the cleaved Caspase-3, TUNEL assay, and cell apoptosis by ELISA. Methods have been described previously.21,27 Briefly, cell apoptosis ratio was measured by the TUNEL percentage (TUNEL/Hoechst 33,342 ×100%). Results shown were expressed of at least 200 cells in 5 random scope fields per treatment. Cell apoptosis was also examined by Cell Apoptosis ELISA Detection Kit (Roche, Palo Alto, CA) after indicated treatments. The detailed protocols were previously described.22,24
Reactive Oxygen Species (ROS) Detection
As described in our previous studies,23–25 the cellular ROS content was assessed by fluorescence-activated cell sorting (FACS; Beckton Dickinson FACScan, Suzhou, China) using the fluorescent dye dihydrorhodamine (DHR). All fluorescent intensities were normalized to an untreated control group.
Cell Transfection
The procedure has been previously described.21 In brief, short hairpin RNA (shRNA) against TXN 1 (Sigma) and non-target control (NTC) shRNA (Sigma) was transfected into human skin keratinocytes using Lipofectamine 2000 Transfection Reagent (Thermo Fisher) according to the manufacturer’s instructions. In all studies, cells were subjected to different treatments 48 hours after the transfection.
TXN and TXNRD Activity Assays
Human skin keratinocytes (1–10 x 106) were washed with PBS and then sonicated in TE Buffer (50 mM Tris-Cl and 1 mM EDTA, pH 7.5). Protein concentration was measured using the Bradford assay (BioRad). Equivalent amounts of total protein (20 µg) were subjected to TXN and TXNRD activity assessment by TXN activity fluorescent assay kit (IMCO, Sweden) and TXNRD assay kit (Sigma, MO, USA) following the manufacturer’s instructions. TXN activity was measured by detecting the reduction of insulin disulfides by reduced TXN with TXNRD and NADPH as the electron donor. Excess TXNRD and NADPH were provided to keep TXN at reduced state. During this reaction, fluorescence intensity at 515–525 nm will be altered. Human recombinant thioredoxin 1 (hTXN-1) was used to generate a standard curve and TXN activity of the sample was determined using the formula given by this standard curve. TXNRD activity was assessed using a colorimetric assay based on the reduction of 5, 5ʹ-dithiobis (2-nitrobenzoic) acid (DTNB) with NADPH to 5-thio-2-nitrobenzoic acid (TNB), which is measured at 412 nm. TXNRD activity of samples was determined by measuring the increase in absorption at 412 nm.
Measure of DNA Single-Strand Breaks (SSBs)
The procedure was described in our previous study.25 Briefly, cells were washed and lysed with the described lysis buffer and 1.5 mL of SDS-EDTA lysis solution supplemented with 0.5 mg/mL proteinase K (Sigma). DNA was then eluted with tetrapropyl-ammonium hydroxide-EDTA (pH 12.1) with 0.1% SDS. Fractions were collected at 20-min intervals for 2 hours. Filters were treated with HC1 (1 M) for 1 hour at 60°C, and 0.4 M NaOH was added prior to scintillation counting. SSBs were normalized to the control.
NF-kB DNA Binding Assay
The activation of NF-κB was examined by use of a NF-κB (p65) Transcription Factor Assay Kit (Cayman) that detects specific NF-κB (p65) DNA binding activity in nuclear extracts. 10 μg of nuclear fractions extracted from human skin keratinocytes was used in duplicate with positive and negative controls. After incubation with the primary and secondary antibody, samples were added with developing solution. The absorbance of the samples was then measured at 450nm.
Statistical Analysis
All data shown in this study were represented as mean values ± SEM of at least three independent experiments. P values were calculated with the appropriate statistical tests using GraphPad Prism software 7.0 (GraphPad Software Inc.). Statistical tests were indicated in the figure legends. Significance was chosen as p< 0.05.
