Back to Journals » International Journal of Nanomedicine » Volume 17
Targeted Therapy for Inflammatory Diseases with Mesenchymal Stem Cells and Their Derived Exosomes: From Basic to Clinics
Authors Wang S, Lei B, Zhang E, Gong P, Gu J, He L, Han L, Yuan Z
Received 23 December 2021
Accepted for publication 9 April 2022
Published 19 April 2022 Volume 2022:17 Pages 1757—1781
DOI https://doi.org/10.2147/IJN.S355366
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Yan Shen
Shuo Wang,1 Biyu Lei,1 E Zhang,2 Puyang Gong,1 Jian Gu,1 Lili He,1 Lu Han,1 Zhixiang Yuan1
1College of Pharmacy, Southwest Minzu University, Chengdu, 610041, Sichuan, People’s Republic of China; 2Department of Basic Sciences, Officers College of People’s Armed Police, Chengdu, Sichuan, 610213, People’s Republic of China
Correspondence: Zhixiang Yuan; Lu Han, Email [email protected]; [email protected]
Abstract: Inflammation is a beneficial and physiological process, but there are a number of inflammatory diseases which have detrimental effects on the body. In addition, the drugs used to treat inflammation have toxic side effects when used over a long period of time. Mesenchymal stem cells (MSCs) are pluripotent stem cells that can be isolated from a variety of tissues and can be differentiate into diverse cell types under appropriate conditions. They also exhibit noteworthy anti-inflammatory properties, providing new options for the treatment of inflammatory diseases. The therapeutic potential of MSCs is currently being investigated for various inflammatory diseases, such as kidney injury, lung injury, osteoarthritis (OA), rheumatoid arthritis (RA), and inflammatory bowel disease (IBD). MSCs can perform multiple functions, including immunomodulation, homing, and differentiation, to enable damaged tissues to form a balanced inflammatory and regenerative microenvironment under severe inflammatory conditions. In addition, accumulated evidence indicates that exosomes from extracellular vesicles of MSCs (MSC-Exos) play an extraordinary role, mainly by transferring their components to recipient cells. In this review, we summarize the mechanism and clinical trials of MSCs and MSC-Exos in various inflammatory diseases in detail, with a view to contributing to the treatment of MSCs and MSC-Exos in inflammatory diseases.
Keywords: mesenchymal stem cells, exosome, inflammatory diseases, clinic data, drug delivery, targeted therapy
Introduction
Inflammation is a natural biophylaxis of the human body that occurs when tissues and cells are attacked by hazardous substances that have certain adverse effects on the human body (such as pathogens, chemical substances, or physical damage) that can repair damaged tissues, restore organ function, and maintain homeostasis through various degrees of fever, leukocytosis, and the enhancement of one’s metabolism.1 The cardinal symptoms of inflammation include redness, edema, and leukocytosis due to hyperemia of the inflammatory lesion, cell and tissue proliferation, enhanced metabolism, and localized infection.2 Although inflammation is predominantly a physiological and beneficial process, uncontrolled or unresolved inflammation can lead to tissue damage, resulting in a plethora of chronic inflammatory diseases.3 These include inflammatory bowel disease, neurodegenerative disease, and rheumatoid arthritis. Although inflammation is not the major cause of these diseases, it is instrumental in the study of their pathogenesis.4 Currently, the drugs used to treat inflammation are mostly steroids, glucocorticoids, and nonsteroidal anti-inflammatory drugs.5 Nevertheless, long-term use of these drugs can induce adverse effects, such as renal, gastrointestinal, and cardiovascular toxicity. Hence, it is imperative to explore new anti-inflammatory drugs with less toxicity and selective effects.6
MSCs are a heterogeneous cell population originating from the mesoderm in the early stage of embryonic development,7 and include stem cells and differentiated progeny from the early stage of embryonic development and subsequent developmental stages.8 As one of the most easily obtained primary cells, MSCs can be isolated from a variety of tissues, such as the umbilical cord, adipose tissue, dermis, tendons, bone marrow, muscles, and dental pulp.9 Given their therapeutic potential, MSCs and MSC-Exos have been widely studied in order to evaluate their availability. As somatic precursor cells or stem cells, MSCs are one of the most widely studied pluripotent stem cells. In addition, the ability to differentiate into multiple lineages and regulate inflammation is the main criterion for defining MSCs.10 MSCs can act in many ways, but are generally believed to enable damaged tissues to form a balanced inflammation and regeneration microenvironment in the presence of severe inflammation.11 Nevertheless, the implantation and differentiation of MSCs at the injury site are very short-lived. MSCs do not affect tissue repair through cell replacement but through paracrine factors.12 Among them, MSC-Exos have attracted much attention because they are capable of carrying complex proteins, nucleic acids, and lipids, containing abundant miRNAs, and delivering these contents to recipient cells.13 MSC-Exos are nanoscale extracellular vesicles with a lipid bilayer membrane structure secreted by MSCs. Physiological and pathological processes can be regulated by affecting the survival, proliferation, migration, and gene expression of recipient cells while also reprogramming the behavior of targeted cells. When compared with MSCs, MSC-Exos are easier to store and transport.14
As a result, we have provided a comprehensive and critical update on MSCs and MSC-Exos as potential options for the treatment of inflammation. In this review, we summarize the functions of MSCs and MSC-Exos, the mechanisms for the clinical application of inflammation-related diseases, and discuss their therapeutic potential as anti-inflammatory drug delivery platforms in the hope of providing inspiration for future investigations.
