Back to Journals » Therapeutics and Clinical Risk Management » Volume 18
The Association Between Circulating Sex Hormones and Central Serous Chorioretinopathy: A Case-Control Study
Authors Zhao C , Huang Y, Chen L, Ye S, Liu XQ
Received 20 April 2022
Accepted for publication 9 August 2022
Published 25 August 2022 Volume 2022:18 Pages 855—865
DOI https://doi.org/10.2147/TCRM.S370133
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Deyun Wang
Chun Zhao1 *, Yan Huang2 *, Lei Chen,1 Sheng Ye,1 Xiao-Qiang Liu1 *
1Department of Ophthalmology, Tenth People’s Hospital Affiliated to Shanghai Tongji University School of Medicine, Shanghai, 200072, People’s Republic of China; 2Affiliated Hospital of Jiangsu University, Zhenjiang, 212001, People’s Republic of China
* These authors contributed equally to this work
Correspondence: Xiao-Qiang Liu, Email [email protected]
Background: Central serous chorioretinopathy (CSC) is preferential cocurated in males, however the associations between sex hormones and CSC incidence or progression remains unclear. The sex hormone concentration assessments in CSC cases and healthy controls will update the knowledge in CSC management.
Methods: This case-control study included 59 CSC cases and 30 healthy controls, from January 2019 to December 2020. The CSC cases would be defined as spontaneous resolved if the subretinal fluid were absorbed within three months. The concentrations of total testosterone (TT), free testosterone (FT), estradiol (E2), sex hormone-binding globulin (SHBG), progesterone, leuteinizing hormone (LH) and dehydroepiandrosterone sulfate (DHEA-S) were detected in all the participants. The relationships between sex hormone concentrations and CSC-related characteristics were analyzed with Pearson correlation analyses.
Results: Significantly increased TT, FT, FT/E2 ratio, SHBG concentrations as well as decreased DHEA-S level were detected in non-resolved CSC group compared with the control group. Comparing with the resolved ones, it was found that TT, FT and SHBG concentrations were increased in the non-resolved CSC. A significant positive correlation between TT concentrations and CMT (R2=0.168, P=0.031) as well as SRF height (R2=0.146, P=0.045) were detected in the non-solved CSC group.
Conclusion: Different concentrations of TT, FT, FT/E2 ratio, DHEA-S and SHBG were detected in resolved and non-resolved CSC cases. Sex hormones were related to CSC symptom durations and related parameters.
Keywords: central serous chorioretinopathy, sex hormones, cortisol, testosterone, case-control study
Introduction
Central serous chorioretinopathy (CSC), which is characterized by the accumulation of transparent liquid under the retinal neuroepithelial layer in the posterior pole, is one of the most common retinal diseases. It could lead to painless visual impairment, visual object deformation, relative scotoma of visual field, visual object shrinkage or visual object discoloration. Increased permeability of choroidal capillary causes huge leakage and impaired retinal pigment epithelium (RPE) function, resulting in serous RPE detachment. Along with increased choroidal hydrostatic pressure, RPE gradually bulges, and the continuity of RPE is interrupted due to mechanical force, resulting in retinal pigment epithelial detachment (PED) and leakage. The subretinal fluid (SRF) concentrated under the neuroretina and then CSC was induced.1 Most CSC cases remitted spontaneously within 3 months, but it’s easy to relapse and cause visual impairment.2
The classifications of CSC are based on duration of the disease as well as multimodal imaging-based examinations,3 and both classification methods were helpful in advanced CSC management. The initial CSC classification is simply based on CSC duration and the CSC cases are divided into two forms, the acute and chronic ones, according to the duration of the SRF presentation.4 Acute CSC lesions can subside within 3 months, and usually no obvious visual impairments were detected. Chronic CSC lesions are potentially destructive, which can lead to long-term localized retinal neuroepithelial detachment and thus cause photoreceptor damage and visual impairment.5
