Back to Journals » International Journal of Nanomedicine » Volume 14
The acute toxic effects of platinum nanoparticles on ion channels, transmembrane potentials of cardiomyocytes in vitro and heart rhythm in vivo in mice
Received 18 March 2019
Accepted for publication 12 June 2019
Published 22 July 2019 Volume 2019:14 Pages 5595—5609
DOI https://doi.org/10.2147/IJN.S209135
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Linlin Sun
Cai-Xia Lin,1,2 Jing-Li Gu,2 Ji-Min Cao1
1Key Laboratory of Cellular Physiology (Shanxi Medical University), Ministry of Education, Department of Physiology, Shanxi Medical University, Taiyuan, People’s Republic of China; 2Department of Physiology, Institute of Basic Medical Sciences, Chinese Academy of Medical Sciences, School of Basic Medicine, Peking Union Medical College, Beijing, People’s Republic of China
Background: Platinum nanoparticles (PtNPs) have been considered a nontoxic nanomaterial and been clinically used in cancer chemotherapy. PtNPs can also be vehicle exhausts and environmental pollutants. These situations increase the possibility of human exposure to PtNPs. However, the potential biotoxicities of PtNPs including that on cardiac electrophysiology have been poorly understood.
Methods: Ion channel currents of cardiomyocytes were recorded by patch clamp. Heart rhythm was monitored by electrocardiogram recording. Morphology and characteristics of PtNPs were examined by transmission electron microscopy, dynamic light scattering and electrophoretic light scattering analyses.
Results: In cultured neonatal mice ventricular cardiomyocytes, PtNPs with diameters 5 nm (PtNP-5) and 70 nm (PtNP-70) concentration-dependently (10–9, – 10–5, g/mL) depolarized the resting potentials, suppressed the depolarization of action potentials and delayed the repolarization of action potentials. At the ion channel level, PtNPs decreased the current densities of INa, IK1 and Ito channels, but did not affect the channel activity kinetics. In vivo, PtNP-5 and PtNP-70 dose-dependently (3–10 mg/kg, i.v.) decreased the heart rate and induced complete atrioventricular conduction block (AVB) at higher doses. Both PtNP-5 and PtNP-70 (10–9, – 10–5, g/mL) did not significantly increase the generation of ROS and leak of lactate dehydrogenase (LDH) from cardiomyocytes within 5 mins after exposure except that only very high PtNP-5 (10–5, g/mL) slightly increased LDH leak. The internalization of PtNP-5 and PtNP-70 did not occur within 5 mins but occurred 1 hr after exposure.
Conclusion: PtNP-5 and PtNP-70 have similar acute toxic effects on cardiac electrophysiology and can induce threatening cardiac conduction block. These acute electrophysiological toxicities of PtNPs are most likely caused by a nanoscale interference of PtNPs on ion channels at the extracellular side, rather than by oxidative damage or other slower biological processes.
Keywords: platinum nanoparticle, transmembrane potential, ion channel, arrhythmia, nanotoxicity, heart
Background
Platinum-based preparations/drugs, including platinum nanoparticles (PtNPs) and special PtNPs-delivering systems, have been regularly used in cancer chemotherapy.1–9 PtNPs are considered a nontoxic anticancer nanomaterial relative to other chemotherapeutics.1 In vitro, PtNPs may kill cancer cells via biological and physical ways. For example, PtNPs inhibit the proliferation and induce apoptosis of cancer cells by means of DNA fragmentation10 or via photothermal ablation.8,11,12
Although PtNPs are generally considered nontoxic in cancer chemotherapy, substantial in vitro studies have reported the toxic effects of PtNPs on genes and a variety of cells. For example, PtNPs induce apoptosis on Raw264.7 macrophages.13 PtNPs size-dependently induce DNA strand breaks in human bronchial epithelial cells.14 PtNPs inhibit matrix metalloproteinases and decrease cell viability in L929 fibroblast cells.15 PtNPs induce DNA damage and p53-mediated growth arrest in human lung fibroblasts.16 A few studies also revealed biotoxicity of PtNPs in vivo. In zebrafish embryos, PtNPs induce hatching delay, heart rate (HR) reduction, touch response and axis curvatures.17 PtNPs (1 nm) are hepatotoxic and nephrotoxic and can also exacerbate drug (carbon tetrachloride or cisplatin) toxicity in mice.18
Now that platinum- and PtNP-based therapeutic agents are widely used in cancer therapy, patients with tumors may have more chance to be exposed to these agents. In addition, PtNPs and platinum-group elements are vehicle-exhaust catalysts and are new kinds of environmental pollutants which are hazardous to organisms including humans.19 Environmental PtNP pollution increases the chance of PtNP exposure not only to tumor patients but also to healthy people and all other organisms. Therefore, a thorough understanding of PtNP biotoxicity is warranted. PtNPs may exert acute and chronic toxic effects on organisms including humans. We noticed that PtNPs decrease the HR in zebrafish.17 This report encourages us to investigate the potential toxic effects of PtNPs on cardiac electrophysiology which are seldom investigated till now, also taking into account that cancers usually occur in elder people who have more incidence of cardiovascular diseases and are susceptible to abnormal cardiac electrophysiology and heart rhythm.
