Back to Journals » Clinical Ophthalmology » Volume 14
The Central Subfoveal Bouquet in Idiopathic Epiretinal Membranes
Authors Damasceno NA , Damasceno EF , Yannuzzi NA, Crane AM, Relhan N, Smiddy WE , Flynn HW Jr
Received 31 March 2020
Accepted for publication 27 May 2020
Published 17 August 2020 Volume 2020:14 Pages 2353—2359
DOI https://doi.org/10.2147/OPTH.S254544
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Nadyr A Damasceno,1,2 Eduardo F Damasceno,1,3 Nicolas A Yannuzzi,1 Ashley M Crane,1 Nidhi Relhan,1 William E Smiddy,1 Harry W Flynn Jr1
1Department of Ophthalmology, Bascom Palmer Eye Institute, University of Miami Miller School of Medicine, Miami, FL, USA; 2Department of Ophthalmology, Hospital Naval Marcilio Dias, Rio de Janeiro, RJ, Brazil; 3Department of Ophthalmology, Universidade Federal Fluminense, Niteroi, RJ, Brazil
Correspondence: Harry W Flynn Jr
Bascom Palmer Eye Institute, 900 N.W. 17th Street, Miami, FL 33136, USA
Tel +1 305-243-2020
Email [email protected]
Purpose: To report both the unoperated clinical course and the surgical outcomes of eyes with a central foveal bouquet (CB) secondary to idiopathic epiretinal membranes (iERMs).
Design: Retrospective, consecutive, and observational case series.
Methods: All patients examined between January 1, 2014, and December 31, 2019, for evaluation of epiretinal membrane with a CB lesion identified on spectral domain optical coherence tomography (SD-OCT) were included. Exclusion criteria included vitreoretinal comorbidities associated with secondary ERMs and an absence of CB lesions on SD-OCT. Patients were divided into two groups: those who were followed with observation (Group I) and those who received surgery (Group II). Each group had 3 different types of mechanical abnormalities of the CB previously described as cotton ball sign, subfoveal detachment, or acquired vitelliform lesion, without a subanalysis discrimination.
Main Outcome Measures: Best-corrected visual acuity (BCVA) at baseline and last follow-up, subjective metamorphopsia, central retinal thickness (CMT), mechanical stress lesions of the CB, and resolution or evolution of the CB lesions during the follow-up interval.
Results: Two hundred seventy-six eyes with iERM were reviewed, and 46 eyes met the inclusion criteria. Among these, 21 of 46 (46%) were observed, and 25 of 46 (54%) underwent surgery. Metamorphopsia was identified in 61.9% of patients in Group I and 81.2% of patients in Group II, at baseline. The mean BCVA was 0.19 ± 0.17 (20/30) in Group I and 0.31 ± 0.33 (20/40) in Group II at presentation. At the final exam, patients in Group I achieved a mean BCVA of 0.24 ± 0.18 (20/30), while patients in Group II obtained a mean BCVA of 0.15 ± 0.21 (20/30). Spontaneous resolution of the CB sign occurred in 5 of 21 eyes (23.8%) that were observed, whereas, after surgery, the CB sign resolved in 16 of 25 eyes (61.5%). Mean CMT was 422 ± 84.2μm in Group I and 531 ± 143.9μm in Group II, at baseline, while at the latest follow-up, the mean CMT was 400 ± 40.8μm in the cases followed with observation and 454 ± 148.7μm in the surgical cases.
Conclusion: The clinical course and surgical outcomes of CB findings in iERM are favorable in terms of visual acuity. However, those receiving surgery had an increase in visual acuity and resolution of the CB abnormality.
