Back to Journals » Cancer Management and Research » Volume 12
The Correlation Between Computed Tomography Volumetry and Prognosis of Advanced Gastric Cancer Treated with Neoadjuvant Chemotherapy
Authors Chen C, Dong H, Shou C, Shi X, Zhang Q, Liu X, Zhu K, Zhong B, Yu J
Received 19 September 2019
Accepted for publication 7 January 2020
Published 3 February 2020 Volume 2020:12 Pages 759—768
DOI https://doi.org/10.2147/CMAR.S231636
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Chien-Feng Li
Chao Chen,1 Hao Dong,1 Chunhui Shou,1 Xiaoxiao Shi,1 Qing Zhang,1 Xiaosun Liu,1 Kankai Zhu,1 Baishu Zhong,2 Jiren Yu1
1Department of Gastrointestinal Surgery, The First Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou, People’s Republic of China; 2Department of Radiology, The First Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou, People’s Republic of China
Correspondence: Jiren Yu
Department of Gastrointestinal Surgery, The First Affiliated Hospital, Zhejiang University School of Medicine, 79 Qingchun Road, Hangzhou 310003, Zhejiang Province, People’s Republic of China
Tel + 86 571 8723 6147
Fax + 86 571 8707 2577
Email [email protected]
Purpose: To investigate the feasibility and utility of computer tomography (CT) volumetry in evaluating the tumor response to neoadjuvant chemotherapy (NAC) in advanced gastric cancer (AGC) patients.
Patients and Methods: One hundred and seventeen Patients with AGC who received NAC followed by R0 resection between January 2006 and December 2012 were included. Tumor volumes were quantified using OsiriX software. The volume reduction rate (VRR) was calculated as follows: VRR = [(pre-chemotherapy total volume) − (post-chemotherapy total volume)]/(pre-chemotherapy total volume) × 100%. The optimal cut-off VRR for differentiating favorable from unfavorable prognosis was determined by receiver operating characteristic (ROC) analysis. Overall survival was calculated using Kaplan-Meier analysis and values were compared using the Log-rank test. Multivariate analysis was determined by the Cox proportional regression model.
Results: The optimal cut-off VRR was 31.95% according to ROC analysis, with a sensitivity of 70.4% and a specificity of 71.7%. Based on the cut-off VRR, patients were divided into the VRR-High (VRR ≥ 31.95%, n = 63) and VRR-Low (VRR < 31.95%, n = 54) groups. The VRR-Low group exhibited a worse prognosis than that of the VRR-High group (HR, 2.85; 95% CI, 1.69– 4.82, P < 0.001), with 3-year survival rates of 40.7% and 79.4%, and 5-year survival rates of 31.5% and 63.5%, respectively.
Conclusion: CT volumetry is a feasible and reliable method for assessing the tumor response to NAC in patients with AGC.
Keywords: advanced gastric cancer, neoadjuvant chemotherapy, computed tomography volumetry
Introduction
Gastric cancer is the third-leading cause of cancer-related mortality worldwide, accounting for about 754,000 deaths annually.1 Although the 5-year overall survival (OS) rate of advanced gastric cancer (AGC) has improved from 23% to 40%, the prognosis of AGC remains unsatisfactory.2–4 Neoadjuvant chemotherapy (NAC) is widely regarded as an effective multidisciplinary approach to AGC therapy, with evidence of improved survival of patients with AGC compared with surgery alone.4–6 Furthermore, the adverse events associated with NAC in AGC were found to be tolerable and manageable, with a low perioperative morbidity.5,7 However, the methods used in these studies to evaluate the tumor response to NAC were not well established and remain controversial.