The present study aimed to investigate the potential acute toxic effects of PtNPs on cardiac electrophysiology at the molecular, cellular and integrative levels in mice both in vitro and in vivo and the potential underlying mechanisms. As the biological effects of PtNPs may be size-dependent, we selected two sizes of PtNPs to do the experiments: diameters 5 nm (PtNP-5) and 70 nm (PtNP-70). We first observed the acute hazardous effects of PtNPs on the transmembrane potential (TMP) including the resting potential (RP) and the action potential (AP). As ion channel activity is the basis of TMP, we then examined the acute effects of PtNPs on several major ion channels in the cardiomyocytes, including the inwardly rectifying potassium channel (IK1 channel) which takes a major role in the maintenance of RP and late-phase repolarization of AP, the voltage-gated sodium channel (INa channel) which is responsible for the depolarization of AP and the transient outward potassium channel (Ito channel) which is the key channel for the early repolarization of AP and takes a major role in AP repolarization especially in mice cardiomyocytes.20 At the integrative level, we recorded the surface electrocardiogram (ECG) and observed whether these PtNPs would affect HR and rhythm. Results showed that PtNPs of both sizes similarly inhibited the activities of above ion channels, and thus depolarized the RP, suppressed the depolarization and decelerated the repolarization of AP. To our expectation, PtNPs decreased the sinus HR at lower doses and induced complete atrioventricular conduction block (AVB) at higher doses in vivo.
To explore the potential mechanisms of the above electrophysiological toxicities of PtNPs, we observed the time-dependent endocytosis of PtNPs in cardiomyocytes and measured the generation of ROS and leak of lactate dehydrogenase (LDH) in cardiomyocytes after acute exposure to PtNPs. Results revealed that endocytosis, oxidative stress and membrane injury are not responsible for the acute toxicities of PtNPs on cardiac electrophysiology. We suggest that patients with cardiac diseases especially with high susceptibility to AVB should be cautious when using PtNPs for cancer therapy or when encountering serious air pollution with PtNP-containing vehicle exhausts.
Materials and methods
Ethical approval
The cell and animal use protocols were approved by the Life Ethics Committee of Shanxi Medical University and were in compliance with the US National Institutes of Health Guidelines for the Care and Use of Laboratory Animals (NIH Publication 85–23, revised 1985).
Preparation and characterization of PtNPs
Citrate-coated PtNPs with diameters 5 nm (PtNP-5) and 70 nm (PtNP-70) were purchased from nanoComposix (San Diego, CA, USA). Both PtNP-5 and PtNP-70 had a purity of 99.99% according to the manufacturer. The PtNP concentration of the stock solution was 1 mg/mL (Pt) in sodium citrate. Prior to experiments, PtNPs were filter-sterilized, and then lipopolysaccharide (LPS) was examined with limulus reagent (Genscript Biotech. Co., Hong Kong, China). Ultrasonic vibration was performed for 20 mins to disperse the PtNP stock solution before use. A certain amount of stock PtNPs was diluted with 5% isotonic glucose solution for in vivo experiments, or diluted with water or respective extracellular solutions (culture medium, Tyrode’s solution or external solutions for recordings of TMP and channel currents for in vitro experiments).
The morphology of PtNPs was observed using a JEM-1400 plus transmission electron microscope (JEOL Ltd., Tokyo, Japan). A small amount of PtNP stock solution was diluted to 10–5 g/mL with distilled water, and then the diluted PtNP solution was dropped onto a copper grid and TEM was performed. Images of PtNPs were taken with a CCD camera (Veleta, Olympus Soft Imaging Solutions GmbH, Münster, Germany) attached to the electron microscope.
The hydrodynamic diameters and zeta potentials of PtNP suspensions were, respectively, determined with the methods of dynamic light scattering (DLS) and electrophoretic light scattering (ELS) analyses, using a ZetasizerNano ZS90 analyzer (Malvern Instruments Ltd, Malvern, UK). Briefly, stock solutions of both PtNP-5 and PtNP-70 were dispersed in 1 mL of various solutions including distilled water, serum-containing culture medium, Tyrode’s solution and bath solutions for recording TMP and channel currents, incubated for 5 mins, and then DLS and ELS were performed at room temperature. Three independent experiments were performed for each treatment.
Isolation and culture of neonatal ventricular myocytes
Cardiomyocytes were isolated from the ventricles of neonatal (1–2 days old) C57BL6/J mice using an enzymatic method as we reported previously.21 Briefly, mice were decapitated, and hearts were harvested and ventricular tissues were trimmed; then they were put into cold PBS and digested with 0.25% trypsin at 37°C for 8 mins. The digestion was stopped with FBS and cell pellets were harvested. The residual tissues were again digested for another 8 mins. These steps were repeated several times until most ventricular tissues were well digested. The collected cell suspension was filtered with a cell strainer (aperture 74 μm) and then centrifuged at 1,200 rpm for 5 mins. All collected cell pellets were resuspended in high-glucose DMEM added with 100 U/mL penicillin, 100 μg/mL streptomycin and 15% FBS, and primarily cultured in 3.5-cm culture dishes. After culture for 48 hrs, cardiomyocytes were used for patch clamp study.
Patch clamp
Whole-cell recording configuration of the patch clamp technique was used to record channel currents or TMPs of the cultured cardiomyocytes using an EPC-10 amplifier and PatchMaster software (Heka Elektronik, Lambrecht, Germany). Current clamp mode was used to record the TMP, and voltage clamp mode was applied to record channel currents. The pipettes (resistance 5–7 MΩ) were pulled with the P-97 horizontal puller (Sutter Instruments, Novato, CA, USA) using standard-wall borosilicate glass (inner diameter 0.86 mm, outer diameter 1.5 mm). All patch clamp experiments were conducted at room temperature (23±2 °C), and the series resistance was mostly compensated by about 70%.
