Back to Journals » International Journal of Nanomedicine » Volume 10 » Issue 1
The effect of aspirin nanoemulsion on TNFα and iNOS in gastric tissue in comparison with conventional aspirin
Authors Mahmoud F, Hashem K , Elkelawy A
Received 19 April 2015
Accepted for publication 8 July 2015
Published 24 August 2015 Volume 2015:10(1) Pages 5301—5308
DOI https://doi.org/10.2147/IJN.S86947
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Thomas Webster
Fatma Abd Elhalim Mahmoud,1,2 Khalid S Hashem,3 Asmaa Mohammed M Hussein Elkelawy2
1Medical Pharmacology Department, Faculty of Medicine, Cairo University, Giza, 2Clinical Pharmacology Department, Faculty of Medicine, 3Biochemistry Department, Faculty of Veterinary Medicine, Beni-Suef University, Beni-Suef, Egypt
Background: No dose of aspirin is free of bleeding risk. Even at a dose as low as 75 mg/day, the risk of upper gastrointestinal bleeding is twice as high as among nonusers. Nanoemulsions (NEs) are emulsion systems with droplet size in nanometer scale in which oil or water droplets are finely dispersed in the opposite phase with the help of a suitable surfactant to stabilize the system.
Objectives: The objective of this study was to determine the effect of aspirin NE in comparison to conventional aspirin.
Materials and methods: A total of 24 male rats were used in the study and arbitrarily assigned to four groups. Group 1 was the control group, and was given saline. Group 2 was given blank NE 1.5 mL/kg orally. Group 3 was given aspirin 30 mg/kg body weight orally. Group 4 was given aspirin NE 30 mg/kg body weight orally. Rats were killed, and gastric tissue was quickly excised after dissection of the animals. The tissues were divided into three pieces. The first one was kept in formalin 10% for pathological investigation. The second piece was kept in liquid nitrogen for molecular investigation. The third piece was homogenized in ten volumes of ice-cold phosphate-buffered saline (pH 7) using a Teflon homogenizer until a uniform suspension was obtained. The homogenate was centrifuged at 4,000 rpm for 30 minutes at 4°C to separate the supernatant from cellular debris. The supernatant was then used for the estimation of biochemical assays.
Results: The present study shows that aspirin has a toxic effect on the stomach as a result of inducing marked oxidative damage and the release of reactive oxygen species. This was shown by the significant increase in TNFα, iNOS, prostaglandin E2, and malondialdehyde levels, and also a significant decrease in glutathione, glutathione reductase, glutathione peroxidase, catalase, and superoxide dismutase. In the aspirin-treated group compared to the control group, the NE had a protective effect on the stomach and caused less injury than aspirin, indicated by significant decreases in TNFα, iNOS, prostaglandin E2, and malondialdehyde levels, and also significant increases in glutathione, glutathione reductase, glutathione peroxidase, catalase, and superoxide dismutase. The biochemical results were confirmed by histopathological studies.
Conclusion: Aspirin nanoemulsion has less toxic effect on the gastric mucosa compared to ordinary aspirin. This can be indicated by the increase of the antioxidant activity and the decrease of the inflammatory mediators in the gastric tissue.
Keywords: aspirin, aspirin nanoemulsion, blank nanoemulsion, stomach
Introduction
Nanoemulsions (NEs) are composed of nanoscale droplets of one immiscible liquid dispersed within another. NEs can be considered to be a conventional emulsion that contains very small particles.1 NEs can improve the drug solubility, rate of absorption, and targeted drug delivery. In the case of drug solubility, both hydrophilic and lipophilic drugs can be solubilized in either oil-in-water or water-in-oil NEs, which in turn ensure a better dissolution, because of the extremely small size of the particles. Furthermore, these small particles can easily penetrate through the epithelial layer to ensure a good rate of absorption of the drug. Small particle size facilitates scientists in fixing and controlling optimum dosing, which prevents dose-related toxicities. Therefore, NE has created a new opportunity for scientists to design poorly soluble and less bioavailable drugs in a more accurate way that could not be formulated through conventional methods.2
Aspirin is one of the most widely used therapeutic substances in humans, being used clinically and by the general population (as a nonprescription drug) for its analgesic, antipyretic, and anti-inflammatory properties.3 No dose of aspirin is free of bleeding risk. Even at a dose as low as 75 mg/day, the risk of upper gastrointestinal bleeding is twice as high as among nonusers.4 Therefore, we aimed to investigate the ability of an aspirin NE preparation to reduce the damage effects of ordinary aspirin on the gastric mucosa.