Choroidal thickening is related to higher hydrostatic pressure as well as choroidal permeability, resulting in neuroretina detachment. Choroidal thickening can be affected by various factors and thus promotes the incidence or progression of PED and CSC. CSC is a multifactorial disease and can be influenced by both systemic and local risk factors. For systemic factors, it is well accepted that male gender, increased age and sleep disorders are related to the persistence or recurrence of CSC.6 Sleep disorder is related to the changes of cortisol and catecholamine secretion, which leads to increased activities of hypothalamus pituitary adrenal axis and autonomic sympathetic adrenal system.7 In addition, increased age, steroid use, pregnancy, post-traumatic stress disorder, antipsychotics (anti-anxiety or antidepressants), smoking and psychological stress promote the incidence of CSC.5,8–11 There are also local risk factors for CSC incidence. Local corticosteroid use could affect the choroidal function and thus promote the incidence or progression of CSC.4 CSC incidence could also be influenced by refractive error. It was found that hyperopia are risk factors for CSC, while myopia is a protective factor.4,12
Considering that male gender is a key hazard factor for CSC, it is natural to determine the correlation between sex hormones and CSC incidence. Understanding the relationship between sex hormone concentration and CSC status would provide improved knowledge in CSC management. Testosterone is related to choroid thickening and thus promotes the development of CSC, however the exact role of testosterone in CSC development remains unclear.13 As the sex hormone concentrations would change with aging and CSC-related drug use, circulating sex hormones might be used in CSC diagnosis, classification and prognosis. In an age- and sex-matched case-control study, 17 steroid hormones in 46 chronic CSC patients and relevant healthy controls were detected and it was found that the concentrations of androstenedione, androsterone, etiocholanolone and estrone (E1) were significantly increased in the serum samples of CSC cases.14 As the prognoses of CSC cases are dependent on the symptom durations and spontaneous remission, detection of the relationship between circulating sex hormones and CSC severity would provide novel evidence in both clinical management and biological mechanism of CSC. In this pilot case-control study, the association between sex hormones in serum samples and CSC status was detected within one year of follow-up. In addition, detailed correlation analyses would provide more evidence on the effects of sex hormones in CSC prognoses.
Materials and Methods
Study Design and Participants
A case-control study design was adopted in this study. All procedures in this study were in accordance with the Helsinki Declaration of 1975. This study was approved by the Ethics Committee of the Shanghai Tenth People’s Hospital and Affiliated Hospital of Jiangsu University. All participants were informed and written consents were obtained.
From January 2019 to December 2020, a total of 59 newly-diagnosed initial CSC patients (CSC group) were included in this study. The inclusion criteria were as follows: (1) age ≥18 years old; (2) meet the diagnostic criteria of active CSC based on typical CSC symptoms and fundus manifestations based on OCT, fluorescein angiography (FFA), and indocyanine green angiography. OCT shows localized or diffuse retinal neuroepithelial detachment with plasma exudation in the detachment area, which is consistent with the leakage point shown by FFA; and (3) cooperate with clinical research and complete relevant inspection operations. The exclusion criteria of CSC cases were: (1) age <18 years old; (2) vitreoretinal diseases and glaucoma history; (3) other macular diseases that cause serous detachment of the neuroepithelial layer in the macular region, including choroidal neovascularization (CNV) and polypoidal choroidal vasculopathy (PCV); and (4) any patients with other serious systemic diseases, such as tumor, reproductive disorders history, severe kidney disease and hormone replacement therapy. The diagnoses of CSC was following a diagnostic criteria by CSC group.3 The healthy controls were included from the out-patient department after fundus photography and OCT examination to excluding any potential cases with retinal disorders.
For all the participants, the age, hypertension status, diabetes incidence, body mass index (BMI) value, axial length (AL) and spherical equivalent (SE) were recorded. All the CSC cases finished a monthly follow-up and the cases with aborted SRF liquid within three months were recorded as the resolved ones. CSC with a persistent SRF for over three months were recorded in non-resolved group. The blood samples were obtained in the first diagnosis and used in sex hormone analyses after all the cases included. The detailed flow diagram of study design and participant screening are presented in Figure 1.