To record TMP including RP and AP using current clamp mode, the bath solution contained (mmol/L): NaCl 140, KCl 4, CaCl2 2, MgCl2 2, HEPES 5, glucose 5, pH 7.4 adjusted with NaOH. Pipette solution contained (mmol/L): NaCl 10, KCl 140, ethylene glycol diethyl ether diamine tetraacetic acid(EGTA) 2, MgCl2 1, Mg-ATP 5, HEPES 10, pH 7.3 adjusted with KOH. APs were elicited by rectangular current pulses of 1 Hz and 1 ms duration at 125% threshold level. TMPs at baseline and after exposure to PtNP-5 or PtNP-70 (both 10–9 – 10–5 g/mL) for 5 mins were recorded and compared.
Voltage clamp experiments were performed to record the INa, IK1 and Ito of cultured cardiomyocytes at baseline and after PtNP-5 or PtNP-70 exposure at different concentrations (10–9, 10–8, 10–7, 10–6 and 10–5 g/mL).
To record the INa and to establish the I–V curves and activation curves of INa channels, the bath solution contained (in mM): NaCl 15, N-methyl-D-glucamine (NMDG) 125, KCl 2.5, CaCl2 1, MgCl2 1, HEPES 10, pH 7.4 adjusted with HCl. Pipette solution contained (in mmol/L): CsF 105, NaCl 10, EGTA 10, HEPES 10, pH 7.3 adjusted with CsOH. The recording protocols were the same as we reported previously.21 Briefly, the holding potential was set to −90 mV followed by 50-ms step depolarization pulses from −90 mV to +20 mV in 10-mV steps. The measured INa was divided by cell capacitance to obtain current densities. The I–V curves and the activation, inactivation and recovery curves of INa channels were accordingly established using protocols as we described previously.21
To record the IK1, the bath solution (modified Tyrode’s solution) contained (in mmol/L): NaCl 135, KCl 5.4, CaCl2 1.8, MgCl2 1.0, NaH2PO4 0.33, HEPES 10, glucose 10, CdCl2 0.2, pH 7.4 adjusted with NaOH. The pipette solution contained (in mmol/L): EGTA 5, KCl 150, K2-ATP 3, HEPES 5, 4-aminopyridine (4-AP) 5, MgCl2 1, Mg-ATP 1, pH 7.3 adjusted with KOH. The membrane potential was held at –20 mV, and 500-ms step depolarizing voltages ranging from −120 mV to 20 mV were delivered to cells to get the serial IK1. The steady-state activation curves of IK1 channels were established using our reported method.21
To record the Ito, the bath solution (modified Tyrode’s solution) contained (in mmol/L): NaCl 135, KCl 5.4, CaCl2 1.8, MgCl2 1.0, NaH2PO4 0.33, HEPES 10, glucose 10, CdCl2 0.1, BaCl2 0.2, pH adjusted to 7.4 with NaOH. The pipette solution contained (in mmol/L): KCl 150, MgCl2 1, EGTA 5, HEPES 5, K2-ATP 3, pH 7.3 adjusted with KOH. Ito was elicited by 500 ms voltage steps from a holding potential of −40 mV to between −40 and 80 mV in a 10-mV step. The I–V curves and activation curves of Ito channels were accordingly established.
Recording of surface ECG
To evaluate the potential arrhythmogenic effect of PtNPs in vivo, adult male C57BL6/J mice (18–20 g) were anesthetized with intraperitoneal injection of 10% chloral hydrate solution (0.03 mL/kg body weight), and surface ECGs were recorded using the BL-420E physiological data acquisition system (Chendu Taimeng Software Inc., Chengdu, China) at baseline and after intravenous infusion of PtNPs at 3–10 mg/kg with PtNP-5 or PtNP-70 dispersed in 100 μL of 5% isotonic glucose solution when use.
Transmission electron microscopy (TEM)
TEM was performed to observe the morphology of PtNP preparations, and PtNP internalization and intracellular localization in cultured neonatal cardiomyocytes after exposure to PtNP-5 and PtNP-70 for 5 mins and 1 hr. Briefly, cardiomyocytes were incubated with 10–7 g/mL PtNP-5 or PtNP-70 at 37°C for 5 mins and 1 hr, respectively. Cells treated with culture medium (without PtNP treatment) were served as a control. After exposure to PtNPs for 5 mins or 1 hr, cells were washed with PBS, detached with 0.25% trypsin and collected by centrifugation at 1,000 rpm for 4 mins. Detailed TEM procedures could be found in our previous report.21
Measurement of intracellular ROS generation
Intracellular ROS levels were measured using a ROS assay kit (Yeasen Biotechnology, Shanghai, China) to determine whether short-time (5 mins) exposure to PtNPs would lead to oxidative stress in the cardiomyocytes. Briefly, the isolated neonatal cardiomyocytes were plated in 96-well culture plates and cultured for 24–48 hrs. Cells were then incubated with PtNP-5 or PtNP-70 at 10–9 – 10–5 g/mL for 5 mins at 37°C, and then were washed with PBS. Cells without PtNP treatment served as controls. Cells were then incubated with 10 μM 2,7-dichlorodihydrofluorescein diacetate (DCFH-DA) at 37°C for 30 mins and washed with PBS for at least twice. Cell fluorescence was measured with the fluorescence microplate (Synergy 4, BioTek Instruments, Winooski, VT, USA).
Measurement of extracellular lactic dehydrogenase (LDH)
The LDH levels in the culture medium were measured using LDH assay kit (Yeasen Biotechnology) to determine whether short-time (5 mins) exposure to PtNPs would injure the membrane integrity and lead to LDH leaking from the cardiomyocytes. The cultured cardiomyocytes were treated with PtNPs at 10–9 – 10–5 g/mL for 5 mins at 37°C and then washed with PBS. Cells without PtNP treatment served as a control. The procedure was provided by the kit manufacturer.
