Back to Archived Journals » Journal of Vascular Diagnostics and Interventions » Volume 2
The Difoprev® system in treatment of the skin of the elderly: preliminary data obtained in patients with dryness and hyperkeratosis
Authors Aluigi L
Received 22 June 2013
Accepted for publication 24 January 2014
Published 16 April 2014 Volume 2014:2 Pages 15—21
DOI https://doi.org/10.2147/JVD.S50320
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Leonardo Aluigi
Center and School of Echography, Department of Internal Medicine, Maggiore Hospital, Bologna, Italy
Background: Aging brings with it a number of structural and functional modifications of the skin and adnexa, particularly in the lower extremities, which may determine a gradual reduction in elasticity, with associated functional alterations which may determine preulcerative lesions. The aim of this study was to test the efficacy of the Difoprev® system, consisting in a sock functioning as a vehicle for microcapsules containing moisturizing agents (Pseudoalteromonas ferment extract, a glycoprotein synthesized by the bacterium, Pseudoalteromonas antarctica NF3). The system is already widely used for cutaneous treatment in cases of neuropathic diabetic foot, and in prevention/treatment of dryness and hyperkeratosis of the skin of the lower extremities in elderly persons (shin/foot).
Methodology: Sixteen patients aged >70 years (group A) were treated with the Difoprev® system for 8 weeks. This group was compared with 8 patients of the same age (group B) who wore the same type of sock without microcapsules. Following initial assessment at the time of recruitment, the patients underwent controls at 15, 30, and 60 days to assess the degree of hydration of the skin derived from an objective evaluation of degree of keratosis according to an arbitrary scale of seriousness ranging from 1 (absent) to 4 (marked). The changes were rated according to a scale ranging from 1 (no improvement) to 5 (marked improvement). A comparative study of all cases was conducted by means of photographs taken as the treatment commenced and on termination of treatment.
Results: Most patients treated with Difoprev® presented a reduction in the degree of keratosis of the skin (90%, of whom 15% had marked improvement), compared to the patients who wore only the sock with no microcapsules (10% with slight improvement). None of the patients undergoing Difoprev® system treatment displayed signs of intolerance or untoward reactions.
Conclusion: While the clinical assessment was conducted on a small number of patients, the results obtained nevertheless demonstrate that the Difoprev® system is a safe, simple method for obtaining improvements to the skin of elderly people by reduction of the degree of keratosis through enhanced hydration.
Keywords: nanotechnologies, cutaneous hydration, hyperkeratosis, ulcerative lesions
Background
As we age, also in the absence of clear venous, arterial, or neurological diseases, our skin will tend to become less elastic and will dry. It may also produce symptomatology of pruriginous conditions and of desquamation both spontaneous and induced by scratching.1–3 The resulting thinning of the epidermis brings with it a reduction of protective capacities, and exposes the skin to desquamation phenomena and, occasionally, to preulcerative lesions. Above all, in the shins and feet, the skin displays a gradual reduction in elasticity, with a reduction in the thickness of the matrix of the dermis and also of the proliferative capacity of the basal cells.4,5 The stratum corneum is harmed by crazing, through which microorganisms may penetrate and this determines inflammation at the site. Crazing may also determine preulcerative and hyperkeratotic lesions which may become chronic, especially when associated with vascular or peripheral neurological conditions,6–8 and the cutaneous adnexa undergoes gradual rarefaction until a point at which they disappear entirely.9
These alterations are frequently underestimated either because they are considered “physiological” or because less attention is paid by elderly patients to this kind of problem. Initial intervention is usually limited to use of cold creams (frequently through self care, using widely consumed products, as opposed to specific active principles in accordance with the indications provided by specialized dermatologists). If applied regularly (above all, after bathing) the active principles may contribute to preventing further pathological developments. However, in elderly patients, cold creams are not always used in a constant manner. A nanotechnology application (Difoprev® system; LVM Technologies, Bologna, Italy) has