Back to Journals » Therapeutics and Clinical Risk Management » Volume 16
Tension Pneumocephalus from Endoscopic Endonasal Surgery: A Case Series and Literature Review
Authors Li W, Liu Q, Lu H, Wang H, Zhang H, Hu L, Sun X , Gu Y, Li H, Zhao W , Wang D
Received 24 April 2020
Accepted for publication 7 June 2020
Published 19 June 2020 Volume 2020:16 Pages 531—538
DOI https://doi.org/10.2147/TCRM.S258890
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Deyun Wang
Wanpeng Li,* Quan Liu,* Hanyu Lu, Huan Wang, Huankang Zhang, Li Hu, Xicai Sun, Yurong Gu, Houyong Li, Weidong Zhao, Dehui Wang
Department of Otolaryngology-Head and Neck Surgery, Affiliated Eye Ear Nose and Throat Hospital, Fudan University, Shanghai 200031, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Weidong Zhao; Dehui Wang Email [email protected]; [email protected]
Purpose: Tension pneumocephalus (TP) caused by endoscopic endonasal surgery is a serious complication. We report four cases of TP caused by endoscopic surgery and review other cases in the literature, with special attention devoted to symptoms, imaging features, and therapeutic approaches.
Methods: A retrospective chart review of patients who experienced TP caused by endoscopic surgery in our institution between 2015 and 2018 was performed. Additionally, the MEDLINE database was searched for all case series or reports of TP caused by endoscopic surgery.
Results: Eighteen articles were identified for review, including four cases from the authors’ institution; ultimately, 26 cases were included in the present study. The main symptoms of TP were headache and a change in mental status. Cerebrospinal fluid (CSF) leakage was reported in 21 of the 26 patients (80.8%). Eight of the 26 patients (30.8%) presented with the “Mount Fuji sign” imaging feature. Twenty-four patients were treated with surgical intervention for TP (endoscopic multilayer closure of skull base defect, cranial burr hole, or bifrontal craniotomy). In addition, the present study is the first to report two patients with TP who were successfully treated conservatively.
Conclusion: The therapeutic method for treating TP should depend on the degree of the mass effect and clinical symptoms. When patients with TP present with obvious symptoms of CSF leakage and intracranial hypertension, urgent surgical multilayer repair of the skull base defects and/or release of the intracranial pressure are keys to treating these patients. However, conservative treatment under close observation is also feasible when the related symptoms are not overtly obvious.
Keywords: tension pneumocephalus, endoscopic surgery, cerebrospinal fluid, skull base
Introduction
Pneumocephalus is defined as the presence of air in the cranial cavity, which may develop after neurosurgery or head trauma; however, it may spontaneously progress and resolve adiabatically. Tension pneumocephalus (TP) is a clinical manifestation characterized by the continuous accumulation of air in the cranial cavity, which leads to neurological signs and symptoms due to a mass effect by the creation of intracranial hypertension.1 Understanding TP-related risk factors, imaging findings, and clinical signs/symptoms is essential for rapid identification and treatment. Delayed clinical diagnosis and treatment may cause neurological deterioration and mortality.
The occurrence of intracranial complications in endoscopic transnasal surgery depends primarily on the presence of defects in the skull base.2 These complications (cerebrospinal fluid rhinorrhea, meningitis, pneumocephalus, TP, etc.) are very rare.3 In addition, there are only a few studies in the literature describing the development of TP in patients who undergo endoscopic surgery for various reasons (chronic rhinosinusitis, nasal septum deviation, pituitary macroadenoma).4–6 Accordingly, the present study collected data from all previously reported cases of TP caused by endoscopic endonasal surgery in the literature, as well as four additional cases treated by the authors, to collectively review the diagnosis, management, and treatment outcomes of this rare disease.
Patients and Methods
Study Design and Literature Review
A retrospective chart review of four patients who were diagnosed with TP caused by endoscopic endonasal surgery and treated at our institution between 2015 and 2018 was performed. The diagnosis of TP requires the fulfilment of three criteria: clear imaging changes on computed tomography (CT) after surgery revealing intracranial air compression; recorded changes in nerve function, including decreased alertness, obtundation, altered mental state, or focal neurological deficits; and specific documentation of the diagnosis of TP. This study was approved by the Institutional Review Board of the Affiliated Eye Ear Nose and Throat Hospital (AEENTH) at Fudan University (Shanghai, China).