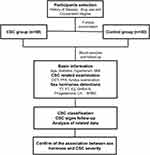 |
Figure 1 The flow diagram of participant screening, study conduction and experiment process. |
Ophthalmic Examination and Definitions
The AL was measured by experienced technicians using the IOLMaster 500 (Carl Zeiss, Germany). A standard fundus synchronous FFA and ICG examination was conducted using a Spectralis HRA instrument (Heidelberg, Germany) for any potential CSC cases in this study. The acquisition of SD-OCT was carried out by Cirrus HD-OCT 5000 imager (Carl Zeiss, Germany) to obtain the central macular thickness (CMT) and SRF height. The PED morphology was evaluated by two ophthalmologists (Z.C and L.X.Q) with OCT images. PED was defined as epithelium pigment detachment from choroid and CNV/PCV was excluded from this study. The leakage types were analyzed using FFA and thus the leakage spots were recorded for advanced analyses.
Blood Sample Collection
Morning fasting blood samples were collected from all participants prior to any treatments or examinations at the time of recruitment. The serum from all the blood samples was separated from the supernatant after centrifugation and stored at −80 °C until use. The biological assay and detections were completed in 8 months after the serum samples were collected.
Measurement of Serum Sex Hormones
The protocols of sex hormone measurements were following a previous report with slight modifications.15 Sex hormones were detected using competitive electro-chemiluminescence immunoassays with the Cobase 601 System (Roche Diagnostics, Mannheim, Germany) at the biochemistry laboratory of the Tenth People’s Hospital. The concentrations of total testosterone (TT), free testosterone (FT), estradiol (E2), sex hormone-binding globulin (SHBG), leuteinizing hormone (LH) and dehydroepiandrosterone sulfate (DHEA-S) were detected in this study. The catalog numbers of these assays were 11776061 for TT, 03000079 for E2, 12145383 for progesterone, 03000095 for DHEA-S,11732234 for LH and 03052028 for SHBG. FT was detected using a commercial kit (GOY-T4300, DRG, Germany) and the operation was conducted in accordance with instructions of the supplier.
Statistical Analysis
Graphpad statistical software (GraphPad Software Inc., San Diego, CA, USA) was used for the statistical analysis. The continuous data are expressed in mean ± SD. Independent sample t-tests were used for the difference between two groups and one-way ANOVA analyses for three groups. The Tukey method was used in multiple comparison analyses. The chi square test was used in the analyses of categorical data. Pearson correlation analyses between sex hormone concentrations and CSC-related parameters were analyzed. The difference was considered statistically significant when P<0.05.
Results
Basic Information and CSC-Related Characteristics of Participants with and without CSC
A total of 59 CSC cases and 30 healthy controls were included in this study. Through follow-up of the SRF status in the CSC cases, 31 cases were identified in the resolved CSC group and 28 cases were included in the non-resolved CSC group (SRF not absorbed within three months). The detailed demographics, clinical characteristics and CSC related parameters are provided in Table 1.
 |
Table 1 Demographics, Clinical Characteristics and CSC-Related Parameters in CSC Cases and Relevant Controls |
In addition, the CSC-related characteristics were analyzed in this study. Both OCT and FFA/ICG were used in CMT thickness, and SRF height, choroidal thickness, presence of pigment epithelium detachment (PED) as well as retinal leakage hot spots were reported and analyzed. It was found that there were no significant differences in age distribution, hypertension status, diabetes incidence, BMI value, AL range or SE degrees among healthy controls, resolved CSC and non-resolved CSC cases (P>0.05). A significantly longer symptom duration was detected in the non-resolved CSC group compared to the resolved CSC group (7.46±2.80 vs 2.26±0.62 months, P<0.001). Following advanced analyses of leakage types, PED status, CMT value and SRF height, no significant differences were detected between the resolved and non-resolved CSC groups (P>0.05).
