Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
The Clinical Effect of Electroconvulsive Therapy and Its Relationship with Serum Levels of MMP-9 and CXCL12 in Patients with Mania
Authors Kashefi B, Mohammadi M, Rezaei F, Ghadami N , Jalili K, Jalili A
Received 10 October 2019
Accepted for publication 5 March 2020
Published 5 April 2020 Volume 2020:16 Pages 909—914
DOI https://doi.org/10.2147/NDT.S234013
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Roger Pinder
Babak Kashefi,1 Mehdi Mohammadi,2 Farzin Rezaei,1 Narges Ghadami,1 Khashaiar Jalili,3 Ali Jalili2
1Neurosciences Research Center, Research Institute for Health Development, Kurdistan University of Medical Sciences, Sanandaj, Iran; 2Cancer and Immunology Research Center, Research Institute for Health Development, Kurdistan University of Medical Sciences, Sanandaj, Iran; 3Faculty of Sciences, University of Waterloo, Waterloo, ON, Canada
Correspondence: Ali Jalili
Cancer and Immunology Research Center, Research Institute for Health Development, Kurdistan University of Medical Sciences, Sanandaj, Iran
Email [email protected]
Introduction: Electroconvulsive therapy (ECT) is a non-pharmacological method for the treatment of psychiatric disorders. The precise biochemical mechanism of the effects of ECT is not clear, and since the two factors including matrix metalloproteinase-9 (MMP-9) and stromal cell-derived factor-1 alpha (CXCL12) play an important role in improving nerve damage, the effects of ECT and its relation with serum levels of MMP-9 and CXCL12 in patients with mania were investigated in this study.
Methods: In this before and after intervention study, the patients with mania, referring to the Qods Hospital in Sanandaj, were selected by the census method during the years 2015– 2018. Young’s test was performed 24 hrs before and after the first, third, and sixth sessions of ECT. For biochemical analysis, 3 mL of peripheral blood were taken prior to any anesthesia and 6 hrs after the first, third, and sixth sessions. Data were analyzed by two-way ANOVA and Pearson correlation coefficient by using the SPSS16 software.
Results: The results showed a significant decrease in Young’s test scores during the first to the sixth session of ECT (P≤ 0.05). Although the levels of CXCL12 were slightly increased after the sixth course of ECT, they were not significant. Moreover, there were no significant relationship between the Young’s test score and the serum levels of both MMP-9 and CXCL12 (P≥ 0.05).
Conclusion: ECT improved patients clinically, but this effect was independent of serum levels of MMP-9 and CXCL12, and possibly other biochemical factors are involved in this pathway.
Keywords: matrix metalloproteinase, stromal cell-derived factor-1 alpha, electroconvulsive therapy
Introduction
Electroconvulsive therapy (ECT) is a non-pharmacological method for treating psychiatric disorders such as depression, bipolar disorder, and schizophrenia. ECT is an electrical stimulus in the brain, which causes cellular membrane depolarization and convulsion. This treatment is much more reliable and, in terms of therapeutic, has a shorter period of treatment than chemotherapy.1,2 Although pharmacotherapy is the first line for management of patients with major psychotic, ECT is the choice therapeutic invention for treatment-resistance patients. ECT has also been used in life-threatening illnesses, patients with severe suicidal symptoms and catatonia.3,4 Although the effectiveness of ECT to treat patients with psychiatric disorders has been demonstrated, the underlying cerebral mechanism remains unclear. However, three mechanistic hypotheses, albeit in part, for ECT have been proposed: (i) immune regulation; accumulating evidence has shown that during neuroinflammation, the release of inflammatory cytokines activates microglia and recruits them to the site of lesion. Of note, microglia also take part in the resolution of inflammation by secreting anti–inflammatory cytokines.5,6 Moreover, it has been reported that a single dose of ECT induces an acute and transient inflammation, while repetitive ECT treatment leads to a long-term immune suppression, at least in the brain.7 (ii) Neuroplastic changes; ECT treatment enhances neurogenesis, synaptogenesis, and proliferation of glial cells.8,9 In addition, increased levels of some cytokines such as IL-6 and brain-derived neurotrophic factor (BDNF) following ECT could result in hippocampal neurogenesis and clinical responses.10,11 (iii) Monoamine pathway; ECT treatment has been shown to enhance neurological levels of serotonin, dopamine, norepinephrine, and acetylcholine.2 Inflammation increases activities of indoleamine 2,3-dioxygenase and degrades tryptophan to kynurenine. As tryptophan is the precursor of serotonin, the levels of serotonin are decreased in neuroinflammation.12 In terms of the neurophysiological mechanism, increased blood flow and metabolic rate of the brain due to the permeability of the blood-brain barrier during convulsion have been introduced.13,14 Even though a recent study reported that ECTacts through increasing thyroid hormone,15 the precise biochemical mechanisms of ECT actions are not yet fully understood.