Additionally, a comprehensive search of the MEDLINE database for all articles describing TP caused by endoscopic endonasal surgery was performed. The query terms in the search included “tension pneumocephalus,” “endoscopic,” “endonasal,” “sinus surgery,” “surgery,” and “skull base.” Specifically, information regarding age, sex, time to presentation (acute: ≤72 h; delayed:delayed >72 h), imaging manifestations, primary lesions associated with TP, location of skull base defect, symptoms of TP, therapeutic approach to TP, and clinical outcomes were extracted.
Case Presentations
Case 1
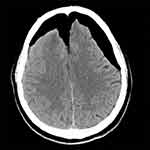
A patient underwent endoscopic resection of a craniopharyngioma in the sellar region via the sellar tubercle approach. An artificial dura mater and septal mucosal flap were used for multilayered reconstruction of the skull base. Thirty-two days postoperatively, the patient reported severe headache and cough. Due to these complaints, the patient was admitted and underwent CT, which revealed intracranial TP at the top of the forehead and compression of the bilateral frontal lobes (Figure 1). Thus, an urgent cranial burr hole was performed. Four days later, the patient presented with distinct clear fluid rhinorrhea, and endoscopic examination revealed a small fissure on the left margin of the septal flap. Along the fissure to the floor and front of the sella, a small defect of the dura mater and active cerebrospinal fluid (CSF) outflow was visible. Multilayer closure was accomplished using abdominal fat, fascia, and free middle turbinate mucosal graft above the small defects. Fibrin glue was used to improve adherence of the graft, and nasal packs were placed beneath to support the repair. The patient did not voice any further complaints at the 33-month follow-up.
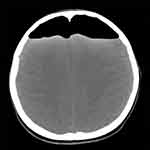 |
Figure 1 Computed tomography revealing intracranial pneumocephalus, evident at the top of the forehead, and compression of the bilateral frontal lobes. |
Case 2
A patient underwent endoscopic resection of chordoma in the clival region and multilayer reconstruction of the skull base without apparent complications. Sixteen days postoperatively, the patient presented with clear fluid rhinorrhea and headache. The symptoms of clear fluid rhinorrhea gradually recovered, while the headache persisted. Thirty days postoperatively, the patient was admitted, and magnetic resonance imaging (MRI) revealed pneumocephalus in the bilateral ventricles, with slight enlargement of the anterior horn of the lateral ventricles (Figure 2). The patient was administered conservative treatment (intravenous antibiotics, mannitol for lowering intracranial pressure, loop diuretics, bed rest, elevation of the head of the bed, and avoidance of positive pressure events), and the symptoms of headache gradually improved. On hospital day 11, the patient was discharged without any complaints. MRI revealed that bilateral ventricular pneumocephalus decreased significantly (Figure 3). The patient continued to do well 56 months after surgery.
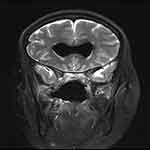 |
Figure 2 Magnetic resonance imaging revealing pneumocephalus in the bilateral ventricles, with slight enlargement of the anterior horn of the lateral ventricles. |
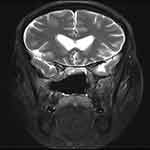 |
Figure 3 Magnetic resonance imaging revealing a significant decrease in bilateral ventricular pneumocephalus. |
Case 3
A patient underwent endoscopic resection of a giant chordoma in the skull base via transnasal and transoral approaches. Artificial dura mater and septal mucosal flaps were used for multilayered reconstruction of the skull base. Seventeen days after surgery, the patient developed clear fluid rhinorrhea. Nineteen days after surgery, the patient developed serious headache and disturbances of consciousness after severe vomiting. CT revealed obvious pneumocephalus in the intracranial lobe. The brain tissue was compressed, and the ventricles and fissures were markedly enlarged (Figure 4). Consequently, emergency endoscopic endonasal exploration was performed. Endoscopic visualization revealed a small fissure in the left margin of the septal flap. Along the fissure to the clival region of the skull base, there was a small defect in the dura mater, and CSF leakage was also visible. The small defect was repaired by multilayer closure, which included abdominal fat, fascia, and free middle turbinate mucosal graft. Fibrin glue was used to improve adherence of the graft, and nasal packs were placed beneath to support the repair. The patient gradually recovered within 2 weeks. At the 12-month follow-up, no leakage or other complications occurred.