Circulating Sex Hormones in Controls, Resolved and Non-Resolved CSC Cases
To evaluate whether sex hormones were associated with CSC progeneses, we analyzed TT, FT, E2, TT/E2 ratio, FT/E2 ratio DHEA-S and progesterone among the three groups (Table 2). Compared to controls, higher FT and DHEA-S concentrations were detected (P=0.007 and P=0.040, respectively) in the resolved CSC cases. In addition, increased TT, FT, FT/E2 and SHBG concentrations and decreased DHEA-S were detected in the non-resolved CSC group (P=0.001, P<0.001, P=0.045, P=0.003, and P=0.045, respectively). Moreover, the sex hormone levels in resolved and non-resolved CSC were analyzed and it was found that higher FT and SHBG were detected in the non-resolved CSC cases (P=0.028, P=0.026 and P=0.018, respectively).
 |
Table 2 The Circulating Sex Hormones in Controls, Resolved and Non-Resolved CSC |
Correlations Between Circulating Sex Hormones and CSC-Related Parameters in CSC Cases
The sex hormones, respectively TT and its related hormones, were significantly associated with CSC incidence and progression. Advanced correlation matrix analyses between sex hormone levels and CSC-related parameters in all the CSC cases are presented in Figure 2. TT, FT, FT/E2 and SHBG levels were positively associated with symptom durations, with R values of 0.463, 0.474, 0.344 and 0.358, respectively (P<0.5). In addition, it was found that TT level was positively correlated with CMT (r=0.29, P=0.023). Even slight significance was reported and it should be noticed that there were limited liner correlations with R values less than 0.5 in all the groups.
 |
Figure 2 The associations between sex hormones and CSC-related parameters in the CSC cases. Red to blue: the R value from −1 to 1. The R value is presented in the box. |
Sex Hormone Levels in the Progression of CSC
As the correlation analyses highlighted the correlations between sex hormones and symptom duration, the values of four sex hormone indexes, including TT, FT, FT/E2 and SHBG levels, were presented for CSC cases with different durations. As shown in Figure 3A, higher TT concentrations were detected in the CSC group with 9–12 month duration compared to the 1–3 month and 4–6 month groups (P<0.001 and P=0.004, respectively). When the FT level was considered, a decreased FT level was detected in the 9–12 month duration group compared to the 1–3 month group and 4–6 month group (P<0.001 and P=0.003, respectively; Figure 3B). In addition, a significantly lower FT/E2 value was detected in the 9–12 month group compared to all other groups (P<0.001; Figure 3C). As demonstrated in Figure 3D, no significant differences in SHBG levels were detected among all the groups (P>0.05).
Association Between TT Concentration and CMT/SRF Height in Resolved and Non-Resolved CSC Cases
TT concentrations were different among various CSC symptom duration groups, however no significant correlation between TT concentration and SRF height was detected among all the CSC cases. Thus, the associations between TT concentrations and CMT/SRF height in resolved and non-resolved CSC cases were analyzed. As shown in Figure 4, no significant association between the TT concentrations and CMT/SRF height in the resolved CSC groups was detected (P>0.05). However, a significant positive correlation between TT concentrations and CMT (R2=0.168, P=0.031) as well as SRF height (R2=0.146, P=0.045) were detected in the non-resolved CSC group.
Sex Hormone Levels in CSC Cases with Different Leakage Types and PED Status
Most previous analyses were based on symptom duration of CSC cases, advanced analyses of leakage types and PED status were provided in this study (Table 3). It was found that all the CSC cases were divided into one spot group (n=23), over two spots group (n=16) and diffuse group (n=20) based on the leakage types. In addition, there were 27 and 32 CSC cases in the with PED and without PED groups, respectively. Comparing the circulating sex hormones in different groups, no significant differences were detected in all the reported sex hormone levels among different CSC groups with leakage types as well as PED status (P>0.05).
