Back to Journals » Clinical Interventions in Aging » Volume 15
Teriparatide in East Asian Postmenopausal Women with Osteoporosis in a Real-World Setting: A Baseline Analysis of the Asia and Latin America Fracture Observational Study (ALAFOS)
Authors Chen CH , Lim SJ , Oh JK, Huang TW, Zeng YH , Wu MT , Yang HL , Cheung JPY , Kim JW , Han JH, Huo L , Lin TJ, Zhou G, Wu WS
Received 26 August 2019
Accepted for publication 26 December 2019
Published 30 January 2020 Volume 2020:15 Pages 111—121
DOI https://doi.org/10.2147/CIA.S228158
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Zhi-Ying Wu
Chung-Hwan Chen, 1 Seung-Jae Lim, 2 Jong-Keon Oh, 3 Tsan-Wen Huang, 4 Yuhong H Zeng, 5 Meng-Ting Wu, 6 Huilin L Yang, 7 Jason Pui-Yin Cheung, 8 Ji Wan Kim, 9 Jeong Hee Han, 10 Li Huo, 11 Tsung-Jung Lin, 12 Gang Zhou, 11 Wen-Shuo Wu 12
1Department of Orthopedics and Orthopedic Research Center, Kaohsiung Municipal Ta-Tung Hospital and Kaohsiung Medical University Hospital, College of Medicine, Kaohsiung Medical University, Kaohsiung City, Taiwan; 2Department of Orthopedic Surgery, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, South Korea; 3Department of Orthopedics, Korea University Guro Hospital, Seoul, South Korea; 4Department of Orthopaedic Surgery, Chang Gung Memorial Hospital, Chiayi, Taiwan; 5Department of Osteoporosis, Honghui Hospital, Xi ‘an Jiaotong University, Xian, People’s Republic of China; 6Division of Neurosurgery, Department of Surgery, Cheng Hsin General Hospital, Taipei, Taiwan; 7Department of Orthopedics, The First Affiliated Hospital of Soochow University, Suzhou, People’s Republic of China; 8Department of Orthopaedics and Traumatology, The University of Hong Kong, Hong Kong, People’s Republic of China; 9Department of Orthopedic Surgery, Asan Medical Center, University of Ulsan, College of Medicine, Seoul, South Korea; 10Department of Medical Quality and Regulatory Affairs, Lilly Korea Ltd., Seoul, South Korea; 11Lilly China Drug Development and Medical Affairs Center, Lilly Suzhou Pharmaceutical Co. Ltd, Shanghai, People’s Republic of China; 12Department of Medical Affairs, Eli Lilly and Company (Taiwan), Inc., Taipei, Taiwan
Correspondence: Wen-Shuo Wu
Eli Lilly and Company (Taiwan), Inc., 11/F, No. 365 Fu Hsing N. Road, Taipei 105 R.O.C., Taiwan
Tel +886 2 2715 2950 ext 2234
Email [email protected]
Purpose: The aim of this analysis is to describe the baseline characteristics of patients who are prescribed teriparatide for the treatment of postmenopausal osteoporosis in a real-world setting in East Asia.
Patients and Methods: The Asia and Latin America Fracture Observational Study (ALAFOS) is a prospective, multinational, observational study designed to evaluate real-world use of teriparatide in the treatment of postmenopausal osteoporosis in 20 countries across Asia, Latin America, the Middle East, and Russia. This subregional analysis focuses on the East Asian subpopulation of the ALAFOS study. Here we report baseline clinical characteristics, details regarding the history of fractures, risk factors for osteoporosis, comorbidities, osteoporosis treatment, and health-related quality of life in patients enrolled in China, Hong Kong, South Korea, and Taiwan.
Results: The East Asian subgroup of ALAFOS included 1136 postmenopausal women, constituting 37.5% (1136/3031) of the overall ALAFOS patient population. The mean (SD) age was 75.0 (9.6) years. The mean (SD) bone mineral density T-scores were − 3.11 (1.54), − 2.58 (1.11), and − 2.86 (1.09) at the lumbar spine, total hip, and femoral neck, respectively; 69.6% of patients had experienced at least one fragility fracture and 40.4% had experienced ≥ 2 fragility fractures after 40 years of age. Overall, 63.3% of patients had used medications for osteoporosis in the past. The mean (SD) EQ-5D-5L Visual Analog Scale (VAS) score at baseline was 59.7 (20.8); the mean (SD) back pain numeric rating scale score for worst pain in the last 24 hrs was 5.2 (3.2).