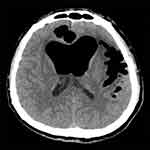 |
Figure 4 Computed tomography revealing obvious pneumocephalus in the intracranial lobe, with compression of the brain tissue, and marked enlargement of the ventricles and fissures. |
Case 4
A patient underwent endoscopic resection of a giant meningioma in the anterior skull base. Artificial dura mater and septal mucosal flaps were used for multilayered reconstruction of the skull base. Postoperatively, the patient presented with clear fluid rhinorrhea and headache, and light hemiplegia appeared in the right limb. Computed tomography revealed a large area of pneumocephalus in the bilateral anterior cranial fossa. The frontal retention of air caused widening of the interhemispheric fissure, leading to a peak appearance of the frontal poles commonly referred to as “Mount Fuji sign” (Figure 5). The patient was administered conservative treatment for 2 weeks (intravenous antibiotics, mannitol for lowering intracranial pressure, loop diuretics, bed rest, elevation of the head of the bed, and avoidance of positive pressure events). During the treatment process, related symptoms gradually decreased, and intracranial pneumocephalus was alleviated. Finally, the patient recovered without further sequelae, with disappearance of intracranial pneumocephalus and no CSF leakage. The patient continued to do well 18 months after surgery.
Results
Eighteen articles were identified using the search strategy outlined above in the study design and literature review.3–20 The earliest report from this search was a single case report published in 1994.3 Including the four cases from our institutions described above, a total of 26 cases were included in the review (Table 1). These cases included 16 men and 10 women, with a median age of 58 years (range, 8–89 years) at diagnosis. Presentation was acute in nine patients and delayed in 17. The imaging characteristic of “Mount Fuji sign” was evident in eight (30.7%) patients. The primary diseases associated with TP were as follows: skull base tumors (10/23 [43.5%]), chronic rhinosinusitis (8/23 [34.8%]), nasal septum deviation (3/23 [13.0%]), CSF leakage (1/23 [4.3%]), and tetraparesia (1/23 [4.3%]). There were two causes for TP: one was iatrogenic injury of the skull base during endoscopic surgery, and the other was an inappropriate flap in the repair of skull base defect.
 |
Table 1 Summary of All Reported Cases of TP Caused by Endoscopic Endonasal Surgery |
The main symptoms of TP were headache and a change in the mental status. Some patients also presented with a wide variety of other symptoms, including lower limb, diplegia aphasia, visual acuity, ataxia, and vomiting. CSF leakage was reported in 21 patients (21/26 [80.8%]). For the treatment of TP, endoscopic multilayer closure of the skull base defect was performed in 13 patients, cranial burr hole surgery in two, cranial burr hole surgery and endoscopic multilayer closure of the skull base defect in five, and bifrontal craniotomy to repair the defect in the skull base in two patients. In addition, this is the first study to report successful conservative treatment of two patients with TP. For the clinical outcome of TP, 24 patients recovered well without obvious complications after active intervention. However, one patient experienced no neurological improvement and one died.
Discussion
There are two mechanisms responsible for the development of TP. The first is the “ball-valve” mechanism, in which air enters a defect while coughing or sneezing, but does not exit the cranial vault. The second is the “inverted pop bottle” mechanism, in which air enters the intracranial compartment to balance the pressure difference when CSF is discharged through the defect.21 TP is a life-threatening neurosurgical emergency with potentially devastating consequences if not treated in a timely manner. One should be highly suspicious of the presence of TP when a patient who has recently undergone skull base surgery or other forms of endoscopic surgery suddenly experiences severe headache or exhibits a change in mental status.
Prompt evaluation using CT or MRI can help in making a timely diagnosis, which usually leads to surgical correction for TP. CT is the preferred diagnostic method for pneumocephalus, and can detect as little as 0.5 cm3 of air.22 For most patients with TP caused by iatrogenic injury of the skull base, CT can also determine the site of bone defect(s). The prominent feature of TP on imaging is the so-called “Mount Fuji sign,” in which the frontal lobe has a peaked appearance, likely as a result of tethering by intact bridging veins.23 Among the sample of 26 patients in this study, the Mount Fuji sign was present in eight (30.8%).
The onset of air accumulation in the intracranial cavity after skull base surgeries can be acute (≤72 h) or delayed (>72 h). A comprehensive literature review by Pillai et al, involving 20 patients with traumatic TP, reported acute onset in most cases.24 However, among patients with TP caused by endoscopic endonasal surgery in the present study, the authors found that those with delayed onset numbered more than those with acute onset. Surprisingly, Matsuda et al reported that one patient unexpectedly incurred prominent TP 5 years after endoscopic endonasal trans-sphenoidal surgery for pituitary adenoma.12 Therefore, it is crucial that otolaryngologists and neurosurgeons understand that TP may also occur many years after endoscopic surgery.