Matrix metalloproteinase-9 (MMP-9) is an enzyme from the family of zinc-dependent metalloproteinases, which is increased in peripheral blood in pathologic conditions such as cardiovascular disease, cancers, and neuropsychiatric disorders.16–18 Currently, it has been shown that MMP-9 plays a very important two-way role in the central nervous system, and that can be either the intermediate between pathological pathways or the central player in the regeneration of the central nervous system.19,20 MMP-9 can open the blood-brain barrier and increase its permeability. It has previously been proposed that ECT increases blood-brain barriers permeability in cerebrovascular.21
Stromal cell-derived factor-1 alpha (CXCL12) is a chemokine that causes the migration of lymphocytes and the stem cells, and also plays an important role in the angiogenesis and the development of the nervous system.22 This chemokine is produced by hematopoietic stromal cells, endothelial cells, osteoblasts, and its receptor CXCR4 is expressed on the surface of various types of leukocytes and stem cells.23
CXCL12 and its receptor not only regulate the growth and migration of the stem cells but may also play a fundamental role in brain development.22 CXCL12 also increases the migration and proliferation of the granuloma cells, activation of microglia and secretions of glutamate by astrocyte, and also causes the migration of the cortical neural progenitor cells.24 Migration to the damaged area is the first important step of stem cells to contribute to reconstruction, and it has been shown that CXCL12 and its receptor are involved in the migration of neural stem cells to the brain lesions.25
Although the effectiveness of ECT to treat patients with mania had been demonstrated, the underlying biochemical and molecular mechanisms have not been yet elucidated. As MMP-9 and CXCL12 play fundamental roles in the nervous system, repair, and cell migration this study was designed to investigate the clinical effects of ECT and its possible relationship with the above biochemical factors.
Materials and Methods
The Population Studied and Sampling Method
This study was a before and after intervention study. The patients with mania referring to Sanandaj Qods Hospital during 2015–2018, who were candidates for receiving ECT, comprised the statistical population.
Entry criteria include:
- Diagnosis of type 1 bipolar disorder (manic episode) based on the endorsed criteria (DSM_IV_TR; American Psychiatric Association, 2000)
- The age of 18 to 45 years
- The patient’s scoring a score of greater or equal to 20 on the Young scale
- Normal IQ score
- Anesthesia class 1 or 2
Exit criteria include:
- History of receiving ECT during the past six months
- Use of non-benzodiazepine anticonvulsant drugs (valproate, carbamazepine, lamotrigine, and topiramate)
- Use of lithium, lidocaine, or theophylline during the last week
- Mixed episode, schizophrenia, or other psychotic disorder that is not part of a mood disorder
- Neurological, cardiac, or severe systemic illness
- Pregnancy
- History of stroke within three months before the start of the study
- History of brain surgery
- History of severe skull injury, addiction to alcohol or any drugs during the past two years
- History of tumors and any inflammatory disease.
The sampling method was census-based and all patients who had the study conditions were sampled. The total number of subjects enrolled were 31, two of which were excluded due to exiting conditions (one due to lack of convulsion and the other because of a positive drug test). Of the 29 specimens obtained, one sample was removed from the study due to lysing; therefore, we practically analyzed 28 samples.
Ethical Considerations
The present study was confirmed by the ethics committee of the Kurdistan University of Medical Sciences with the numbered MUK88622/14 and recorded in IRCT with IRCT201410221556N6 code. All participants in the study were given a full explanation of why and how to do the study, and all participants provided written informed consent.