Computed tomography (CT) is a common procedure for assessing the clinical stage of gastric cancer due to its convenient and non-invasive nature. The Response Evaluation Criteria in Solid Tumors (RECIST) are one of the most widely used methods for evaluating the response to chemotherapy in various tumors.8,9 According to the RECIST for gastric cancer, only lymph nodes with a short-axis diameter greater than 15 mm or other metastatic lesions (such as hepatic lesions) are considered target lesions;9 primary gastric cancer lesions are deemed unsuitable targets because of their irregular shape, although the sensitivity and specificity of CT for detecting lymph node metastases vary in gastric cancer. One study reported a specificity of 99.8% and a positive predictive value of 98.6% for lymph nodes with a short-axis diameter greater than 15 mm; however, the sensitivity of this test was only 22.5%.10 Accordingly, the utility of the RECIST for evaluating the tumor response to NAC in patients with AGC might be limited. The Japanese Classification of Gastric Carcinoma–Response Assessment of Chemotherapy for Gastric Cancer (JCGC criteria) is based on morphological changes in the primary lesions, as determined using barium X-ray or endoscopic examinations,11 but it does not evaluate metastatic lesions such as lymph nodes. Furthermore, patients find the procedure inconvenient and are generally unwilling to undergo repeated endoscopic examinations.
Recent studies have suggested CT volumetry as a useful technique for tumor assessment in gastric cancer,12,13 yielding significantly greater accuracy in predicting T and N3 stages compared with conventional CT,14 with post-chemotherapy tumor volumes significantly correlated with tumor stage in patients with gastric cancer.13 Moreover, two small studies showed a significant association between CT volume reduction in primary gastric lesions after NAC and clinical outcomes.15,16 Here, we sought to investigate the feasibility and utility of CT volumetry for evaluating the tumor response to NAC.
Patients and Methods
Ethics
Ethics approval for this study was obtained from the Research Ethics Committee of the First Affiliated Hospital, College of Medicine, Zhejiang University. Patient consent was not required as this is a retrospective study. Patient data confidentiality was guaranteed. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions.
Patient Selection
Patients with AGC who received NAC followed by R0 resection between January 2006 and December 2012 were considered for this study (Figure 1). The inclusion criteria were as follows: (1) pathologically confirmed gastric adenocarcinoma, (2) a primary tumor invading the serosa (T4a) or adjacent structures (T4b) with or without metastatic lymph nodes according to CT, (3) ambulatory males or females aged 18–80 years, (4) Eastern Cooperative Oncology Group score of 0–2, (5) life expectancy greater than 3 months, (6) and normal cardiac, hepatic, renal, and bone marrow function. The exclusion criteria included (1) distant metastasis (such as lymph nodes 13 and 16, liver, lung, brain, bone, and peritoneal metastases), (2) previous major stomach surgery, (3) previous cytotoxic chemotherapy, radiotherapy, target therapy, or immunotherapy for any tumor, (4) history of another malignancy except cured basal cell carcinoma of the skin and cured carcinoma in-situ of the uterine cervix, (5) and women who were pregnant, breastfeeding, or contemplating pregnancy.
 |
Figure 1 Flowchart shows the study enrollment. Of the 242 initial patients, 117 were finally included. |
NAC Regimen and Surgery
Chemotherapy regimens included XELOX (130 mg/m2 oxaliplatin as a 2-hrs infusion on day 1, followed by 1000 mg/m2 capecitabine twice daily for 14 consecutive days), FOLFOX (130 mg/m2 oxaliplatin as a 2-hrs infusion, 400 mg/m2 leucovorin, and a bolus of 400 mg/m2 5-fluorouracil on day 1, followed by a 46-hrs infusion of 2400 mg/m2 5-fluorouracil), and SOX (130 mg/m2 oxaliplatin as a 2-hrs infusion on day 1, followed by S-1 given orally twice daily for 2 weeks). The dose of S-1 was 80 mg/day for a body surface area (BSA) <1.25 m2, 100 mg/day for a BSA ≥ 1.25 to < 1.5 m2, and 120 mg/day for a BSA ≥ 1.5 m2. Chemotherapy was repeated every 3 weeks.