Results
The Protective Role of BEX in UV-Irradiated Human Skin Keratinocytes
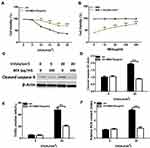
To test the role of BEX in UV-irradiated human skin keratinocytes, cells were irradiated with UV at different intensities (5, 10, 15, 20, and 25 mJ/cm2) with or without BEX pretreatment (100 μg/mL) (Figure 1A). MTT assay results revealed that UV radiation significantly induced the keratinocyte cell death, which was largely inhibited by BEX (100 μg/mL) pretreatment (Figure 1A). Further experiments showed that BEX exhibited a dose-dependent response to UV (20 mJ/cm2) radiation in keratinocytes (Figure 1B). Cell treated with BEX at 100 μg/mL exhibited decent anti-UV activity, thus, this concentration was chosen for our present study.
Apoptosis levels in human skin keratinocytes were analyzed by measuring cleaved caspase-3 levels using Western blot and TUNEL assay. Expression of cleaved caspase-3 was elevated by UV radiation and was significantly reduced by BEX co-treatment (Figure 1C and D). Consistent with the aforementioned results, TUNEL assay results supported that BEX protects keratinocytes from UV-provoked cell apoptosis (Figure 1E). UV radiation is known to cause DNA damage largely attributed to ROS production and oxidative stress.28,29 Therefore, in our study, ROS production was tested after treatments (Figure 1F). UV radiation in human primary keratinocytes provoked significant ROS production and this effect was largely diminished by BEX pretreatment (Figure 1F). These results support that BEX mitigates UV-induced cell apoptosis and ROS production in skin keratinocytes.
BEX Regulates TXN1 Level in Primary Skin Keratinocytes
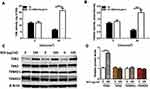
It is well established that TXN system is fundamental in many biological processes including cell proliferation and apoptosis.30 As demonstrated in our previous study, BEX significantly attenuates UV-induced ROS production. Therefore, we sought to test the effects of BEX in the TXN system in UV-irritated keratinocytes. Results revealed that TXN and TXNRD activities of human keratinocytes were significantly reduced after UV radiation. In addition, this effect was largely attenuated by BEX treatment (Figure 2A and B). We then tested whether TXN family proteins would be affected by BEX treatment. Skin keratinocytes were treated with or without BEX at indicated concentration for 24 hours. Expression of TXN1, TXN2, TXNRD1, and TXNRD2 was examined by Western blot analysis (Figure 2C). Remarkably, only TXN1 levels were significantly elevated by BEX treatment (Figure 2D), indicating a crucial role of TXN1 in the cytoprotective function of BEX.
TXN1 Is Important for the Protective Function of BEX in UV-Irradiated Human Skin Keratinocytes
To study the specific role of TXN1 in BEX-mediated cytoprotection, we conducted experiments using keratinocytes transfected with NTC or TXN 1 shRNA. As demonstrated in Figure 3A, although TXN1 deficiency significantly decreased cell viability in the BEX pretreatment group, it could not totally block the protective function of BEX, indicating that BEX may have other alternatives to regulate the UV-induced skin cell death. In line with this result, data from Histone-DNA ELISA assay showed that BEX has little effect on UV-irritated cell apoptosis when TXN1 is knocked down (Figure 3B). These results suggest that TXN1 is pivotal for BEX-mediated cytoprotection against UV in keratinocytes.
BEX Diminishes UV-Induced NF-κB/P65 Activation in Skin Keratinocytes
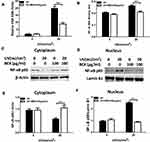
Next, we sought to identify the underlying mechanism in the protective effect of BEX against UV. Recent studies have proposed that TXN activates NF-κB signaling by reducing the cysteine residues using its active redox sites.31 Surprisingly, we discovered that BEX diminishes UV-induced NF-κB/p65 activation in skin keratinocytes. To identify the possible involvement of NF-κB signaling in BEX-induced activity against UV in skin cells, we first tested the effect of BEX in UV-induced DNA damages and NF-κB DNA binding activity (Figure 4A and B). As demonstrated, BEX significantly lowered DNA SSB formation and NF-κB activation following UV radiation (Figure 4A and B). In addition, proteins extracted from cytoplasm (Figure 4C and E) and nucleus (Figure 4D and F) were subjected to Western blot analysis for NF-κB p65 pathway proteins. BEX significantly abrogated the transportation of NF-κB p65 from cytoplasm to nucleus induced by UV-B radiation. These results suggest that NF-κB/p65 pathway is critical for BEX-induced cytoprotection against UV-B radiation in human keratinocytes.
