Back to Journals » Substance Abuse and Rehabilitation » Volume 8
The cannabis withdrawal syndrome: current insights
Received 2 October 2016
Accepted for publication 28 December 2016
Published 27 April 2017 Volume 2017:8 Pages 9—37
DOI https://doi.org/10.2147/SAR.S109576
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Li-Tzy Wu
Udo Bonnet,1,2 Ulrich W Preuss3,4
1Department of Psychiatry, Psychotherapy and Psychosomatic Medicine, Evangelisches Krankenhaus Castrop-Rauxel, Academic Teaching Hospital of the University of Duisburg-Essen, Castrop-Rauxel, 2Department of Psychiatry and Psychotherapy, Faculty of Medicine, LVR-Hospital Essen, University of Duisburg-Essen, Essen, 3Vitos-Klinik Psychiatrie und Psychotherapie Herborn, Herborn, 4Martin Luther University Halle-Wittenberg, Halle (Saale), Germany
Abstract: The cannabis withdrawal syndrome (CWS) is a criterion of cannabis use disorders (CUDs) (Diagnostic and Statistical Manual of Mental Disorders – Fifth Edition) and cannabis dependence (International Classification of Diseases [ICD]-10). Several lines of evidence from animal and human studies indicate that cessation from long-term and regular cannabis use precipitates a specific withdrawal syndrome with mainly mood and behavioral symptoms of light to moderate intensity, which can usually be treated in an outpatient setting. Regular cannabis intake is related to a desensitization and downregulation of human brain cannabinoid 1 (CB1) receptors. This starts to reverse within the first 2 days of abstinence and the receptors return to normal functioning within 4 weeks of abstinence, which could constitute a neurobiological time frame for the duration of CWS, not taking into account cellular and synaptic long-term neuroplasticity elicited by long-term cannabis use before cessation, for example, being possibly responsible for cannabis craving. The CWS severity is dependent on the amount of cannabis used pre-cessation, gender, and heritable and several environmental factors. Therefore, naturalistic severity of CWS highly varies. Women reported a stronger CWS than men including physical symptoms, such as nausea and stomach pain. Comorbidity with mental or somatic disorders, severe CUD, and low social functioning may require an inpatient treatment (preferably qualified detox) and post-acute rehabilitation. There are promising results with gabapentin and delta-9-tetrahydrocannabinol analogs in the treatment of CWS. Mirtazapine can be beneficial to treat CWS insomnia. According to small studies, venlafaxine can worsen the CWS, whereas other antidepressants, atomoxetine, lithium, buspirone, and divalproex had no relevant effect. Certainly, further research is required with respect to the impact of the CWS treatment setting on long-term CUD prognosis and with respect to psychopharmacological or behavioral approaches, such as aerobic exercise therapy or psychoeducation, in the treatment of CWS. The up-to-date ICD-11 Beta Draft is recommended to be expanded by physical CWS symptoms, the specification of CWS intensity and duration as well as gender effects.
Keywords: marijuana, humans, neurobiology, treatment, course, detoxification, symptoms
Introduction
Cannabis is a psychotropic substance with widespread recreational use worldwide, surpassed only by nicotine and alcohol.1 Its use continues to be high in West and Central Africa, Western and Central Europe, Australasia, and North America, where recently an increase in the prevalence of past year cannabis use was recorded in the USA (12.6%).1 In Europe, prevalence rates of annual cannabis use rise in Nordic countries (7%–18%) and France (22%). They decline in Spain, UK, and Germany (currently 12%), and there is an increase in the number of treatment demands for cannabis-related problems across Europe2 and the USA.3 Although such prevalence rates are useful to indicate consumption trends, it is doubted whether these rates are relevant to reflect a health risk. Approximately 1% of European adolescents and young adults use cannabis daily or almost daily (defined as use on ≥20 days in the last month),2 a consumption pattern which is more likely to produce cannabis-related disabling disorders.4,5 The prevalence of cannabis dependence (Diagnostic and Statistical Manual of Mental Disorders – Fourth Edition – Text Revision [DSM-IV-TR]) is highest in Australasia (0.68%), followed by North America (0.60%), Western Europe (0.34%), Asia Central (0.28%), and southern Latin America (0.26%).4 In Germany, ~0.5% of the adult population have a cannabis dependence diagnosis.6 Most of the other regions of the world providing data report a prevalence of cannabis dependence of <0.2%.4 There is a significant positive correlation between the region’s economic situation and the prevalence of cannabis dependence.4 A hallmark of cannabis dependence (Diagnostic and Statistical Manual of Mental Disorders – Fourth Edition [DSM-IV] or International Classification of Diseases [ICD]-10) as well as cannabis use disorder (CUD) (Diagnostic and Statistical Manual of Mental Disorders – Fifth Edition [DSM-5]) is the cannabis withdrawal syndrome (CWS) that characteristically occurs after quitting a regular cannabis use abruptly.
Although there was early evidence from animal experiments7 and despite observations in humans in every decade,8,9 CWS entity was doubted before the 1990s, when a new cannabis wave started to roll in worldwide, particularly in affluent regions.4 This was related with a mounting number of patients seeking treatment due to various cannabis-related disorders, including cognitive deficits, psychosis, and dependence.4,5 Considering these populations and also nontreatment-seeking cannabis-dependent individuals, larger retrospective clinical trials10,11 demonstrated that discontinuation of regular cannabis use is frequently followed by waxing and waning behavioral, mood and physical symptoms such weakness, sweating, restlessness, dysphoria, sleeping problems, anxiety, and craving, which are subsequently positively associated with relapse to cannabis use.11–19 However, other studies did not find this association.20 CWS was further validated by epidemiological,21,22 retrospective,11,19,23 and prospective outpatient12,13,20,24–26 and inpatient laboratory studies27–30 (Table 1). Based on this research, diagnostic criteria of CWS were newly included in DSM-5 (Table 2).31 In ICD-10, CWS is still vaguely defined32 and awaits due definition in ICD-11.33 More recent clinical inpatient detoxification studies arranging controlled abstinence conditions confirmed the entity of CWS.34–36 The CWS was also verified in youths and adolescents (aged 13–19 years), who sought treatment for their disabilitating cannabis dependence.18,37–40
  | Table 2 Marijuana Withdrawal Checklist (MWC) Notes: A total MWC score is obtained by summing the severity ratings, mild = 1, moderate = 2, severe = 3 points; *symptoms listed in DSM-5. There is no valid definition available for assigning a cannabis withdrawal syndrome to be mild, moderate, or severe. An MWC score of 10 points was found to be comparable with 5 points on the Clinical Global Impression – Severity scale (CGI-S), which is a 7-point scale. Four or more withdrawal symptoms were shown to predict the severity of cannabis-related problems at 1-year follow-up among treated adolescents (N=214, 92% retention). Data from previous studies.18,24,26,31,36,37,80 Abbreviation: DSM-5, Diagnostic and Statistical Manual of Mental Disorders – Fifth Edition. |
There is a consistent evidence that CWS occurs in ~90% of the patients being diagnosed with cannabis dependence according to ICD-10 or DSM-IV12,13,38,41,42 (Table 1). Among them, most often, male adolescents and young adults demonstrated a significant loss of quality of life during their cannabis dependence as measured by disability-adjusted life years in the Global Burden of Disease 2010 Study (cf Figures 2 and 3 in http://journals.plos.org/plosone/article?id=info:doi/10.1371/journal.pone.0076635, accessed November 25, 2016).4
Recent studies revealed that 35%–75% patients seeking outpatient cannabis detoxification developed a CWS post-cessation, which usually seemed to be mild to moderate in severity.11–13,15,16,19 However, most of the cannabis dependents developed a CWS of greater severity.36 Adult cannabis dependents were shown to develop a severe CWS likelier than adolescent frequent users.24,37 A prolonged and heavier cannabis use predicts a stronger CWS.12,13,19 It was confirmed again more recently that the occurrence of CWS is a highly specific indicator of a cannabis dependence, particularly in adolescents and young adults.42
This review intends to provide a synthesis of current evidence on the biology and clinical characteristics of the human CWS and its treatment. In addition, it includes information on the role of CWS in the course of CUD31 or cannabis dependence.22,43
Materials and methods
This study is a review of the current literature on human CWS. The search for articles was performed on the PubMed44 (Medline) and Scopus,45 using the a combination of the search terms “cannabis withdrawal,” “humans,” “epidemiology,” “disability,” “clinical studies,” “clinical trials,” “case reports,” “cannabis use disorder,” “cannabis dependence,” “treatment,” “psychotherapy,” “psychosocial,” “exercise,” “occupational therapy,” “pharmacotherapy,” and “potency”. In addition, an active search for related literature was carried out in the reference lists of the selected publications. In total, 2,440 documents were screened, and mainly those studies providing information on human CWS and those published in English or German (N=101) were considered. Articles published up to November 25, 2016, were included.