Surgery was performed after at least two cycles of chemotherapy. Distal, total gastrectomy, or combined resection was performed within 2 weeks after completion of the last cycle of NAC, depending on the location and extent of the primary tumor. D2 lymphadenectomy was conducted according to the criteria established by the Japanese Gastric Cancer Association.17 Postoperative chemotherapy was initiated at 4–6 weeks after surgery. The yield pathological (yp) TNM stage was assessed according to the criteria of the AJCC TNM staging system, 7th edition.18
CT Acquisition and Analysis
A baseline CT workup was obtained up to 2 weeks prior to the initiation of NAC. For response evaluation, CT was also performed after completing at least two cycles of NAC.
Patient preparation included oral administration of 1000 mL water before CT. Patients were placed in the supine position and examined on a 16-row multi-slice spiral computed tomography (MDCT) scanner (Toshiba-MEC CT3; HiSpeed, GE Medical Systems) or a 64-row MDCT scanner (Brilliance 64, Philips Medical Systems). The scanning parameters used for 16-, and 64-row MDCT scanners were detector configuration of 16 × 0.75 mm and 64 × 0.625 mm, respectively, slice thickness of 5 mm, table speed of 15 and 40 mm/rotation, respectively, rotation time of 0.5 s, effective mAs of 200, tube voltage of 120 kVp, and matrix size of 512 × 512. The images of the portal venous phase were used for volume measurements. Volume measurement was performed using the CT image post-processing software OsiriX v7.5.1 (Pixmeo, Switzerland).
Measurable lesions were defined using the following criteria: primary lesion covering at least four consecutive CT scanning layers and measurable lymph nodes covering at least four CT scanning layers or those greater than 15 mm in the short-axis diameter at the maximum cross-section.
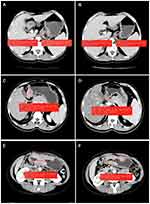
In the portal venous phase, the enclosed area along the edge of the primary tumor or lymph node was considered a region of interest (ROI, Figure 2). The ROI area was calculated automatically using OsiriX software. The volume of the target lesion was calculated by multiplying the slice thickness of the CT scan by the sum of each ROI area using the following formula:
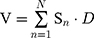
(The layers of the target lesions covered in the CT scan were defined as N, the ROI area for each layer was defined as Sn, slice thickness was defined as D, and target lesion volume was defined as V).
The number of measurable lesions was limited to a maximum of total five. The total volume was calculated by summing the volumes of the primary lesion and target lymph nodes. The percentage volume reduction rate (VRR) was calculated using the following equation:
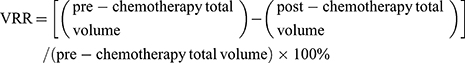
Follow-Up
All patients returned for follow-up every 3–6 months for the first 2 years, every 6–12 months during years 3–5, and annually thereafter. Standard follow-up included complete blood count, chemistry profile, and tumor marker measurements and endoscopic and radiological imaging examinations (including CT, magnetic resonance imaging, and positron emission tomography-CT if necessary).
Statistical Analysis
Quantitative values, which were analyzed by the Mann–Whitney U-test, were expressed as means ± the standard deviation. Categorical variables were expressed as absolute and relative frequencies (count and percentage) by the χ2 test. Thirty patients were randomized to investigate the interobserver variability of the tumor volume. Volumetric measurements for these patients were independently performed by two experienced doctors. The data were compared using the Wilcoxon test and Spearman correlation analysis.
The diagnostic accuracy of VRR in predicting the prognosis of patients receiving NAC was evaluated by receiver operating characteristic (ROC) analysis. The optimal cut-off VRR for differentiating favorable from unfavorable prognosis was defined as the point on the ROC curve closest to the 0% false-positive and 100% true-positive mark. Patients who survived more than 3 years were considered to have had a favorable prognosis, and a survival of less than 3 years was considered an unfavorable prognosis. The area under the ROC curve and the corresponding 95% confidence interval (CI) were determined accordingly.
OS was defined as the time from the beginning of chemotherapy to death from any cause. Patients, who were alive or lost to follow-up on June 2, 2017 were censored for the analysis of OS. The Kaplan–Meier method was used for calculation of survival time, and the resulting survival curves were compared by the Log-rank test. Cox’s proportional hazards model was used for multivariate analysis.