Statistics
Data are presented as mean ± SEM. Student’s t-test or ANOVA were used for statistical analysis where appropriate. P<0.05 was considered to be statistically significant.
Results
Characterization of PtNPs
TEM images showed that the two kinds of citrate-coated PtNPs were homogeneously spherical, and the average diameters for PtNP-5 (Figure 1A) and PtNP-70 (Figure 1B) well-matched their size scales (5 nm and 70 nm).
Both PtNP-5 and PtNP-70 were well-dispersed in various solutions including water, cell culture medium containing 15% FBS and external solutions for recording AP and INa currents after incubation for 5 mins. PtNP-5 showed zeta potentials (mV) of –54.20±0.44, –14.27±0.16, –24.53±0.22, –14.30±0.15 and –11.40±0.40, respectively, in water, culture medium, Tyrode’s solution, external solution for recording of AP and external solution for INa recording (Figure 1C). PtNP-70 showed zeta potentials (mV) of –39.43±0.62, –18.90±0.18, –26.80±0.07, –10.28±0.33 and –10.04±0.17, respectively, in water, culture medium, Tyrode’s solution, external solution for AP recording and external solution for INa recording (Figure 1D). The zeta potentials of both PtNP-5 and PtNP-70 were more negative in water than in other solutions.
The hydrodynamic diameters for PtNP-5 (Figure 1E) and PtNP-70 (Figure 1F) in different solutions were basically consistent with Gaussian distribution. The variations of zeta potentials and sizes of PtNPs in different solutions might be contributed to adsorption of medium components onto the PtNP surface or mild degree of aggregation of PtNPs in different solutions.
PtNPs depolarize the RPs, suppress the depolarization and delay the repolarization of APs in cultured neonatal mice cardiomyocytes
The TMPs were recorded using the current clamp mode of patch clamp technique in cultured neonatal mice cardiomyocytes. The baseline RP was about –70 mV, the action potential amplitude (APA) was about 110 mV and the APD90 was about 90 ms (Figure 2). PtNP-5 (Figure 2A) concentration (10−9–10−5 g/mL)-dependently depolarized the RP, suppressed the depolarization and delayed the repolarization of AP after exposure for 5 mins. PtNP-5 at 10−9 g/mL only slightly depolarized the RP, while it did not significantly affect the shape of AP. Higher concentrations of PtNP-5 induced more obvious changes of AP shape, including more obvious RP depolarization, APA reduction and APD prolongation (Figure 2A). PtNP-70 (Figure 2B) exerted similar effects on the RP and AP as that induced by PtNP-5 after exposure for 5 mins. Statistical results of TMP parameters showed the quantitative changes of TMPs of cardiomyocytes after exposure to PtNP-5 or PtNP-70 for 5 mins (Figure S1).
PtNPs decrease the current densities of INa channels but do not significantly alter the channel activation, inactivation and recovery kinetics in cultured neonatal cardiomyocytes
Voltage clamp mode was used to record the INa currents. PtNP-5 concentration (10−9–10−5 g/mL)-dependently decreased the INa currents compared with baseline (control) INa currents after exposure for 5 mins (Figure 3A). PtNP-70 exerted similar inhibitory effects on the INa currents as that did by PtNP-5 (Figure 3B). The I–V curves of INa channels showed that both PtNP-5 and PtNP-70 significantly decreased the INa current densities after exposure for 5 mins (Figure 3C and D).
Either PtNP-5 or PtNP-70 at 10−7 g/mL did not significantly affect the activation (Figure S2), inactivation (Figure S3) and recovery (Figure S4) kinetics of INa channels.
PtNPs decrease the current densities but do not significantly alter the activation kinetics of IK1 channels in cultured neonatal mice cardiomyocytes
As PtNPs depolarized the RP of cardiomyocytes (shown in Figure 2), we estimated that PtNPs may suppress the IK1 channels which are known to be responsible for the maintenance of RP. Results confirmed our estimation that both PtNP-5 and PtNP-70 at 10−7 g/mL moderately but significantly decreased the IK1 (Figure 4A and B). However, PtNP-5 and PtNP-70 at 10−7 g/mL did not significantly affect the activation curves of IK1 channels in the cardiomyocytes (Figure 4C).
PtNPs decrease the current densities but do not significantly alter the activation kinetics of Ito channels in cultured neonatal cardiomyocytes
As we observed that PtNPs blunted the early repolarization of AP (shown in Figure 2), and Ito channels are known responsible for the early repolarization of AP, we thus examined the effects of PtNPs on Ito channels. Results showed that both PtNP-5 and PtNP-70 at 10−7 g/mL moderately but significantly suppressed the Ito currents and current densities (Figure 5A and B) but did not significantly affect the activation kinetics of Ito channels in the cardiomyocytes (Figure 5C).