Materials and methods
Chemicals
Aspirin (acetylsalicylic acid) was purchased from Sigma Aldrich Co (St Louis, MO, USA). Chemicals used in the preparation of aspirin NE, including propylene glycol monolaurate type II (Lauroglycol), diethylene glycol monoethyl ether (Transcutol), polyoxyl 35 castor oil (Cremophor EL), deionized water, and λ-carrageenan were also purchased from Sigma Aldrich. Transmission electron microscopy (TEM) was used for characterization of nanoparticles (shape and size) (Figure 1).
Preparation of aspirin nanoemulsion
Aspirin (200 mg), 10% w/w Lauroglycol 90, 10% w/w Transcutol, 3.52% w/w Cremophor EL, and 76.48% w/v deionized water, treated with 15 minutes’ premixing, 20% ultrasonic amplitude, and 70 seconds’ irradiation, were presented to provide the most desired physicochemical properties. This technique produces aspirin NEs with a mean droplet diameter of about 200 nm.5,6
Aspirin NE was prepared by using Lauroglycol 90 as the dispersed phase and deionized water as the continuous phase. Aspirin was first dissolved in Transcutol. The resultant solution was then added into the aqueous phase containing Cremophor EL as the emulsifier to form the emulsion. The premixed emulsion was then further subjected to a second stage of ultrasound treatment by an ultrasonic horn processor.
Characterization of aspirin nanoemulsion
Particle-size analyzes
Mean particle-size diameter and polydispersity index were all measured in a solution form directly after synthesis, using photon correlation spectroscopy (Malvern Instruments, Malvern, UK). Aspirin NEs (2 mL) were added to the quartz cell of the spectroscopy. Measurements were taken in triplicate at 90° to the incident light source.
Nanoparticle observation
A droplet of aspirin NE was dropped onto copper grids and allowed to dry in air before observation under high-resolution TEM. The TEM-measured droplet size corroborated the results obtained from the particle-size analysis. Both measurements indicated that the average size of nanoparticles was 200 nm (range 150–200 nm). The TEM revealed that the aspirin NE droplets were almost spherical in shape with a homogeneous nanometric size distribution.
Experimental animals
This study was approved by the Committee of Scientific Ethics at Beni-Suef University, Egypt, and was carried out in accordance with its guidelines for animal use. A total of 24 male albino Wistar rats weighing on average 160±20 g were used in this study. They were obtained from the animal house of the Research Institute of Ophthalmology, Giza, Egypt. They were kept under suitable conditions for 1 week for adaptation. They were maintained in stainless steel cages in a well-ventilated animal house at normal temperature (22°C±5°C) under a 12:12-hour light–dark cycle. They were fed with a standard diet and given water ad libitum. The rats were randomly divided into four equal groups (six rats each); triplicate experiments were performed for more accurate data. Group 1 (control) rats were given saline orally. Group 2 rats were given a single dose of 1.5 mL/kg blank NE. Group 3 rats were given a single dose of aspirin (30 mg/kg body weight orally). Group 4 rats were given a single dose of aspirin NE (30 mg/kg body weight orally). At 24 hours after the last treatment, rats were killed and gastric tissue quickly excised after dissection of the animals. The tissues were divided into three pieces. The first was kept in formalin 10% for pathological investigation, and the second kept in liquid nitrogen for molecular investigation. The third piece was homogenized in ten volumes of ice-cold phosphate-buffered saline (pH 7) using a Teflon homogenizer (Tissue-Tearor; BioSpec Products Inc, Bartlesville, OK, USA) until a uniform suspension was obtained. The homogenate was centrifuged at 4,000 rpm for 30 minutes at 4°C to separate the supernatant from cellular debris. The supernatant was then used for the estimation of biochemical assays.