All statistical tests were two-sided, with P values ≤ 0.05 considered to indicate statistical significance. Statistical analyses were performed using SPSS version 19.0 (IBM, Armonk, NY, USA).
Results
Clinical and Pathological Characteristics
A total of 117 eligible patients, consisting of 83 males and 34 females, with a median age of 60 years (range 37–80 years) were included in this study. The chemotherapy regimen was FOLFOX in 49 (41.9%) patients, SOX in 45 (38.5%) patients, and XELOX in 23 (19.6%) patients. The median number of NAC cycles was 3 (range 2–6). The primary lesion was located in the upper, middle, and lower third of the stomach in 27 (23.1%), 34 (29.0%) and 54 (46.2%) patients, respectively. Multiple regions were involved in 2 (1.7%) patients. Fifty-three (45.3%) patients received distal gastrectomy, 57 (48.7%) received total gastrectomy, and 7 (6.0%) received combined resection. Following surgery, 100 (85.5%) patients received postoperative chemotherapy. According to the AJCC TNM staging system (7th edition), 9 (7.7%), 22 (18.8%), and 76 (64.9%) patients were categorized as stage I, II, and III, respectively. Five (4.3%) patients exhibited complete pathological tumor regression after NAC. Of the remaining 5 (4.3%) patients, who could not be classified, 3 were staged as ypT0N1M0 and 2 as ypT0N2M0.
Interobserver Variability
The tumor volumes before and after chemotherapy were independently assessed by two physicians. There were no significant differences between the two physicians in terms of the tumor volume before NAC (44.25 cm3 vs 45.50 cm3, p = 0.43), tumor volume after NAC (28.59 cm3 vs 31.40 cm3, p = 0.14) or the VRR (37.2% vs 33.6%, p =0.31). Spearman correlation coefficients for tumor volume before NAC, tumor volume after NAC, and VRR were 0.94 (p < 0.001), 0.88 (p < 0.001), and 0.88 (p < 0.001) respectively, indicating significant reproducibility between the two observers.
CT Volumetry Analysis
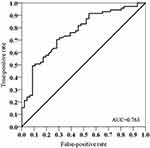
Volumetric analysis revealed a mean tumor volume of 53.8 ± 32.6 cm3 before NAC, which decreased to 35.9 ± 28.4 cm3 after NAC (p < 0.001), with an area under the ROC curve of 0.76 (95% CI, 0.68–0.85). According to ROC analysis, the optimal cut-off VRR was 31.95%, with a sensitivity of 70.4% and a specificity of 71.7% (Figure 3). According to this cut-off level, patients were divided into the VRR-High (VRR ≥ 31.95%, n = 63) and VRR-Low group (VRR < 31.95%, n = 54).
The clinical characteristics, including sex, age, chemotherapy regimen, surgical procedure, and primary tumor site, were not significantly different between the VRR-High and VRR-Low groups (Table 1). The proportion of patients with pathological T0-1 stage was 20.6% in the VRR-High group and 5.6% in the VRR-Low group. The proportion of patients with pathological N0 stage was 36.5% in the VRR-High group and 11.1% in the VRR-Low group. In addition, four patients in the VRR-High group exhibited complete pathological tumor regression after NAC, compared with only one patient in the VRR-Low group (Table 1).
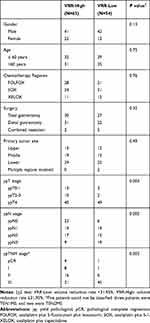 |
Table 1 The Clinical and Pathological Characteristics of the VRR-High and VRR-Low Group |
Survival Analysis
The median follow-up interval was 53.0 months (range 7–120 months). Among all patients, the 3- and 5-year survival rates were 61.5% (95% CI, 52.68–70.32) and 48.7% (95% CI, 39.68–57.72), respectively. The VRR-Low group exhibited a worse prognosis compared with the VRR-High group (HR, 2.85; 95% CI, 1.69–4.82, p <0.001), with 3-year survival rates of 40.7% (95% CI, 27.57–53.83) and 79.4% (95% CI, 69.4–89.40) and 5-year survival rates of 31.5% (95% CI, 19.15–43.85) and 63.5% (95% CI, 51.54–75.46), respectively (Figure 4). The VRR and ypN stage were identified as independent prognostic factors in Cox’s proportional hazards models (Table 2).