 |
Table 3 The Circulating Sex Hormones in CSC Cases with Different Leakage Types and PED Status |
Discussion
This study demonstrated that significantly increased TT, FT, FT/E2 and SHBG levels and decreased DHA-S in resolved or non-resolved CSC cases were detected compared to the controls. In addition, higher TT, FT and SHBG concentrations were detected in non-resolved CSC cases compared to the resolved ones. Also, through analyzing the correlations between sex hormone levels and symptom duration, it was found that TT, FT, FT/E2 and SHBG demonstrated positive correlations with symptom duration. Significant linear correlations between TT concentrations and CMT/SRF heights were also detected in the non-resolved cases. Interestingly, it was found that there were no differences in the sex hormone-related parameters in CSC cases with different leakage types or PED status.
CSC was one of the most frequent retinal disorders observed; however, the classification and management of CSC remained quite insufficient. Based on the persistence of SRF liquid, it was divided into acute and chronic CSC subtypes. The acute CSC cases usually demonstrated a characteristic of spontaneous recovery, and the incidence rate of sequelae, such as visual impairment, was low.16 However, CSC was easy to transform into a chronic course or recurrent attack, which would cause the detachment of RPE in the macular region and eventually lead to the decline of visual function. A previous study had pointed out that about 12.8% of patients with chronic CSC would eventually suffer from permanent blindness in both eyes,2 thus it is important to develop several prognostic factors. It was reported that psychological stress, type-A behavior pattern, steroid use, and pregnancy are reported diagnostic and prognostic factors of CSC.17 The potential contributions of sex hormones on the CSC incidences were inconsistent, and thus the results in this study provided more evidence to apply to targeting sex hormones in CSC management.
As CSC was related to male gender, it was natural to hypothesise that androgen was related to an increased risk of CSC. This hypothesis was supported by CSC cases secondary to exogenous testosterone use.18,19 In addition, previous case control studies demonstrated that serum testosterone level in the CSC group was higher compared to the control group.14,20 However, inconsistent conclusion was detected based on several studies involving the contribution of serum testosterone levels in chronic CSC risk.21 In this current study, our results supported the association between testosterone and CSC risk. In advanced stratified analyses, non-resolved CSC demonstrated increased TT and FT compared to the resolved CSC cases and healthy controls. As TT and FT concentrations were positively associated with symptom duration, it highlighted the potential application of TT/FT on CSC prognoses. As part of CSC cases could resolve spontaneously, a potential prognostic index for CSC would promote the management of CSC.
E2 was another sex hormone related to CSC incidence and progression. Even though the association between estrogen status and CSC incidence remained uncertain, the potential pathological role of estrogen in CSC was reported by a previous review.22 Most previous studies demonstrated an unsignificant difference between estrogen concentrations of CSC cases and controls20,22 and this conclusion was consistent with this current study. Based on several case series studies, the most frequently used estrogen inhibitor, Tamoxifen, had been reported to induce CSC.23,24 However, no solid evidence demonstrated that estrogen would promote progression of CSC. The prevalence of vascular diseases in postmenopausal women was much higher than that in premenopausal women of the same age25,26 and this evidence suggests a relationship between circulating estrogen levels and the change of vascular function in women. Even if CSC was another vascular dysfunction-related disorder, the potential protective effects of estrogen in CSC were not supported by current clinical evidence.
Among all the detected sex hormones, TT and FT demonstrated an increased concentration in CSC cases, especially the non-resolved cases. Considering that another corticosteroid, aldosterone, was reported to be a diagnostic factor and therapeutic target of CSC,27,28 we tried to detect the association between TT/FT and CSC-related parameters. Advanced analyses showed that TT and FT were related to a longer disease duration. In addition, the TT/FT concentrations in CSC with different leakage types and PED status were analyzed; however, no significant difference in any points were detected in the CSC cases. After dividing the CSC cases into resolved and non-resolved cases, a positive linear correlation between TT and CMT/SRF height in the non-resolved CSC group was observed. This interesting finding demonstrated that TT might produce different effects in regulating retinal thickness, thus affecting the prognosis of CSC. However, it should be noted that a low R2 value (R2=0.168 and 0.146, respectively) and slight significance (P=0.031 and 0.045) were detected in the non-resolved group. In addition, it demonstrated that there were negative correlations between TT concentrations and CMT/SRF height, even though no statistical significance was detected. This contradictory finding is caused by the small sample size in this study and we should be cautious before we consider it a new pathological mechanism in CSC progression. More observational studies with larger sample sizes concerning the role of testosterone in CSC are required before applying it as a therapeutic target in CSC treatment.