In the literature, it has been reported that pneumocephalus heals spontaneously with conservative treatment in patients with small defects.13 However, many studies have reported that, regardless of the size of the skull base defect in TP, conservative treatment is ineffective because air will continue to enter the intracranial cavity through the existing skull base defect.3,4,25 In addition, Aksoy et al described a case of TP with minimal skull base defect (only 1 mm), but the patient experienced a herniation table and serious neurological problems.3 Thus, urgent surgical repair of skull base defects and/or release of intracranial pressure are key to treating patients with TP. These surgical methods include endoscopic multilayer closure of skull base defect, cranial burr hole, and bifrontal craniotomy.
In the clinical experience of the authors, the method for managing TP should depend on the degree of mass effect and clinical symptoms. Two patients (cases 1 and 3) exhibited obvious symptoms of CSF leakage and intracranial hypertension. Surgical multilayered reconstruction of skull base defects was performed as soon as possible. In addition, the patient in case 1 experienced significant neurological deterioration. An emergency cranial burr hole was also adopted to relieve intracranial pressure and reduce secondary injury. However, when the symptoms of CSF leakage and intracranial hypertension were not obvious in cases 2 and 4, conservative treatment under close observation was also successfully performed. The conservative method included intravenous antibiotics, mannitol to lower intracranial pressure, loop diuretics, bed rest, elevation of the head of the bed, and avoidance of positive pressure events. These patients recovered well without any sequelae and avoided surgical repair of the skull base defects. In addition, some patients developed TP caused by endoscopic surgery for skull base tumors due to an inappropriate flap in the repair of skull base defects. Most patients experienced CSF leakage. Therefore, a reduction in the amount of CSF leakage and reliable skull base reconstruction during surgery are key to preventing the development of TP.
Conclusion
TP is a life-threatening neurosurgical emergency with potentially devastating consequences if not treated in a timely manner. The therapeutic method should depend on the degree of mass effect and clinical symptoms. When patients with TP present with obvious symptoms of CSF leakage and intracranial hypertension, urgent surgical multilayer repair of the skull base defects or/and release of intracranial pressure are key to treating these patients. However, conservative treatment under close observation was also feasible when the symptoms of CSF leakage and intracranial hypertension were not overtly obvious.
Ethics
This study was approved by the Institutional Review Board of the Affiliated Eye Ear Nose and Throat Hospital (AEENTH) at Fudan University.
Consent for Publication
Written informed consent for publication of anonymized case details, which included accompanying images, was obtained from the patients.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Webber-Jones JE. Tension pneumocephalus. J Neurosci Nurs. 2005;37(5):272–276. doi:10.1097/01376517-200510000-00007
2. Solomiichuk VO, Lebed VO, Drizhdov KI. Posttraumatic delayed subdural tension pneumocephalus. Surg Neurol Int. 2013;4:37. doi:10.4103/2152-7806.109537
3. Aksoy F, Dogan R, Ozturan O, Tugrul S, Yildirim YS. Tension pneumocephalus: an extremely small defect leading to an extremely serious problem. Am J Otolaryngol. 2013;34(6):749–752. doi:10.1016/j.amjoto.2013.07.011
4. Martinez-Capoccioni G, Serramito-Garcia R, Cabanas-Rodriguez E, Garcia-Allut A, Martin-Martin C. Tension pneumocephalus as a result of endonasal surgery: an uncommon intracranial complication. Eur Arch Otorhinolaryngol. 2014;271(5):1043–1049. doi:10.1007/s00405-013-2650-6
5. Iqbal SM, Khan AJ, Zhi C. Tension pneumocephalus: a rare complication of transsphenoidal resection of a pituitary macroadenoma. Cureus. 2019;11(5):e4623.
6. Naraghi M, Ghazizadeh M. Tension pneumocephalus: a life-threatening complication of septoplasty and septorhinoplasty. B-ENT. 2012;8(3):203–205.