 |
Table 2 Univariate and Multivariate Analyses of Prognostic Factors |
 |
Figure 4 Survival analysis of the VRR-High group and VRR-Low group. VRR-Low group had worse survival rates compared with VRR-High group (HR, 2.85; 95% CI, 1.69–4.82, P<0.001). |
Discussion
NAC is widely regarded as a successful therapeutic option for AGC. Accurate and timely evaluation of the tumor response to NAC is of critical importance when making surgical decisions and may help to determine the postoperative chemotherapy regimens.19,20 Therefore, how to evaluate the tumor response to NAC is a critical issue. Until now, the common tumor response evaluation methods in clinical include RECIST, JCGC, and histopathologic tumor regression; however, certain limitations must be considered when using these methods. The RECIST are not recommended for evaluating primary gastric cancer lesions due to the irregular tumor shape, while the JCGC criteria do not address metastatic lesions, including lymph nodes. Therefore, the validity of these two methods in evaluating gastric cancer responses to chemotherapy remains uncertain and inconsistent. In a recent study, Kurokawa et al found that histological evaluation was a more effective assessment of the treatment response than radiological evaluation, and the OS of responders was significantly longer than that of non-responders evaluated by histological criteria.21 Despite these findings, histopathological tumor regression was not identified as an independent prognostic factor, even though it was found to be associated with survival in a large retrospective study of esophagogastric cancer.22
In recent years, CT volumetry has garnered significant attention as a tool for evaluating tumor responses to neoadjuvant treatment in gastrointestinal cancers. The VRR was found to be superior to the RECIST for predicting the pathological response of rectal cancer treated with neoadjuvant chemoradiation,23 while CT volumetry predicted the long-term survival of esophageal cancer patients treated with NAC followed by surgical resection.24 However, only a few studies have used CT volumetry to assess the tumor response to NAC in gastric cancer. One study showed that patients with a VRR greater than 35.6% at 8 weeks after NAC could be categorized as pathologic responders with 100% sensitivity.15 This cut-off level was much higher than that seen in a different study, in which only patients with a VRR greater than 14.8% were considered responders.16 Nevertheless, the sample sizes of both studies were small, and the correlation of CT volumetry with long-term survival was not investigated.
In the present study, no significant interobserver variability in the measurements of CT tumor volumetry was found between the two observers, which indicated that CT volumetry was reproducible. Our sample size (117 patients) makes this the largest study investigating the use of CT volumetry to evaluate the tumor response to NAC in gastric cancer to date. The median follow-up time was 53.0 months (range 7–120 months). The cut-off VRR was determined to be 31.95% by ROC analysis and was used to identify 63 (53.8%) patients as responders. Meanwhile, the maximum number of measurable lesions in this study was five, which was in accordance with the RECIST. Furthermore, the cut-off VRR of 31.95% was very close to the definitive endpoint for a partial response (≥30% decrease in the sum of the target lesion diameters) used in the RECIST.
Validated prognostic factors for patients with AGC treated with NAC followed by R0 resection have not been established. A meta-analysis of patient outcomes found that ypN stage, resection status, and age were all independent predictors for patients who receive NAC, but ypT stage was not.25 Although ypN and ypT stages were strongly associated with OS in the univariate analysis in this study, only ypN stage was an independent prognostic factor, consistent with previous studies.6,25 One possible explanation is that the effects of chemotherapy made it difficult to assess the influence of ypT stage on prognosis.