A significantly increased SHBG was detected in CSC cases compared to controls. In addition, higher SHBG concentration was detected in the non-resolved CSC cases compared to non-resolved ones. No previous reports describe the contribution of SHBG on CSC incidence or progression and limited knowledge exists on this point. SHBG is a glycoprotein synthesized by hepatocytes that has high affinity with steroid hormones and can regulate the concentration of sex hormones and affect their activity.29 SHBG is widely used in the diagnosis of obstetric diseases, precocious puberty, breast development, pregnancy-induced hypertension, diabetes and thyroid disease.30 As SHBG is quite related to the biological functions of each sex hormone, our data provide a clue for advanced application of SHBG in CSC management.
As sex hormones were significantly dysregulated in this study, they might act as therapeutic targets of CSC. Treatments that target serum cortisol levels or block their effects have been proposed. The antifungal drug, ketoconazole, is a glucocorticoid receptor antagonist, which can inhibit the biosynthesis of the adrenal gland and reduce the level of endogenous cortisol.31 In a retrospective cohort study with a total of 41 naive CSC eyes, it was found that ketoconazole treatment could shorten the disease duration through promoting SRF absorbtion.32 Another effective glucocorticoid antagonist, mifepristone, was used in CSC treatment. Mifepristone 200 mg was administered orally to 16 chronic CSC cases and showed that systemic glucocorticoid receptor antagonism promoted improvement and remission of retinal detachment.33 Androgen inhibitor is a key field in prostate cancer and its potential application in CSC treatment demonstrated potential beneficial effects.
A total of nine sex hormones or their related parameters were detected and analyzed in the healthy controls, resolved CSC cases and non-resolved CSC cases. In this study, comprehensive CSC-related parameters were detected in the CSC cases and monthly follow-ups were conducted for one year. However, several limitations should be reported. Firstly, the sample size was relatively small and this was caused partly by the COVID-19 pandemic. Larger sample size would be included in a prospective multicenter cohort study in the future. Secondly, the follow-up duration was relatively short in this study. Considering that several positive outcomes were reported in this study, more well-designed studies are required in the future.
In conclusion, significantly increased androgen concentrations, including TT and FT, were found in the CSC groups, especially in the non-resolved CSC group. Higher TT/FT concentrations were related to longer disease duration. However, no significant difference was detected in the estrogen levels among different groups. Therefore, TT/FT could be considered in the management of CSC, based on the findings of better designed clinical studies.
Acknowledgments
This work was supported by Development Projects in Tenth People’s Hospital Affiliated to Shanghai Tongji University School of Medicine (SYLCYJ15).
Author Contributions
All authors contributed to data analysis, drafting or revising the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors declare no competing interests.