Conclusion: Our results indicate that patients who are prescribed teriparatide in East Asia were elderly women with severe osteoporosis, low bone mineral density, high prevalence of fractures, back pain and poor health-related quality of life. Most of the patients received teriparatide as a second-line treatment.
Keywords: postmenopausal osteoporosis, teriparatide, observational study, fractures, East Asia
Corrigendum for this paper has been published
Introduction
Osteoporosis is a common and well-recognized health problem worldwide with a growing importance in Asian countries.1 Osteoporotic fractures, particularly vertebral and hip fractures, contribute largely to fracture-related morbidity, hospitalization, functional disability, and poor quality of life, as well as mortality.2–4 Worldwide estimates revealed that the number of new osteoporotic fractures in people ≥50 years of age in the year 2000 was approximately 9 million; this included 1.6 million hip fractures and 1.4 million clinical vertebral fractures.5
The number of new vertebral fractures reported from Southeast Asia in the year 2000 was 0.25 million.5 The Asia–Pacific Regional Audit report (2013) predicts that there will be an enormous increase in the number of osteoporotic fractures in Asia in the coming years.3 For example, Asia accounted for 31% of hip fractures worldwide in the year 1990; future predictions reveal that this number will increase to 51% by the year 2050.6 This huge increase in the proportion of osteoporotic fractures in the Asian population can be attributed to changing demographics and a rapid increase in the elderly population in this region.2 Another factor that may contribute to this increase is rapid economic development and urbanization in several parts of Asia.7 The Asian Osteoporosis Study7 found that the incidence of hip fractures in women >50 years was highest in urbanized Asian countries such as Hong Kong and Singapore; in these countries, the incidence of hip fractures is rapidly approaching that observed in Caucasian populations.7,8
Osteoporosis is an underdiagnosed and often suboptimally treated medical condition, which puts a huge burden on resources and healthcare systems in Asia.2,3 Lack of awareness about the disease among patients, non-availability of reliable epidemiological data, limited access to quality healthcare and diagnostic/therapeutic interventions, reimbursement issues, and financial constraints are important unmet needs that require attention.2
The Asia and Latin America Fracture Observational Study (ALAFOS) is a large observational study being conducted in 20 countries across Asia, Latin America, the Middle East, and Russia. This is one of the first studies evaluating real-world use of teriparatide in the treatment of postmenopausal osteoporosis in these geographies. In this report, we focus on the East Asian subregion of the overall ALAFOS study cohort. This subregional analysis includes four East Asian regions (China, Hong Kong, South Korea, and Taiwan), which are generally similar in racial/ethnic backgrounds, lifestyles, and healthcare systems. The aim of the present analysis is to describe the baseline clinical characteristics of patients who are prescribed teriparatide for the treatment of postmenopausal osteoporosis. In view of the increasing burden of osteoporosis in Asia, we believe that this subregional analysis of the ALAFOS study will provide useful insights into patient characteristics, common risk factors, prescribing patterns, fracture outcomes, and health-related quality of life (HRQoL) measures.
Materials and Methods
Study Design and Assessments
ALAFOS is a multinational, prospective, observational study being conducted in teriparatide-naïve, postmenopausal women at a high risk of fracture who are initiating treatment with teriparatide as prescribed by their physicians during normal clinical practice. This means that the decision to prescribe teriparatide was taken by the physicians at their own discretion, based on their clinical judgement. The aim of the ALAFOS study is to evaluate the incidence of new osteoporotic fractures in relation to time on teriparatide, and persistence and adherence to teriparatide treatment in this patient population. Details regarding the ALAFOS study design have been published previously.9 Patients are treated with once-daily teriparatide (20 μg/day) for up to 24 months, the maximum approved treatment duration, during the active treatment phase. Patient assessments and data collection are done during routine clinical visits (at baseline, 3 months, 6 months and approximately every 6 months thereafter; Figure 1). Patients will be followed up for up to 12 months after completion of the active treatment with teriparatide.