Although the findings of our study are encouraging, several limitations should be considered when evaluating these findings. First, although the section thickness of the CT scans was 5 mm, the small lesions (<5 mm) could not be assessed well compared with thin-section CT scanning. Second, although the lesions were still detected by CT after NAC, some patients achieved complete pathological regression of the tumor. Finally, despite a significantly larger sample size compared with previous studies, a much larger multi-center study will be necessary to fully evaluate these findings. Hence, CT tumor volumetry combined with some imaging biomarkers,26,27 such as the apparent diffusion coefficient from diffusion-weighted magnetic resonance imaging and the textural features from contrast-enhanced multidetector computed tomography, may represent a better choice for evaluating tumor response to NAC.
Conclusions
The data presented here indicate that CT volumetry is a feasible and reliable method for assessing the tumor response to NAC in patients with AGC. Patients with a VRR exceeding 31.95% after NAC would be categorized as clinical responders.
Abbreviations
NAC, neoadjuvant chemotherapy; AGC, advanced gastric cancer; VRR, volume reduction rate; ROC, receiver operating characteristic; ROI, region of interest.
Acknowledgments
This work was supported by the Major Project of Science and Technology Department of zhejiang province (grant number 2014C03040-1). The abstract of this paper was presented at the 2018 ASCO (American Society of Clinical Oncology) Annual Meeting as an online publication with interim findings. The poster’s abstract was published in “Meeting Abstract” in JOURNAL OF CLINICAL ONCOLOGY (DOI: 10.1200/JCO.2018.36.15_suppl.e16061).
Disclosure
The authors declare that they have no conflict of interest.
References
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108. doi:10.3322/caac.21262
2. Sasako M, Sano T, Yamamoto S, et al. D2 lymphadenectomy alone or with para-aortic nodal dissection for gastric cancer. N Engl J Med. 2008;359(5):453–462. doi:10.1056/NEJMoa0707035
3. Bang YJ, Kim YW, Yang HK, et al. Adjuvant capecitabine and oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): a Phase 3 open-label, randomised controlled trial. Lancet. 2012;379(9813):315–321. doi:10.1016/S0140-6736(11)61873-4
4. Ychou M, Boige V, Pignon JP, et al. Perioperative chemotherapy compared with surgery alone for resectable gastroesophageal adenocarcinoma: an FNCLCC and FFCD multicenter Phase III trial. J Clin Oncol. 2011;29(13):1715–1721. doi:10.1200/JCO.2010.33.0597
5. Cunningham D, Allum WH, Stenning SP, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006;355(1):11–20. doi:10.1056/NEJMoa055531
6. Becker K, Langer R, Reim D, et al. Significance of histopathological tumor regression after neoadjuvant chemotherapy in gastric adenocarcinomas: a summary of 480 cases. Ann Surg. 2011;253(5):934–939. doi:10.1097/SLA.0b013e318216f449
7. Al-Batran SE, Hofheinz RD, Pauligk C, et al. Histopathological regression after neoadjuvant docetaxel, oxaliplatin, fluorouracil, and leucovorin versus epirubicin, cisplatin, and fluorouracil or capecitabine in patients with resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4-AIO): results from the Phase 2 part of a multicentre, open-label, randomised phase 2/3 trial. Lancet Oncol. 2016;17(12):1697–1708. doi:10.1016/S1470-2045(16)30531-9
8. Therasse P, Arbuck SG, Eisenhauer EA, et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst. 2000;92(3):205–216. doi:10.1093/jnci/92.3.205
9. Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228–247. doi:10.1016/j.ejca.2008.10.026
10. Tokunaga M, Sugisawa N, Tanizawa Y, Bando E, Kawamura T, Terashima M. The impact of preoperative lymph node size on long-term outcome following curative gastrectomy for gastric cancer. Ann Surg Oncol. 2013;20(5):1598–1603. doi:10.1245/s10434-012-2699-2
