Back to Journals » Therapeutics and Clinical Risk Management » Volume 15
The accuracy assessment of presepsin (sCD14-ST) for mortality prediction in adult patients with sepsis and a head-to-head comparison to PCT: a meta-analysis
Authors Zhu Y, Li X, Guo P, Chen Y, Li J, Tao T
Received 18 December 2018
Accepted for publication 16 April 2019
Published 13 June 2019 Volume 2019:15 Pages 741—753
DOI https://doi.org/10.2147/TCRM.S198735
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Deyun Wang
Ying Zhu,1,2 Xuehui Li,1 Peiyan Guo,1 Yuhan Chen,3 Jiandong Li,1 Tianzhu Tao4
1Department of Respiratory Medicine, The Seventh Medical Center of PLA General Hospital, Beijing, People’s Republic of China; 2Department of Respiratory and Critical Care Medicine, Shanghai East Hospital, Tongji University, Shanghai, People’s Republic of China; 3Affiliated BaYi Children’s Hospital, The Seventh Medical Center of PLA General Hospital, Beijing, People’s Republic of China; 4Department of Anesthesiology, Air Force Medical Center, PLA, Beijing, People’s Republic of China
Objective: The soluble cluster of differentiation 14 subtype (sCD14-ST) or presepsin has recently been identified as a promising biomarker in sepsis. The present meta-analysis is performed to assess the prognostic value of presepsin in septic patients. Further, we compare the prognostic performance between presepsin and procalcitonin (PCT) in predicting all-cause mortality in these patients.
Methods: A systemic and comprehensive search was conducted in PubMed, Embase and Cochrane databases by using Exploded Medical Subject Headings and appropriate corresponding keywords. Studies were eligible if they assessed the prognostic value of presepsin in sepsis and provided sufficient information to construct a 2×2 contingency table. A bivariate meta-analysis model was used to calculate the pooled sensitivity, specificity, positive/negative likelihood ratios and diagnostic odds ratio. The Chi-square and I2, index were used to assess the heterogeneity and inconsistency. The Deek’s funnel plot asymmetry test was used to assess the likelihood of publication bias.
Results: Nine publications, comprising 1,561 patients, were included in this study. The overall area under the receiver operating characteristic curve (AUROC) of presepsin was 0.77 (95% CI, 0.73–0.81) with a pooled prognostic sensitivity (SEN) and specificity (SPE) of 0.83 (95% CI, 0.72–0.90) and 0.69 (95% CI, 0.63–0.74), respectively. Additionally, the PLR, NLR and DOR of presepsin were 2.6 (95% CI, 2.1–3.3), 0.25 (95% CI, 0.15–0.44) and 10 (95% CI, 5–22), respectively. The AUROC of PCT was 0.81 (95% CI, 0.78–0.84) with a pooled SEN of 0.76 (95% CI, 0.55–0.89) and SPE of 0.74 (95% CI, 0.33–0.94). There is no statistically significant difference in the performance of pooled SEN and SPE between presepsin and PCT, with a p value of 0.39 and 0.71, respectively.
Conclusions: Based on the results of this meta-analysis, both presepsin and PCT are promising biomarkers for the prognosis of mortality in sepsis.
Keywords: presepsin, sCD14-ST, procalcitonin, prognostic, sepsis, mortality
Introduction
Sepsis is a life-threatening organ dysfunction characterized by a dysregulated host response to infection. Sepsis accounts for about 25% of intensive care unit (ICU) admissions, with a mortality range from 10% to 52%.1–3 Despite advances and breakthroughs in antibiotic treatment or other managements of bundled care for the patients with sepsis, the fatality rate remains unacceptably high and the incidence is still increasing.4,5 To improve sepsis-related survival, it is imperative to early recognize septic patients at high risk of poor clinical outcomes. However, due to the lack of an ideal prognostic indicator, timely identification of patients at risk of dying from the condition is challenging.