Variables assessed at baseline include demographic and clinical characteristics, fracture history, possible risk factors for osteoporosis or fractures, existing comorbidities, concomitant medications, osteoporosis and back pain medications, and HRQoL measures such as EQ-5D-5L, and back pain scores. Outcome measures that will be assessed during subsequent visits include fracture details (based on medical confirmation); adherence to treatment; patients’ knowledge, beliefs, and attitudes about osteoporosis; and back pain, physical function, mobility, and HRQoL measures. Protocol-defined adverse event data will also be collected. Details regarding these assessments have been published previously.9
The present analysis focuses on the baseline characteristics of patients who receive teriparatide for the treatment of postmenopausal osteoporosis.
Statistics
In this analysis, we disclose the baseline data for the East Asian subregion (N=1136 patients), including baseline data for the four participant regions: Taiwan (N=474; 41.7% of the East-Asia subregional group), South Korea (N=379; 33.4%), China (N=235; 20.7%) and Hong Kong (N=48; 4.2%). Descriptive statistics are used to describe the study population (for continuous variables, means and standard deviations [SD] or medians and first and third quartiles [Q1, Q3] are presented, and categorical variables are summarized using counts and percentages). We have not made any statistical comparisons among the four participant regions or with the overall East Asian subregional group. Data analysis was done using SAS v9.4 or higher (SAS Institute Inc., Cary, NC, USA).
Ethics Approval
The ALAFOS study complies with the principles of Good Clinical Practice, the Declaration of Helsinki, and all local laws and regulations as applicable. Appropriate local site-level ethics committees have approved the study protocol. All participants provided written informed consent and were required to sign a Consent to Release Information form.
Results
The overall ALAFOS baseline analysis included 3031 patients across 20 countries.9 The present analysis of the East Asian subregional cohort includes 1136 patients, constituting 37.5% of the overall ALAFOS patient population.
Table 1 shows the baseline demographics of the overall East Asian study cohort and the four participant regions. The mean (SD) age of the overall East Asian cohort was 75.0 (9.6) years and the mean (SD) body mass index (BMI) was 23.1 (3.9) kg/m2. The mean (SD) age at onset of menopause was 49.5 (5.2) years for the overall East Asian cohort, which was generally consistent across all four participant regions. Surgical menopause was reported by 9.4% of patients in the overall East Asian cohort with patients from Hong Kong reporting the highest rate (18.8%).
 |
Table 1 Baseline Characteristics and Reproductive History |
Bone Mineral Density and History of Fractures
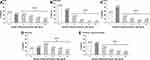
The baseline mean (SD) bone mineral density (BMD) T-scores for the overall East Asian cohort were −3.11 (1.54), −2.58 (1.11), and −2.86 (1.09) at the lumbar spine, total hip, and femoral neck, respectively (Table 1). A majority of patients (69.6%) had experienced at least one fragility fracture after 40 years of age (85.4% in Hong Kong, 75.5% in Taiwan; 74.9% in China, and 57.0% in South Korea; Table 1). The percentage of patients with a history of ≥2 fragility fractures after 40 years of age was 40.4% (66.7% in Hong Kong, 53.8% in Taiwan, 34.9% in China, and 23.7% in South Korea; Figure 2). Previous vertebral fractures were reported by 47.0% of patients; non-vertebral and hip fractures were reported by 28.3% and 13.9% of patients, respectively (Table 1). A prior history of hip fracture was more common in Taiwan (17.3%), South Korea (16.1%), and Hong Kong (14.6%) than in China (3.4%; Table 1).
Risk Factors for Osteoporosis/Fractures
In the overall East Asian subregional cohort, 23.8% of patients reported having a maternal history of osteoporosis or hip fracture; the corresponding rates in the four regions ranged from 2.8% in Hong Kong to 29.9% in China (Table 2). Almost 70% of patients needed to use the support of their arms while standing up from a chair. Approximately 50% of patients reported having problems related to vision and 36.3% reported having at least one fall in the past year. The percentage of patients experiencing falls in the past year was the highest in Hong Kong and Taiwan (43.8% and 43.1%, respectively; Table 2).
 |
Table 2 Risk Factors for Osteoporosis and Fractures, Current Comorbidities, and Concomitant Medications |
Current Comorbid Conditions and Concomitant Medications
Hypertension was the most common comorbid condition in the overall East Asian cohort (44.4% of patients), followed by type 2 diabetes (10.7%) and rheumatoid arthritis/other rheumatologic conditions (7.2%; Table 2). Hypertension was more common in Hong Kong and South Korea (66.7% and 57.3%, respectively) than in China (37.3%) and Taiwan (34.8%). Antihypertensive medications were used by 29% of patients, and glucocorticoid use (prednisone daily equivalent dose ≥7.5 mg for ≥3 months) was reported by 4.0% of patients in the overall East Asian cohort at baseline (Table 2).