Human biological background
The cannabis plant contains >420 chemical compounds of which 61 being cannabinoids themselves being defined to bind to cannabinoid 1 and 2 (CB1, CB2) receptors.46 Regular cannabis use is associated with neuroanatomic abnormalities within brain regions with a high density of CB1 receptors, particularly the hippocampus and prefrontal cortex.47,48 It is assumed that, the main psychoactive ingredient of cannabis, the partial CB1 receptor agonist delta-9-tetrahydrocannabinol (THC) is involved in the etiology of this damage,47 which certainly awaits further study. For instance, a contribution of receptor-independent mechanisms of cannabinoids49,50 as well as distress due to psychiatric CUD or CWS cannot as yet be excluded. A crucial role of THC in the genesis of CWS in humans is demonstrated by 1) pharmacokinetic studies showing a hysteresis effect between the decrease in plasma THC and onset of CWS,51,52 2) an abstinence syndrome following oral THC12,13 and THC analogs,53 3) alleviation of CWS by oral THC and THC analogs,29,54,55 and 4) the occurrence of CWS-like symptoms after quitting recreational intake of synthetic cannabinoid (SC) receptor agonists, often being full CB1 receptor agonists, differing from THC being a partial agonist.56,57 The withdrawal syndrome of SCs binding closer to CB1 receptors than THC seemed to be stronger than CWS and obviously showed characteristics unknown to CWS, such as seizures.58 Otherwise, single cases of patients with diagnosed epilepsy who quit regular cannabis use are reported to exacerbate,59 which is attributed to an anticonvulsive effect of cannabis.46 The psychoactive potency of bred cannabis products sold for recreational use has been increasing in many markets over the past decade,1,2 which could lead to a stronger withdrawal syndrome than usually known for cannabis. Intriguingly, there is one case report regarding improvement of CWS following the administration of cannabidiol,60 another constituent of cannabis, shown to reverse some adverse effects of THC in the laboratory.61 The cardiovascular functioning seemed to be scarcely altered during CWS.62 Although the endocannabinoid system is involved in the regulation of most of the other peripheral organ systems, the immune system and the gut, too, we are unaware of any such study on the contribution of these organs to human CWS. Notably, applying a CB1 receptor antagonist (rimonabant) to cannabis-dependent patients substituted with THC analogs did not precipitate a relevant CWS.63 This may be due to the low doses of rimonabant applied (20 and 40 mg) or the CWS-generating mechanisms that are at least partly independent upon CB1 receptors.49,50 Cannabis users with opioid dependence are less likely to experience CWS,64 which may indicate the contribution of the endogenous opioid system. In a laboratory study, the µ-opioid receptor antagonist naltrexone was recently shown to reduce self-administration of active cannabis and its related subjective positive effects on heavy cannabis users.65 The authors are unaware of any study having directly examined the effect of naltrexone on the CWS under naturalistic conditions.
Abstinence-induced craving is associated with reduced amygdala volumes in frequent adolescent cannabis users, which was also found in adult alcohol and cocaine users.66 Thus, the specificity of this finding for CWS is doubted and may represent a more general precursor of substance abuse itself;66 that is, early stress in life.67,68 With respect to the three “a”s of CWS (anger, aggression, and anxiety) (Table 1), the threat-related amygdala reactivity was shown to be inversely related to the level of cannabis use in adolescents with comorbid cannabis dependence and major depression.69 This finding may reflect the neurobiological basis of these transient, mostly short-lasting CWS symptoms, thus possibly being even rebound “amygdala-related” symptoms after quitting regular cannabis use. Nevertheless, the CWS symptoms could persist even longer in genetically or epigenetically more susceptible individuals upon withdrawal.
Regular cannabis intake is related to a desensitization and downregulation of human cortical and subcortical CB1 receptors. This starts to reverse within the first 2 days of abstinence and the receptors return to normal functioning after ~4 weeks of abstinence,70 which could constitute a neurobiological time frame for the duration of CWS, not taking into account cellular and synaptic long-term neuroplasticity elicited by long-term cannabis use before cessation, for example, being possibly responsible for craving. In support, cannabis dependents were recently shown to have a robust negative correlation between CB1 receptor availability in almost all brain regions and their withdrawal symptoms after 2 days of cannabis abstinence which in turn resolved in the next 28 days of abstinence.71
If compared with nonusers, long-term cannabis users were demonstrated to have greater brain activity during cannabis cues relative to natural reward cues (ie, fruit itself being superior to neutral cues) in the orbitofrontal cortex, striatum, anterior cingulate gyrus, and ventral tegmental area.72 The users had positive correlations between neural response to cannabis cues in the fronto-striatal-temporal regions and subjective craving, cannabis-related problems, serum levels of THC metabolites, and the intensity of CWS. All of which were not found in non-cannabis users,72 suggesting a sensitization and specificity of the brain response to cannabis cues in long-term cannabis users.72
In the San Francisco Family Study, some symptoms of CWS, craving and cannabis-related paranoia were found to be heritable,73 which could have been confounded by the heritability of age at first-ever use, for instance. It was suggested that genetic factors determine whether an individual may try or use cannabis; however, environmental factors are more crucial in determining whether a person develops dependence or not.73 Recent findings provide evidence that the use of nicotine, alcohol, or cannabis shares genetic and environmental pathways on the way to develop a substance use disorder.74 Regular intake of alcohol, nicotine, cannabis, or other drugs of abuse alters the stress response sustainably75 and, thereby, may precipitate a substance use disorder.
Characteristics of CWS
Considering the cannabis research of the last 20 years,12,13,16,18–20,31 there was no doubt that cessation of heavy or prolonged cannabis use is most likely followed by typical symptoms, such as
irritability
nervousness/anxiety
sleep difficulty
decreased appetite or weight loss
depressed mood
one of the following physical symptoms such as abdominal pain, shakiness/tremors, sweating, fever, chills, or headache.
According to DSM-5,31 CWS (292.0) is diagnosed if three or more of these symptoms (1–6) develop within ~1 week after quitting cannabis use abruptly.31 Withdrawal severity and duration can vary widely between individuals and fluctuate depending on the amount of prior cannabis use, context of cessation (eg, outpatient vs inpatient, voluntary vs involuntary), personality traits, psychiatric and somatic comorbidity, current life stressors, previous experiences, expectations, support, and severity of dependence.12,13 Women seeking treatment for CUD were shown to generate more frequent and more severe withdrawal symptoms than men after quitting their frequent cannabis use.36,76,77 However, older studies did not reveal this gender effect (Table 1).
Additional heavy tobacco use was reported to be associated with stronger irritability during the CWS of adolescents.40 Black adolescents were shown to have lower withdrawal complaints and experience less severe depressed mood, sleep difficulty, and nervousness/anxiety than non-Black adolescents.40 In youths with conduct disorder, this disorder antedated cannabis use.38
Currently, psychometrically validated cannabis withdrawal scales are unavailable. Several versions to measure CWS11–13,16,18,24,78 were developed, some of which compared with each user by Gorelick et al.19 All these versions were based on the Marijuana Withdrawal Checklist (MWC) of Budney et al.24 The MWC was originally designed with 22 items that assessed mood, behavioral, and physical symptoms and was revised to a 15-item version comprising these items that had been most frequently endorsed during cannabis withdrawal12,13,26,37 (Table 2). Later, this version builds the construct of the DSM-5 definition of CWS31 (Table 2), which, however, does not consider cannabis craving and nausea.31
Regarding the course of the overall CWS, there were two different types described in the available literature (Figure 1 and Table 1). One peaked between the second and sixth abstinence day (type A)11,15,16,19,20,23,26,27,35,36,56,79 and the other decreased continuously following cannabis cessation (type B).28,34,39 It is assumed that type-A CWS includes more intoxication symptoms which vanished during the first few days post-cessation, thereby unmasking the “pure” CWS.36 A negative correlation with serum levels of THC at admission, which would support this assumption, was found in type A.35,36 Type-B CWS was not investigated to this subject. Alternative explanations are that the contribution of single items (cf Figures 2 and 3) differed between types A and B or more patients without a measurable CWS were included in the group of patients producing a type-B course.