Characteristics of MSCs and MSC-Exos
Studies have shown that MSCs were first discovered in monocytes in bone marrow.15 Owing to different protein expression profiles, the characteristics of MSCs from various sources are slightly different, and they generally have self-renewal ability, multidirectional differentiation potential, immune regulation, and all meet the minimum identification standards established by the International Cell Therapy Association.16,17 The basic definition of MSCs involves the following characteristics: (1) maintaining plastic adhesion in a standard medium; (2) the expression of CD105, CD73, and CD90, but not CD45, CD34, CD14, CD11b, CD79a, CD19, or HLADR surface markers; and (3) the differentiation of osteoblasts, adipocytes, and chondroblasts under standard in vitro conditions.18–21
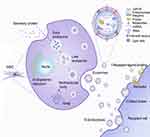
Exosomes are extracellular vesicles (40–150 nm in diameter). Their biogenesis stems from endosomes, with early endosomes maturing into multivesicular bodies (MVBs) and releasing their contents in the form of exosomes.22 Ceramide, sphingomyelin, cholesterol, and various lipid molecules are abundantly present in exosomal membranes. When exosomes are released into the intercellular space, they can be absorbed by recipient cells via endocytosis, and either receptor ligand or direct binding (Figure 1). Exosomes containing a large number of miRNAs are composed of molecules such as lipids, enzymes, nucleic acids, protein structures, and pro-inflammatory and anti-inflammatory cytokines, which can prevent miRNA from being degraded by ribonucleases in body fluids. Consequently, miRNAs in exosomes are more abundant than those found in their parent cells and body fluids.23,24 After entry of MSC-Exos into recipient cells, miRNA is released to further target and silence the mRNA of related proteins, thereby affecting the physiological functions of the recipient cells.25 Exosomes have no cell structure, and are both highly stable and non-carcinogenic. They further benefit from cell-and tissue-specific homing, immune silencing, and a lack in vascular obstruction tendency, all of which give them greater therapeutic advantages than MSCs. Therefore, exosome-based cell-free therapies have been extensively studied.
Mechanism of MSCs and MSC-Exos in the Treatment of Inflammatory Diseases
Immunomodulatory
Macrophages are specialized cells that can engulf and eliminate both foreign organisms and apoptotic cells. M1 macrophages secrete pro-inflammatory cytokines, while M2 type macrophages secrete anti-inflammatory cytokines to promote tissue repair. The imbalance between the two activities can lead to continuous inflammation, hinder the normal repair process, and cause damage to tissue repair.26 MSCs can inhibit M1 macrophages and promote their differentiation into M2 macrophages, downregulate the production of IL-6 and IL-23, and increase the expression levels of interleukin 10 (IL-10) and TGF-β, exerting an anti-inflammatory role and enhancing the repair of damaged tissues.27 Macrophages were found to have an anti-inflammatory M2 phenotype, and the expression of CD206 and secretory cytokine IL-10 increased when they were co-cultured with MSCs. Investigators who injected macrophages co-cultured with MSCs, lipopolysaccharide (LPS)-stimulated macrophages, and normal macrophages into acute kidney injury (AKI) mice found that lipopolysaccharide-stimulated macrophages and normal macrophage-infused mice had more severe functional and histological damage, while macrophages co-cultured with MSC-infused macrophages exhibited less-severe kidney damage. In summary, MSCs can improve inflammation by activating M2 type macrophages.28 In addition, MSCs can affect the dendritic cell (DCs) phenotype, cytokine release, differentiation and maturation, and weaken their antigen presentation ability.29 Activated DCs produce a variety of pro-inflammatory cytokines (TNF-α, IL-1β, IL-6, and IL-12) and express high levels of co-stimulatory molecules (CD40, CD80, and CD86), thereby promoting the development of inflammation. Previous studies have noted the importance of DC maturation and activation in the progression of inflammation. MSCs can downregulate the expression of cyclinD2 and p27Kip1 genes to inhibit DC maturation and change the secretion characteristics