Medications for Osteoporosis and Back Pain
Table 3 summarizes the use of osteoporosis and back pain medications by the study participants. In the overall East Asian cohort, 63.3% of patients had used medications for osteoporosis in the past. Osteoporosis medication use was highest in Hong Kong (97.9%), followed by Taiwan (78.1%), China (59.6%), and South Korea (42.7%; Table 3). The most commonly used antiresorptive drug class in the East Asian cohort was bisphosphonates (29.8% of patients). The frequency of use of bisphosphonates was highest in Hong Kong (Hong Kong [68.8%], Taiwan [38.0%], China [23.0%], and South Korea [19.0%]). Alendronate was the most commonly used bisphosphonate across all four regions (Table 3). Denosumab use was reported most often by patients from Taiwan (22.4%), with a lower use in Hong Kong (8.3%), South Korea (0.3%), and China (0.0%). A similar trend was observed for raloxifene (Taiwan [13.9%], South Korea [2.4%], China [0.0%], and Hong Kong [0.0%]; Table 3). Calcium and vitamin D use was highest in Hong Kong (79.2% and 87.5%, respectively; Table 3).
 |
Table 3 Previous Use of Osteoporosis and Analgesic Medications |
In the overall East Asian cohort, 39.2% of patients had used ≥1 back pain medication over the past 24 hrs. The most commonly used analgesic medication drug class in the overall East Asian cohort was nonsteroidal anti-inflammatory drugs (18.9%). The most commonly used medication for back pain was paracetamol in Hong Kong (29.2% of patients) and opioids in South Korea (23.2% of patients; Table 3).
HRQoL and Back Pain Scores
The HRQoL scores at baseline are summarized in Table 4. The median (Q1, Q3) and mean (SD) EQ-5D-5L Visual Analog Scale (VAS) scores for the overall East Asian cohort were 60.0 (50.0, 75.0) and 59.7 (20.8), respectively. These scores were generally similar across the four participant regions, although slightly higher for Hong Kong (Table 4). The median (Q1, Q3) EQ-5D-5L utility total score was 0.5 (0.2, 0.7) for the East Asian cohort; the corresponding score for South Korea was lower than for the other three regions (Table 4). A considerable percentage of patients in the overall East Asian cohort reported experiencing severe or extreme problems on the EQ-5D-5L individual domains (mobility [36.1%], self-care [28.7%], usual activities [36.7%], pain/discomfort [27.2%], and anxiety/depression [13.2%]; Table 4). The mean (SD) back pain numeric rating scale scores were 3.9 (2.8) for average pain and 5.2 (3.2) for worst pain in the last 24 hrs.
 |
Table 4 Health-Related Quality of Life Scores at Baseline |
Discussion
Osteoporosis is recognized as a healthcare concern of increasing magnitude in East Asia.2 A survey conducted by the China Ministry of Health from the year 2003 to 2006 revealed that ~30% of women from the Chinese mainland, aged ≥50 years, were suffering from osteoporosis based on the World Health Organization diagnostic criteria.10 This percentage, when correlated with the large population in China, translates to ~54.1 million women suffering from osteoporosis in China alone.10 The increasing burden of osteoporosis in East Asia underscores the importance of studying the epidemiology of osteoporosis in this region with a focus on risk factors, diagnostic and therapeutic options, common prescription patterns for osteoporosis medications, and preventive strategies.
The approved labels for teriparatide in major geographies such as the United States and Europe state that teriparatide is indicated in postmenopausal women with osteoporosis at high risk for fracture.11,12 Local labels for teriparatide in the East Asian countries also support the use of teriparatide in this patient population.13–16 In addition, teriparatide is also approved for the treatment of men with osteoporosis who are at an increased risk of fracture and osteoporosis associated with sustained systemic glucocorticoid therapy in Hong Kong and South Korea.15,16 Our analysis revealed that patients receiving teriparatide in East Asia were typically elderly, postmenopausal women (mean age: 75.0 years). East Asian patients enrolled in the ALAFOS study were older than patients from Latin America (mean age: 71.0 years) and the Middle East (mean age: 69.3 years).9 Within the East Asian cohort, patients from Hong Kong (mean age: 78.4 years) were older than those from other regions.