  | Figure 1 Courses of overall CWS post-cessation. The CWS usually lasts up to 3 weeks and its average peak severity (burden) is comparable to that of a moderate depression or alcohol withdrawal syndrome or in outpatient settings, similar to that of a tobacco withdrawal syndrome. Data from previous studies.14,36,79 Abbreviation: CWS, cannabis withdrawal syndrome. |
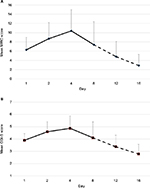  | Figure 2 Mean and standard deviation of the (A) CWS checklist (MWC score according to previous studies24,26,37) and (B) the Clinical Global Impression Scale (CGI-S Score80) during the course of the study. Reduced sample sizes on day 12 (n=35) and day 16 (n=28) due to regular dismissals and missed assessments are indicated by dashed lines. The effect size according to Cohen (Cohen’s d) was 1.1 for the CWS (day 1 to day 16), Cohen’s d ≥0.8 is defined to reflect a strong effect.130 Vertical imaginary Y-axis: severity scores. Horizontal imaginary X-axis: time course. Note: Reproduced from Drug Alcohol Depend, 143, Bonnet U, Specka M, Stratmann U, Ochwadt R, Scherbaum N, Abstinence phenomena of chronic cannabis-addicts prospectively monitored during controlled inpatient detoxification: cannabis withdrawal syndrome and its correlation with delta-9-tetrahydrocannabinol and -metabolites in serum, 189–197. Copyright (2014), with permission from Elsevier.36 Abbreviations: CWS, cannabis withdrawal syndrome; MWC, Marijuana Withdrawal Checklist. |
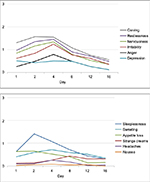  | Figure 3 Mean rating of single symptoms of the MWC (MWC score according to previous studies24,26,37); 4-point scale (0 = none, 1 = mild, 2 = moderate, 3 = heavy). Note the delayed occurrence of strange dreams.25 Vertical imaginary Y-axis: severity scores. Horizontal imaginary X-axis: time course. Note: Reproduced from Drug Alcohol Depend, 143, Bonnet U, Specka M, Stratmann U, Ochwadt R, Scherbaum N, Abstinence phenomena of chronic cannabis-addicts prospectively monitored during controlled inpatient detoxification: cannabis withdrawal syndrome and its correlation with delta-9-tetrahydrocannabinol and -metabolites in serum, 189–197. Copyright (2014), with permission from Elsevier.36 |
In the following, the course of a CWS (Figures 2 and 3) is presented, which was recorded during a controlled inpatient detoxification treatment of a sample consisting of long-term cannabis users (N=39, 38 Caucasians, 8 females, median age: 27 years, median daily cannabis use: 2.5 g, median duration of daily cannabis use: 36 months).36 Their cannabis consumption was in the upper range if compared with other clinical studies on CWS.11,12,16,20,34 All patients of this sample developed a considerable CWS.36 Although this study was conducted in a large detoxification ward of a university hospital residing in a metropolitan area (Ruhr Area, Germany), it lasted 5 years (2006–2011) to find 39 appropriate patients seeking treatment due to a current sole cannabis dependence without a competing additional substance use (except for tobacco) or active comorbidity to measure a CWS as pure as possible.36 This study aimed to apply both MWC24 and the Clinical Global Impression Scale (CGI-S)80 to this sample to make the severity of CWS comparable to the severity of other psychiatric disorders.36 The course of the single items is shown in Figure 3. Cronbach’s α coefficients were 0.67, 0.78, and 0.73 on days 4, 8, and 12, respectively.36 The MWC applied to this sample achieved α coefficients comparable to that of the CWS criteria proposed for DSM-5 (0.75)19 and to that of the original MWC of Budney et al (0.77).12,24
CWS severity in comparison with other psychiatric conditions
As outlined earlier, the severity of CWS is positively related to the cumulative amount and potency of cannabis used before cessation,12,13,19 gender,36,76,77 and several environmental12,13,73 as well as heritable73 factors. Therefore, its naturalistic severity varies a lot. There are some evidences that the discomfort due to CWS is similar to that found during tobacco withdrawal14,79 or a moderate alcohol withdrawal syndrome.36 Inpatients detoxifying from heavy cannabis use were rated to be “moderately ill” at the peak of CWS according to CGI-S.36 For a first orientation, inpatients suffering from acute schizophrenic episodes and patients with acute depressive episodes in outpatient settings have been rated in the majority to be “severely ill” and “moderately ill,” respectively.36 Strong CWS can mimic eating disorders associated with gastrointestinal symptoms, food avoidance, and weight loss of adolescents.81
The role of nausea in CWS
There is one case report of severe nausea being associated with CWS.82 In the last years, increasing cases with a cannabis-hyperemesis syndrome were noticed, which characteristically occurred in frequent and long-term cannabis users and vanished in their next 5–20 abstinent days.83-–85 In order to differentiate this condition from CWS, we studied the course of the item “nausea” in the “cannabis burdened” sample mentioned earlier and found no correlation (r=–0.14 to 0.19) with the other items of MWC, whose internal consistency did not change, if “nausea” was excluded from the scale.36 Thus, we found no evidence of nausea being a characteristic element in the orchestra of the CWS, which confirms the previous results of others.11,12,19
Nevertheless, nausea seems to be a less common cannabis withdrawal symptom than chills, shaking, sweating, depressed mood, and stomach pain.13 This is supported by the observation that nausea can occur more pronounced in the female CWS than in the male CWS.76,77
A retrospective chart review found preliminary evidence that “nausea and vomiting” might emerge more frequently in the withdrawal syndrome of SC agonists (Table 1)56 often being full agonists at the CB1 receptor, other than the partial agonist THC.57 Is this a clue that nausea breaks through when very potent agonists are removed from CB1 receptors? On the other hand, “severe nausea and vomiting” were key symptoms of the intoxication syndrome following the intravenous application of crude marijuana extracts86 and are typical signs for the overdosing of smoking or swallowing cannabis preparations,46,86 including the first-ever intake experience. However, low to moderate amounts of cannabis preparations or THC analogs have well-known antiemetic properties.46
Treatment of CWS
Cannabis detoxification treatment is usually performed in outpatient settings. However, in the case of a moderate or severe dependence syndrome, low psychosocial functioning or moderate or severe psychiatric comorbidity, an inpatient treatment is required. In Germany, inpatient cannabis detoxification is ideally performed in specialized wards following a “qualified detoxification” protocol. This includes supportive psychosocial interventions, psychoeducation, non-pharmacological symptom management, occupational and exercise therapy, professional care, as well as medical and psychiatric diagnostics and therapy of comorbid conditions. The treatment duration is related to the severity of the comorbidity or the CUD. An inpatient detoxification treatment on grounds of the diagnosis of “cannabis dependence” alone is mostly not accepted by the German health care providers, and they prima vista doubted the existence of a treatment-relevant CWS. This point of view may have a historical background, because many physicians consider cannabis to be a “soft drug,” as probably 20 years ago it contained lower THC contents.87–90 This view may change with a clear definition of CWS in current diagnostic classification systems, such as DSM-5.31 As a rule of thumb, an “acute” inpatient detoxification treatment lasts between a few days and up to 3 weeks. In case of a too high psychiatric comorbidity and too low psychosocial functioning for an outpatient treatment, the patients could be transferred into specialized inpatient rehabilitation wards. Because this post-acute treatment approach is paid by the German Person Fund (DRV), a substantial formal request is required. The rehabilitation treatment normally lasts for several weeks and is a special feature of the German health care system. In Germany, most of the cannabis patients entering outpatient (28,000 individuals in 2014) and inpatient (3,367 individuals in 2014) rehab programs show additional problems with the co-use of alcohol (7%–14%), opioids (30%–55%), cocaine (45%–60%), stimulants (45%–70%), and pathological gambling (6%–18%).91 This pattern of comorbidity is common in other high-income countries.1
The effects of behavioral approaches on the mitigation of CWS were not intentionally studied , even though a beneficial action of aerobic exercise therapy can be assumed.92
Currently, there are no approved medications for the treatment of CUD. Nevertheless, various pharmaceuticals have been studied in small (N<80) controlled, mostly outpatient or laboratory pilot trials: lithium, antidepressants (bupropion, nefazodone, venlafaxine, fluoxetine, escitalopram, and mirtazapine), anticonvulsants (divalproex and gabapentin), norepinephrine reuptake inhibitor (atomoxetine), glutamate modulator and mucolytic agent (N-acetylcysteine), muscle relaxants (baclofen), anxiolytic (buspirone), antipsychotics (quetiapine), and CB receptor agonists (dronabinol and nabiximols).55,93 The antidepressants, atomoxetine, lithium, buspirone, and divalproex had no relevant effect on the CWS or had worsened it.55,93 For instance, venlafaxine was shown to aggravate CWS and, thus, was accused to uphold cannabis smoking.94 Quetiapine (200 mg/day) improved appetite and sleep quality during the CWS but worsened marijuana craving and drove self-administration of marijuana.95 A more recent open pilot study reported a decrease of cannabis use within 8 weeks of quetiapine treatment (25–600 mg).96
Putatively beneficial agents