To identify mortality risk and ensure the appropriate therapeutic interventions, clinical scores have been introduced. In clinical practice, the most commonly used clinical scores are the Acute Physiology and Chronic Health Evaluation (APACHE) II score and Sequential Organ Failure Assessment (SOFA) score.6,7 They are validated as the most recognized tools to stratify the severity of the condition. However, with the increasing controversies and complicated methods for using these clinical scoring systems, a growing body of evidence has proposed blood biomarkers as promising alternatives.8
Hundreds of the circulating biomarkers have been investigated for potential use in improving early diagnosis and monitoring the outcome of septic patients.9–11 However, due to the controversial and uncertain factors in clinical value, fewer biomarkers were used in the practical medical work. Among the most extensively studied in recent years, PCT, CRP, pentraxin-3, IL-6 and myeloid cells expressing triggering receptor-1 (TREM-1) have been identified as probable predictors in sepsis.12–14 Among those, procalcitonin (PCT) is considered as the most preferable biological predictors and is a widely recognized biomarker in predicting outcomes in septic patients.15,16
Presepsin, a truncated N-terminal fragment of CD14, also known as soluble cluster of differentiation 14 subtype (sCD14-ST), has recently attracted great clinical interest and risen up as a promising prognostic serum marker in sepsis. The generation and release of the presepsin are cleaved from the monocyte/macrophage-specific CD14 receptor complex which is binding with bacterial lipopolysaccharide (LPS) and LPS binding protein (LPB) during the pro-inflammatory signaling cascade.17–20 Previous studies showed that presepsin existed in much higher concentration in septic patients than that in healthy individuals and those with Systemic Inflammatory Response Syndrome (SIRS).1,17,21 Moreover, publications revealed that the level of presepsin increased within 2 hrs and peaked at 3 hrs after infection, which has proved to be reliable for early stage sepsis diagnosis and prognostic stratification.18,19 However, some researchers proposed that presepsin may not outweigh traditional biomarkers in prognosis of survival rate.22,23 Yang et al recently published a meta-analysis to assess the prognostic value of presepin in adult sepsis patients, which demonstrated that presepsin did have some mortality prediction value as evidenced by a significantly higher level of presepsin in non-survivors.24 We conducted this systematic review of the literature and a meta-analysis to further evaluate the relationship between presepsin and hospital mortality and provide additional information of pooled sensitivity and specificity to determine its prognostic performance. Since PCT is a widely used predictor in mortality, we further performed a head-to-head comparison with this well-known serum marker in the prognostic value of septic patients.
Materials and methods
Search strategy and selection criteria
We systematically searched studies using PubMed, Embase, and the Cochrane Library without language limitation through 15th August, 2018. The search terms were as follows: (presepsin OR “sCD14-ST” OR “sCD14-subtype” OR “soluble cluster of differentiation 14 subtype”) AND (sepsis OR SIRS OR “systemic inflammatory response syndrome” OR “septic shock” OR “severe sepsis” OR “bacteria infection”) AND (mortality OR prediction OR prognosis OR “prognostic value” OR “prognostic biomarker”). Additionally, to obtain potentially relevant articles, we reviewed the reference list of the selected articles, and one was included in this study.2,25
Eligible studies were required to have a well-defined reference standard for patients recruited (sepsis or severe sepsis or septic shock) based on the criteria of the American College of Chest Physicians/Society of Critical Care Medicine in 1991/2001 (sepsis 1.0 and 2.0), International Guidelines for Management of Severe Sepsis and Septic Shock 2012 or the third international consensus definitions for sepsis and septic shock (sepsis 3.0).3,26–28 Further, a 2*2 contingency table should be available. If multiple studies reused the same patient sample, only the most recent article or the most informative article was included. For studies that evaluated the prognostic accuracy of presepsin levels at multiple time points, we chose the data relied on the initial presepsin levels of patients admitted to the ED or ICU. For studies predicting mortality in different follow-up periods across studies, we basically used the data on 28 days, 30 days and in-hospital stays if no exact time course was mentioned. Followed modalities such as reviews, letters, editorials, case reports, meeting abstracts, animal experiments or meta-analysis, and articles involving pediatric patients were considered ineligible. Two investigators (Zhu Y and Li JD) independently conducted the search strategy and assessed the studies. Any disagreements were resolved by a third reviewer (Tao TZ).
Data extraction and quality evaluation
Descriptive data were extracted from the selected preferences, including the following items: 1) Surname of the first author and year of publication, 2) country and clinical setting, 3) study design, 4) characteristics of participants such as sample size, gender and age, 5) cut-off point, 6) true positives (TP), 7) false positives (FP), 8) false negatives (FN), 9) true negatives (TN), 10) the overall area under the ROC (AUROC), 11) sensitivity (SEN) and 12) specificity (SPE). Quality Assessment of Diagnostic Accuracy Studies (QUADAS-2) was performed to assess the risk of bias in each study, which was recommended by the Cochrane collaboration for the quality assessment of diagnostic studies.29,30
Statistical analysis
All statistical and meta-analyses were conducted by using the MIDAS module in the software of STATA version 13 (Stata Corporation, College Station, TX, USA) Based on the hierarchical summary receiver operating characteristic (HSROC) method, the pooled sensitivity (SEN), specificity (SPE), diagnostic odds ratio (DOR), positive likelihood ratio (PLR), negative likelihood ratio (NLR) and the area under the receiver operating characteristic curve (AUC) were calculated for meta-analysis in the prognostic value of presepsin and PCT.