East Asian patients had low BMD T-scores at baseline, similar to the Latin America and the Middle East subregions of the ALAFOS study.9 Low BMD is a well-established risk factor for fractures. Most treatment guidelines use BMD values as a guide to therapeutic decisions.2 It is important to note that the BMD values for Asians differ from those for Caucasians. Population-specific BMD reference databases have been developed for several East Asian countries using dual-energy X-ray absorptiometry.17–21
A previous history of fracture is another well-known risk factor that can predict possible fractures.2 A majority of patients in the East Asian ALAFOS cohort (69.6%) had experienced at least 1 fragility fracture after 40 years of age and 40.4% had experienced ≥2 fractures at baseline; these percentages were marginally higher than those reported for the overall ALAFOS population (62.9% and 28.8%, respectively) and the Latin American subpopulation of the ALAFOS (62.8% and 27.2%, respectively).9 These findings suggest that patients who initiate teriparatide in East Asia may be at a higher risk of fractures at baseline than those in other geographies, based on their previous fracture history. Within the East Asian cohort as well, there were differences across regions with respect to the percentage of patients with more than one fragility fracture at baseline; Hong Kong reported the highest percentage (85.4%), followed by Taiwan (75.5%), China (74.9%) and South Korea (57.0%). These differences could be related to the different reimbursement criteria in these regions. For example, in Taiwan, patients would be reimbursed if they had a BMD T-score < −3, or ≥2 fractures, or intolerance to or previous treatment with at least one antiresorptive medication for 1 year.22 In South Korea, teriparatide is reimbursed for patients with postmenopausal osteoporosis who have had two fragility fractures with a BMD T-score < −2.5 and who have shown no improvement after receiving antiresorptive treatment for at least 1 year,23 while in China, teriparatide is not reimbursed at all.
Vertebral fractures were reported by 47.0% of patients in the East Asian cohort, while non-vertebral fractures were reported by 28.3%. In the overall ALAFOS study cohort, a history of vertebral and non-vertebral fractures was present in 33.0% and 32.2% of patients, respectively.9 Hip fractures were reported less often in China (3.4% of patients) than in the other East Asian regions. These differences may be related to the different medical specialties of the participating study sites (primary care, orthopedics, endocrinology). This observation may also be explained by the fact that the Chinese label for teriparatide supports the use of teriparatide in reducing the risk of vertebral and non-vertebral fractures (but not hip fractures) in postmenopausal women.13
Certain comorbid conditions such as diabetes mellitus and chronic kidney disease are known to increase the risk of osteoporosis and fractures.24 In these cases, osteoporosis is mainly due to issues with bone quality, while BMD may be relatively preserved.24 Conditions such as rheumatoid arthritis are also associated with a higher fracture risk due to increased bone resorption and reduced BMD as a result of cytokine activation, immobilization and prolonged glucocorticoid treatment.24 In the ALAFOS East Asian cohort, hypertension (44.4% of patients), type 2 diabetes mellitus (10.7%) and rheumatoid arthritis or other rheumatologic conditions (7.2%) were the most common comorbidities. This finding was similar to the overall ALAFOS study population.9 Glucocorticoid use was slightly lower in the East Asian cohort (4.0%) than in the overall ALAFOS study population (5.3%).9
The use of osteoporosis medications in the past in the East Asian ALAFOS cohort (63.3% of patients) was lesser than that reported in similar studies in Europe (91.7%), the United States (88.4%), and Latin America (70.9%), but it was higher than that reported in Japan (51.3%) and the Middle East (19.9%).9,25–27 Bisphosphonates were the most commonly used class of antiresorptive drugs across most geographies.9,25,27 These findings indicate that a large proportion of patients in East Asia had already received antiresorptive therapies prior to initiating teriparatide. Country-specific treatment guidelines regarding teriparatide use are available for several East Asian countries. The Japanese guidelines for the treatment and prevention of osteoporosis recommend the use of teriparatide in patients who are at high risk of fractures (patients who have a fracture while on bisphosphonate or selective estrogen receptor modulators therapy, elderly patients with ≥1 vertebral fracture or proximal femoral fracture, or patients with significantly reduced BMD).24 The 2013 guidelines for the management of postmenopausal osteoporosis from the Osteoporosis Society of Hong Kong recommend teriparatide as a first-line therapy, especially in patients with ≥1 osteoporotic fracture, BMD T-score < −3, or those who do not respond to antiresorptive therapy.28 The Taiwanese guidelines also state that there is good evidence regarding the beneficial effects of teriparatide in the prevention of vertebral and non-vertebral fractures as well as in glucocorticoid-induced osteoporosis.4 The 2017 Chinese guidelines from the Osteoporosis and Bone Mineral Research Society of the Chinese Medical Association recommend the use of teriparatide in postmenopausal women at a high risk of fractures (patients with >1 vertebral fracture or significantly reduced BMD) and patients who have contraindications or are unable to tolerate oral drugs.29 In South Korea, teriparatide is indicated for the treatment of postmenopausal osteoporosis, osteoporosis in men and glucocorticoid-induced osteoporosis.16 These healthcare policies have a great impact on the patterns of treatment of osteoporosis and on real-world evidence in East Asia.