There is evidence for an improvement of CWS with gabapentin (1200 mg/day).97 The efficacy of the THC analogs dronabinol and nabiximols (plus cannabidiol) in reducing CWS is demonstrated in three small but well-controlled outpatient studies.98–101 An improvement of the dependence syndrome or craving was not found.98–101 Although innovative compounds, high costs of dronabinol or nabiximols may limit not only the use but also the abuse of these drugs. A significant effect of the THC substitution on the severity of cannabis dependence, craving, or cannabis-related problems was not found yet.98–101 Nabiximols is a drug containing two of the main active cannabinoids, namely, THC and cannabidiol, and has been approved in some countries for the treatment of spasticity of multiple sclerosis (MS).99–101 It is an oral spray formulation, and each puff of 100 μL contains 2.7 mg of THC and 2.5 mg of cannabidiol. A treatment with six puffs over the day revealed >10 times smaller blood THC concentrations than the blood concentrations known to produce psychotropic effects.102 For the nabiximols regimes in the treatment of CWS, cannabis users had been instructed to take eight sprays qid99,100 or a maximum of four sprays every hour (up to 40 sprays/day).101 Oral dronabinol had been administered in 20 mg doses three times a day.98 The effectiveness of N-acetylcysteine (1200 mg/day) on CWS was not assessed directly, but this agent was shown to reduce relapse markers in the urine alongside a large (N=116) well-controlled study.103 A recent laboratory study demonstrated the efficacy of the THC analog nabilon.104
Because sleep difficulty is the withdrawal symptom that is assumed to be most associated with relapse to cannabis use,105 a few sleeping medications were tested in the CWS treatment with first promising results for mirtazapine30 and zolpidem106,107 during the first days of abstinence, necessarily taking care for zolpidem’s potential misuse. Nevertheless, both the drugs had no effects on CWS in general or relapse prevention.30,107
The withdrawal syndrome of SC receptor agonists56 awaits further characterization and may respond to benzodiazepines and quetiapine.108
Impact on CUD or dependence
The CWS is part of a CUD (DSM-5)31 and dependence-syndrome (ID-10, DSM-IV-R)32,43 being characterized by frequent, heavy, or prolonged cannabis use. The importance of the treatment of CWS on the maintenance of cannabis use or substance use trajectories over time is unclear and awaits further study. From previous literature, there is small evidence for both 1) CWS treatment initiated abstinence or dose reduction12,13 and 2) CWS treatment does not influence cannabis use in the following.18,20,42,109 Frequent cannabis users had reported that withdrawal symptoms negatively influence their desire and ability to quit.12,13,18,105 Two actual studies on adolescents and young adults found no association of CWS with abstinence rates being monitored up to 3 months20 and 1 year posttreatment,109 which confirmed the previous finding of Arendt et al.20 Furthermore, patients with CWS relapsed sooner than those without CWS.20 Patients recognizing a problem with CWS were associated with better abstinence rates than patients not recognizing a problem with CWS42 pointing to a potential value of psychoeducation, an approach to be further studied in the management of CUD.
Abstaining from cannabis was reported to be followed by an increase of alcohol and tobacco use, which decreased again after continuation of cannabis use.110 CWS in people with schizophrenia is associated with behavioral change, including relapse with cannabis and increased tobacco use.111 “Religious support” and “prayer” were self-identified by cannabis users to be the most helpful quitting strategies and both were associated with higher 1-month and 1-year abstinence rates in these population.111 Furthermore, the symptom severity of patients with posttraumatic stress disorder was positively associated with the use of cannabis (probably taken as a “self-medication”), cannabis use problems, and severity of CWS.112
Studies that compared the effectiveness of outpatient versus inpatient treatments with respect to the severity and prognosis of CUD, especially their differential efficacy on long-term relapse prevention, dose reduction, or psychosocial functioning, are missing. At the end of a 16-day lasting inpatient detoxification treatment (qualified detox) of heavy cannabis users, the following effect sizes were found (Cohen’s d): 1.1 (CWS), 1.4 (cognition), psychiatric symptoms (0.8–0.9).113 The bothersome global distress of this sample had improved significantly during the qualified detox (Figure 4).
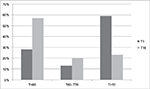  | Figure 4 Significant improvement (p<0.001) of the subjective global distress of adult heavy cannabis users during inpatient qualified detoxification as measured by the Symptom Checklist 90, revised version (SCL-90-R).129 Y-axis: percent of the sample (N=35); X-axis: global distress according to T-values: T<60: normal global distress; T>70: severe global distress;129 T1 = admission day and T16 = last day (day 16) of the controlled inpatient qualified detoxification treatment.113 Note: Reproduced from Dtsch Med Wochenschr, 141(2), Bonnet U, Specka M, Scherbaum N, Häufiger Konsum von nichtmedizinischem Cannabis, 126–131. Copyright (2016), with permission from Georg Thieme Verlag.113 |
At present, the effectiveness of different cannabis detoxification treatments on the course of the CUD has not been studied in depth. Outpatient treatment programs improved the psychosocial functioning and dropped the cannabis use for a while.12,13 Currently, only a few long-term follow-ups are available, so far showing no sustaining improvement of CUD subsequent to outpatient detoxification attempts.18,20,42,109
Discussion
With the definition of the DSM-5 criteria,31 the CWS comes of age. Genetic influences on cannabis withdrawal were described to be the same as those affecting cannabis abuse and dependence.114 Beyond that, the existence of the CWS has solid neurobiological underpinnings since it was found that the availability of brain CB1 receptor in cannabis dependents was inversely associated with the occurrence of CWS .70,71 After 4 weeks of abstinence, the anomalies of CB1 receptors binding had been normalized in cannabis dependents, thus giving a rough time frame of the duration of CWS,70,71 which apt to the clinical observations that have been obtained in the past 20 years (Table 1). Two different courses of CWS might result from the different contribution of cannabis residual symptoms assumed to be initially more prominent in the type A than in type B CWS.36 Looking at a key symptom of cannabis use, such as “increase in appetite,”5,7 this was indeed reported more often in the first days of the type A CWS (Table 1).19,20,35
Mood and behavioral symptoms, namely, insomnia, dysphoria, and anxiety, are the key symptoms of the CWS (Tables 1 and 2). Similar symptoms occurred in the obesity treatment with the CB-1 receptor antagonist rimonabant (also known as SR14171) and were the reason why rimonabant was withdrawn from the market in 2008.115 Possibly, a “sustained CWS” had been precipitated when the effects of the physiological tone of endogenous endocannabinoids on brain and peripheral CB1 receptors were impeded by receptor antagonists, even in non-addicted but susceptible individuals. In support, the neurocircuitries involved in the regulation of stress, anxiety, and mood (such as the serotonergic, noradrenergic, and dopaminergic systems) were demonstrated to be sensitive to CB-1 receptor antagonists.115
Influence of alcohol and tobacco
Regular alcohol drinking might influence the clinical expression of the CWS, and this is not through the overlapping alcohol withdrawal symptoms. Continuous exposure to ethanol, in either cell culture or rodent models, led to an increase in endocannabinoid levels that resulted in downregulation of the CB1 receptor and uncoupling of this receptor from downstream G protein signaling pathways.116 A similar downregulation of CB1 receptors was found in multiple brain regions of chronic drinkers.116 Alcoholic drinks were reported to be co-used by 33%–46% of regular cannabis users. The rates of co-use for cocaine, stimulants, and hallucinogens were 37%–43%, 30%–52%, and 36%–42%, respectively1,2,116 – all putatively being able to influence the course and intensity of the CWS. Approximately 90% of cannabis users are also tobacco smokers, possibly reflecting the common route of administration, and even synergistic and compensatory actions of cannabis and tobacco as well as genetic and epigenetic factors assumed to mediate addiction vulnerability.117,118 More specifically, smoking tobacco use was shown to increase the number of cannabis dependence symptoms119 and precipitated cannabis relapse.120 Vice versa, cannabis use decreased the likelihood of abstaining from tobacco.117,118 There is a preliminary evidence that simultaneous tobacco and cannabis abstinence predicts better psychosocial treatment outcomes.117,118 There is still a paucity of clinical studies on this important subject, although alcohol, tobacco, and cannabis were consistently identified to be the substances with earliest onset of use, the highest prevalence of lifetime use, and the highest prevalence of lifetime disorder.1–6,74
Choice of treatment setting
In comparison with outpatient programs, inpatient detoxifications can provide strict abstinence conditions and, thus, can be used to better differentiate CWS from comorbidity, but are much more expensive and usually not the first choice of patients seeking treatment due to CUD. However, 1) the inability to initiate cannabis abstinence due to bothersome CWS, 2) the continuous co-use of other harmful drugs of dependence, or 3) the coexistence of other disabling psychiatric or somatic complaints give reasons for the medical necessity of an inpatient detoxification program, the duration of which depends on the intensity of the withdrawal symptoms and concomitant complaints.5,113 At this juncture, the duration of an inpatient detoxification program of heavy cannabis users is recommended to be not less than 14 days, ideally 21 days, taken into account that their pure CWS itself usually lasted up to 14–21 days (Table 1),12,13,36,113 and the diagnosing of potentially underlying comorbidity is more sensitive from then on. It remains a challenge of future in-depth studies to compare the impact of outpatient and inpatient treatment programs on the long-term course and disability of substance use disorders, which applies to CUD, too.