To further evaluate the performance of the predicting assessment, a Chi-square (x2) test and the inconsistency (I2) metric were used to check heterogeneity of the included studies. The latter expresses the variability caused by heterogeneity between the studies in the form of a percentage. A value of I2 greater than 50% was considered sustainable heterogeneity among selected studies.31 The Deek’s funnel plot asymmetry test was used to assess the likelihood of publication bias. In addition, Fagan’s nomogram was conducted to calculate the post-test probability (PTP).32
Results
Characteristics of included studies
A total of nine publications including 1,561 patients were selected from 206 records by searching Pubmed (n=21), Cochrane Library databases (n=10) and Embase (n=175). The included studies were published from 2011 to 2017 (Figure 1). Three studies were conducted in Europe,22,33,34 two in Africa,35,36 two in Eastern Asia,34,37 one in South America38 and one was missing this information.25 All included studies were published in English. The mean age of the populations varied between 55.2 and 71.5 years, and the proportion of men ranged from 30.3% to 72.4%. Five studies were performed in the ICU,22,33,35,36,39 three in the ED37,38,40 and one in both the ICU and ED.34 There are six prospective studies,22,35–39 two were designed as retrospective recruitments33,34 and one is unknown.25 Of these, diagnosis as sepsis was defined as “gold standard” by the criteria in 1991 ACCP/SCCM consensus conference (1992 criteria) in four studies,22,25,38,39 by 2001 ACCP/SCCM consensus conference in two,35,37 by the third international consensus definitions for sepsis and septic shock (sepsis-3) in two34,36 and one was carried out according to the Surviving Sepsis Campaign: International Guidelines for Management of Severe Sepsis and Septic Shock 2012.33 Levels of the circulating presepsin were measured by a chemiluminescent immunoassay on a PATHFAST immunoanalyzer in all studies (Mitsubishi Chemical or LSI Medience Corporation, Tokyo, Japan or Mitsubishi Chemical Europe GmbH, Dusseldorf, Germany). Details of included studies involving data of presepsin and PCT are shown in Table 1.
 | Table 1 Chracteristics of presepsin and PCT and performance for predicting mortality of sepsis patients |
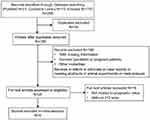 | Figure 1 Flow chart of identification, inclusion and exclusion of studies for the meta-analysis. |
Assessment of studies quality
Details of the methodological quality assessments of nine included studies are summarized by using QUADAS-2 tool in Table 2 and Figure 2. All included studies showed acceptable quality based on the methodological assessment. All studies scored “low” in the domain of risk of bias in the reference standard since the 1991/2001 ACCP/SCCM consensus conference, sepsis-3.0 and Surviving Sepsis Campaign: International Guidelines for Management of Severe Sepsis and Septic Shock were all recognized for diagnosing sepsis in different periods.3,26–28 In patient selection, seven studies providing the criteria of inclusion and exclusion were scored “low” risk, two studies that did not show enough information marked “unclear” and another study that was designed as case-control scored “high”. Regarding the risk of bias of index tests, six studies not pre-specifying a threshold were defined as “high” risk, for two being designed as retrospective recruitment and one missing the information given “unclear”. As for patient flow and timing domain, seven studies scored “low” and two were “high” since some of the patients were lost to follow up.
 | Table 2 Risk of bias and applicability concerns |
 | Figure 2 Risk of bias and applicability concerns. |
Egger regression, investigation of heterogeneity and publication bias
The Egger’s regression model was performed to assess the likelihood of publication bias for nine included studies of presepsin and seven of PCT. The Deek’ funnel plot revealed that the slope coefficient for presepsin was associated with a p-value of less than 0.001 and 0.1 for PCT,suggesting the existence of significant publication bias for presepsin but not PCT in this meta-analysis (Figure 3). The overall I2 values of presepsin were 82% (95% CI, 62–100) and 99% (95% CI, 98–99), indicating the significant presence of heterogeneity for both biomarkers.
 | Figure 3 Deek’s funnel plot asymmetry test for publication bias of presepsin (A) (p=0.001) and PCT (B) (p=0.1). |
Analysis of the prognostic value of presepsin in predicting mortality
A total of 9 studies with 1,561 patients were included in this group. The pooled SEN and SPE for presepsin were 0.83 (95% CI, 0.72–0.90) and 0.69 (95% CI, 0.63–0.74), respectively (Figure 4) with the overall area under the ROC (AUROC) of 0.77 (95% CI, 0.73–0.81) (Figure 5A). The PLR and NLR were 2.6 (95% CI, 2.1–3.3) and 0.25 (95% CI, 0.15–0.44), respectively. The Diagnostic Odds Ratio (DOR) was 10 (95% CI, 5–22). Fagan’s nomogram for likelihood ratios indicates that predicting mortality in sepsis enhanced the post-probability to 40% by presepsin when the consequences were positive and decreased the post-probability to 6% for presepsin while the results were turning to negative (Figure 6A).
 | Figure 4 Forest plots of the pooled sensitivity and specificity for presepsin. |
 | Figure 5 The AUROC for prognostic value of presepsin (A) and PCT (B) in the mortality of sepsis patients. |
 | Figure 6 Fagan’s nomogram for likelihood ratios of presepsin (A) and PCT (B). |
Of all the studies included in our study, only 8 had provided a cut-off value for presepsin and 6 for PCT. Three studies had a cut-off value lower than 1,000 pg/mL, ranging from 556 pg/mL to 957.5 pg/mL. Two studies provided similar concentrations as 1,622 and 1,631 pg/mL, one as 1,853 pg/mL and one was higher than 2,000 pg/mL. As for PCT, the concentrations ranged from 0.16 ng/mL to 14.27 ng/mL (Table 1).