Osteoporosis is known to have a detrimental effect on the quality of life of patients. Also, evidence shows that quality of life often improves after the treatment of osteoporosis.2 Patients from the East Asian ALAFOS cohort reported a low HRQoL at baseline (median EQ-5D-5L VAS scores for perceived overall health status of 60.0); this was lower than that observed in Latin America (70.0) and the Middle East (70.0),9 while it was higher than that reported in Europe (50.0).25 The proportion of patients reporting severe or extreme problems in East Asia was higher than that reported in other regions from the ALAFOS study.9 A similar trend was observed in the mean (SD) back pain numeric rating scale scores.9 The ALAFOS study will evaluate patients’ response to teriparatide over time and provide useful data regarding fracture benefits and quality of life improvements in the real world.
Data suggest that East Asia is likely to bear a huge burden of osteoporosis in the next few decades. In fact, the East Asian region has some unique challenges which contribute considerably to the increased burden and management gap for osteoporosis. These challenges include a rapidly increasing elderly population, high fracture rates, underestimation of disease burden due to scarcity of epidemiological data especially from rural areas, unequal distribution of health resources, non-availability of diagnostic and therapeutic options in rural areas, and poor patient awareness and compliance. In addition, factors such as limited access to medical insurance/reimbursement policies and financial considerations further magnify the issue.2,3,10
Better knowledge of epidemiology, risk factors, diagnostic and therapeutic options, and quality of life will help governments and physicians to channelize their efforts in the right direction. Strategies such as timely diagnosis and treatment, lifestyle modification, maintenance of physical activity, and fall prevention will be required to reduce the burden of osteoporosis in the coming years.30 This East Asian subregional analysis is the first observational study evaluating real-world use of teriparatide in a community setting in East Asia. The current baseline analysis provides useful information regarding demographic characteristics, risk factors, fracture history, and HRQoL measures for patients who are currently being prescribed teriparatide in the real world in East Asia.
The limitations of this analysis are those that are commonly seen with non-randomized, observational studies. The study does not have a comparator arm hence this study does not generate any data comparing teriparatide with other treatments. All historical data, including fracture history, risk factors, comorbid conditions, and concomitant medications, are patient-reported, giving rise to the possibility of recall bias. In addition, patients included in this analysis were not distributed uniformly across the four East Asian regions and were also from different medical specialties. Given the heterogeneity of the East Asian subregion and the relatively small sample size for each participating region, our sample may not be truly representative of the general population.
Conclusions
Our analysis revealed that patients who are initiating treatment with teriparatide in East Asia are elderly women who have a high risk of fracture, due to their low BMD and history of fractures. Typically, these patients had associated comorbidities and were receiving several concomitant medications. Many patients had been treated with osteoporosis medications prior to initiating teriparatide and had back pain and poor HRQoL at baseline. This information would help physicians, payers and health policy makers to make informed decisions regarding the use of this osteoanabolic drug in their regions.