References
1. Ersoz MG, Karacorlu M, Arf S, Sayman Muslubas I, Hocaoglu M. Retinal pigment epithelium tears: classification, pathogenesis, predictors, and management. Surv Ophthalmol. 2017;62(4):493–505. doi:10.1016/j.survophthal.2017.03.004
2. Vilela M, Mengue C. Central Serous Chorioretinopathy Classification. Pharmaceuticals. 2020;14(1):26. doi:10.3390/ph14010026
3. Chhablani J, Cohen FB, Aymard P. Central serous chorioretinopathy international, multimodal imaging-based central serous chorioretinopathy classification. Ophthalmol Retina. 2020;4(11):1043–1046. doi:10.1016/j.oret.2020.07.026
4. Kaye R, Chandra S, Sheth J, Boon CJF, Sivaprasad S, Lotery A. Central serous chorioretinopathy: an update on risk factors, pathophysiology and imaging modalities. Prog Retin Eye Res. 2020;79:100865. doi:10.1016/j.preteyeres.2020.100865
5. van Rijssen TJ, van Dijk EHC, Yzer S, et al. Central serous chorioretinopathy: towards an evidence-based treatment guideline. Prog Retin Eye Res. 2019;73:100770. doi:10.1016/j.preteyeres.2019.07.003
6. Pan CK, Vail D, Bhattacharya J, Cao M, Mruthyunjaya P. The effect of obstructive sleep apnea on absolute risk of central serous chorioretinopathy. Am J Ophthalmol. 2020;218:148–155. doi:10.1016/j.ajo.2020.05.040
7. Sakurada Y, Leong BCS, Parikh R, Fragiotta S, Freund KB. Association between choroidal caverns and choroidal vascular hyperpermeability in eyes with pachychoroid diseases. Retina. 2018;38(10):1977–1983. doi:10.1097/IAE.0000000000002294
8. Ersoz MG, Arf S, Hocaoglu M, Sayman Muslubas I, Karacorlu M. Patient characteristics and risk factors for central serous chorioretinopathy: an analysis of 811 patients. Br J Ophthalmol. 2019;103(6):725–729. doi:10.1136/bjophthalmol-2018-312431
9. Gundlach BS, Tsui I. Post-traumatic stress disorder is a significant risk factor for central serous chorioretinopathy in male veterans. Am J Mens Health. 2021;15(5):15579883211034990. doi:10.1177/15579883211034990
10. Dhanji S, Fung AT. Central serous chorioretinopathy secondary to subtenon triamcinolone injection: a new hypothesis for the mechanism. Clin Exp Ophthalmol. 2020;48(7):1007–1009. doi:10.1111/ceo.13807
11. Zhou X, Komuku Y, Araki T, et al. Risk factors and characteristics of central serous chorioretinopathy with later development of macular neovascularisation detected on OCT angiography: a retrospective multicentre observational study. BMJ Open Ophthalmol. 2022;7(1):e000976. doi:10.1136/bmjophth-2022-000976
12. Chatziralli I, Kabanarou SA, Parikakis E, Chatzirallis A, Xirou T, Mitropoulos P. Risk factors for central serous chorioretinopathy: multivariate approach in a case-control study. Curr Eye Res. 2017;42(7):1069–1073. doi:10.1080/02713683.2016.1276196
13. Spaide RF, Gemmy Cheung CM, Matsumoto H, et al. Venous overload choroidopathy: a hypothetical framework for central serous chorioretinopathy and allied disorders. Prog Retin Eye Res. 2022;86:100973. doi:10.1016/j.preteyeres.2021.100973
14. Schellevis RL, Altay L, Kalisingh A, et al. Elevated steroid hormone levels in active chronic central serous chorioretinopathy. Invest Ophthalmol Vis Sci. 2019;60(10):3407–3413. doi:10.1167/iovs.19-26781
15. Villard C, Roy J, Bogdanovic M, Eriksson P, Hultgren R. Sex hormones in men with abdominal aortic aneurysm. J Vasc Surg. 2021;74(6):2023–2029. doi:10.1016/j.jvs.2021.06.020
16. Nkrumah G, Paez-Escamilla M, Singh SR, et al. Biomarkers for central serous chorioretinopathy. Ther Adv Ophthalmol. 2020;12:2515841420950846. doi:10.1177/2515841420950846