Performance comparison with PCT in predicting value on mortality
Seven studies with 1,407 patients were recruited for extracting the data of biomarker PCT, and the other 2 of the 9 studies were eliminated for not providing the information. The AUC of PCT was 0.81 (95% CI, 0.78–0.84) with a pooled SEN of 0.76 (95% CI, 0.55–0.89) and SPE of 0.74 (95% CI, 0.33–0.94) (Figures 5B–7). The PLR, NLR and DOR were 2.9 (95% CI, 0.9–9.5), 0.32 (95% CI, 0.17–0.62) and 9 (95% CI, 2–41), accordingly. We found that in contrast to the PCT, the pooled sensitivity of presepsin was slightly higher (0.83, 95% CI, 0.72–0.90 vs 0.76, 95% CI, 0.55–0.89) and the pooled specificity was a bit lower (0.69, 95% CI 0.63–0.74 vs 0.74, 95% CI 0.33–0.94). However, the analytic consequences showed no statistically significant difference between two predictors (p-value=0.39 and 0.71, respectively, for presepsin and PCT). Additionally, Fagan’s nomogram for likelihood ratios indicated that predicting mortality in sepsis enhanced the post-probability to 42% by PCT when the results were positive and decreased the post-probability to 7% as it was turning to negative, which showed the similar predicting ability of mortality with presepsin as well (Figure 6B). The summary analysis of prognostic information between presepsin and PCT is presented in Table 3, indicating the comparable prognostication in sepsis outcome of presepsin with PCT.
 | Table 3 Summaries of performance statistics of presepsin and PCT for predicting mortalities in sepsis |
 | Figure 7 Forest plots of the pooled sensitivity and specificity for PCT. |
Discussion
Sepsis is more a complicated pathophysiological condition rather than a specific syndrome. Despite advanced modern life support, fatality and morbidity remain high in the past two decades.41,42 Early recognition of high-risk-to-die patients may prevent the high morbidity and improve the clinical outcome in this subpopulation. Hence, it is imperative to find a reliable prognostic biomarker with high sensitivity and specificity that can help aid in differentiating septic patients in a fast and cost-effective manner. Yang et al recently reported a meta–analysis showing that presepsin concentrations in decedents outweigh that in survivors.24 However, our research presented additional evidence with sensitivity and specificity for assessing the accuracy of presepsin in predicting mortality of septic patients and further made a head-to-head comparison with PCT, a widely used biomarker in assessing the predictive value. We found that there is no statistically significant difference between the two predictors, implying that both presepsin and PCT are promising prognostic biomarkers of sepsis.
In our study, the results showed that the presepsin reached a pooled sensitivity of 0.83 and specificity of 0.69, suggesting a relatively high rate of missed prognosis by 17% and misprognosis of 31%. The overall PLR was 2.6, indicating that the likelihood of a positive presepsin test result was almost 3-fold higher in septic non-survivors than in patients who survived. The pooled NLR was 0.25, suggesting that the possibility of a negative result was only a chance of one-fourth. Further, the AUC of 0.77 indicated an acceptable prognostic accuracy that presepsin could be a new and promising biomarker for predicting sepsis mortality.
We further compared the results with PCT, a useful but also controversial biomarker in predicting mortality for sepsis. Some suggested that the levels of PCT showed no significant difference between survivors and decedents and PCT could also rise transiently in patients with non-septic situations such as trauma, surgery or systemic inflammatory response syndrome (SIRS).43–46 In addition, studies suggested that the peak of PCT level was reached later than 24 hrs, which might not be reasonably early enough as biomarkers for sepsis diagnosis and stratification of the sepsis severity. However, to date, PCT is extensively accepted for mirroring the poor outcome in infectious diseases47–49 and is also recognized for diagnosis, guided therapy and prognostication of patients with sepsis.15,16 In a recent meta-analysis, Prkno et al found that under a PCT-guided medicine treatment, the use of antibiotic therapy decreased remarkably from 8 days in the control group to 6 days. The lesser duration of the antibiotic consumption appeared to be associated with a reduction in the mortality, morbidity, length of hospitalization and health care costs.50 More importantly, recent studies show that PCT might confer positive prediction value in mortality.15,16 In Liu’s meta-analysis exploring the effect of PCT on mortality, the pooled SEN was 0.72 (95% CI, 0.58–0.82) compared to 0.76 (95% CI, 0.55–0.89) in our result and SPE was 0.77 (95% CI, 0.55–0.90) compared to our data of 0.74 (95% CI, 0.33–0.94).16 The AUC in Liu’s publication was 0.70 (95% CI, 0.75–0.83) and that in our study is 0.81 (95% CI, 0.78–0.84). In spite of the fact that publication bias existed for the limited number of articles about PCT included in the present study, through the comparison with the published meta-analysis, our results demonstrated that PCT is a stable and reliable biomarker in the prognostic value on mortality. Therefore, it is credible for us to make the contrast of the predicting value in these two biomarkers. Though the pooled SEN for presepsin is higher while the SPE is inferior than that of PCT, there showed no statistically significant difference in both groups, with a p-value of 0.39 and 0.71. Hence, presepsin could be another promising predictor, which appears to be comparable to PCT in predicting mortality in septic patients.