Influence of high potency cannabis preparations, gender, and so on
Similar to the cannabis addiction syndrome itself,121 one of its hallmark, the CWS, is based upon complex interactions between drug-induced neurobiological changes, environmental factors, genetic and epigenetic factors, comorbidity, personality traits, gender influences, and stress responsivity, all of which contributing to the high inter- and intrapersonal variations in the composition, annoyance, and duration of the CWS (Table 1).12,13,114,121
In addition to an increasing awareness of the existence of the CWS, its increasing emergence in the last 20 years might result from the increasing psychotropic potency of the used marijuana originating from the breeding of strains with high THC (10%–18.5%) and low cannabidiol concentrations (<0.15%) being found especially in high-income countries.1,88–90,122 In the Netherlands, the recent cannabidiol content of imported resin was ~7%.90 According to animal experiments, cannabidiol can counterbalance some adverse effects of cannabis,61 and in patient populations, there is mounting evidence of anticonvulsive and antipsychotic properties of cannabidiol.123 Since the early 1950s, it is known that the chemical composition of the resin itself varies with cannabidiol activities between 0% and 50% depending on the provenance of the drug.87 Whether the users of more potent cannabis strains adjust their intake according to the potency is still unclear.88 However, there is first evidence that the occurrence of first-episode psychosis as well as the intensity of the CUD increased alongside the use of high potency cannabis preparations.124,125 This throws an extremely critical light on emerging modern cannabis ingestion methods (“dabbing” or “cannavaping” of cannabis concentrates with 20%–80% THC) used by individuals seeking a more rapid and even bigger than being possible with smoking flowers that THC contents are usually in the range of 2% and 6%.126,127 Marijuana users who had turned to “dabbing” reported higher tolerance and withdrawal experiences.126
The CWS could have an measurement bias regarding a recent finding that it was endorsed more likely by the US than by Dutch cannabis users, which applies to other CUD criteria, such as tolerance, and gender effects on CWS, too.77 In this context, recent studies revealed a consistent gender impact on CWS, because women experienced a stronger CWS (Table 1)36,76 and were shown to have a greater susceptibility to developing CUD than men.121,128 Women were also found to be more sensitive to the cannabis than men.121,128 Remarkably, women reported physical CWS complaints more likely, such as nausea and stomach pain (Table 1).19,76
CWS in the ICD-11 Beta Draft
In 2018, the 11th revision of the ICD-11 is planned to be published. The so-called Beta-Draft of the chapter about “Mental and Behavioral Disorders” is already available online at http://apps.who.int/classifications/icd11/browse/f/en#/http%3a%2f%2fid.who.int%2ficd%2fentity%2f637576511 (accessed November 25, 2016).33 The current version of this ICD-11 Beta Draft33 lists the usual mood and behavioral CWS symptoms according to DSM-5"> (accessed November 25, 2016).33 The current version of this ICD-11 Beta Draft33 lists the usual mood and behavioral CWS symptoms according to DSM-5xs31 but does not consider physical CWS symptoms.33 We recommend to include at least “nausea” and “stomach pain” into the final version because these symptoms were recently found to be more prominent in the female CWS,76,77,121,128 and yet, it seems likely that the increasing use of high potency cannabis preparations are associated with more physical CWS symptoms. It is also recommended to include a note on the high intra- and interpersonal variability of the CWS intensity and the observation that – if a CWS occurs – it is extra distressing between the first and the third week after quitting a frequent, heavy, or prolonged cannabis use (Table 1).12,13,36 Heavy users were shown to experience a CWS whose average severity is comparable to the burden of a moderate depression or moderate alcohol withdrawal syndrome.36 In outpatient settings, the average discomfort of CWS was similar to that of tobacco withdrawal.14,79
Certainly, it awaits future study whether the inhalation of very potent cannabis concentrates126,127 is indeed associated with a further decrease of psychosocial functioning, higher comorbidity, and a stronger CUD and CWS – eventually with more physical features (eg, hyperalgesia, nausea, sweating, tremor, flu-like symptoms)31 than occurring after the cessation of a heavy or prolonged use of traditional non-concentrated cannabis preparations.
Conclusion
The CWS is a criterion of CUDs (DSM-5) and cannabis dependence (DSM-IV-R, ICD-10). Several lines of evidence from human studies indicate that cessation from long-term and regular cannabis use precipitates a specific withdrawal syndrome with mainly mood and behavioral symptoms of light to moderate intensity, which can usually be treated in an outpatient setting. However, comorbidity with mental or somatic disorders, severe CUD, and low social functioning may require an inpatient treatment (preferably a qualified detox) and post-acute rehabilitation or long-term outpatient care. There are promising results with gabapentin and THC analogs in the treatment of CWS. Mirtazapine could improve insomnia, and venlafaxine was found to worsen the CWS. Certainly, further research is required with respect to the impact of the CWS treatment setting on long-term CUD prognosis and with respect to psychopharmacological or behavioral approaches, such as aerobic exercise therapy or psychoeducation, in the CWS treatment. The preliminary up-to-date content for the ICD-1133 (intended to be finally published in 2018) is recommended to be expanded by physical CWS-symptoms, the specification of CWS severity and duration as well as gender effects.
Disclosure
The authors report no conflicts of interest in this work.