Data Sharing Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Approval and Informed Consent
The ALAFOS study complies with the principles of Good Clinical Practice and all local laws and regulations as applicable. Appropriate local site-level ethics committees have approved the study protocol. All participants provided written informed consent and were required to sign a Consent to Release Information form.
Acknowledgments
The authors thank Fernando Marin (MD, PhD), global study physician, Thomas Moll (PhD, MBA), clinical trial manager, Alan Brnabic (MSc), study statistician, and Sheetal Pradhan (MD), medical writer, for their contributions to this manuscript. All are employees of Eli Lilly and Company. The authors also thank Ms. Sin Ting Lau for her help in study support.
Author Contributions
C-HC, S-JL, J-KO, T-WH, YZ, M-TW, HY, JPYC, and JWK were involved in the recruitment of patients who participated in the study. All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
Eli Lilly and Company sponsored the ALAFOS study. Unique study identifier for the ALAFOS study: B3D-MC-B026.
Disclosure
The following authors did not report any conflict of interest related with the current manuscript: C-HC, S-JL, J-KO, T-WH, YHZ, HLY, JPYC, M-TW and JWK. JHH is an employee of Lilly Korea Ltd., T-JL is an employee of Eli Lilly and Company, LH and GZ are employees of Lilly Suzhou Pharmaceutical Co. Ltd. W-SW is an employee and stockholder of Eli Lilly and Company.
References
1. Dhanwal DK, Cooper C, Dennison EM. Geographic variation in osteoporotic hip fracture incidence: the growing importance of Asian influences in coming decades. J Osteoporos. 2010;2010:757102. doi:10.4061/2010/757102
2. Cheung EYN, Tan KCB, Cheung CL, Kung AWC. Osteoporosis in East Asia: current issues in assessment and management. Osteoporos Sarcopenia. 2016;2(3):118–133. doi:10.1016/j.afos.2016.07.001
3. Mithal A, Ebeling P. Asia pacific regional audit. Epidemiology, costs & burden of osteoporosis in 2013; 2013. Available from: https://www.iofbonehealth.org/sites/default/files/media/PDFs/Regional%20Audits/2013-Asia_Pacific_Audit_0_0.pdf.
4. Hwang JS, Chan DC, Chen JF, et al. Clinical practice guidelines for the prevention and treatment of osteoporosis in Taiwan: summary. J Bone Miner Metab. 2014;32(1):10–16. doi:10.1007/s00774-013-0495-0
5. Johnell O, Kanis JA. An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos Int. 2006;17(12):1726–1733. doi:10.1007/s00198-006-0172-4
6. Cooper C, Campion G, Melton LJ
7. Lau EM, Lee JK, Suriwongpaisal P, et al. The incidence of hip fracture in four Asian countries: the Asian Osteoporosis Study (AOS). Osteoporos Int. 2001;12(3):239–243. doi:10.1007/s001980170135
8. Lau EMC, Sambrook P, Seeman E, Leong KH, Leung PC, Delmas P. Guidelines for diagnosing, prevention and treatment of osteoporosis in Asia. APLAR J Rheumatol. 2006;9(1):24–36. doi:10.1111/j.1479-8077.2006.00161.x
9. Chen CH, Elsalmawy AH, Ish-Shalom S, et al. Study description and baseline characteristics of the population enrolled in a multinational, observational study of teriparatide in postmenopausal women with osteoporosis: the Asia and Latin America Fracture Observational Study (ALAFOS). Curr Med Res Opin. 2019;35(6):1041–1049. doi:10.1080/03007995.2018.1552576
10. Yu F, Xia W. The epidemiology of osteoporosis, associated fragility fractures, and management gap in China. Arch Osteoporos. 2019;14(1):32. doi:10.1007/s11657-018-0549-y
11. Forteo (teriparatide) [rDNA origin] injection) for subcutaneous use [US prescribing information]. Indianapolis, IN: Eli Lilly and Company; 2012. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2013/021318s036lbl.pdf.
12. European Medicines Agency. Forsteo summary of product characteristics; 2013. Available from: https://www.ema.europa.eu/en/documents/product-information/forsteo-epar-product-information_en.pdf.
13. Forsteo (teriparatide) [rDNA origin] injection) for subcutaneous use [China prescribing information]. 2019.
14. Forteo (teriparatide) [rDNA origin] injection) for subcutaneous use [Taiwan prescribing information]. 2012. Available from: https://www.fda.gov.tw/MLMS/ShowFile.aspx?LicId=10000787&Seq=052&Type=9.