Keywords: foveal tractional lesions; idiopathic epiretinal membrane lesions; central foveal bouquet; clinical outcomes; surgical outcomes,
Introduction
Idiopathic epiretinal membrane (iERM) is a spontaneous fibrocellular proliferation of tissue on the surface of the internal limiting membrane (ILM) which can cause decreased visual acuity, metamorphopsia, and aniseikonia.1
Idiopathic ERMs may affect the subfoveal space less than 100 µm in diameter called the central bouquet (CB). This structure is composed of nearly 2000 photoreceptors, all cones.2,3 The CB is vulnerable to mechanical traction as in vitreomacular traction (VMT) and ERM.4,5 These changes have been observed using spectral-domain optical coherence tomography (SD-OCT) and have been given various terms including the cotton ball sign, subfoveal detachment, and acquired vitelliform lesion (AVL).4,6-8 The cotton ball sign has been identified as a thickened blurred hyperreflective structure placed between the ellipsoid zone (EZ) and the interdigitation zone.4 In relation to the subfoveal detachment, Pison et al recognized this structure as a subretinal hyperreflective area.7 Additionally, the SD-OCT allowed to localize the vitelliform material as a dome-shaped subretinal highly reflective lesion between the EZ and the retinal pigment epithelium (RPE).8–10
Previously, Govetto et al hypothesized that Muller cells enveloping the floor of the fovea centralis transmit mechanical forces to the photoreceptors.8 Other studies have reported the persistence of subfoveal tractional abnormalities despite surgical removal of tractional membranes.11 The purpose of the current study is to report the clinical and anatomic outcomes of eyes with tractional abnormalities of the central foveal bouquet (CB) due to idiopathic epiretinal membranes (iERMs).
Methods
The current study is a retrospective, case series of patients with idiopathic epiretinal membrane (iERM) who presented with central foveal bouquet (CB) at Bascom Palmer Eye institute between January 1, 2014 and December 31, 2019. Patients with iERM were searched from the medical records, and, then, were included in the present study if they presented CB lesion on the SD-OCT. This study complied with the Health Insurance Portability and Accountability Act (HIPAA) and was approved by the Institutional Review Board of the University of Miami Miller School of Medicine. The present study and data collected complied with the Declaration of Helsinki. All patients signed the written informed consent for the surgical procedure.
In the current study, patients diagnosed with iERM and tractional abnormalities of the CB were included, independently if they had undergone to surgical or clinical treatment of the iERM. The minimum follow-up period was 1 year and the maximum 2 years. The exclusion criteria included any previous intraocular surgery (excluding eyes that had received phacoemulsification and pars plana vitrectomy with membrane peel), any other macular disease, advanced glaucoma or optic neuropathy, visually significant cataract, and any cause of secondary ERM.
In this study, the indication for surgery was metamorphopsia, aniseikonia, low visual acuity, the patient’s professional needs, and the patient’s request. In this study, the BCVA ranged from 20/50 to 20/25 (Snellen chart).
The collected data comprised demographic factors such as age and gender, best-corrected visual acuity (BCVA) at initial and last clinical examination, presence of metamorphopsia as determined by Amsler Grid testing, and central macular thickness (CMT) measured using spectral domain tomography (SD-OCT). SD-OCT was performed using either Cirrus (Carl Zeiss Meditec AG, Jena, Germany) or a Heidelberg Spectralis device (Heidelberg Engineering, Heidelberg, Germany). For statistical analysis, the visual acuities were converted into logMAR.
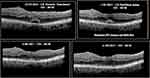
According to the images on SD-OCT, central foveal bouquets were classified into 3 categories as the Govetto et al classification: cotton ball sign, subfoveal detachment and AVL.8
The surgical procedure involved 25-gauge pars plana vitrectomy plus ERM and internal limiting membrane peeling. Neither dye nor tamponade were used.
Patients were divided into 2 groups: Group I (observed clinically) and Group II (operated with pars plana vitrectomy with membrane peeling). Each group included the 3 different types of abnormalities of the CB as represented in Figures 1–3. CB abnormality changes or resolution were evaluated at the baseline and at the last clinical examination.
Statistical analysis was performed with IBM-SPSS, 2015, Chicago, USA, version 23.0 IBM. The level of significance was established as 5% (p<0.05). Continuous values such as age, BCVA, and CMT were expressed as mean ± standard deviation (SD). Frequencies of incident variables were analyzed using a Chi-squared test to nominal variable as metamorphopsia. The Kruskal–Wallis test was employed to calculate ordinal variables as improved and worsened BCVA and CMT after the follow-up, and CB resolution, Means were compared with two-sample t test as BCVA and CMT baseline and latest data and Follow-up times.