Detection of MMP-9 and CXCL12 and Determination of Young’s Test
Young’s test (Young Mania Rating Scale) was performed 24 hrs before and after the first, third and sixth sessions of ECT. For biochemistry analysis, 3 mL of peripheral blood were taken prior to any anesthesia, 10 mins and 6 hrs after the first, third, and sixth sessions. Blood samples were placed in a centrifuge device at a rate of 1500 rpm for 5 mins to separate the plasma. The obtained serum was then transferred to the microtubes and placed in a freezer at −70°C until the tests were carried out. Serum levels of MMP-9 and CXCL12 were measured by the ELISA method according to the manufacturer’s instructions (R & D systems, MN). The clinical symptoms of the patient were also assessed by our resident (BK) using Young’s test, which is a mania severity assessment table, scored based on the patient reports and evaluation during the interview.
Statistical Analysis
SPSS16 software was used for statistical analysis. Two-Way ANOVA test (repeated measure) was used to analyze ECT over time in terms of matrix MMP-9 and CXCL12. One-Way ANOVA test was used to analyze changes in Young’s test scores over time, and also for determining the relationship between the above factors and the Young’s test, the Pearson correlation coefficient was used. The significant level was considered to be as P≤ 0.05.
Results
In this study, 28patients with mania were examined (14 male and 14 female). The mean age of participants was 32 years old. In Table 1, the mean and standard deviation of the two measured biochemical parameters have been shown. The findings of this study indicated that no changes in MMP-9 have been observed following ECT, but the amount of CXCL12 increased after the third course of the treatment, although this increase was not significant (P>0.05). Table 2 describes the mean of scores based on the Young Mania Rating Scale (YMRS). As can be seen, the scores, according to the Scale, have been declining, indicating the effects of the ECT in patients with mania.
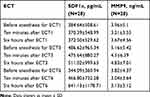 |
Table 1 The Levels of MMP-9 and CXCL12 in Various Courses of ECT |
 |
Table 2 Description of the Mean Scores Based on the Young Clinical Questionnaire in Mania Patients |
Table 3 shows that there was a significant relationship between ECT and the scores obtained from YMRS at different times. Statistical analysis showed no significant relationship between the ECT and the levels of MMP-9 and CXCL12. This information is presented in Table 4. Also, there was no significant relationship between YMRS (clinical findings) and the levels of MMP-9 and CXCL12.
 |
Table 3 Comparison of the Mean Score of Young’s Questionnaires in Patients with Mania |
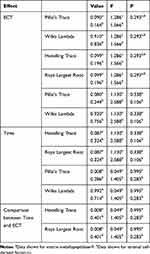 |
Table 4 Comparison of MMP-9 and CXCL12 at Different Times of ECT in Patients with Mania (Multivariate Tests) |
Discussion
The main objective of this study was to evaluate the clinical effect of ECT and its association with MMP-9 and CXCL12 in patients with bipolar disorder in manic phases. The important results of this study were the reduction of Young’s test score during the first to the sixth session of ECT.
Although ECT was effective in improving the patients in manic phases which was consistent with a recent study has shown that early ECT reduces the length of hospitalization and the cost of treatment,26 but the levels of MMP-9 did not show a significant difference during the ECT courses. However, this effect is independent of MMP-9 levels during the treatment process and other factors in the mechanism of recovery by ECT may be involved. However, a recent study had demonstrated that the levels of MMP-9, but not BDNF, are significantly higher in the schizophrenic patients.27 BDNF regulates the survival and growth of neurons and affects effects on neurogenesis and neuroplasticity. BDNF is initially sanitized as pro-BDNF and eventually converted to mature BDNF by MMP-9. In contrast to pro-BDNF which elicit apoptosis, mature BDNF preferentially binds to TrkB and increases cell survival, outgrowth of neuritis, and synaptic plasticity,28,29 implying that MMP-9 could play an important role in neuronal cells survival and development by coveting of Pro-BDNF to mature BDNF. However, Yamamori et al previously demonstrated that neither mature BDNF nor MMP-9 plasma levels were associated with clinical variables.27 In the line of this later observation, we found that the recovery of patients with bipolar disorder in the manic phase was independent of MMP-9; thus, implying that enzymes might be involved in the recovery process of patients by ECT. On the other hand, due to the fact that there are 23 types of metalloproteinases in the human body,30 and in many cases these enzymes have overlapping effects, we envision that metalloproteinases other than MMP-9, could be involved in the biochemical and molecular effects of ECT. Another study showed that ECT may induce its treatment through the effect on the hypothalamic-pituitary-thyroid axis in patients with bipolar disorder. The results of this study showed that the levels of thyroid hormones (T3 and T4) increased after treatment compared with pre-treatment, while TSH decreased.15 On the other hand, the change in MMP-9 is not tangible in the bloodstream because MMP-9 is not produced by certain tissues, unlike thyroid hormones that are produced by many cells, including brain cells,16 and its increase may be localized.