17. Daruich A, Matet A, Dirani A, et al. Central serous chorioretinopathy: recent findings and new physiopathology hypothesis. Prog Retin Eye Res. 2015;48:82–118. doi:10.1016/j.preteyeres.2015.05.003
18. Conway MD, Noble JA, Peyman GA. Central serous chorioretinopathy in postmenopausal women receiving exogenous testosterone. Retin Cases Brief Rep. 2017;11(2):95–99. doi:10.1097/ICB.0000000000000298
19. Nudleman E, Witmer MT, Kiss S, Williams GA, Wolfe JD. Central serous chorioretinopathy in patients receiving exogenous testosterone therapy. Retina. 2014;34(10):2128–2132. doi:10.1097/IAE.0000000000000198
20. Ciloglu E, Unal F, Dogan NC. The relationship between the central serous chorioretinopathy, choroidal thickness, and serum hormone levels. Graefes Arch Clin Exp Ophthalmol. 2018;256(6):1111–1116. doi:10.1007/s00417-018-3985-x
21. Zakir SM, Shukla M, Simi ZU, Ahmad J, Sajid M. Serum cortisol and testosterone levels in idiopathic central serous chorioretinopathy. Indian J Ophthalmol. 2009;57(6):419–422. doi:10.4103/0301-4738.57143
22. Nuzzi R, Scalabrin S, Becco A, Panzica G. Gonadal Hormones and Retinal Disorders: a Review. Front Endocrinol. 2018;9:66. doi:10.3389/fendo.2018.00066
23. Bolukbasi S, Kandemir Gursel O, Cakir A, Erden B, Karatas G. Retinal structural changes in patients receiving tamoxifen therapy by spectral-domain optical coherence tomography. Cutan Ocul Toxicol. 2020;39(2):115–121. doi:10.1080/15569527.2020.1734816
24. Koulisis N, Moysidis SN, Olmos de Koo LC, Russell CA, Kashani AH. The tipping point: tamoxifen toxicity, central serous chorioretinopathy, and the role of estrogen and its receptors. Am J Ophthalmol Case Rep. 2016;3:8–13. doi:10.1016/j.ajoc.2016.05.004
25. Pauls SD, Du Y, Clair L, et al. Impact of age, menopause, and obesity on oxylipins linked to vascular health. Arterioscler Thromb Vasc Biol. 2021;41(2):883–897. doi:10.1161/ATVBAHA.120.315133
26. Moss ME, Carvajal B, Jaffe IZ. The endothelial mineralocorticoid receptor: contributions to sex differences in cardiovascular disease. Pharmacol Ther. 2019;203:107387. doi:10.1016/j.pharmthera.2019.06.009
27. Wilkinson-Berka JL, Suphapimol V, Jerome JR, Deliyanti D, Allingham MJ. Angiotensin II and aldosterone in retinal vasculopathy and inflammation. Exp Eye Res. 2019;187:107766. doi:10.1016/j.exer.2019.107766
28. Allingham MJ, Mettu PS, Cousins SW. Aldosterone as a mediator of severity in retinal vascular disease: evidence and potential mechanisms. Exp Eye Res. 2019;188:107788. doi:10.1016/j.exer.2019.107788
29. Bourebaba N, Ngo T, Smieszek A, Bourebaba L, Marycz K. Sex hormone binding globulin as a potential drug candidate for liver-related metabolic disorders treatment. Biomed Pharmacother. 2022;153:113261. doi:10.1016/j.biopha.2022.113261
30. Simons P, Valkenburg O, Stehouwer CDA, Brouwers M. Sex hormone-binding globulin: biomarker and hepatokine? Trends Endocrinol Metab. 2021;32(8):544–553. doi:10.1016/j.tem.2021.05.002
31. Marques JVO, Boguszewski CL. Medical therapy in severe hypercortisolism. Best Pract Res Clin Endocrinol Metab. 2021;35(2):101487. doi:10.1016/j.beem.2021.101487
32. Chantarasorn Y, Rasmidatta K, Pokawattana I, Silpa-Archa S. Effects of ketoconazole on the clinical recovery in central serous chorioretinopathy. Clin Ophthalmol. 2022;16:1871–1882.
33. Nielsen JS, Jampol LM. Oral mifepristone for chronic central serous chorioretinopathy. Retina. 2011;31(9):1928–1936. doi:10.1097/IAE.0b013e31821c3ef6
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