15. Forteo (teriparatide) [rDNA origin] injection) for subcutaneous use [Hong Kong prescribing information]. 2014. Available from: https://www.mims.com/hongkong/drug/info/forteo.
16. Forsteo (teriparatide) [rDNA origin] injection) for subcutaneous use [South Korea prescribing information]. 2015. Available from: https://nedrug.mfds.go.kr/pbp/CCBBB01/getItemDetail?itemSeq=200209623.
17. Cheng XG, Yang DZ, Zhou Q, et al. Age-related bone mineral density, bone loss rate, prevalence of osteoporosis, and reference database of women at multiple centers in China. J Clin Densitom. 2007;10(3):276–284. doi:10.1016/j.jocd.2007.05.004
18. Cui LH, Choi JS, Shin MH, et al. Prevalence of osteoporosis and reference data for lumbar spine and hip bone mineral density in a Korean population. J Bone Miner Metab. 2008;26(6):609–617. doi:10.1007/s00774-007-0847-8
19. Iki M, Kagamimori S, Kagawa Y, Matsuzaki T, Yoneshima H, Marumo F. Bone mineral density of the spine, hip and distal forearm in representative samples of the Japanese female population: Japanese Population-Based Osteoporosis (JPOS) Study. Osteoporos Int. 2001;12:529–537. doi:10.1007/s001980170073
20. Lynn HS, Lau EM, Au B, Leung PC. Bone mineral density reference norms for Hong Kong Chinese. Osteoporos Int. 2005;16(12):1663–1668. doi:10.1007/s00198-005-1899-z
21. Yeh LR, Chen CK, Lai PH. Normal bone mineral density in anteroposterior, lateral spine and hip of Chinese men in Taiwan: effect of age change, body weight and height. J Chin Med Assoc. 2004;67(6):287–295.
22. National Health Insurance Administration (Taiwan). Chapter 5. Hormones & drugs affecting hormonal mechanism; 2019. Available from: https://www.nhi.gov.tw/Content_List.aspx?n=E70D4F1BD029DC37&topn=3FC7D09599D25979.
23. Korea Insurance Charge Review Association. 2017. Available from: http://www.hicra.or.kr/sub_asp/04_data01.html?mode=read&read_no=1282&now_page=2&menu.
24. Orimo H, Nakamura T, Hosoi T, et al. Japanese 2011 guidelines for prevention and treatment of osteoporosis–executive summary. Arch Osteoporos. 2012;7:3–20. doi:10.1007/s11657-012-0109-9
25. Rajzbaum G, Jakob F, Karras D, et al. Characterization of patients in the European Forsteo Observational Study (EFOS): postmenopausal women entering teriparatide treatment in a community setting. Curr Med Res Opin. 2008;24(2):377–384. doi:10.1185/030079908X261087
26. Silverman S, Miller P, Sebba A, et al. The Direct Assessment of Nonvertebral Fractures in Community Experience (DANCE) study: 2-year nonvertebral fragility fracture results. Osteoporos Int. 2013;24(8):2309–2317. doi:10.1007/s00198-013-2284-y
27. Soen S, Fujiwara S, Takayanagi R, et al. Real-world effectiveness of daily teriparatide in Japanese patients with osteoporosis at high risk for fracture: final results from the 24-month Japan Fracture Observational Study (JFOS). Curr Med Res Opin. 2017;33(11):2049–2056. doi:10.1080/03007995.2017.1354826
28. The Osteoporosis Society of Hong Kong (OSHK) Task Group. 2013 OSHK guideline for clinical management of postmenopausal osteoporosis in Hong Kong. Hong Kong Med J. 2013;19(Suppl 2):S1–S140.
29. Guidelines for primary osteoporosis diagnosis and management. Osteoporosis and bone mineral research society of the Chinese Medical Association; 2017. Available from: http://kns.cnki.net/kcms/detail/13.1222.R.20171027.1618.002.html.
30. Hagino H, Katagiri H, Okano T, Yamamoto K, Teshima R. Increasing incidence of hip fracture in Tottori Prefecture, Japan: trend from 1986 to 2001. Osteoporos Int. 2005;16(12):1963–1968. doi:10.1007/s00198-005-1974-5
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.