Methods
Biochemical studies
The following parameters were measured in the gastric tissue of the four tested rat groups, according to the instructions of the used kits. Detection of TNFα and iNOS7 in gastric tissue was performed by real-time polymerase chain reaction (RT-PCR): total RNA was extracted with Trizol reagent and purified with an RNeasy column (Qiagen NV, Venlo, the Netherlands) according to the manufacturer’s instructions. Total RNA was reverse-transcribed with murine leukemia virus reverse transcriptase and oligo-dT primers (Table 1).
  | Table 1 Primers of the tested parameters |
SYBR Green Power PCR Master Mix (Thermo Fisher Scientific, Waltham, MA, USA) was used for RT-PCR analysis. Relative differences in expression between groups were expressed using cycle-time values, and these values for genes of interest were first normalized with that of β-actin in the same sample, and then relative differences between groups were expressed as relative increases, setting control as 100% using an SV Total RNA Isolation System (Promega Corporation, Fitchburg, WI, USA) and an RT-PCR kit (Stratagene; Agilent Technologies, Santa Clara, CA, USA).
Determination of prostaglandin E2 (PGE-2) in gastric tissue was performed with an enzyme-linked immunosorbent assay kit (Cayman Chemical Company, Ann Arbor, M).8,9 Reduced glutathione (GSH),10 glutathione reductase (GR),11 glutathione peroxidase (GPx),11 catalase,12 superoxide dismutase (SOD),13 and malondialdehyde (MDA)14,15 were measured in gastric tissue using colorimetric assay kits (BioDiagnostic, Giza, Egypt).
Histopathological examination of the gastric tissue
Hematoxylin and eosin stains were used for histopathological examination of the general structure of the gastric tissue.16
Statistical analysis
Results are expressed for each parameter investigated as means ± standard deviation. One-way analysis of variance was used to compare the different groups, with Tukey’s multiple-comparison test used as a post hoc range test. All statistics were carried out using SPSS version 15. Differences were considered statistically significant when P<0.05.
Results
Biochemical changes
Measurement showed a significant increase in TNFα in the aspirin-treated group compared to the control group (Figure 2). Moreover, the blank NE group showed a significant decrease in TNFα compared to the aspirin group and a nonsignificant increase compared to the control group. The aspirin NE-treated group showed a significant decrease in TNFα compared to aspirin group (P<0.05).
iNOS in the gastric tissue of the aspirin group showed a significant increase compared to the control group (P<0.05) (Figure 3). The aspirin NE-treated group showed a significant decrease in iNOS level compared to aspirin group. Both blank NE and aspirin NE showed a nonsignificant difference compared to the control group (P<0.05).
Measurement of PGE-2 in the gastric tissue of the four tested rat groups revealed that there was a significant decrease in its level (P<0.05) in group 3 (aspirin-treated group) compared with group 1 (control), while its level significantly increased (P<0.05) in group 4 compared to group 3 (Figure 4).
There was a significant increase in the gastric tissue level of GSH, GR, GPx, catalase and SOD in group 4 compared with group 3. In addition, measurement of the gastric contents of MDA showed a significant increase in its level (P<0.05) in group 4 compared with group 3 (Tables 2 and 3).
Histopathological changes
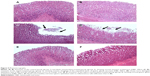
Stomachs of saline-treated control rats (group 1) and blank NE-treated rats (group 2) revealed normal architecture (Figure 5A and B). Stomachs of aspirin-treated rats (group 3) showed massive sloughing to the superficial parts of gastric glands (Figure 5C and D). Stomachs of aspirin NE-treated rats (group 4) revealed normal architecture (Figure 5E and F).