Presepsin was a newly identified marker for sepsis in 2004, and the use of presepsin to make an assessment of severity and prognosis was first reported in 2011.21,25 The prominent advantage of this indicator was the early secretion after infection, while the peak concentration of its counterpart was observed later than 24 hrs after infection. This could mean that when presepsin is secreted at its maximum, the level of PCT has not increased completely or reached its summit. This earlier increase in the concentration of presepsin may also partially explain why presepsin expresses a slightly higher sensitivity than PCT in our present study. In 2013 Guidelines of the Surviving Sepsis Campaign, it was recommended that confirming the potential infectious site within 6 hrs of presentation was critical to the outcomes in sepsis patients and that broad-spectrum antibiotic intervention must be treated within 1 hr after sepsis identification.28 Presepsin was also considered as more of an independent mortality predictor than PCT.37,51 However, there is some inherent limitation for this novel serum biological marker. Presepsin is detected while being releasing into the circulation after activating host cells by recognition of bacterial lipopolysaccharide (LPS), which is more of a characteristic of gram-negative bacteria. Thus, in general, the levels of presepsin were often examined elevated most in the gram-negative organisms, followed by fungi and lesser in gram-positive bacteria.33,39,52–59 In addition, in the same way, that PCT could be detected in other critical conditions such as multiple trauma, pancreatitis and extensive burns, several studies revealed that high levels of presepsin were also found in patients with cardiac surgery.22 Particularly, presepsin was found to be elevated in patients with renal or liver dysfunction such as severe chronic kidney disease or liver cirrhosis without infection.60 This could be explained by the fact that presepsin was detained in the serum when the function of kidneys had failed and the degradation ability of liver was impaired. Therefore, more studies including sepsis patients with or without kidney and liver dysfunction should be conducted to fully exploit the diagnostic and prognostic value of presepsin. Considering the merits and demerits of both biomarkers, the combination use of presepsin and PCT appears to provide greater clinical value.
Significant publication bias was found after being visually tested by the Deek’s funnel plot. The exclusion of the following modalities, including review, letters, case reports, abstracts of conference and negative studies, etc., are the probable sources attributed to the publication bias in this review. In addition, language bias could be a factor for the reason that we include articles written in English.
This systematic review and meta-analysis has several limitations as well. First, the study presented some heterogeneity which is inherent in the present available data. The cut-off value revealed a large gap varying from 556 to 2455 pg/mL among different studies despite the same presepsin assay method. This threshold effect caused by diverse cut-off values could be one of the reasons and thus the optimal cut-off could not be defined. In addition, the reference standard was not uniform, ranging from ACCP/SCCM 1992 criteria, 2001 version (Sepsis 1.0 or 2.0), International Guidelines, to the newly defined sepsis 3.0. Based on the new guideline (sepsis 3.0), SIRS with infection is no longer defined as sepsis which might explain the highest presepsin level of 2,455 pg/mL in Kim’s article. And a further reason for this discrepancy could be from the study designs, with five in PR, two in RR, and one being unknown. With a limited number and high heterogeneity between selected studies, these results should be carefully and cautiously interpreted.
Conclusion
According to the results of our study, both presepsin and PCT are prospective predictors for contrasting decedents and survivors with sepsis. However, with the limitation of both biomarkers, the combination of presepsin and PCT might provide a more accurate prognostic value. Finally, continuous re-evaluation should be conducted to determine a promising mortality predictor with high sensitivity and specificity in the clinical practice and define an optimized cut-off value.
Data sharing statement
The data used to manage and analyses are archived and be available from the PubMed, Embase and Cochrane databases.
Acknowlegments
Our deepest gratitude goes to the Elizabeth C. Scheyder, Ph.D., University of Pennsylvania, for the English language improvement and anonymous reviewers for their careful work and thoughtful suggestions that have helped improve this paper substantially. This work was supported by the National Natural Science Foundation of China (Grant No. 81700069 and No. 81701964) and The Clinical Research Project supported by Beijing Municipal Science & Technology Commission (Z181100001718024).