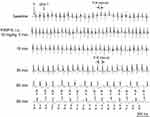
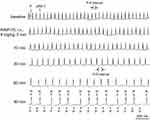
PtNPs dose-dependently decrease the HR, prolong the P-R intervals and finally lead to complete A-V conduction block in adult mice in vivo
The suppressing effects of PtNPs on RP and AP (shown in Figure 2) suggest that PtNPs may induce cardiac conduction block and bradyarrhythmias. We, therefore, observed the effects of PtNPs (i.v.) on the ECGs of mice in vivo. PtNP-5 i.v. at 3 mg/kg did not affect the ECG (Figure S5). PtNP-5 i.v. at 4 mg/kg slightly decreased the sinus HR and prolonged the P-R intervals (degree I AVB) but did not induce other types of arrhythmias within the observed 90 mins (Figure S6). PtNP-5 i.v. at 6 mg/kg decreased the HR and significantly prolonged the P-R intervals but did not induce complete AVB within 90 mins (Figure S7). PtNP-5 i.v. at 8 mg/kg decreased the HR and severely prolonged the P-R intervals, but also did not induce complete AVB within 90 mins (Figure S8). PtNP-5 i.v. at 10 mg/kg significantly decreased the HR and further induced complete AVB but did not lead to cardiac asystole and animal death within 90 mins (Figure 6). PtNP-70 i.v. at doses of 3–10 mg/kg exerted similar effects on the ECGs including the induction of complete AVB at 8–10 mg/kg, and also did not induce cardiac asystole and animal death within the observed 90 mins after intravenous injection (Figure 7; Figures S9–S12). No tachyarrhythmias (such as ventricular tachycardia and ventricular fibrillation) were observed after PtNP-5 or PtNP-70 intravenous injection.
Note that both PtNP-5 and PtNP-70 also slowed down the intraventricular conduction. For example, PtNP-5 widened QRS complex at higher doses (shown in Figures 6 and 7).
PtNPs do not significantly induce oxidative stress and membrane injury after short-time exposure in cultured neonatal cardiomyocytes
To identify whether the above acute toxic effects of PtNPs on cardiac electrophysiology were caused by oxidative stress and cell membrane injury, we measured the acute effects of PtNPs (5 mins after exposure) on the generation of intracellular ROS and LDH leak from the cardiomyocytes which represents surface membrane injury. Results showed that both PtNP-5 and PtNP-70 at a concentration range of 10–9 – 10–5 g/mL did not significantly increase the intracellular ROS levels (Figure S13A). PtNP-5 and PtNP-70 at a concentration range of 10–9 – 10–6 g/mL also both did not significantly increase LDH leaking, except that PtNP-5 at 10–5 g/mL slightly but significantly increased LDH leak (Figure S13B). These results suggest that oxidative stress and membrane injury was not a leading mechanism underlying the acute toxic effects of PtNPs on cardiac electrophysiology.s
The rapid hazardous effects of PtNPs on cardiac electrophysiology are internalization independent
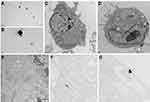
Nanoparticles may impair the cell function through an extracellular way or an intracellular way. Some studies have demonstrated that nanoparticles can be internalized by cells after certain time of exposure. To determine whether the rapid hazardous effects of PtNPs on cardiac electrophysiology were actually caused by extracellular or intracellular PtNPs, we used TEM to examine the internalization of PtNPs in cultured neonatal cardiomyocytes after cell exposure to PtNP-5 or PtNP-70 (both 10–5 g/mL) for 5 mins and 1 hr. Figure 8A and B showed the appearance of PtNP-5 or PtNP-70, respectively, in the culture medium (extracellular space); note that the sizes of these two types of PtNPs just matched the scale. Figure 8C shows a control cardiomyocyte without exposure to PtNPs. Both PtNP-5 (Figure 8D) and PtNP-70 (Figure 8E) could not be internalized within 5 mins after exposure at 37ºC, and neither PtNP-5 nor PtNP-70 was found inside the cell. After exposure to PtNP-5 or PtNP-70 for 1 hr, both PtNP-5 (Figure 8F) and PtNP-70 (Figure 8G) were found internalized by the cardiomyocytes.
Discussion
Along with the increased clinical use of PtNP-based cancer therapeutic agents and the worsened air pollution with vehicle PtNP exhausts, attention ought to be paid to the potential acute and chronic biotoxicities of PtNPs. However, investigations into the toxicities of PtNPs are insufficient at present. Most of the studies on PtNP toxicities are conducted on the cellular level, seldom studies have been done at the integrative level, and no report is available to show the potential toxic effects of PtNPs on cardiac electrophysiology. One of the reasons may be that Pt/PtNP-based therapeutic agents are basically not very toxic and are even considered nontoxic.1 Even so, there are still some reports showing the toxicities of PtNPs at the cellular and integrative levels as described above.13–18 The present study focused on the acute toxic effects of PtNPs on the cardiac electrophysiology in a mouse model both in vitro and in vivo. We did find substantial toxicities of PtNPs on the cardiac electrophysiology at different levels. PtNPs exerted these toxic effects in a concentration/dose-dependent manner both in vitro and in vivo, and the lowest toxic concentration/dose was 10−9 g/mL in vitro and 4 mg/kg i.v. in mice in vivo. Generally, PtNPs suppressed the activities of several ion channels, affected the depolarization and repolarization of AP and thus changed the shape of TMP and decreased the HR and induced complete AVB at higher doses in vivo. Cardiac ion channel activities endow the maintenance of RP and the formation of AP in the cardiomyocytes. AP conduction in the myocardium triggers the excitation–contraction coupling and thus accomplish the pumping function of the heart. The effects of PtNPs on ion channels better explain the effects of PtNPs on TMP and heart rhythm in the present study.