References
UNDOC (United Nations Office on Drugs and Crime). World Drug Report 2015. United Nations publication, Sales No. E.15.XI.6. Available from: https://www.unodc.org/documents/wdr2015/World_Drug_Report_2015.pdf. Accessed September 28, 2016. | ||
EMCDDA (European Monitoring Centre for Drugs and Drug Addiction). European Drug Report 2015. Trends and Developments. Luxembourg Publication Office of the European Union 2015. Available from: http://www.emcdda.europa.eu/attachements.cfm/att_239505_EN_TDAT15001ENN.pdf. Accessed September 28, 2016. | ||
Hasin DS, Saha TD, Kerridge BT, et al. Prevalence of Marijuana Use Disorders in the United States Between 2001-2002 and 2012-2013. Prevalence of Marijuana Use Disorders in the United States Between 2001-2002 and 2012-2013. JAMA Psychiatry. 2015;72(12):1235–1242. | ||
Degenhardt L, Ferrari AJ, Calabria B, et al. The global epidemiology and contribution of cannabis use and dependence to the global burden of disease: results from the GBD 2010 study. PLoS One. 2013;8(10):e76635. | ||
Hoch E, Bonnet U, Thomasius R, Ganzer F, Havemann-Reinecke U, Preuss UW. Risks associated with the non-medicinal use of cannabis. Dtsch Arztebl Int. 2015;112(16):271–278. | ||
Pabst A, Kraus L, Gomes de Matos E, Piontek D. Substanzkonsum und substanzbezogene Störungen in Deutschland im Jahr 2012. Sucht. 2013;59:321–331. | ||
Paton WD. Cannabis and its problems. Proc R Soc Med. 1973;66(7):718–721. | ||
Fraser JD. Withdrawal symptoms in cannabis-indica addicts. Lancet. 1949;2(6582):747. | ||
Bensuan AD. Marihuana withdrawal symptoms. Br Med J. 1971;3(5766):112. | ||
Wiesbeck GA, Schuckit MA, Kalmijn JA, Tipp JE, Bucholz KK, Smith TL. An evaluation of the history of a marijuana withdrawal syndrome in a large population. Addiction. 1996;91(10):1469–1478. | ||
Levin KH, Copersino ML, Heishman SJ, et al. Cannabis withdrawal symptoms in non-treatment-seeking adult cannabis smokers. Drug Alcohol Depend. 2010;111:120–127. | ||
Budney AJ, Hughes JR, Moore BA, Vandrey R. Review of the validity and significance of cannabis withdrawal syndrome. Am J Psychiatry. 2004;161:1967–1977. | ||
Budney AJ, Hughes JR. The cannabis withdrawal syndrome. Curr Opin Psychiatry. 2006;19(3):233–238. | ||
Budney AJ, Vandrey RG, Hughes JR, Thostenson JD, Bursac Z. Comparison of cannabis and tobacco withdrawal: severity and contribution to relapse. J Subst Abuse Treat. 2008;35(4):362–368. | ||
Allsop DJ, Norberg MM, Copeland J, Fu S, Budney AJ. The cannabis withdrawal scale development: patterns and predictors of cannabis withdrawal and distress. Drug Alcohol Depend. 2011;119:123–129. | ||
Allsop DJ, Copeland J, Norberg MM, et al. Quantifying the clinical significance of cannabis withdrawal. PLoS One. 2012;7(9):e44864. | ||
Cornelius JR, Chung T, Martin C, Wood DS, Clark DB. Cannabis withdrawal is common among treatment-seeking adolescents with cannabis dependence and major depression, and is associated with rapid relapse to dependence. Addict Behav. 2008;33(11):1500–1505. | ||
Chung T, Martin CS, Cornelius JR, Clark DB Cannabis withdrawal predicts severity of cannabis involvement at 1-year follow-up among treated adolescents. Addiction. 2008;103(5):787–799. | ||
Gorelick DA, Levin KH, Copersino ML, et al. Diagnostic criteria for cannabis withdrawal syndrome. Drug Alcohol Depend- 2012;123:141–147. | ||
Arendt M, Rosenberg R, Foldager L, Sher L, Munk-Jørgensen P. Withdrawal symptoms do not predict relapse among subjects treated for cannabis dependence. Am J Addict. 2007;16:461–467. | ||
Agrawal A, Pergadia ML, Lynskey MT. Is there evidence for symptoms of cannabis withdrawal in the national epidemiologic survey of alcohol and related conditions? Am J Addict. 2008;17:199–208. | ||
Hasin DS, Keyes KM, Alderson D, Wang S, Aharonovich E, Grant BF. Cannabis withdrawal in the United States: results from NESARC. J Clin Psychiatry. 2008;69:1354–1363. | ||
Copersino ML, Boyd SJ, Tashkin DP, et al. Cannabis withdrawal among non-treatment-seeking adult cannabis users. Am J Addict. 2006;15(1):8–14. | ||
Budney AJ, Novy PL, Hughes JR. Marijuana withdrawal among adults seeking treatment for marijuana dependence. Addiction. 1999;94(9):1311–1322. | ||
Budney AJ, Hughes JR, Moore BA, Novy PL. Marijuana abstinence effects in marijuana smokers maintained in their home environment. Arch Gen Psychiatry. 2001;58(10):917–924. | ||
Budney AJ, Moore BA, Vandrey RG, Hughes JR. The time course and significance of cannabis withdrawal. J Abnorm Psychol. 2003;112(3):393–402. | ||
Kouri EM, Pope HG Jr. Abstinence symptoms during withdrawal from chronic marijuana use. Exp Clin Psychopharmacol. 2000;8(4):483–492. | ||
Haney M, Ward AS, Comer SD, Foltin RW, Fischman MW. Abstinence symptoms following smoked marijuana in humans. Psychopharmacology (Berl). 1999;141(4):395–404. | ||
Haney M, Hart CL, Vosburg SK, Comer SD, Reed SC, Foltin RW. Effects of THC and lofexidine in a human laboratory model of marijuana withdrawal and relapse. Psychopharmacology (Berlin). 2008;197:157–168. | ||
Haney M, Hart CL, Vosburg SK, et al. Effects of baclofen and mirtazapine on a laboratory model of marijuana withdrawal and relapse. Psychopharmacology (Berlin). 2010;211(2):233–244. | ||
APA (American Psychiatric Association). Diagnostic and Statistical Manual of Mental Disorders, Fifth ed. (DSM-5TM). Washington, DC: American Psychiatric Association; 2013. | ||
Dilling H, Mombour W, Schmidt MH. Internationale Klassifikation psychischer Störungen. ICD-10 Kapitel V (F). Diagnostische Kriterien für Forschung und Praxis. Bern, Switzerland: Huber; 2004. | ||
ICD-11 Beta Draft (Foundation). Cannabis withdrawal. Available from: http://apps.who.int/classifications/icd11/browse/f/en#/http%3a%2f%2fid.who.int%2ficd%2fentity%2f637576511. Accessed November 25, 2016. | ||
Preuss UW, Watzke AB, Zimmermann J, Wong JW, Schmidt CO. Cannabis withdrawal severity and short-term course among cannabis-dependent adolescent and young adult inpatients. Drug Alcohol Depend. 2010;106:133–134. | ||
Lee D, Schroeder JR, Karschner EL, et al. Cannabis withdrawal in chronic, frequent smokers during sustained abstinence within a closed residential environment. Am J Addict. 2014;23:234–242. | ||
Bonnet U, Specka M, Stratmann U, Ochwadt R, Scherbaum N. Abstinence phenomena of chronic cannabis-addicts prospectively monitored during controlled inpatient detoxification: cannabis withdrawal syndrome and its correlation with delta-9-tetrahydrocannabinol and -metabolites in serum. Drug Alcohol Depend. 2014;143:189–197. | ||
Vandrey R, Budney AJ, Kamon JL, Stanger C. Cannabis withdrawal in adolescent treatment seekers. Drug Alcohol Depend. 2005;78(2):205–210. | ||
Crowley TJ, Macdonald MJ, Whitmore EA, Mikulich SK. Cannabis dependence, withdrawal, and reinforcing effects among adolescents with conduct symptoms and substance use disorders. Drug Alcohol Depend. 1998;50(1):27–37. | ||
Milin R, Manion I, Dare G, Walker S. Prospective assessment of cannabis withdrawal in adolescents with cannabis dependence: a pilot study. J Am Acad Child Adolesc Psychiatry. 2008;47(2):174–178. | ||
Soenksen S, Stein LA, Brown JD, Stengel JR, Rossi JS, Lebeau R. Cannabis withdrawal among detained adolescents: exploring the impact of nicotine and race. J Child Adolesc Subst Abuse. 2015;24(2):119–124. | ||
Swift W, Hall W, Teesson M. Characteristics of DSM-IV and ICD-10 cannabis dependence among Australian adults: results from the National Survey of Mental Health and Wellbeing. Drug Alcohol Depend. 2001;63(2):147–153. | ||
Greene MC, Kelly JF. The prevalence of cannabis withdrawal and its influence on adolescents’ treatment response and outcomes: a 12-month prospective investigation. J Addict Med. 2014;8(5):359–367. | ||