Discussions
Over the past several decades, great effort has been applied towards studies of multiple interrelated physiological and pathological factors behind UV-related skin disorders, including genetics, age, and protective reagents, etc. A direct correlation between solar UV radiation and oxidative damage in skin tissues has been clearly defined.7 Repetitive exposure to UV radiation is observed to induce cytotoxic effects, such as solar erythema, edema, skin sensibility, skin inflammation, eventually photoaging, and even skin cancers.28,29 Therefore, a growing interest exists in investigating new therapeutic reagents that can effectively prevent UV-provoked cellular toxicity. In the present study, we discover a new compound extracted from our local plant, Acidosasa longiligula, that can counteract UV-induced ROS generation (Figure 1F), DNA damage (Figure 4A), and cell death (Figure 1) in keratinocytes. Remarkably, data presented herein indicates that TXN1 is an important mediator in BEX-regulated protective effect.
TXN-regulated redox signaling during the last several decades has been developed very fast. The TXN systems are undoubtedly an important counteractant of ROS generation through the utilization TNX cysteine residues in the Cys–X–X–Cys active site. TXNs are strong antioxidants and fundamental for cell growth and apoptosis in different organs.32 TXNs are upregulated in many cancer types and negatively regulates apoptosis in various cancer cells, such as colon, melanoma, liver, and lung cancers.33–35 Among TXN family proteins, TXN1 is known to be involved in the aging process and some age-related diseases such as neurodegenerative disorders and cancers.33,34,36,37 It can interact with redox-sensitive signaling molecules containing redox-sensitive cysteines in their DNA binding domain including activator protein 1 (AP-1), protein 21 (p21), protein 53 (p53), hypoxia-inducible transcription factor-1 alpha (HIF-1), nuclear factor κB (NF-κB), etc. The NF-κB pathway has a documented role in immune responses via regulation of the gene expression of a large number of cytokines and other immune response genes.38 NF-κB is located in the cytoplasm and will translocate into the nucleus in response to various stimuli, including ROS.35 Recent studies have indicated that TXNs facilitate NF-κB signaling by reducing the cysteine residues.10 Surprisingly, in the present study, we found that BEX diminishes UV-induced NF-κB/p65 activation even when BEX upregulates the TXN 1 expression, indicating BEX may have multiple downstream targets. We here verified that BEX inhibits UV-triggered NF-κB/p65 activation and translocation from cytoplasm to nucleus.
Herein, our findings provide novel evidence revealing that BEX from this special species, Acidosasa longiligula, can effectively protect human skin keratinocytes from UV radiation-mediated ROS production and apoptosis via regulation of thioredoxin 1 and inhibition of NF-κB pathway. Thus, BEX could serve as a potential and promising therapeutic reagent in UV-related disorders.
Disclosure
The authors declare no conflicts of interest.