We first observed that PtNPs of both sizes depolarized the RP in a concentration-dependent manner. It is known that mild-to-moderate RP depolarization increases the excitability and the susceptibility to delayed after-depolarization and triggered activity, and severe RP depolarization would lead to decrease and even loss of excitability in the cardiomyocytes. Both PtNP-5 and PtNP-70 at the highest tested concentration (10−5 g/mL) depolarized the RP by about 20 mV (roughly from −70 mV to −50 mV), and this intensity of RP depolarization did not reach the membrane potential threshold for Na+ channel inactivation (usually at −40 mV). In other words, the RP-depolarizing effect of PtNPs was not as severe as silver nanoparticles (AgNPs), AgNPs could severely depolarize the RP and lead to total loss of cardiomyocyte excitability at higher concentration (10−6 g/mL).21 This may be one reason why PtNPs could not lead to loss of cardiomyocyte excitability and cardiac asystole even at higher concentration in vitro or higher dosage in vivo. The RP-depolarizing effect of PtNPs was mainly contributed to the inhibitory effect of PtNPs on IK1 channels, as we observed the suppressing effect of PtNPs on the IK1 currents, and IK1 currents are considered the primary conductance controlling the RP.22
We then observed that both PtNP-5 and PtNP-70 significantly suppressed the depolarization of AP with similar efficacies, as shown by the reduction of APA (shown in Figure 2 and Figure S1). These effects of PtNPs contributed to the repressing effects of PtNPs on voltage-gated Na+ channels as we present here. It is known that normal conduction of myocardial excitation depends on a normal depolarization which is reflected by APA. Fall of APA to a certain extent would harm the excitation conduction and lead to conduction block as we observed here.
Except for suppressing the depolarization, PtNPs also delayed the repolarization of AP, as shown by the prolongations of APD50 and APD90 (shown in Figure 2 and Figure S1). Generally, APD decides the effective refractory period (ERP). Prolongation of APD means longer ERP and increases the severity of functional conduction block. Therefore, suppressions of AP depolarization and repolarization may both contribute to cardiac conduction block. We observed the inhibitory effects of PtNPs on Ito and IK1 channels which can explain why PtNPs prolonged the APD, because these two channels provide the main outward currents which accelerate the AP repolarization in mice cardiomyocytes. We did not check the effect of PtNPs on the delayed rectifier K+ (IK) channels (including IKr and IKs), because the expressions of IK channels are rapidly declined in mouse myocardium after birth, and both IKr and IKs could not be detected in adult cardiomyoctes.23 In addition, we observed that PtNPs did not affect the activation, inactivation and recovery kinetics of INa channels, and also had no effect on the activation kinetics of IK1 and Ito channels. These properties of PtNPs on channel kinetics were different from that of AgNPs; AgNPs not only decreased the channel current densities but also changed the channel kinetics as we previously reported in cardiomyocytes.21 This may be one reason why PtNPs were less toxic than AgNPs.
The inhibitions of INa, Ito and IK1 by PtNPs and subsequent changes of RP and AP suggest that PtNPs may induce cardiac conduction block and bradyarrhythmias. Results proved our speculation that PtNPs prolonged the P-R intervals (degree I AVB) at lower doses and caused complete (degree III) AVB at higher doses. In addition to causing AVB, PtNPs also slowed down the intraventricular conduction, as indicated by the widened QRS complex at higher doses (shown in Figures 6 and 7). Repression of Na+ channels by PtNPs could underlie this effect and partially explained why AVB and hampering of ventricular conduction happened. In addition, direct inhibition of IK1 and INa channels by PtNPs may also disturb the voltage-dependent characters of other channels and indirectly affect the activities of these channels. These direct and indirect effects of PtNPs on ion channels might together cause bradyarrhythmias as we observed.
We noticed that PtNP-induced arrhythmias were dominantly bradyarrhyrthmias which mainly exhibited decelerated sinus HR and degrees I and III AVB. No tachyarrhythmia was observed after PtNP exposure. The inhibitory effect of PtNPs on INa channels may be one reason, because tachyarrhythmias require rapid excitation conduction, while inhibition of INa channels would prohibit the high-efficacy AP conduction in the myocardium. In addition, APD prolongation also hampers the high-frequency AP initiation which is necessary for tachyarrhythmias.
It is known that the toxicity of nanoparticles is size dependent. For example, Isoda et al.18 reported that PtNP-1 (diameter 1 nm, i.v.), but not PtNP-8, showed acute hepato-renal toxicity in mice; Konieczny et al.19 showed that smaller PtNPs (5.8 nm) exhibited a more deleterious effect on DNA stability than bigger PtNPs (57 nm) in primary keratinocytes. In the present study, it is likely that PtNP-5 showed slightly higher inhibitory effects on INa, Ito and IK1 than PtNP-70 in cardiomyocytes in vitro, while PtNP-5 and PtNP-70 exerted similar effects on the TMP in vitro and heart rhythm in vivo. Overall, we only find mild, but not striking, difference in the ion channel toxicity between PtNP-5 and PtNP-70.
Upon the mechanisms underlying the acute toxic effects of PtNPs on cardiac electrophysiology, we noticed that the toxic effects appeared rapidly (within 5 mins) either at the cellular AP level (shown in Figure 2) or at the ion channel level (see Figures 3–5). Even at the integrative level, HR showed rapid (within 5–10 mins) and a significant decrease after PtNP exposure (see Figures 6 and 7). The in vivo effects of nanoparticles usually appear slower than their in vitro effects, as nanoparticles need time to travel and diffuse to the target cells under in vivo condition. These phenomena suggest that PtNPs exerted the acute toxic effects in an endocytosis-independent manner. The TEM results confirmed this viewpoint: PtNP endocytosis could not occur within 5 mins but occurred 1 hr after exposure. Usually, the completion of endocytosis process needs at least 10 mins even in cells with high phagocytic capability such as macrophages as we previously observed,21,24 and cells with lower phagocytic capability need longer time to complete the internalization of nanoparticles such as 3T3 L1 cells25 and cardiomyocytes. Furthermore, potential changes of gene expression and other relatively slower biological processes also unlikely underlie the acute toxic effects of PtNPs, as these processes need more than 5 mins. This quick action pattern of PtNPs on cells is similar to AgNPs21 but is different from multiwalled carbon nanotubes, the latter affect cells on the intracellular side and thus is endocytosis dependent as we have reported previously.26 Another potential mechanism might be oxidative stress caused by PtNPs.27,28 However, we observed here that short-time exposure (5 mins) of cardiomyocytes to PtNPs did not significantly increase the production of ROS and membrane injury as indicated by LDH leak. A third potential mechanism might be that Pt ions released from PtNPs might exert the acute toxicity. However, Pt is a well-known inert metal. It is extremely stable and cannot be oxidized or dissolved in most regular mediums such as water and physiological solutions, while it can only be dissociated to release Pt ions at extreme conditions such as in aqua regia.29,30 Therefore, Pt ions could not be a contributor to the acute toxicity of PtNPs. Taken together, in terms of mechanism, we suggest that PtNPs exerted the acute toxic effects on cardiac electrophysiology via a nanoscale-level interference with ion channels at the extracellular side, and not likely via changes of protein expression or other slower biological processes, oxidative stress and membrane injury.