APA (American Psychiatric Association). Diagnostic and Statistical Manual of Mental Disorders, 4th ed. Washington, DC: American Psychiatric Association; 1994. | ||
PubMed. US National Library of Medicine National Institutes of Health Search database. Available from: https://www.ncbi.nlm.nih.gov/pubmed. Accessed November 25, 2016. | ||
Scopus. Document search. Elsevier. Available from: https://www.scopus.com/home.uri. Accessed November 25, 2016. | ||
Ligresti A, De Petrocellis L, Di Marzo V. From phytocannabinoids to cannabinoid receptors and endocannabinoids: pleiotropic physiological and pathological roles through complex pharmacology. Physiol Rev. 2016;96(4):1593–1659. | ||
Bhattacharyya S, Atakan Z, Martin-Santos R, Crippa JA, McGuire PK. Neural mechanisms for the cannabinoid modulation of cognition and affect in man: a critical review of neuroimaging studies. Curr Pharmaceut Des. 2012;18:5045–5054. | ||
Lorenzetti V, Solowij N, Yücel M. The role of cannabinoids in neuroanatomic alterations in cannabis users. Biol Psychiatry. 2016;79(7):e17–e31. | ||
Oz M. Receptor-independent actions of cannabinoids on cell membranes: focus on endocannabinoids. Pharmacol Ther. 2006;111(1):114–144. | ||
Manjarrez-Marmolejo J, Franco-Pérez J. Gap junction blockers: an overview of their effects on induced seizures in animal models. Curr Neuropharmacol. 2016;14(7):759–771. | ||
Cone EJ, Huestis MA. Relating blood concentrations of terahydrocannabinoland metabolites to pharmacologic effects and time of marijuana usage. Ther Drug Monit. 1993;15:527–532. | ||
Laprevote V, Gambier N, Cridlig J, et al. Early withdrawal effects in a heavy cannabis smoker during hemodialysis. Biol Psychiatry. 2015;77(5):e25–e26. | ||
Muramatsu RS, Silva N, Ahmed I. Suspected dronabinol withdrawal in an elderly cannabis-naïve medically ill patient. Am J Psychiatry. 2013;170(7):804. | ||
Haney M, Cooper ZD, Bedi G, Vosburg SK, Comer SD, Foltin RW. Nabilone decreases marijuana withdrawal and a laboratory measure of marijuana relapse. Neuropsychopharmacology. 2015;40(11):2489–2498. | ||
Balter RE, Cooper ZD, Haney M. Novel pharmacologic approaches to treating cannabis use disorder. Curr Addict Rep. 2014;1(2):137–143. | ||
Macfarlane V, Christie G. Synthetic cannabinoid withdrawal: a new demand on detoxification services. Drug Alcohol Rev. 2015;34(2):147–153. | ||
Wiley JL, Marusich JA, Huffman JW. Moving around the molecule: relationship between chemical structure and in vivo activity of synthetic cannabinoids. Life Sci. 2014;97(1):55–63. | ||
Sampson CS, Bedy SM, Carlisle T. Withdrawal seizures seen in the setting of synthetic cannabinoid abuse. Am J Emerg Med. 2015;33(11):1712.e3. | ||
Hegde M, Santos-Sanchez C, Hess CP, Kabir AA, Garcia PA. Seizure exacerbation in two patients with focal epilepsy following marijuana cessation. Epilepsy Behav. 2012;25(4):563–566. | ||
Crippa JA, Hallak JE, Machado-de-Sousa JP, et al. Cannabidiol for the treatment of cannabis withdrawal syndrome: a case report. J Clin Pharm Ther. 2013;38(2):162–164. | ||
Niesink RJ, van Laar MW. Does cannabidiol protect against adverse psychological effects of THC? Front Psychiatry. 2013;4:130. | ||
Bonnet U. Abrupt quitting of long-term heavy recreational cannabis use is not followed by significant changes in blood pressure and heart rate. Pharmacopsychiatry. 2016;49(1):23–25. | ||
Gorelick DA, Goodwin RS, Schwilke E, et al. Antagonist-elicited cannabis withdrawal in humans. Clin Psychopharmacol. 2011;31(5):603–612. | ||
Chauchard E, Goncharov O, Krupitsky E, Gorelick DA. Cannabis withdrawal in patients with and without opioid dependence. Subst Abuse. 2014;35(3):230–234. | ||
Haney M, Ramesh D, Glass A, Pavlicova M, Bedi G, Cooper ZD. Naltrexone maintenance decreases cannabis self-administration and subjective effects in daily cannabis smokers. Neuropsychopharmacology.2015;40(11):2489–2498. | ||
Padula CB, McQueeny T, Lisdahl KM, Price JS, Tapert SF. Craving is associated with amygdala volumes in adolescent marijuana users during abstinence. Am J Drug Alcohol Abuse. 2015;41(2):127–132. | ||
McEwen BS. Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol Rev. 2007;87(3):873–904. | ||
Aleksić D, Aksić M, Radonjić NV, et al. Long-term effects of maternal deprivation on the volume, number and size of neurons in the amygdala and nucleus accumbens of rats. Psychiatr Danub. 2016;28(3):211–219. | ||
Cornelius JR, Aizenstein HJ, Hariri AR. Amygdala reactivity is inversely related to level of cannabis use in individuals with comorbid cannabis dependence and major depression. Addict Behav. 2010;35(6):644–646. | ||
Hirvonen J, Goodwin RS, Li C-T, et al. Reversible and regionally selective downregulation of brain cannabinoid CB1-receptors in chronic daily cannabis smokers. Mol Psychiatry. 2012;17:642–649. | ||
D’Souza DC, Cortes-Briones JA, Ranganathan M, et al. Rapid changes in CB1 receptor availability in cannabis dependent males after abstinence from cannabis. Biol Psychiatry Cogn Neurosci Neuroimaging. 2016;1(1):60–67. | ||
Filbey FM, Dunlop J, Ketcherside A, et al. fMRI study of neural sensitization to hedonic stimuli in long-term, daily cannabis users. Hum Brain Mapp. 2016;37(10):3431–3443. | ||
Ehlers CL, Gizer IR, Vieten C, et al. Cannabis dependence in the San Francisco Family Study: age of onset of use, DSM-IV symptoms, withdrawal, and heritability. Addict Behav. 2010;35(2):102–110. | ||
Richmond-Rakerd LS, Slutske WS, Lynskey MT, et al. Age at first use and later substance use disorder: shared genetic and environmental pathways for nicotine, alcohol, and cannabis. J Abnorm Psychol. 2016;125(7):946–959. | ||
Fosnocht AQ, Briand LA. Substance use modulates stress reactivity: behavioral and physiological outcomes. Physiol Behav. 2016;166:32–42. | ||
Herrmann ES, Weerts EM, Vandrey R. Sex differences in cannabis withdrawal symptoms among treatment-seeking cannabis users. Exp Clin Psychopharmacol. 2015;23(6):415–421. | ||
Delforterie M, Creemers H, Agrawal A, et al. Functioning of cannabis abuse and dependence criteria across two different countries: the United States and the Netherlands. Subst Use Misuse. 2015;50(2):242–250. | ||
Brown SA, Myers MG, Lippke L, Tapert SF, Stewart DG, Vik PW. Psychometric evaluation of the Customary Drinking and Drug Use Record (CDDR): a measure of adolescent alcohol and drug involvement. J Stud Alcohol. 1998;59(4):427–438. | ||
Vandrey RG, Budney AJ, Hughes JR, Liguori A. A within-subject comparison of withdrawal symptoms during abstinence from cannabis, tobacco, and both substances. Drug Alcohol Depend. 2008;92(1–3):48–54. | ||
Busner J, Targum SD. The clinical global impression scale: applying a research tool in clinical practice. Psychiatry (Edgemont). 2007;4:28–37. | ||
Chesney T, Matsos L, Couturier J, Johnson N. Cannabis withdrawal syndrome: an important diagnostic consideration in adolescents presenting with disordered eating. Int J Eat Disord. 2014;47(2):219–223. | ||
Lam PW, Frost DW. Nabilone therapy for cannabis withdrawal presenting as protracted nausea and vomiting. BMJ Case Rep. 2014;2014. | ||
Allen JH, de Moore GM, Heddle R, Twartz JC. Cannabinoid hyperemesis: cyclical hyperemesis in association with chronic cannabis abuse. Gut. 2004;53(11):1566–1570. | ||
Bonnet U, Chang DI, Scherbaum N. Cannabis-Hyperemesis-Syndrom [Cannabis hyperemesis syndrome]. Fortschr Neurol Psychiatr. 2012;80(2):98–101. German. | ||
Heise L. Cannabinoid hyperemesis syndrome. Adv Emerg Nurs J. 2015;37(2):95–101. | ||
Vaziri ND, Thomas R, Sterling M, et al. Toxicity with intravenous injection of crude marijuana extract. Clin Toxicol. 1981;18:353–366. | ||
Grlic L. A comparative study on some chemical and biological characteristics of various samples of cannabis resin. UNDOC 1962. Available from: https://www.unodc.org/unodc/en/data-and-analysis/bulletin/bulletin_1962-01-01_3_page005.html. Accessed November 25, 2016. | ||