A previous study on the people with schizophrenia and depression showed that before ECT, MMP-9 level in people with depression was more than those in the control group and people with schizophrenia;, and after ECT, MMP-9 level significantly decreased in people with depression.31 Conversely, MMP-2 levels in mood disorder patients, but not in schizophrenia patients were scientifically lower than the healthy, and increased after ECT treatment,31 suggesting that for each certain type of psychiatric disorder a specific MMP might play a role. In the present study, after ECT, MMP9 level was also initially reduced during the first sessions and then increased in subsequent sessions, but these changes were not statistically significant. Also, there was no significant relationship between the clinical signs and the changes in MMP-9.
CXCL12 and its cognitive receptor CXCR4 are expressed by neural stem cells are involved in adult neurogenesis.32 Enhanced environment induces neuronal plasticity and hippocampal neurogenesis which is accompanied by was accompanied by the increased expressions of SDF-1 and CXCR4 in the hippocampus,33 indicating that CXCL12 is a neurogenesis inducer factor. This notion was supported by another observation which demonstrated that CXCR4 antagonist, AMD3100 reversed the effect of EE on neurogenesis.34 In the present study, the positive effect of ECT on the recovery of people with bipolar disorder in the manic phase was observed but there was no significant correlation between this recovery and CXCL12 levels. However, in the third period of treatment, a relatively small increase in serum CXCL12 levels was observed after 10 mins and 6 hrs of treatment, but this difference was not significant (P>0.05), and probably the low volume of sample may be the reason for this result. Moreover, Deng et al reported that the bone marrow stromal cells (in vitro) receiving electric shock showed more angiogenesis and neurogenesis compared to the control group; and CXCL12 and its receptor (CXCR4) were effective in this phenomenon.35 In the present study, similar to the previous study, the positive effect of electric shock on the recovery of patients with bipolar disorder in the maniac phase was observed. However, the results of the current study showed that this case is probably independent of CXCL12.
In the current study, we encountered several limitations. Lacking enough budget for buying reagents and kits to measure other potential biomarkers was the main obstacle for us. Of course, drawing blood sample at various time course of ECT treatment was another limitation of this study.
Conclusion
In the present study, with respect to Young Mania Rating Scale, the positive effect of ECT in people with bipolar mania was found. The study on MMP-9 and CXCL12 showed that this effect is independent of changes in these two factors, and possibly these effects are through other factors. It is suggested that molecular factors along with important hormones in psychoneuroimmunological pathways should be investigated in a larger statistical study.
Acknowledgments
This work was supported by grants from Kurdistan University of Medical Sciences to AJ and FR for supporting the BK’s thesis as a resident in Psychiatry.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Stripp TK, Jorgensen MB, Olsen NV. Anaesthesia for electroconvulsive therapy–new tricks for old drugs: a systematic review. Acta Neuropsychiatr. 2018;30(2):61–69. doi:10.1017/neu.2017.12
2. Rudorfer MV, Henry ME, Sackeim HA. Electroconvulsive therapy. Psychiatry. 2003;2:1865–1901.
3. Coffey MJ, Cooper JJ. Therapeutic uses of seizures in neuropsychiatry. Focus (Am Psychiatr Publ). 2019;17(1):13–17. doi:10.1176/appi.focus.20180023
4. Salik I, Marwaha R. Electroconvulsive Therapy. StatPearls. Treasure Island (FL): StatPearls Publishing StatPearls Publishing LLC.; 2020.