enabled incorporation into the fabric of a sock of a molecular film containing a glycoprotein endowed with marked moisturizing action: Pseudoalteromonas ferment extract. When worn, the sock gradually releases the active principle and enables the hydration action required for the skin over the entire period in which the sock is worn, thus doing away with the need for creams. The Difoprev® system is based on a technology derived from nanostructured lipid carriers, which has been demonstrated to be highly effective as a vehicle for both cosmetic and dermatological preparations.10 It consists of a sock made of a special synthetic fiber, polyamide fiber (Meryl® [Nylstar S.P.A Yarn Manufacturer - Italy, Cesano Maderno, Italy]), which is smooth and does not irritate the skin, and which, because of negative/positive surface charges interaction, can be loaded with the microcapsules containing the active moisturizer. When the socks are worn, the contained microcapsules constantly release the agent to the patient for the whole time the sock is in contact with the skin. Each charge lasts for 36 hours. This eliminates the need to moisturize skin, while also maximizing the time of application of the active agent. The sock is recharged with the microcapsules at each washing. The moisturizing agent is an emulsion of liposomes, made up of a core of lipids (phosphatidylcholine) coated by the glycoprotein secreted by the bacterium Pseudoalteromonas antarctica.11,12 This protein, along with other proteins found in microorganisms, is able to coat liposomes, protecting them from surfactant agents, thus increasing their solubility and availability.13,14 The glycoprotein (Pseudoalteromonas ferment extract), which is synthesized by a bacterium (P. antarctica NF3), is endowed with properties particularly indicated for homeostatic maintenance of the skin and subcutaneous tissue (Figures 1 and 2).
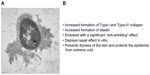  | Figure 1 Properties of the Pseudoalteromonas antarctica NF3 glycoprotein. |
The liposomes, positively charged, can exert a range of activities in the skin. They interact with the extracellular lipid junctions in the stratum corneum and isolate the deeper layer from the outer ones. It is also reported that the liposomes have a positive effect on fibroblast functions.14
This system was designed initially as a means of prevention and treatment of cutaneous trophism, especially for patients with diabetic pathological conditions, vascular pathological conditions, or wounds or sequelae of amputation.15,16 In view of the product’s characteristics and demonstrated efficacy, the indications were then broadened to include also perilesional skin treatment and treatment of skin lesions induced by bandages or elastic compression. The aim of this work is to assess the efficacy of this system in the treatment of the skin of elderly patients.
Materials and methods
Over a period of 8 weeks, a consecutive series of 16 male and female outpatients (age >70 years) underwent treatment with the Difoprev® system (consisting in microcapsules containing a glycoprotein synthesized by the bacterium, P. antarctica NF3, which are anchored to the threads of a very comfortable sock made of a special polyamide synthetic fiber, Meryl®). A comparison was conducted with 8 patients of the same age bracket who wore the same type of sock, but without the microfilm. The patients were assigned to one of the two treatment groups on the basis of a randomization list: group A, managed with the full system and group B, managed with socks with no active principle. The duration of the effect of application is approximately 36 hours. During this period, the glycoprotein contained in the microcapsule, released following rupture of the microcapsules during walking, exerts its moisturizing action as it spreads within the skin; the peculiarity of the system consists in the possibility of “recharging” the sock after washing by dipping it in a solution containing microcapsules. The solution was provided as part of the system. The patients were duly provided with instructions concerning use of the system, with attention paid in particular to the charging procedure. Of course, during the study periods no creams or ointments were used, and use of a neutral soap was recommended for personal hygiene. The patients displayed no symptomatology indicative of vascular, arterial, venous, or neurological diseases. Regardless, all patients underwent preliminary instrumental echo with color Doppler examination. If arteriopathy or chronic venous insufficiency was noted, the patient was not included in the study. Diabetic patients were also excluded.