Cressey D. The cannabis experiment. Nature. 2015;524(7565):280–283. | ||
Swift W, Wong A, Li KM, Arnold JC, McGregor IS. Analysis of cannabis seizures in NSW, Australia: cannabis potency and cannabinoid profile. PLoS One. 2013;8(7):e70052. | ||
Niesink RJ, Rigter S, Koeter MW, Brunt TM. Potency trends of Δ9-tetrahydrocannabinol, cannabidiol and cannabinol in cannabis in the Netherlands: 2005-15. Addiction. 2015;110(12):1941–1950. | ||
Braun B, Künzel J, Brand H. Kapitel 3.1.: Jahresstatistik 2014 der professionellen Suchtkrankenhilfe. In: Deutsche Hauptstellen für Suchtfragen e.V., editor. Jahrbuch Sucht 2016. Lengerich, Germany: Pabst Verlag; 2016: 173–199. | ||
Buchowski MS, Meade NN, Charboneau E, Park S, Dietrich MS, Cowan RL, Martin PR. Aerobic exercise training reduces cannabis craving and use in non-treatment seeking cannabis-dependent adults. PLoS One. 2011;6(3):e17465. | ||
Copeland J, Pokorski I. Progress toward pharmacotherapies for cannabis-use disorder: an evidence-based review. Subst Abuse Rehab. 2016;7:41–53. | ||
Kelly MA, Pavlicova M, Glass A, et al. Do withdrawal-like symptoms mediate increased marijuana smoking in individuals treated with venlafaxine-XR? Drug Alcohol Depend. 2014;144:42–46. | ||
Cooper ZD, Foltin RW, Hart CL, Vosburg SK, Comer SD, Haney M. A human laboratory study investigating the effects of quetiapine on marijuana withdrawal and relapse in daily marijuana smokers. Addict Biol. 2013;18(6):993–1002. | ||
Mariani JJ, Pavlicova M, Mamczur AK, Bisaga A, Nunes EV, Levin FR. Open-label pilot study of quetiapine treatment for cannabis dependence. Am J Drug Alcohol Abuse. 2014;40(4):280–284. | ||
Mason BJ, Crean R, Goodell V, et al. A proof-of-concept randomized controlled study of gabapentin: effects on cannabis use, withdrawal and executive function deficits in cannabis-dependent adults. Neuropsychopharmacology. 2012;37(7):1689–1698. | ||
Levin FR, Mariani JJ, Brooks DJ, Pavlicova M, Cheng W, Nunes EV. Dronabinol for the treatment of cannabis dependence: a randomized, double-blind, placebo-controlled trial. Drug Alcohol Depend. 2011;116(1–3):142–150. | ||
Allsop DJ, Copeland J, Lintzeris N, et al. Nabiximols as an agonist replacement therapy during cannabis withdrawal: a randomized clinical trial. JAMA Psychiatry. 2014;71(3):281–291. | ||
Allsop DJ, Lintzeris N, Copeland J, Dunlop A, McGregor IS. Cannabinoid replacement therapy (CRT): nabiximols (Sativex) as a novel treatment for cannabis withdrawal. Clin Pharmacol Ther. 2015;97(6):571–574. | ||
Trigo JM, Lagzdins D, Rehm J, et al. Effects of fixed or self-titrated dosages of Sativex on cannabis withdrawal and cravings. Drug Alcohol Depend. 2016;161:298–306. | ||
Indorato F, Liberto A, Ledda C, Romano G, Barbera N. The therapeutic use of cannabinoids: forensic aspects. Forensic Sci Int. 2016;265:200–203. | ||
Gray KM, Carpenter MJ, Baker NL, et al. A double-blind randomized controlled trial of N-acetylcysteine in cannabis-dependent adolescents. Am J Psychiatry. 2012;169(8):805–812. | ||
Haney M, Cooper ZD, Bedi G, Vosburg SK, Comer SD, Foltin RW. Nabilone decreases marijuana withdrawal and a laboratory measure of marijuana relapse. Neuropsychopharmacology. 2015;40(11):2489–2498. | ||
Budney AJ, Vandrey RG, Stanger C. Pharmacological and psychosocial interventions for cannabis use disorders. Rev Bras Psiquiatr. 2010;32(Suppl 1):S46–S55. | ||
Vandrey R, Smith MT, McCann UD, Budney AJ, Curran EM. Sleep disturbance and the effects of extended-release zolpidem during cannabis withdrawal. Drug Alcohol Depend. 2011;117(1):38–44. | ||
Herrmann ES, Cooper ZD, Bedi G, et al. Effects of zolpidem alone and in combination with nabilone on cannabis withdrawal and a laboratory model of relapse in cannabis users. Psychopharmacology (Berl). 2016;233(13):2469–2478. | ||
Cooper ZD. Adverse effects of synthetic cannabinoids: management of acute toxicity and withdrawal. Curr Psychiatry Rep. 2016;18(5):52. | ||
Davis JP, Smith DC, Morphew JW, Lei X, Zhang S. Cannabis withdrawal, posttreatment abstinence, and days to first cannabis use among emerging adults in substance use treatment: a prospective study. J Drug Issues. 2016;46(1):64–83. | ||
Allsop DJ, Dunlop AJ, Saddler C, Rivas GR, McGregor IS, Copeland J. Changes in cigarette and alcohol use during cannabis abstinence. Drug Alcohol Depend. 2014;138:54–60. | ||
Koola MM, Boggs DL, Kelly DL, et al. Relief of cannabis withdrawal symptoms and cannabis quitting strategies in people with schizophrenia. Psychiatry Res. 2013;209(3):273–278. | ||
Boden MT, Babson KA, Vujanovic AA, Short NA, Bonn-Miller MO. Posttraumatic stress disorder and cannabis use characteristics among military veterans with cannabis dependence. Am J Addict. 2013;22(3):277–284. | ||
Bonnet U, Specka M, Scherbaum N. Häufiger Konsum von nicht-medizinischem Cannabis [Frequent non-medical cannabis use: health sequelae and effectiveness of detoxification treatment]. Dtsch Med Wochenschr. 2016;141(2):126–131. German. | ||
Verweij KJ, Agrawal A, Nat NO, et al. A genetic perspective on the proposed inclusion of cannabis withdrawal in DSM-5. Psychol Med. 2013;43(8):1713–1722. | ||
Gamaleddin IH, Trigo JM, Gueye AB, et al. Role of the endogenous cannabinoid system in nicotine addiction: novel insights. Front Psychiatry. 2015;6:41. | ||
Henderson-Redmond AN, Guindon J, Morgan DJ Roles for the endocannabinoid system in ethanol-motivated behavior. Prog Neuropsychopharmacol Biol Psychiatry. 2016;65:330–339. | ||
Agrawal A, Budney AJ, Lynskey MT. The co-occurring use and misuse of cannabis and tobacco: a review. Addiction. 2012;107(7):1221–1233. | ||
Rabin RA, George TP. A review of co-morbid tobacco and cannabis use disorders: possible mechanisms to explain high rates of co-use. Am J Addict. 2015;24(2):105–116. | ||
Ream GL, Benoit E, Johnson BD, et al. Smoking tobacco along with marijuana increases symptoms of cannabis dependence. Drug Alcohol Depend. 2008;95:199–208. | ||
Haney M, Bedi G, Cooper ZD, et al. Predictors of marijuana relapse in the human laboratory: robust impact of tobacco cigarette smoking status. Biol Psychiatry. 2013;73:242–248. | ||
Fattore L. Considering gender in cannabinoid research: a step towards personalized treatment of marijuana addicts: Drug Test Anal. 2013;5(1):57–61. | ||
Bonnet U. Rauschzustände: Risiken und Nebenwirkungen. Im Focus: nicht-medizinisches Cannabis und synthetische Cannabinoide [States of intoxication: risks and adverse effects: focus on non-medical cannabis and synthetic cannabinoids]. Suchttherapie. 2016;17(2):61–70. German. | ||
Leweke FM, Mueller JK, Lange B, Rohleder C. Therapeutic potential of cannabinoids in psychosis. Biol Psychiatry. 2016;79(7):04–612. | ||
Di Forti M, Marconi A, Carra E, et al. Proportion of patients in South London with first-episode psychosis attributable to use of high potency cannabis: a case-control study. Lancet Psychiatry. 2015;2(3):233–238. | ||
Freeman TP, Winstock AR. Examining the profile of high-potency cannabis and its association with severity of cannabis dependence. Psychol Med. 2015;45:3181–3189. | ||
Loflin M, Earleywine M. A new method of cannabis ingestion: the dangers of dabs? Addict Behav. 2014;39(10):1430–1433. | ||
Varlet V, Concha-Lozano N, Berthet A, et al. Drug vaping applied to cannabis: is “cannavaping” a therapeutic alternative to marijuana? Sci Rep. 2016;6:25599. | ||
Cooper ZD, Haney M. Investigation of sex-dependent effects of cannabis in daily cannabis smokers. Drug Alcohol Depend. 2014;136:85–91. | ||
Franke G. SCL-90-R. Die Symptomcheckliste von Derogatis – Deutsche Version – Manual (2.Auflage). Göttingen, Germany: Hogrefe; 2002. | ||
Cohen J. Statistical Power for the Behavioral Sciences. New York: Academic Press; 1988. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