Discussion
Although aspirin is the first drug in the nonsteroidal anti-inflammatory class and has been used for its anti-inflammatory, antipyretic, and analgesic properities,17 every dose causes some superficial loss of cells from the gastric mucosa, leading to mucosal damage in most people, and interferes with gastric ulcer healing.18 This was obvious in the present study, as a single dose of aspirin (30 mg/kg) given orally caused massive injury to the stomach, indicated by significant effects on TNFα, iNOS, and PGE-2 (Figures 2–4). In addition, aspirin resulted in oxidative stress, as the level of MDA significantly increased in rats receiving aspirin. Also, aspirin significantly decreased the activity of the antioxidants GSH, GR, GPx, catalase, and SOD (Tables 2 and 3), and histopathological examination of the stomach revealed marked injury (Figure 5C and D). Results of the present work are in accordance with Naito et al19 who summarized that the gastric content of TNFα increased and the expression of TNFα messenger RNA was upregulated after aspirin treatment. TNFα is a proinflammatory cytokine, and has been shown to be a crucial mediator of aspirin-induced gastric mucosal injury.20 Aspirin induces the reactive oxygen metabolites in animal models, which may contribute to mucosal injury.21 Oxygen-derived free radicals play a key role in the mechanism of aspirin-induced acute gastric mucosal lesions.22
A number of strategies have been adopted in an attempt to reduce the gastrointestinal side effects of aspirin with the development of selective COX-2 inhibitors, which exhibit anti-inflammatory activity (COX-2 inhibition) but with reduced gastrointestinal toxicity (COX-1 inhibition).23 However, studies of the selective COX-2 inhibitors celecoxib and rofecoxib indicated that these agents still produce significant gastrointestinal injury.24 Also, the efficacy of enterically coated aspirin to reduce serious upper gastrointestinal complications when compared to plain aspirin showed a nonsignificant difference.25
However, there is a lack of studies evaluating the potential protective effect of NE preparation of nonsteroidal anti-inflammatory drugs (including aspirin) on the stomach. Therefore, the current study was constructed to evaluate the effect of an aspirin NE preparation on the stomach in comparison with aspirin.
The results of the present study showed that aspirin NE 30 mg/kg resulted in more protection and less injury to the stomach than aspirin. This was indicated by the significant decrease in TNFα (Figure 2), iNOS (Figure 3), and MDA (Table 2) levels and a significant increase in PGE-2 (Figure 4) and GSH levels and GR, GPx, catalase, and SOD activities (Tables 2 and 3) than conventional aspirin. With regard to the histopathological examination of the stomach, aspirin NE-treated groups showed less tissue injury (Figure 5E and F).
The obtained results of the current study indicated that aspirin NE is more protective of the stomach than aspirin. As one of the advantages of NEs is that they do not damage healthy human or animal cells, NEs are suitable for human and veterinary therapeutic purposes.26 In addition, fine oil droplets empty rapidly from the stomach and promote a wide distribution of the drug throughout the intestinal tract, thereby minimizing irritation.27 The NE positively influences drug transport and delivery, along with targeting to specific sites.28,29 Interestingly, NE increases the drug-retention time in the target region, and thus it causes less side effects or toxicities because it does not act on unwanted areas of the body, increases bioavailability and retention time, and decreases drug loss.30 This may explain the protective effect of aspirin NE on the stomach compared to conventional aspirin.
Conclusion and recommendations
We can conclude that aspirin NE 30 mg/kg results in more protection and less injury to the stomach than aspirin. This was indicated by the significant decrease in TNFα, iNOS, and MDA levels and significant increase in PGE-2 and GSH levels and GR, GPx, catalase, and SOD activities than conventional aspirin. Histopathological examination of the stomach confirmed our biochemical and molecular findings, and showed less tissue injury. Further investigations should be undertaken to study accurately the mechanisms of aspirin NE on the stomach wall.
Disclosure
The authors report no conflicts of interest in this work.