The present study may have a translational potential. By scanning the concentration/dosage ranges and respective cardiomyocyte/heart electrophysiological responses both in vitro and in vivo, we found that the lowest toxic concentration of PtNPs was 10–9 g/mL in vitro and the lowest toxic dose of PtNPs was 4 mg/kg (i.v.) in mice in vivo. In real life, people may encounter risk of PtNP exposure in situations of medical PtNP administration, occupational exposure, severe environmental pollution and accidentally, poisoning. The above measures in mice shown here might be used as references when evaluating PtNP toxicities and exposure level–toxicity relations in human. In addition, blood Pt levels after exposure are also valuable in the assessment of exposure-toxicity associations and thus need to be measured both in animal and human in the future.
Conclusion
PtNPs (PtNP-5 and PtNP-70) rapidly (within 5 mins) and concentration-dependently reduced the current densities of IK1, INa and Ito channels with similar efficacies but did not significantly affect the kinetics of these channels in cultured neonatal mice cardiomyocytes. These channel effects of PtNPs could better explain the hazardous effects of PtNPs on the TMP, including depolarization of RP and repressions of AP depolarization and repolarization. In vivo, PtNPs dose-dependently decreased the sinus HR and led to degrees I and III AVB, but could not cause cardiac asystole and animal death at the highest tested dose (10 mg/kg, i.v.) in adult mice. Overall, the cardiac electrophysiological toxicity of PtNPs is lower than AgNPs as we previously reported.21 PtNPs exerted these rapid toxicities on cardiac electrophysiology directly via a nanoscale-level interference on ion channels at the extracellular side, but not via ROS generation, membrane injury and other slower biological processes. The study warns that PtNPs are toxic to cardiac electrophysiology and can lead to threatening cardiac conduction block. Attention should be paid to patients especially those with heart diseases and those encountering PtNP exposure.
Limitations
The first limitation of this study was that we could not demonstrate the exact mechanism by which PtNPs affect the cardiac electrophysiology, although results suggest that extracellular PtNPs directly interfere with ion channels at the nanoscale level. The second limitation was that we did not observe the effects of PtNPs on other ion channels such as ICa-L, IK and IKur channels in the cardiomyocytes. A third limitation was that we did not measure the blood Pt level after intravenous PtNP injection. Investigations into the association between blood Pt level and exposure dosage are warranted in future studies both in animal and human.
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (NSFC) (81670313) and Shanxi “1331 Project” Key Subjects Construction (1331KSC).
Author contributions
CXL and JMC designed the study; CXL performed major parts of the experiments and analyzed the data and JLG performed the transmission electron microscopy (TEM) experiments. All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Samadi A, Klingberg H, Jauffred L, Kjær A, Bendix PM, Oddershede LB. Platinum nanoparticles: a non-toxic, effective and thermally stable alternative plasmonic material for cancer therapy and bioengineering. Nanoscale. 2018;10:9097–9107. doi:10.1039/c8nr02275e
2. Pasha SS, Fageria L, Climent C, et al. Evaluation of novel platinum(ii) based AIE compound-encapsulated mesoporous silica nanoparticles for cancer theranostic application. Dalton Trans. 2018;47:4613–4624. doi:10.1039/c7dt04232a
3. Parker JP, Ude Z, Marmion CJ. Exploiting developments in nanotechnology for the preferential delivery of platinum-based anti-cancer agents to tumours: targeting some of the hallmarks of cancer. Metallomics. 2016;8:43–60. doi:10.1039/c5mt00181a
4. Desale SS, Soni KS, Romanova S, Cohen SM, Bronich TK. Targeted delivery of platinum-taxane combination therapy in ovarian cancer. J Control Release. 2015;220:651–659. doi:10.1016/j.jconrel.2015.09.007
5. Shi T, Jiang R, Yu J, et al. SGOG-OV/AICE investigators. Addition of intraperitoneal cisplatin and etoposide to first-line chemotherapy for advanced ovarian cancer: a randomised, phase 2 trial. Br J Cancer. 2018;119:12–18. doi:10.1038/s41416-018-0036-7
6. Sarkar A. Novel platinum compounds and nanoparticles as anticancer agents. Pharm Pat Anal. 2018;7:33–46. doi:10.4155/ppa-2017-0036
7. Xiao H, Yan L, Dempsey EM, et al. Recent progress in polymer-based platinum drug delivery systems. Prog Polym Sci. 2018;87:70–106. doi:10.1016/j.progpolymsci.2018.07.004