5. van Rossum D, Hanisch U-K. Microglia. Metab Brain Dis. 2004;19(3–4):393–411. doi:10.1023/B:MEBR.0000043984.73063.d8
6. Hanisch UK. Microglia as a source and target of cytokines. Glia. 2002;40(2):140–155. doi:10.1002/glia.10161
7. Guloksuz S, Rutten BP, Arts B, van Os J, Kenis G. The immune system and electroconvulsive therapy for depression. J ECT. 2014;30(2):132–137. doi:10.1097/YCT.0000000000000127
8. Chen F, Madsen TM, Wegener G, Nyengaard JR. Repeated electroconvulsive seizures increase the total number of synapses in adult male rat hippocampus. Eur Neuropsychopharmacol. 2009;19(5):329–338. doi:10.1016/j.euroneuro.2008.12.007
9. Wennström M, Hellsten J, Tingström A. Electroconvulsive seizures induce proliferation of NG2-expressing glial cells in adult rat amygdala. Biological psychiatry. 2004;55(5):464–471. doi:10.1016/j.biopsych.2003.11.011
10. van Buel EM, Patas K, Peters M, Bosker FJ, Eisel UL, Klein HC. Immune and neurotrophin stimulation by electroconvulsive therapy: is some inflammation needed after all? Transl Psychiatry. 2015;5(7):e609–e. doi:10.1038/tp.2015.100
11. Rush G, O’donovan A, Nagle L, et al. Alteration of immune markers in a group of melancholic depressed patients and their response to electroconvulsive therapy. J Affect Disord. 2016;205:60–68. doi:10.1016/j.jad.2016.06.035
12. Miller AH, Raison CL. The role of inflammation in depression: from evolutionary imperative to modern treatment target. Nat Rev Immunol. 2016;16(1):22. doi:10.1038/nri.2015.5
13. Hermida AP, Glass OM, Shafi H, McDonald WM. Electroconvulsive therapy in depression: current practice and future direction. Psychiatric Clin. 2018;41(3):341–353. doi:10.1016/j.psc.2018.04.001
14. Oltedal L, Narr KL, Abbott C, et al. Volume of the human hippocampus and clinical response following electroconvulsive therapy. Biological Psychiatry. 2018;84(8):574–581. doi:10.1016/j.biopsych.2018.05.017
15. Akbari M, Afkhami-ardekani M, Eslami M. Effects of electroconvulsive therapy on thyroid function parameters in depressed patients. J Shahid Sadoughi Univ Med Sci. 2008;16(3):312.
16. Kaminari A, Tsilibary EC, Tzinia A. A new perspective in utilizing MMP-9 as a therapeutic target for Alzheimer’s disease and type 2 diabetes mellitus. J Alzheimer’s Dis. 2018;64(1):1–16. doi:10.3233/JAD-180035
17. Ellies LG, Hingorani DV, Lippert CN, et al. Impact of MMP-2 and MMP-9 activation on wound healing, tumor growth and RACPP cleavage. BioRxiv. 2018;327791.