References
Tadros T, Izquierdo P, Esquena J, Solans C. Formation and stability of nano-emulsions. Adv Colloid Interface Sci. 2004;108–109:303–318. | ||
Prakash UR, Thiagarajan P. Nanoemulsions for drug delivery through different routes. Res Biotechnol. 2011;2:1–13. | ||
Miners JO. Drug interactions involving aspirin (acetylsalicylic acid) and salicylic acid. Clin Pharmacokinet. 1989;17:327–344. | ||
Weil J, Colin-Jones D, Langman M, et al. Prophylactic aspirin and risk of peptic ulcer bleeding. BMJ. 1995;310:827–830. | ||
Tang SY, Manickam S, Wei TK, Nashiru B. Formulation development and optimization of a novel Cremophore EL-based nanoemulsion using ultrasound cavitation. Ultrason Sonochem. 2012;19:330–345. | ||
Tang SY, Sivakumar M, Ng AM, Shridharan P. Anti-inflammatory and analgesic activity of novel oral aspirin-loaded nanoemulsion and nano multiple emulsion formulations generated using ultrasound cavitation. Int J Pharm. 2012;430:299–306. | ||
Pfaffl MW. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001;29:e45. | ||
Maxey KM, Maddipati KR, Birkmeier J. Interference in enzyme immunoassays. J Clin Immunoassay. 1992;15:116–120. | ||
Ujihara M, Urade Y, Eguchi N, Hayashi H, Ikai K, Hayaishi O. Prostaglandin D2 formation ad characterization of its synthetases in various tissues of adult rats. Arch Biochem Biophys. 1988;260:521–531. | ||
Beutler E, Duron O, Kelly BM. Improved method for the determination of glutathione. J Lab Clin Med. 1963;61:882–888. | ||
Ursini F, Maiorino M, Gregolin C. The selenoenzyme phospholipid hydroperoxide glutathione peroxidase. Biochim Biophys Acta. 1985;839:62–70. | ||
Atebi H. Catalase in vitro. Methods Enzymol. 1984;105:121–126. | ||
Nishikimi M, Appaji N, Yagi K. The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem Biophys Res Commun. 1972;46:849–854. | ||
Ohkawa H, Ohishi W, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 1979;95:351–358. | ||
Satoh K. Serum lipid peroxide in cerebrovascular disorders determined by a new colorimetric method. Clin Chim Acta. 1978;90:37–43. | ||
Drury RA, Wallington EA. Carleton’s Histological Techniques. 6th ed. London: Oxford University Press; 1980. | ||
Kapetanovic IM, Bauer KS, Tessier DM, et al. Comparison of pharmacokinetic and pharmacodynamic profiles of aspirin following oral gavage and diet dosing in rats. Chem Biol Interact. 2009;179(2–3):233–239. | ||
Penney AG, Andrews FJ, O’Brien PE. Effects of misoprostol on delayed ulcer healing induced by aspirin. Dig Dis Sci. 1994;39(5):934–939. | ||
Naito Y, Takagi T, Matsuyama K, Yoshida N, Yoshikawa T. Pioglitazone, a specific PPAR-γ ligand, inhibits aspirin-induced gastric mucosal injury in rats. Aliment Pharmacol Ther. 2001;15(6):865–873. | ||
Santucci L, Fiorucci S, Di Matteo FM, Morelli A. Role of tumor necrosis factor α release and leukocyte margination in indomethacin-induced gastric injury in rats. Gastroenterology. 1995;108(2):393–401. | ||
McAlindon ME, Muller AF, Fillipowicz B, Hawkey CJ. Effect of allopurinol, sulphasalazine and vitamin C on aspirin induced gastroduodenal injury in human volunteers. Gut. 1996;38(4):518–524. | ||
McCord JM. The evolution of free radicals and oxidative stress. Am J Med. 2000;108(8):652–659. | ||
Fung HB, Kirschenbaum HL. Selective cyclooxygenase-2 inhibitors for the treatment of arthritis. Clin Ther. 1999;21(7):1131–1157. | ||
Silverstein FE, Faich G, Goldstein JL, et al. Gastrointestinal toxicity with celecoxib vs nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis: the CLASS study: a randomized controlled trial. Celecoxib Long-Term Arthritis Safety Study. JAMA. 2000;284(10):1247–1255. | ||
Floyd CN, Ferro A. Mechanism of aspirin resistance. Pharmacol Ther. 2014;141(1):69–78. | ||
Rutvij JP, Gunjan JP, Bharadia PD, Pandya VM, Modi DA. Nanoemulsion: an advanced concept of dosage form. Int J Pharm Cosmetol. 2011;1(5):122–133. | ||
Pouton CW. Self-emulsifying drug delivery systems: assessment of the efficiency of emulsification. Int J Pharm. 1985;27(2–3):335–348. | ||
Anton N, Vandamme TF. The universality of low-energy nano-emulsification. Int J Pharm. 2009;377(1–2):142–147. | ||
Ravi TP, Padma T. Nanoemulsions for drug delivery through different routes. Res Biotechnol. 2011;2(3):1–13. | ||
Sharma N, Bansal M, Visht S, Sharma PK, Kulkarni GT. Nanoemulsion: a new concept of delivery system. Chron Young Sci. 2010;1(2):2–6. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.