7. Pruss H, Klingebiel R, Endres M. Tension pneumocephalus with diplegia and deterioration of consciousness. Case Rep Neurol. 2011;3(1):48–49. doi:10.1159/000324824
8. Clevens RA, Bradford CR, Wolf GT. Tension pneumocephalus after endoscopic sinus surgery. Ann Otol Rhinol Laryngol. 1994;103(3):235–237. doi:10.1177/000348949410300312
9. Emmez H, Durdag E, Uslu S, Pasaoglu A, Ceviker N. Intracerebral tension pneumocephalus complicating endoscopic sinus surgery: case report. Acta Neurochir (Wien). 2009;151(8):1001–1002. doi:10.1007/s00701-009-0347-9
10. Iplikcioglu AC, Bek S, Bikmaz K, Basocak K. Tension pneumocyst after transsphenoidal surgery for Rathke’s cleft cyst: case report. Neurosurgery. 2003;52(4):
11. Mammis A, Agarwal N, Eloy JA, Liu JK. Intraventricular tension pneumocephalus after endoscopic skull base surgery. J Neurol Surg A Cent Eur Neurosurg. 2013;74(Suppl 1):e96–e99. doi:10.1055/s-0032-1328954
12. Matsuda T, Horiguchi K, Higuchi Y, Hanazawa T, Okamoto Y, Iwadate Y. Unexpected prominent tension pneumosella five years after endoscopic endonasal transsphenoidal surgery. Laryngoscope. 2018;128(8):1798–1801. doi:10.1002/lary.27060
13. Whitmore RG, Bonhomme G, Balcer LJ, Palmer JN. Tension pneumocephalus after endoscopic sinus surgery: case report of repair and management in absence of obvious skull base defect. Ear Nose Throat J. 2008;87(2):96–99. doi:10.1177/014556130808700212
14. Simmons J, Luks AM. Tension pneumocephalus: an uncommon cause of altered mental status. J Emerg Med. 2013;44(2):340–343. doi:10.1016/j.jemermed.2012.01.055
15. Yin C, Chen BY. Tension pneumocephalus from skull base surgery: A case report and review of the literature. Surg Neurol Int. 2018;9:128. doi:10.4103/sni.sni_91_18
16. Polster SP, Obuobi S, Brutto VJD, Avner K, Markopoulou A, Wong RH. An unusual presentation of dystonia and chorea from intraventricular pneumocephalus. Surg Neurol Int. 2018;9:193. doi:10.4103/sni.sni_190_18
17. Celikoglu E, Hazneci J, Ramazanoglu AF. Tension pneumocephalus causing brain herniation after endoscopic sinus surgery. Asian J Neurosurg. 2016;11(3):309–310. doi:10.4103/1793-5482.179646
18. Yorgason JG, Arthur AS, Orlandi RR, Apfelbaum RI. Endoscopic decompression of tension pneumosella following transsphenoidal pituitary tumor resection. Pituitary. 2004;7(3):171–177. doi:10.1007/s11102-005-3410-4
19. Bly RA, Morton RP, Kim LJ, Moe KS. Tension pneumocephalus after endoscopic sinus surgery: a technical report of multiportal endoscopic skull base repair. Otolaryngol Head Neck Surg. 2014;151(6):1081–1083. doi:10.1177/0194599814547502
20. Kahilogullari G, Meco C, Zaimoglu M, et al. Pneumocephalus after endoscopic odontoidectomy in a pediatric patient: the lesson learned. Childs Nerv Syst. 2015;31(9):1595–1599. doi:10.1007/s00381-015-2740-4
21. DelGaudio JM, Ingley AP. Treatment of pneumocephalus after endoscopic sinus and microscopic skull base surgery. Am J Otolaryngol. 2010;31(4):226–230. doi:10.1016/j.amjoto.2009.02.012
22. Becker WJ. Pneumocephalus as a cause for headache. Can J Neurol Sci. 2002;29(3):278–281. doi:10.1017/S0317167100002079
23. Sadeghian H. Mount Fuji sign in tension pneumocephalus. Arch Neurol. 2000;57(9):1366. doi:10.1001/archneur.57.9.1366
24. Pillai P, Sharma R, MacKenzie L, et al. Traumatic tension pneumocephalus – two cases and comprehensive review of literature. Int J Crit Illn Inj Sci. 2017;7(1):58–64. doi:10.4103/IJCIIS.IJCIIS_8_17
25. Aferzon M, Aferzon J, Spektor Z. Endoscopic repair of tension pneumocephalus. Otolaryngol Head Neck Surg. 2001;124(6):688–689. doi:10.1177/019459980112400618
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.