Results
There were 276 eyes with iERM were reviewed of which 46 were found to have a CB and met the inclusion criteria. There were 46 patients of which 32 (70%) were male. The average (SD) age was 73.6 ± 7.9 years. Of the entire cohort of eyes with mechanical stress abnormalities of the CB, 33 (72%) had a cotton ball sign, 8 (17%) had subfoveal detachment, and 5 (11%) had an AVL. Of these 46 eyes, 21 (45.6%) were followed clinically (Group I) and 25 (54.4%) underwent surgery (Group II).
The average (SD) age of patients in Group I was 77.3 ± 8.2 years while in Group II was 69.8 ± 7.6 (p = 0.29). There was a statistical significance when comparing the baseline metamorphopsia between Group I and Group II (p = 0.04), Group I had 13 of 21 eyes (61.9%) with metamorphopsia and Group II had 22 of 25 eyes (88%) with metamorphopsia.
In Group I, spontaneous resolution of the CB lesions occurred in 5 eyes (23.8%) while the CB disappearance was observed in 15 eyes (60%) of Group II, demonstrating significance (p = 0.0001) as shown in Table 1.
 |
Table 1 BCVA, iERM Characteristics and Symptoms: VA and CMT Changes, CB Spontaneous Resolution and CB Disappearance After iERM Removing. Distributions of Groups II and I |
In Group I, CMT worsened in 4 eyes (19%), remained stable in 15 eyes (71.4%) and improved in 2 eyes (9.6%). In Group II, CMT worsened in 3 eyes (12%), remained stable in 11 eyes (44%) and improved in 11 eyes (44%), as illustrated in Table 1.
For Group I, the logMAR initial and final mean BCVA were 0.19 ± 0.17 (20/30) and 0.24 ± 0.18 (20/30), respectively. For Group II, the logMAR initial and final mean BCVA were 0.31 ± 0.33 (20/40) and 0.15 ± 0.21 (20/30), respectively. When comparing the baseline and last visual acuity between Group I and Group II, there was a statistical significance (p=0.04).
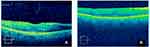
Figures 1–3 are examples of CB types found before surgery (A) and the results after surgery (B). Figure 4A and B shows aspects of iERM under OCTA analysis.
 |
Figure 4 (A) 6×6 mm SS-OCTA En face structure showing ERM and (B) scan showing bouquet. |
Discussion
To the best of our knowledge, this is the first study comparing the foveal tractional abnormalities in iERMs that were either observed or treated surgically. The current study discloses that patients with tractional abnormalities of the CB report high rates of metamorphopsia.
The tractional CB abnormalities observed in iERM have been considered as subsequent stages of the same disorders, beginning with the cotton ball sign, followed by the subfoveal detachment and later, the acquired vitelliform lesion.8 A sequential morphologic progression was identified in 7 eyes by Govetto et al.8 They also observed that cases with cotton ball sign were associated with higher BCVA, while cases with AVL had lower BCVA.
Freund and associates observed the AVL in variable macular disorders, including in 3 eyes with vitreomacular traction. The authors hypothesized that these deposits resulted from dysfunction of the RPE and loss of apposition between the photoreceptors tips and RPE, impeding outer segment phagocytosis.12 In the present study, an analysis of visual outcomes with the various types of CB lesions was not performed due to the small number of cases in each group.
Nevertheless, Optical coherence tomography angiography (OCTA) of selected eyes with CB lesions demonstrated absence of a vascular abnormality, re-emphasizing that traction on the photoreceptors is the main factor involved on the etiology of CB lesions.13 The authors observed the same in the present study (Figure 4A and B).
In terms of visual acuity over time, gains or stability were achieved in 95% of eyes followed clinically and 88% of eyes that underwent to PPV/MP by the end of the study. However, the surgical cases had larger logMAR absolute gains in BCVA but also tended to have worse initial BCVA as eyes with poor BCVA were often selected for surgery in this retrospective series. Damasceno et al reported retrospectively a propitious BCVA in patients with iERM who had been submitted to a clinical follow-up.14
Surgery enhanced the anatomical resolution of the CB sign. In the present study, 23.8% in Group I and 96% of cases in Group II had resolution of the CB sign.
The limitations of the present study include the small sample size, the limited postoperative period of clinical examination, and the study’s retrospective design.