Author contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors have declared that no competing interests exist in this work.
References
1. Ulla M, Pizzolato E, Lucchiari M, et al. Diagnostic and prognostic value of presepsin in the management of sepsis in the emergency department: a multicenter prospective study. Crit Care. 2013;17:R168. doi:10.1186/cc12734
2. Zou Q, Wen W, Zhang XC. Presepsin as a novel sepsis biomarker. World J Emerg Med. 2014;5:16–19. doi:10.5847/wjem.j.1920-8642.2014.01.002
3. Singer M, Deutschman CS, Seymour CW, et al. The third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA. 2016;315:801–810. doi:10.1001/jama.2016.0287
4. Zhang X, Liu D, Liu YN, et al. The accuracy of presepsin (sCD14-ST) for the diagnosis of sepsis in adults: a meta-analysis. Crit Care. 2015;19:323. doi:10.1186/s13054-015-1032-4
5. Heublein S, Hartmann S, Hagel S, et al. Epidemiology of sepsis in German hospitals derived from administrative databases. Infection. 2013;41:S71.
6. Escarce JJ, Kelley MA. Admission source to the medical intensive care unit predicts hospital death independent of APACHE II score. J Am Med Assoc. 1990;264:2389–2394. doi:10.1001/jama.1990.03450180053028
7. Ferreira FL, Bota DP, Bross A, et al. Serial evaluation of the SOFA score to predict outcome in critically ill patients. J Am Med Assoc. 2001;286:1754–1758. doi:10.1001/jama.286.14.1754
8. Vincent JL, Opal SM, Marshall JC. Ten reasons why we should NOT use severity scores as entry criteria for clinical trials or in our treatment decisions. Crit Care Med. 2010;38:283–287. doi:10.1097/CCM.0b013e3181b785a2
9. Lee YT, Gong M, Chau A, et al. Pentraxin-3 as a marker of sepsis severity and predictor of mortality outcomes: a systematic review and meta-analysis. J Infect. 2018;76:1–10. doi:10.1016/j.jinf.2017.10.016
10. Enguix-Armada A, Escobar-Conesa R, Garcia-De La Torre A, et al. Usefulness of several biomarkers in the management of septic patients: C-reactive protein, procalcitonin, presepsin and mid-regional pro-adrenomedullin. Clin Chem Lab Med. 2016;54:163–168. doi:10.1515/cclm-2015-0243
11. Pierrakos C, Vincent JL. Sepsis biomarkers: a review. Crit Care. 2010;14:R15. doi:10.1186/cc8872
12. Wu Y, Wang F, Fan X, et al. Accuracy of plasma sTREM-1 for sepsis diagnosis in systemic inflammatory patients: a systematic review and meta-analysis. Crit Care. 2012;16:R229. doi:10.1186/cc11884
13. Simon L, Gauvin F, Amre DK, et al. Serum procalcitonin and C-reactive protein levels as markers of bacterial infection: a systematic review and meta-analysis. Clin Infect Dis. 2004;39:206–217. doi:10.1086/421997
14. Harbarth S, Holeckova K, Froidevaux C, et al. Diagnostic value of procalcitonin, interleukin-6, and interleukin-8 in critically ill patients admitted with suspected sepsis. Am J Respir Crit Care Med Hypotheses. 2001;164:396–402. doi:10.1164/ajrccm.164.3.2009052
15. Arora S, Singh P, Singh PM, et al. Procalcitonin levels in survivors and nonsurvivors of sepsis: systematic review and meta-analysis. Shock. 2015;43:212–221. doi:10.1097/SHK.0000000000000305
16. Liu D, Su L, Han G, et al. Prognostic value of procalcitonin in adult patients with sepsis: a systematic review and meta-analysis. PLoS One. 2015;10:e0129450. doi:10.1371/journal.pone.0129450
17. Shozushima T, Takahashi G, Matsumoto N, et al. Usefulness of presepsin (sCD14-ST) measurements as a marker for the diagnosis and severity of sepsis that satisfied diagnostic criteria of systemic inflammatory response syndrome. J Infect Chemother. 2011;17:764–769. doi:10.1007/s10156-010-0197-7
18. Okamura Y, Yokoi H. Development of a point-of-care assay system for measurement of presepsin (sCD14-ST). Clin Chim Acta. 2011;412:157–161. doi:10.1016/j.cca.2011.07.024
19. Shirakawa K, Naitou K, Hirose J, et al. Presepsin (sCD14-ST): development and evaluation of one-step ELISA with a new standard that is similar to the form of presepsin in septic patients. Clin Chem Lab Med. 2011;49:937–939. doi:10.1515/CCLM.2011.066
20. Sugie Y, Igami K, Shoji K, et al. Performance evaluation of the new rapid fertility assays in whole blood and plasma on PATHFAST. Clin Lab. 2011;57:99–106.