11. Hallinan JT, Venkatesh SK. Gastric carcinoma: imaging diagnosis, staging and assessment of treatment response. Cancer Imaging. 2013;13:212–227. doi:10.1102/1470-7330.2013.0023
12. Li H, Chen XL, Li JR, et al. Tumor volume of resectable gastric adenocarcinoma on multidetector computed tomography: association with N categories. Clinics (Sao Paulo). 2016;71(4):199–204. doi:10.6061/clinics
13. Wang ZC, Wang C, Ding Y, Ji Y, MS Z, Rao SX. CT volumetry can potentially predict the local stage for gastric cancer after chemotherapy. Diagn Interv Radiol. 2017;23(4):257–262. doi:10.5152/dir.2017.16517
14. Hallinan JT, Venkatesh SK, Peter L, Makmur A, Yong WP, So JB. CT volumetry for gastric carcinoma: association with TNM stage. Eur Radiol. 2014;24(12):3105–3114. doi:10.1007/s00330-014-3316-5
15. Lee SM, Kim SH, Lee JM, et al. Usefulness of CT volumetry for primary gastric lesions in predicting pathologic response to neoadjuvant chemotherapy in advanced gastric cancer. Abdom Imaging. 2009;34(4):430–440. doi:10.1007/s00261-008-9420-8
16. Beer AJ, Wieder HA, Lordick F, et al. Adenocarcinomas of esophagogastric junction: multi-detector row CT to evaluate early response to neoadjuvant chemotherapy. Radiology. 2006;239(2):472–480. doi:10.1148/radiol.2391050043
17. Japanese Gastric Cancer A. Japanese gastric cancer treatment guidelines 2010 (ver. 3). Gastric Cancer. 2011;14(2):113–123. doi:10.1007/s10120-011-0042-4
18. Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010;17(6):1471–1474. doi:10.1245/s10434-010-0985-4
19. Ott K, Lordick F, Herrmann K, Krause BJ, Schuhmacher C, Siewert JR. The new credo: induction chemotherapy in locally advanced gastric cancer: consequences for surgical strategies. Gastric Cancer. 2008;11(1):1–9. doi:10.1007/s10120-007-0448-1
20. Tang L, Li ZY, Li ZW, et al. Evaluating the response of gastric carcinomas to neoadjuvant chemotherapy using iodine concentration on spectral CT: a comparison with pathological regression. Clin Radiol. 2015;70(11):1198–1204. doi:10.1016/j.crad.2015.06.083
21. Kurokawa Y, Shibata T, Sasako M, et al. Validity of response assessment criteria in neoadjuvant chemotherapy for gastric cancer (JCOG0507-A). Gastric Cancer. 2014;17(3):514–521. doi:10.1007/s10120-013-0294-2
22. Schmidt T, Sicic L, Blank S, et al. Prognostic value of histopathological regression in 850 neoadjuvantly treated oesophagogastric adenocarcinomas. Br J Cancer. 2014;110(7):1712–1720. doi:10.1038/bjc.2014.94
23. Xiao J, Tan Y, Li W, et al. Tumor volume reduction rate is superior to RECIST for predicting the pathological response of rectal cancer treated with neoadjuvant chemoradiation: results from a prospective study. Oncol Lett. 2015;9(6):2680–2686. doi:10.3892/ol.2015.3101
24. Tamandl D, Gore RM, Fueger B, et al. Change in volume parameters induced by neoadjuvant chemotherapy provide accurate prediction of overall survival after resection in patients with oesophageal cancer. Eur Radiol. 2016;26(2):311–321. doi:10.1007/s00330-015-3860-7
25. Ronellenfitsch U, Schwarzbach M, Hofheinz R, et al. Predictors of overall and recurrence-free survival after neoadjuvant chemotherapy for gastroesophageal adenocarcinoma: pooled analysis of individual patient data (IPD) from randomized controlled trials (RCTs). Eur J Surg Oncol. 2017;43(8):1550–1558. doi:10.1016/j.ejso.2017.05.005
26. Giganti F, Tang L, Baba H. Gastric cancer and imaging biomarkers: part 1–a critical review of DW-MRI and CE-MDCT findings. Eur Radiol. 2019;29(4):1743–1753. doi:10.1007/s00330-018-5732-4
27. Jiang Y, Chen C, Xie J, et al. Radiomics signature of computed tomography imaging for prediction of survival and chemotherapeutic benefits in gastric cancer. EBioMedicine. 2018;36:171–182. doi:10.1016/j.ebiom.2018.09.007
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.