18. Akbarzadeh M, Movassaghpour AA, Ghanbari H, et al. The potential therapeutic effect of melatonin on human ovarian cancer by inhibition of invasion and migration of cancer stem cells. Sci Rep. 2017;7(1):17062. doi:10.1038/s41598-017-16940-y
19. Qu J, Zhao H, Li Q, et al. MST1 suppression reduces early brain injury by inhibiting the NF-κB/MMP-9 pathway after subarachnoid hemorrhage in mice. Behav Neurol. 2018;2018. doi:10.1155/2018/6470957
20. Chelluboina B, Nalamolu KR, Klopfenstein JD, et al. MMP-12, a promising therapeutic target for neurological diseases. Mol Neurobiol. 2018;55(2):1405–1409. doi:10.1007/s12035-017-0418-5
21. Sweeney MD, Sagare AP, Zlokovic BV. Blood–brain barrier breakdown in Alzheimer disease and other neurodegenerative disorders. Nat Rev Neurol. 2018;14(3):133. doi:10.1038/nrneurol.2017.188
22. Zhang Y, Zhang H, Lin S, et al. SDF-1/CXCR7 chemokine signaling is induced in the peri-infarct regions in patients with ischemic stroke. Aging Dis. 2018;9(2):287. doi:10.14336/AD.2017.1112
23. Kara T, Akaltun İ, Cakmakoglu B, Kaya İ, Zoroğlu S. An investigation of SDF1/CXCR4 gene polymorphisms in autism spectrum disorder: a family-based study. Psychiatry Investig. 2018;15(3):300. doi:10.30773/pi.2017.05.31.2
24. Chu T, Shields LB, Zhang YP, Feng S-Q, Shields CB, Cai J. CXCL12/CXCR4/CXCR7 chemokine axis in the central nervous system: therapeutic targets for remyelination in demyelinating diseases. Neuroscientist. 2017;23(6):627–648. doi:10.1177/1073858416685690
25. Shroff G, Titus JD, Shroff R. A review of the emerging potential therapy for neurological disorders: human embryonic stem cell therapy. Am J Stem Cells. 2017;6(1):1.
26. Patel RS, Jain SB, Youssef NA. Electroconvulsive treatment utilization for the inpatient management of severe manic episodes of bipolar disorder. J ECT. 2019;35(3):195–200. doi:10.1097/YCT.0000000000000587
27. Yamamori H, Hashimoto R, Ishima T, et al. Plasma levels of mature brain-derived neurotrophic factor (BDNF) and matrix metalloproteinase-9 (MMP-9) in treatment-resistant schizophrenia treated with clozapine. Neurosci Lett. 2013;556:37–41. doi:10.1016/j.neulet.2013.09.059
28. Hashimoto K. Sigma-1 receptor chaperone and brain-derived neurotrophic factor: emerging links between cardiovascular disease and depression. Prog Neurobiol. 2013;100:15–29. doi:10.1016/j.pneurobio.2012.09.001
29. Martinowich K, Manji H, Lu B. New insights into BDNF function in depression and anxiety. Nat Neurosci. 2007;10(9):1089–1093. doi:10.1038/nn1971
30. Jabłońska-trypuć A, Matejczyk M, Rosochacki S. Matrix metalloproteinases (MMPs), the main extracellular matrix (ECM) enzymes in collagen degradation, as a target for anticancer drugs. J Enzyme Inhib Med Chem. 2016;31(sup1):177–183. doi:10.3109/14756366.2016.1161620
31. Shibasaki C, Takebayashi M, Itagaki K, et al. Altered serum levels of matrix metalloproteinase-2,-9 in response to electroconvulsive therapy for mood disorders. Int J Neuropsychopharmacol. 2016;19:9. doi:10.1093/ijnp/pyw019
32. Cheng X, Wang H, Zhang X, et al. The role of SDF-1/CXCR4/CXCR7 in neuronal regeneration after cerebral ischemia. Front Neurosci. 2017;11:590. doi:10.3389/fnins.2017.00590
33. Zhang X, Liu T, Zhou Z, et al. Enriched environment altered aberrant hippocampal neurogenesis and improved long-term consequences after temporal lobe epilepsy in adult rats. j mol neurosci. 2015;56(2):409–421. doi:10.1007/s12031-015-0571-0
34. Zhou Z, Liu T, Sun X, et al. CXCR4 antagonist AMD3100 reverses the neurogenesis promoted by enriched environment and suppresses long-term seizure activity in adult rats of temporal lobe epilepsy. Behav Brain Res. 2017;322(Pt A):83–91. doi:10.1016/j.bbr.2017.01.014
35. Deng QJ, Xu XF, Ren J. Effects of SDF-1/CXCR4 on the repair of traumatic brain injury in rats by mediating bone marrow derived mesenchymal stem cells. Cell Mol Neurobiol. 2018;38(2):467–477. doi:10.1007/s10571-017-0490-4
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