21. Yaegashi Y, Shirakawa K, Sato N, et al. Evaluation of a newly identified soluble CD14 subtype as a marker for sepsis. J Infect Chemother. 2005;11:234–238. doi:10.1007/s10156-005-0400-4
22. Brodska H, Valenta J, Pelinkova K, et al. Diagnostic and prognostic value of presepsin vs. established biomarkers in critically ill patients with sepsis or systemic inflammatory response syndrome. Clin Chem Lab Med. 2017;56:658–668.
23. de la Torre-Prados MV, García-de la Torre A, Escobar-Conesa R, et al. Accuracy prognostic using presepsin and proadrenomedullin in critically septic patients. Intensive Care Med. 2014;40(1 SUPPL. 1):S210.
24. Yang HS, Hur M, Yi A, et al. Prognostic value of presepsin in adult patients with sepsis: systematic review and meta-analysis. PLoS One. 2018;13:e0191486. doi:10.1371/journal.pone.0191486
25. Spanuth E, Ebeit H, Ivandic B, et al. Diagnostic and prognostic value of soluble CD14 subtype (sCD14-ST) in emergency patients with early sepsis using the new assay pat HFAST presepsin. Clin Chem Lab Med. 2011;49(1):S361. doi:10.1515/CCLM.2011.066
26. Levy MM, Fink MP, Marshall JC, et al. 2001 SCCM/ESICM/ACCP/ATS/SIS international sepsis definitions conference. Intensive Care Med. 2003;29:530–538. doi:10.1007/s00134-003-1662-x
27.
28. Dellinger RP, Levy MM, Rhodes A, et al. Surviving sepsis campaign guidelines committee including the pediatric subgroup: surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock. Crit Care Med. 2013;41:580–637. doi:10.1097/CCM.0b013e31829133d6
29. Whiting P, Rutjes AW, Reitsma JB, et al. Thedevelopment of QUADAS: a tool for the quality assessment ofstudies of diagnostic accuracy included in systematic reviews. BMC Med Res Methodol. 2003;3:25. doi:10.1186/1471-2288-3-25
30. Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics. 1977;33:159–174. doi:10.2307/2529310
31. Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21:1539–1558. doi:10.1002/sim.1186
32. Fagan TJ. Letter: nomogram for Bayes theorem. N Engl J Med. 1975;293:257. doi:10.1056/NEJM197507312930513
33. Masson S, Caironi P, Spanuth E, et al. Presepsin (soluble CD14 subtype) and procalcitonin levels for mortality prediction in sepsis: data from the Albumin Italian Outcome Sepsis trial. Crit Care. 2014;18:R6. doi:10.1186/cc13712
34. Kim H, Hur M, Moon HW, et al. Multi-marker approach using procalcitonin, presepsin, galectin-3, and soluble suppression of tumorigenicity 2 for the prediction of mortality in sepsis. Ann Intensive Care. 2017;7:27. doi:10.1186/s13613-017-0252-y
35. El-Shafie ME-S, Taema KM, El-Hallag MM, et al. Role of presepsin compared to C-reactive protein in sepsis diagnosis and prognostication. Egypt J Crit Care Med. 2017;5:1–12. doi:10.1016/j.ejccm.2017.02.001
36. Ali FT, Ali MA, Elnakeeb MM, et al. Presepsin is an early monitoring biomarker for predicting clinical outcome in patients with sepsis. Clin Chim Acta. 2016;460:93–101. doi:10.1016/j.cca.2016.06.030
37. Liu B, Chen YX, Yin Q, et al. Diagnostic value and prognostic evaluation of Presepsin for sepsis in an emergency department. Crit Care. 2013;17:R244. doi:10.1186/cc12734
38. Carpio R, Zapata J, Spanuth E, et al. Utility of presepsin (sCD14-ST) as a diagnostic and prognostic marker of sepsis in the emergency department. Clin Chim Acta. 2015;450:169–175. doi:10.1016/j.cca.2015.08.013
39. Matera G, Quirino A, Peronace C, et al. Soluble CD14 subtype-a new biomarker in predicting the outcome of critically Ill septic patients. Am J Med Sci. 2017;353:543–551. doi:10.1016/j.amjms.2017.03.036
40. Spanuth E, Ebelt H, Ivandic B, et al. Diagnostic and prognostic value of soluble CD14 subtype (sCD14-ST) in emergency patients with early sepsis using the new assay pat HFAST presepsin. Clin Chem Lab Med. 2011;49(SUPPL. 1):S361. doi:10.1515/CCLM.2011.066
41. Martin GS, Mannino DM, Eaton S, et al. The epidemiology of sepsis in the United States from 1979 through 2000. N Engl J Med. 2003;348:1546–1554. doi:10.1056/NEJMicm020037
42. Angus DC, Linde-Zwirble WT, Lidicker J, et al. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome and associated costs of care. Crit Care Med. 2001;29:1303–1310. doi:10.1097/00003246-200107000-00002
43. Kibe S, Adams K, Barlow G. Diagnostic and prognostic biomarkers of sepsis in critical care. J Antimicrob Chemother. 2011;66:ii33–ii40.