References
1. Breitkreutz D, Mirancea N, Nischt R. Basement membranes in skin: unique matrix structures with diverse functions? Histochem Cell Biol. 2009;132:1–10. doi:10.1007/s00418-009-0586-0
2. Iozzo RV. Basement membrane proteoglycans: from cellar to ceiling. Nat Rev Mol Cell Biol. 2005;6:646–656. doi:10.1038/nrm1702
3. Masaki H, Atsumi T, Sakurai H. Detection of hydrogen peroxide and hydroxyl radicals in murine skin fibroblasts under UVB irradiation. Biochem Biophys Res Commun. 1995;206:474–479. doi:10.1006/bbrc.1995.1067
4. Hattori Y, et al. 8-hydroxy-2ʹ-deoxyguanosine is increased in epidermal cells of hairless mice after chronic ultraviolet B exposure. J Invest Dermatol. 1996;107:733–737. doi:10.1111/1523-1747.ep12365625
5. Heck DE, Vetrano AM, Mariano TM, Laskin JD. UVB light stimulates production of reactive oxygen species: unexpected role for catalase. J Biol Chem. 2003;278:22432–22436. doi:10.1074/jbc.C300048200
6. Terra VA, et al. Genistein prevents ultraviolet B radiation-induced nitrosative skin injury and promotes cell proliferation. J Photochem Photobiol B. 2015;144:20–27. doi:10.1016/j.jphotobiol.2015.01.013
7. Narayanan DL, Saladi RN, Fox JL. Ultraviolet radiation and skin cancer. Int J Dermatol. 2010;49:978–986. doi:10.1111/j.1365-4632.2010.04474.x
8. Sime S, Reeve VE. Protection from inflammation, immunosuppression and carcinogenesis induced by UV radiation in mice by topical Pycnogenol. Photochem Photobiol. 2004;79:193–198. doi:10.1562/0031-8655(2004)079<0193:pfiiac>2.0.co
9. Sample A, He YY. Mechanisms and prevention of UV-induced melanoma. Photodermatol Photoimmunol Photomed. 2018;34:13–24. doi:10.1111/phpp.12329
10. Lee S, Kim SM, Lee RT. Thioredoxin and thioredoxin target proteins: from molecular mechanisms to functional significance. Antioxid Redox Signal. 2013;18:1165–1207. doi:10.1089/ars.2011.4322
11. Karlenius TC, Tonissen KF. Thioredoxin and cancer: a role for thioredoxin in all states of tumor oxygenation. Cancers. 2010;2:209–232. doi:10.3390/cancers2020209
12. Arner ES. Focus on mammalian thioredoxin reductases–important selenoproteins with versatile functions. Biochim Biophys Acta. 2009;1790:495–526. doi:10.1016/j.bbagen.2009.01.014
13. Holmgren A. Thioredoxin. Annu Rev Biochem. 1985;54:237–271. doi:10.1146/annurev.bi.54.070185.001321
14. Gromer S, Urig S, Becker K. The thioredoxin system–from science to clinic. Med Res Rev. 2004;24:40–89. doi:10.1002/med.10051
15. Anand P, Stamler JS. Enzymatic mechanisms regulating protein S-nitrosylation: implications in health and disease. J Mol Med. 2012;90:233–244. doi:10.1007/s00109-012-0878-z
16. Kim A, Im M, Yim NH, Jung YP, Ma JY. Aqueous extract of Bambusa caulis in taeniam inhibits PMA-induced tumor cell invasion and pulmonary metastasis: suppression of NF-kappaB activation through ROS signaling. PLoS One. 2013;8:e78061. doi:10.1371/journal.pone.0078061
17. Liu JX, et al. Bamboo leaf extract improves spatial learning ability in a rat model with senile dementia. J Zhejiang Univ Sci B. 2015;16:593–601. doi:10.1631/jzus.B1400249
18. Kim NR, Nam SY, Ryu KJ, Kim HM, Jeong HJ. Effects of bamboo salt and its component, hydrogen sulfide, on enhancing immunity. Mol Med Rep. 2016;14:1673–1680. doi:10.3892/mmr.2016.5407
19. Choi MH, Jo HG, Yang JH, Ki SH, Shin HJ. Antioxidative and anti-melanogenic activities of bamboo stems (Phyllostachys nigra variety henosis) via PKA/CREB-Mediated MITF downregulation in B16F10 melanoma cells. Int J Mol Sci. 2018;19:409. doi:10.3390/ijms19020409
20. Yoou MS, Nam SY, Wan Yoon K, Jeong HJ, Kim HM. Bamboo salt suppresses skin inflammation in mice with 2, 4-dinitrofluorobenzene-induced atopic dermatitis. Chin J Nat Med. 2018;16:97–104. doi:10.1016/S1875-5364(18)30035-9