In conclusion, the presence of tractional abnormalities in the central foveal bouquet in eyes with idiopathic epiretinal membranes portends a high rate of metamorphopsia. These eyes can be managed successfully with either observation or surgery with favorable visual outcomes with respect to measured visual acuity. The resolution of the CB was more common in this retrospective series in eyes receiving surgery than those treated with observation in a maximum follow-up of up to 2 years yet the visual implications of this finding remain uncertain.
Disclosure
Nicolas A Yannuzzi reports advisory work for Novartis and genentech, outside the submitted work. Harry W. Flynn report grants from NIH, Bethesda, Maryland and Research to Prevent Blindness, during the conduct of the study. The authors report no other potential conflicts of interest for this work.
References
1. Takabatake M, Higashide T, Udagawa S, Sugiyama K. Postoperative changes and prognostic factors of visual acuity, metamorphopsia, and aniseikonia after vitrectomy for epiretinal membrane. Retina. 2018;38:2118. doi:10.1097/IAE.0000000000001831
2. Rochon-Duvigneaud A. Recherches Sur La Fovea de la Retine Humaine et Particuliérement Sur le Bouquet des Cones Centraux. Arch Anat Microsc. 1907;9:315–342.
3. Polyak SL. The Retina. Chicago: University Press; 1941:455–489.
4. Tsunoda K, Watanabe K, Akiyama K, Usui T, Noda T. Highly reflective foveal region in optical coherence tomography in eyes with vitreomacular traction or epiretinal membrane. Ophthalmology. 2012;119:581–587. doi:10.1016/j.ophtha.2011.08.026
5. Gass JD. Muller cell cone, an overlooked part of the anatomy of the fovea centralis: hypotheses concerning its role in the pathogenesis of macular hole and foveomacular retinoschisis. Arch Ophthalmol. 1999;117(6):821–823. doi:10.1001/archopht.117.6.821
6. Dupas B, Tadayoni R, Erginay A, Massin P, Gaudric A. Subfoveal deposits secondary to idiopathic epiretinal membrane. Ophthalmology. 2009;116:1794–1798. doi:10.1016/j.ophtha.2009.06.005
7. Pison A, Dupas B, Couturier A, Rothschild P-R. Evolution of subfoveal detachments secondary to idiopathic epiretinal membranes after surgery. Ophthalmology. 2016;123:583–589. doi:10.1016/j.ophtha.2015.10.020
8. Govetto A, Bhavsar KV, Virgili G, et al. Tractional abnormalities of the central foveal bouquet in epiretinal membranes: clinical spectrum and pathophysiological perspectives. Am J Ophthalmol. 2017;184:167–180. doi:10.1016/j.ajo.2017.10.011
9. Querques G, Regenbogen M, Quijano C, Delphin N, Soubrane G, Souied EH. High-definition optical coherence tomography features in vitelliform macular dystrophy. Am J Ophthalmol. 2008;146:501–507. doi:10.1016/j.ajo.2008.05.029
10. Querques G, Regenbogen M, Soubrane G, Souied E. High-resolution spectral domain optical coherence tomography findings in multifocal vitelliform macular dystrophy. Surv Ophthalmol. 2009;54:311–316. doi:10.1016/j.survophthal.2008.12.010
11. Lombargo M, Scarinci F, Giannini D, et al. High-resolution multimodal imaging after idiopathic epiretinal membrane surgery. Retina. 2016;36:171–180. doi:10.1097/IAE.0000000000000679
12. Freund B, Laud K, Lima L, Spaide R, Zweifel S, Yannuzzi L. Acquired vitelliform lesions: correlation of clinical findings and multiple imaging analyses. Retina. 2011;31:13. doi:10.1097/IAE.0b013e3181ea48ba
13. Okawa Y, Maruko I, Kawai M, Hasegawa T, Arakawa H, Iida T. Foveal structure and vasculature in eyes with idiopathic epiretinal membrane. PLoS One. 2019;14(4):e0214881. doi:10.1371/journal.pone.0214881
14. Damasceno EF, Damasceno NA, Crane AM, et al. The clinical course of patients with idiopathic epiretinal membranes and good visual acuity managed without surgery. Clin Ophthalmol. 2019;13:
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.