Echo color Doppler
We performed a whole duplex echo scan for every patient of both the arterial and venous systems by Esaote MyLab™70 XV (Esaote Group, Genova, Italy) according to the ultrasound investigation guidelines of the Italian Society for Vascular Investigation17 to select patients without peripheral artery disease and/or venous disease (mostly acute venous thrombosis, chronic venous insufficiency, and post-thrombotic syndrome).
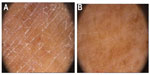
At the start and on termination of treatment, clinical assessment was conducted for the degree of hydration of skin and the corresponding degree of keratosis given in terms of an arbitrary scale of seriousness ranging from 1 (absent) to 4 (marked) (Table 1), corresponding to the clinical appearance of the skin as represented in Figure 3.
  | Table 1 Scale of the degree of keratosis |
  | Figure 3 Clinical appearance of skin. |
After an initial assessment at the time of recruitment, the patients were assessed twice at 15 day intervals and then after 30 days on termination of treatment as planned. During the control examinations and at the end of treatment, objective assessment was conducted relative to the degree of improvement of the appearance of the skin in accordance with an arbitrary scale ranging from 1 (no variation) to 5 (marked improvement) (Table 2). The variations of the conditions of the skin were photographically recorded at the start and on termination of treatment.
  | Table 2 Scale of degree of improvement obtained through use of the Difoprev® system |
Patients were instructed to carry on with their regular activities and to record in a log book any eventual adverse event, even if not apparently related to the treatment. A telephone number was provided for any emergency. Patients were also requested to keep all the remnants from the used boxes of the refills, so that in the event, a calculation of effective usage of the system could be made retrospectively.
Results
There were no drop outs in the study. None of the subjects suffered from irritation or allergic phenomena. In all treated patients, an improvement in skin condition could be observed.
Keratosis
In group A, the degree of keratosis decreased from an average value derived from the mean of the visual scale (Figure 3) of 3.31±0.54 at the initial examination to 2.59±0.61 at the second examination and to the value of 1.21±0.31 at the end of the observational period. Group B showed no change in the degree of keratosis. The corresponding keratosis values for the control group were 3.25±0.46 at the start of the study and 3.06±0.56 during the third examination at 2 months. The improvement in keratosis can be clearly seen in Table 3 and Figure 4.
  | Table 3 Degree of keratosis noted on the first examination and after 15, 30, and 60 days |
General conditions of the skin
Assessment of general conditions according to the arbitrary scale (Table 2) revealed improvement in the skin conditions of all members of group A, most of whom (more than 60%) displayed degree 4 or 5 improvement when compared to the basically stable conditions noted in the members of group B (Figure 5). Most patients treated with Difoprev® displayed a reduction in the degree of keratosis of the skin (90%, of whom 15% had marked improvement) when compared to patients who used the sock alone with no microcapsules (only 10%, with slight improvement).
  | Figure 5 Percentage of those who displayed improvement, subdivided in degrees of the general state of the skin at the end of the treatment period. |
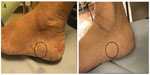
All treated patients recorded a sensation of improvement and agreeability of the device, and no problems in complying with the request for daily usage and in regard to the refilling process. By way of exemplification, comparisons may be made of the photographs regarding one treated case among the patients (15%) displaying marked improvement (Figures 6 and 7).