21. Ji C, et al. Gremlin inhibits UV-induced skin cell damages via activating VEGFR2-Nrf2 signaling. Oncotarget. 2016;7:84748–84757. doi:10.18632/oncotarget.12454
22. Ji C, et al. Exogenous cell-permeable C6 ceramide sensitizes multiple cancer cell lines to Doxorubicin-induced apoptosis by promoting AMPK activation and mTORC1 inhibition. Oncogene. 2010;29:6557–6568. doi:10.1038/onc.2010.379
23. Ji C, et al. Ultra-violet B (UVB)-induced skin cell death occurs through a cyclophilin D intrinsic signaling pathway. Biochem Biophys Res Commun. 2012;425:825–829. doi:10.1016/j.bbrc.2012.07.160
24. Ji C, et al. Trans-Zeatin attenuates ultraviolet induced down-regulation of aquaporin-3 in cultured human skin keratinocytes. Int J Mol Med. 2010;26:257–263. doi:10.3892/ijmm_00000460
25. Ji C, et al. Perifosine sensitizes UVB-induced apoptosis in skin cells: new implication of skin cancer prevention? Cell Signal. 2012;24:1781–1789. doi:10.1016/j.cellsig.2012.05.003
26. Gong T, et al. Celecoxib suppresses cutaneous squamous-cell carcinoma cell migration via inhibition of SDF1-induced endocytosis of CXCR4. Onco Targets Ther. 2018;11:8063–8071. doi:10.2147/OTT.S180472
27. Ji C, Yang B, Huang SY, Huang JW, Cheng B. Salubrinal protects human skin fibroblasts against UVB-induced cell death by blocking endoplasmic reticulum (ER) stress and regulating calcium homeostasis. Biochem Biophys Res Commun. 2017;493:1371–1376. doi:10.1016/j.bbrc.2017.10.012
28. de Gruijl FR, van Kranen HJ, Mullenders LH. UV-induced DNA damage, repair, mutations and oncogenic pathways in skin cancer. J photochem photobiol B, Biol. 2001;63:19–27. doi:10.1016/s1011-1344(01)00199-3
29. Ichihashi M, et al. UV-induced skin damage. Toxicology. 2003;189:21–39. doi:10.1016/s0300-483x(03)00150-1
30. Arner ES, Holmgren A. Physiological functions of thioredoxin and thioredoxin reductase. Eur j Biochem. 2000;267:6102–6109. doi:10.1046/j.1432-1327.2000.01701.x
31. Kelleher ZT, et al. Thioredoxin-mediated denitrosylation regulates cytokine-induced nuclear factor kappaB (NF-kappaB) activation. J Biol Chem. 2014;289:3066–3072. doi:10.1074/jbc.M113.503938
32. Matsui M, et al. Early embryonic lethality caused by targeted disruption of the mouse thioredoxin gene. Dev Biol. 1996;178:179–185. doi:10.1006/dbio.1996.0208
33. Gasdaska PY, Oblong JE, Cotgreave IA, Powis G. The predicted amino acid sequence of human thioredoxin is identical to that of the autocrine growth factor human adult T-cell derived factor (ADF): thioredoxin mRNA is elevated in some human tumors. Biochim Biophys Acta. 1994;1218:292–296. doi:10.1016/0167-4781(94)90180-5
34. Berggren M, et al. Thioredoxin and thioredoxin reductase gene expression in human tumors and cell lines, and the effects of serum stimulation and hypoxia. Anticancer Res. 1996;16:3459–3466.
35. Grogan TM, et al. Thioredoxin, a putative oncogene product, is overexpressed in gastric carcinoma and associated with increased proliferation and increased cell survival. Hum Pathol. 2000;31:475–481. doi:10.1053/hp.2000.6546
36. Mitsui A, et al. Overexpression of human thioredoxin in transgenic mice controls oxidative stress and life span. Antioxid Redox Signal. 2002;4:693–696. doi:10.1089/15230860260220201
37. Matthews JR, Wakasugi N, Virelizier JL, Yodoi J, Hay RT. Thioredoxin regulates the DNA binding activity of NF-kappa B by reduction of a disulphide bond involving cysteine 62. Nucleic Acids Res. 1992;20:3821–3830. doi:10.1093/nar/20.15.3821
38. Kabe Y, Ando K, Hirao S, Yoshida M, Handa H. Redox regulation of NF-kappaB activation: distinct redox regulation between the cytoplasm and the nucleus. Antioxid Redox Signal. 2005;7:395–403. doi:10.1089/ars.2005.7.395
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.