44. Tang BM, Eslick GD, Craig JC, et al. Accuracy of procalcitonin for sepsis diagnosis in critically ill patients: systematic review and metaanalysis. Lancet Infect Dis. 2007;7:210–217. doi:10.1016/S1473-3099(07)70052-X
45. Wacker C, Prkno A, Brunkhorst FM, et al. Procalcitonin as a diagnostic marker for sepsis: a systematic review and meta-analysis. Lancet Infect Dis. 2013;13:426–435. doi:10.1016/S1473-3099(12)70323-7
46. Song X, Song Y, Yuan Y, et al. Prognostic value of presepsin for outcomes and complications in enterocutaneous fistula complicated by abdominal sepsis. Int J Surg. 2016;33(Pt A):96–101. doi:10.1016/j.ijsu.2016.07.070
47. Huang DT, Weissfeld LA, Kellum JA, et al. Risk prediction with procalcitonin and clinical rules in community-acquired pneumonia. Ann Emerg Med. 2008;52:48–58 e42. doi:10.1016/j.annemergmed.2008.01.003
48. Kruger S, Ewig S, Marre R, et al. Procalcitonin predicts patients at low risk of death from community-acquired pneumonia across all CRB-65 classes. Eur Respir J. 2008;31:349–355. doi:10.1183/09031936.00054507
49. Park JH, Wee JH, Choi SP, et al. The value of procalcitonin level in community-acquired pneumonia in the ED. Am J Emerg Med. 2012;30:1248–1254. doi:10.1016/j.ajem.2011.08.009
50. Prkno A, Wacker C, Brunkhorst FM, et al. Procalcitonin-guided therapy in intensive care unit patients with severe sepsis and septic shock–a systematic review and meta-analysis. Crit Care. 2013;17:R291. doi:10.1186/cc12734
51. Ruiz-Alvarez MJ, García-Valdecasas S, de Pablo R, et al. Diagnostic efficacy and prognostic value of serum procalcitonin concentration in patients with suspected sepsis. J Intensive Care Med. 2009;24:63–71. doi:10.1177/0885066608327095
52. Tong X, Cao Y, Yu M, et al. Presepsin as a diagnostic marker for sepsis: evidence from a bivariate meta-analysis. Ther Clin Risk Manag. 2015;11:1027–1033. doi:10.2147/TCRM.S84811
53. Wu J, Hu L, Zhang G, et al. Accuracy of presepsin in sepsis diagnosis: a systematic review and meta-analysis. PLoS One. 2015;10:e0133057. doi:10.1371/journal.pone.0133057
54. Zhang J, Hu ZD, Song J, et al. Diagnostic value of presepsin for sepsis: a systematic review and meta-analysis. Medicine (Baltimore). 2015;94:e2158. doi:10.1097/MD.0000000000000874
55. Zheng Z, Jiang L, Ye L, et al. The accuracy of presepsin for the diagnosis of sepsis from SIRS: a systematic review and meta-analysis. Ann Intensive Care. 2015;5:48. doi:10.1186/s13613-015-0089-1
56. Liu Y, Hou JH, Li Q, et al. Biomarkers for diagnosis of sepsis in patients with systemic inflammatory response syndrome: a systematic review and meta-analysis. Springerplus. 2016;5:2091. doi:10.1186/s40064-016-3591-5
57. Hayashida K, Kondo Y, Hara Y, et al. Head-to-head comparison of procalcitonin and presepsin for the diagnosis of sepsis in critically ill adult patients: a protocol for a systematic review and meta-analysis. BMJ Open. 2017;7:e014305. doi:10.1136/bmjopen-2016-014305
58. Wu CC, Lan HM, Han ST, et al. Comparison of diagnostic accuracy in sepsis between presepsin, procalcitonin, and C-reactive protein: a systematic review and meta-analysis. Ann Intensive Care. 2017;7:91. doi:10.1186/s13613-017-0316-z
59. Romualdo LG, Torrella PE, González MV, et al. Diagnostic accuracy of presepsin(solubleCD14subtype)for prediction of bacteremia in patients with systemic inflammatory response syndrome in the Emergency Department. Clin Biochem. 2014;47:S505–S508. doi:10.1016/j.clinbiochem.2014.02.011
60. Nakamura Y, Ishikura H, Nishida T, et al. Usefulness of presepsin in the diagnosis of sepsis in patients with or without acute kidney injury. BMC Anesthesiol. 2014;14:88. doi:10.1186/1471-2253-14-88
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