  | Figure 7 Macro-detail of the same case as that illustrated in Figure 6. |
Conclusion
Dryness, accompanied by hyperkeratosis, is the most typical sign of peripheral neuropathy in diabetic patients; both conditions are considered preulcerative. Irrespective of the presence of any vascular or neurological pathological conditions, elderly patients shall still frequently display alterations of the skin which fully resemble the alterations noted in diabetic patients (with the same characteristics, preulcerative lesions, and also with the same potential intercurrent diseases). The possibility of preventing the development and the evolution of anhydrosis usually relies on local application of moisturizers in the foot and the shin. Such application has proven to be an effective measure in preventing complications, but it depends on the effectiveness of the moisturizing agent and on compliance on the part of the patient which is essential for guaranteeing the success of this approach.18
Recently, a biomedical application of nanotechnology (Difoprev® system) has made it possible to design and realize an instrument which enables not only maximized moisturizing action but also the bypassing of patient compliance questions. In view of the optimal results obtained by this system in the treatment of preulcerative lesions of the shin and foot in diabetic patients,15,16,19 and in view of the characteristics of the lesions frequently noted in elderly patients which are similar, it was decided that a test be conducted regarding the efficacy of this system also under these latter conditions (which may be defined or considered “paraphysiological”). Elderly persons, especially those who are not well off, or who are socially deprived, frequently neglect personal hygiene. They therefore also pay little attention to alterations in the skin of the shin and foot, which if untreated, may develop into ulcerative lesions and/or intercurrent diseases which are infectious in nature. Alongside the above considerations, it is noted that elderly persons tend to consume liquids less regularly and this potentially leads to dehydration which is particularly evident in the skin. The possibility of using a system which enables moisturization of the skin of the shin and foot by simple means such as socks endowed with the characteristics of the Difoprev® system may thus provide a convenient remedy for elderly patients who are otherwise forced to apply creams, oils, or ointments (which for the reasons mentioned above, rarely provide guarantees of continuity of treatment).
The simple daily action of putting on a routine accessory such as socks can provide a readily applicable solution to the problem of dryness of the skin and a tendency to hyperkeratosis, while also eliminating the risk of intercurrent diseases and the need for application of topical preparations. Constant release of the active principle contained in the microcapsules of the Difoprev® system throughout the day (the duration of each refill is about 36 hours) ensures hydration of the skin and concurrently makes for greater sock–skin contact comfort. Well hydrated skin is more elastic and resistant and can reduce the risks of skin disruption and therefore ulceration. Positively charged nanoemulsions, like the one which we used, are particularly effective because of the electrical attraction between the particles and the negatively charged structures of the skin. Moreover, the small size of the particles and large surface area of application may help maximize the interaction between the skin and the lipids coated with glycoproteins.20,21 It goes without saying that the results obtained in this study can only be considered preliminary, given the few patients sampled on the basis of a distinctly clinical approach. Further in depth study and validation are required. However, the data obtained point to a significant improvement in the skin of patients treated with the Difoprev® system (compared to patients who wore the sock without microcapsules). A reduction in keratosis and a distinct improvement in the appearance of the skin were noted in group A (compared to the group B patients in whom improvement was practically absent). These findings indicate that the efficacy noted is owing to the properties of the system. Prevention is crucial to avoid progression of the disease of the skin from the early stages in which only predisposing conditions are present. The charged socks have the potential to aid prevention. As already seen for the skin of diabetic patients, Difoprev® may readily contribute to preventing skin lesions and the associated derived discomfort also experienced by elderly patients. The system is active, safe, and enables improved conditions of the skin of elderly patients by enhancing hydration and elasticity.
Disclosure
The author reports no conflicts of interest in this work.