8. Yu Y, Xu Q, He S, et al. Recent advances in delivery of photosensitive metal-based drugs. Coord Chem Rev. 2019;387:154–179. doi:10.1016/j.ccr.2019.01.020
9. He S, Li C, Zhang Q, et al. Tailoring platinum(IV) amphiphiles for self-targeting all-in-one assemblies as precise multimodal theranostic nanomedicine. ACS Nano. 2018;12:7272−7281. doi:10.1021/acsnano.8b03476
10. Şahin B, Aygün A, Gündüz H, et al. Cytotoxic effects of platinum nanoparticles obtained from pomegranate extract by the green synthesis method on the MCF-7 cell line. Colloids Surf B Biointerfaces. 2018;163:119–124. doi:10.1016/j.colsurfb.2017.12.042
11. Wang C, Cai X, Zhang J, et al. Trifolium-like platinum nanoparticle-mediated photothermal therapy inhibits tumor growth and osteolysis in a bone metastasis model. Small. 2015;11:2080–2086. doi:10.1002/smll.201403315
12. Cong Y, Xiao H, Xiong H, et al. Dual drug backboned shattering polymeric theranostic nanomedicine for synergistic eradication of patient-derived lung cancer. Adv Mater. 2018;30:1706220. doi:10.1002/adma.201706220
13. Loan TT, Do LT, Yoo H. Platinum nanoparticles induce apoptosis on Raw 264.7 macrophage cells. J Nanosci Nanotechnol. 2018;18:861–864. doi:10.1166/jnn.2018.14874
14. Lebedová J, Hedberg YS, Odnevall Wallinder I, Karlsson HL. Size-dependent genotoxicity of silver, gold and platinum nanoparticles studied using the mini-gel comet assay and micronucleus scoring with flow cytometry. Mutagenesis. 2018;33:77–85. doi:10.1093/mutage/gex027
15. Hashimoto M, Yamaguchi S, Sasaki J, et al. Inhibition of matrix metalloproteinases and toxicity of gold and platinum nanoparticles in L929 fibroblast cells. Eur J Oral Sci. 2016;124:68–74. doi:10.1111/eos.12235
16. Asharani PV, Xinyi N, Hande MP, Valiyaveettil S. DNA damage and p53-mediated growth arrest in human cells treated with platinum nanoparticles. Nanomedicine (Lond). 2010;5:51–64. doi:10.2217/nnm.09.85
17. Asharani PV, Lianwu Y, Gong Z, Valiyaveettil S. Comparison of the toxicity of silver, gold and platinum nanoparticles in developing zebrafish embryos. Nanotoxicology. 2011;5:43–54. doi:10.3109/17435390.2010.489207
18. Isoda K, Daibo T, Yushina K, et al. Hepatotoxicity, nephrotoxicity, and drug/chemical interaction toxicity of platinum nanoparticles in mice. Pharmazie. 2017;72:10–16. doi:10.1691/ph.2017.6758
19. Konieczny P, Goralczyk AG, Szmyd R, et al. Effects triggered by platinum nanoparticles on primary keratinocytes. Int J Nanomed. 2013;8:3963–3975.
20. Priest BT, McDermott JS. Cardiac ion channels. Channels (Austin). 2015;9:352–359. doi:10.1080/19336950.2015.1076597
21. Lin CX, Yang SY, Gu JL, Meng J, Xu HY, Cao JM. The acute toxic effects of silver nanoparticles on myocardial transmembrane potential, INa and IK1 channels and heart rhythm in mice. Nanotoxicology. 2017;11:827–837. doi:10.1080/17435390.2017.1367047
22. Anumonwo JM, Lopatin AN. Cardiac strong inward rectifier potassium channels. J Mol Cell Cardiol. 2009;48:45–54. doi:10.1016/j.yjmcc.2009.08.013
23. Wang L, Feng ZP, Kondo CS, Sheldon RS, Duff HJ. Developmental changes in the delayed rectifier K+ channels in mouse heart. Circ Res. 1996;79:79–85.
24. Gu J, Xu H, Han Y, et al. The internalization pathway, metabolic fate and biological effect of superparamagnetic iron oxide nanoparticles in the macrophage-like RAW264.7 cell. Sci China Life Sci. 2011;54:793–805. doi:10.1007/s11427-011-4215-5
25. Xu H, Dai W, Han Y, et al. Differential internalization of superparamagnetic iron oxide nanoparticles in different types of cells. J Nanosci Nanotechnol. 2010;10:7406–7410.
26. Li H, Tan XQ, Yan L, et al. Multi-walled carbon nanotubes act as a chemokine and recruit macrophages by activating the PLC/IP3/CRAC channel signaling pathway. Sci Rep. 2017;7:226. doi:10.1038/s41598-017-00386-3
27. Nejdl L, Kudr J, Moulick A, et al. Platinum nanoparticles induce damage to DNA and inhibit DNA replication. PLoS One. 2017;12:e0180798. doi:10.1371/journal.pone.0180798
28. Sørensen SN, Engelbrekt C, Lützhøft HH, et al. A multimethod approach for investigating algal toxicity of platinum nanoparticles. Environ Sci Technol. 2016;50:10635–10643. doi:10.1021/acs.est.6b01072
29. Hodnik N, Baldizzone C, Polymeros G, et al. Platinum recycling going green via induced surface potential alteration enabling fast and efficient dissolution. Nat Commun. 2016;7:13164. doi:10.1038/ncomms13164
30. Marinho RS, Afonso JC, da Cunha JW. Recovery of platinum from spent catalysts by liquid-liquid extraction in chloride medium. J Hazard Mater. 2010;179:488–494. doi:10.1016/j.jhazmat.2010.03.029
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.