References
Norman R. The aging of the world’s population. In: Norman R, editor. Geriatric Dermatology. New York, NY: Parthenon Publishing; 2001:1–4. | |
Rogers J, Harding R, Mayo C, Rawlings A. Stratum corneum lipids: the effect of ageing and the seasons. Arch Dermatol Res. 1996;288(12):765–770. | |
Norman R. Xerosis and pruritus. Ann Longterm Care. 2001;9(12):35–40. | |
Habif T, Campbell J Jr, Quitadamo M, Zug K. Skin Disease: Diagnosis and Treatment. St Louis, MO: Mosby; 2001. | |
Moschella S. Skin diseases of the elderly. In: Norman R, editor. Geriatric Dermatology. New York, NY: Parthenon Publishing; 2001:17–34. | |
Engelke M, Jensen J, Ekanayake-Mudiyanselage S, Proksch E. Effects of xerosis and ageing on epidermal proliferation and differentiation. Br J Dermatol. 1997;137(2):219–225. | |
Simon M, Bernard D, Minondo AM, et al. Persistence of both peripheral and non-peripheral corneodesmosomes in the upper stratum corneum of winter xerosis skin versus only peripheral in normal skin. J Invest Dermatol. 2001;116(1):23–30. | |
Sakai S, Kikuchi K, Satoh J, Tagami H, Inoue S. Functional properties of the stratum corneum in patients with diabetes mellitus: similarities to senile xerosis. Br J Dermatol. 2005;153:319–323. | |
Verdier-Sévrain S, Bonté F. Skin hydration: a review of its molecular mechanisms. J Cosmet Dermatol. 2007;6:75–82. | |
Muller RH, Petersen RD, Hommoss A, Pardeike J. Nanostructured lipid carriers (NLC) in cosmetic dermal products. Adv Drug Deliv Rev. 2007;59:522–530. | |
Sung JK, Joung HY. Cryoprotective properties of exopolysaccaride (p-21653) produced by the Antarctic bacterium, Pseudoalteromonas antarctica KOPRI 21653. J Microbiol. 2007;45:510–514. | |
Nevot M, Deroncelé V, Messner P, Guinea J, Mercadé E. Characterization of outer membrane vesicles released by the psychrotolerant bacterium Pseudoalteromonas antarctica NF3. Environ Microbiol. 2006;8:1523–1533. | |
de La Maza A, Parra JL, Congregado F, Bozal N, Guinea J. Interaction of the glycoprotein excreted by Pseudoalteromonas antarctica NF3 with phosphatidylcholine liposomes. Colloids Surf A Physicochem Eng Asp. 1998;137:181–188. | |
de La Maza A, Codech L, Lopez O, Parra JL, Sabes M, Guinea J. Ability of the exopolymer excreted by Pseudoalteromonas antarctica NF3 to coat liposomes and to protect these structures against octyl glucoside. J Biomater Sci Polym Ed. 1999;10:557–572. | |
Manfredelli S, Martire I, Passafiume F, et al. Trattamento della Neuropatia autonomica nella prevenzione delle ulcere del piede diabetico. [Autonomic neuropathy treatment to prevent diabetic foot ulcers]. Acta Vulnologica. 2010;8(2):73–79. Italian. | |
Banchellini E, Macchiarini S, Dini V, et al. Use of nanotechnology-designed footsock in the management of preulcerative conditions in the diabetic foot: results of a single, blind randomized study. Int J Low Extrem Wounds. 2008;7(2):82–87. | |
Antignani PL, Benedetti-Valentini F, Aluigi L, et al. Diagnosis of vascular disease ultrasound investigations – guidelines. Int Angiol. 2012;31(5 Suppl 1):1–77. | |
Johnston MV, Pogach L, Rajan M, et al. Personal and treatment factors associated with foot self-care among veterans with diabetes. J Rehabil Res. 2006;43:227–238. | |
Piaggesi A, Iacopi E, Banchellini E, Ambrosini Nobili L. Nanotechnology footsocks for diabetic foot. In: Le LA, Hunter RJ, Preedy VR, editors. Nanotechnology and Nanomedicine in Diabetes. Enfield: Science Publishers; 2012:371–385. | |
Rojanasakul Y, Wang LY, Bhat M, Glover DD, Malagna CJ, Ma JK. The transport barrier of epithelia: a comparative study on membrane permeability and charge selectivity in the rabbit. Pharm Res. 1992;9:1029–1034. | |
Yilmaz E, Borchert H. Effect of lipid-containing, positively charged nanoemulsion on skin hydration, elasticity and erythema – an in vivo study. Int J Pharm. 2006;307:232–238. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.