of its tolerogenic phenotypes, impairing its antigen presentation and migration functions, and regulating the secretion of related cytokines (such as upregulating IL-10 levels and down-regulating IL-12 levels30,31). Inhibiting the proliferation and differentiation of natural killer cells (NKs) and reducing the expression of NKs activation receptors (such as 2B4 and NKG2D) also reduce the production of pro-inflammatory cytokines.32 The mechanism involves inducing apoptosis of T lymphocytes, inhibiting their proliferation, and inhibiting the expression of chemokine receptors on B lymphocytes. Secretion of soluble factors such as CC-chemokine ligand 2 (CCL2), TNF stimulating gene 6 (TSG6), prostaglandin E2 (PGE2), nitric oxide (NO), indoleamine 2,3-dioxygenase (IDO), and IL-10 and then play a regulatory role in the immune system.11,33 An imbalance in the immune expression of Th17 and Treg cells is an important reason for the long-term inflammatory state. Th17 and Treg cells can transform and restrict each other, and regulate intestinal immune homeostasis. Therefore, regulating the balance of Th17/Treg cells is crucial for the treatment of inflammatory diseases. MSCs inhibit the transformation of T cells into Th17 cells by inhibiting the expression of ROR γt and simultaneously induce the expression of Treg cells by upregulating Foxp3+ Tregs.34 Once the Th17/Treg cell balance is disrupted, MSCs can further inhibit the number and function of Th1 and Th17 cells, increase the number and promote the function of Treg cells in mesenteric lymph nodes, and finally balance Th17/Treg cells in a dynamic manner, thereby alleviating inflammation.35
Neutrophils, one of the earliest cells to develop after inflammation, are recruited to the infection site to perform defense functions at an early stage. Nevertheless, excessive recruitment of neutrophils may amplify inflammatory effects and aggravate inflammatory damage.36 Deleterious reactive oxygen species (ROS), neutrophil extracellular traps (NETs), and proteases such as neutrophil elastase (NE), myeloperoxidase (MPO), and matrix metalloproteinases were produced by activated neutrophils, which complicated the inflammatory microenvironment. NETs have a certain effect on the removal of pathogenic bacteria from lung injury sites. However, excessive amounts of NETs have a damaging effect on epithelial cells and vascular endothelial cells of lung tissue owing to the various antibacterial peptides and proteases contained within them.37 MSCs can reduce the production of pro-inflammatory cytokines, increase the production of IL-10, reduce the production of TNF-α, and reduce the influx and accumulation of neutrophils; MSCs, similarly, can inhibit the activation of T cells by regulating several immune regulatory factors such as TGF-β, PGE2, HLA-G5, and membrane-bound molecules (PD-L1, VCAM-1, and Gal-1),38,39 while also increasing Treg and anti-inflammatory TH2 cells. In addition, IDO released by MSCs can inhibit the production of T cell factors and conversion of monocytes and CD34+ hematopoietic progenitor cells to DCs in vitro, highlighting the immunomodulatory effect of MSCs.40
Homing
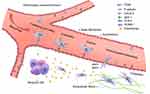
The current definition of homing in regard to MSCs is the retention of MSCs in the vasculature of the tissue and the subsequent migration between endothelial cells.41 The homing process can be divided into five steps: (1) tethering and rolling; (2) activation; (3) arrest; (4) transmigration or diapedesis; and (5) migration. Selectins in endothelial cells are mainly involved in the first step of homing. MSCs express CD44, which binds to selectin and causes the cells to start rolling along the vessel wall. The second step is facilitated by G protein-coupled chemokine receptors, and the expression of SDF-1 in endothelial cells is thought to be critical for this step. In the third step of homing, integrins play an important role in the stable activation-dependent arrest. Very late antigen 4 (VLA-4) expressed by MSCs interacts with vascular cell adhesion molecule 1 (VCAM-1) on endothelial cells. During transmigration or diapedesis, MSCs pass through the endothelial cell layer and basement membrane under the action of matrix metalloproteinases (MMPs). Finally, MSCs migrate through the extracellular matrix, guided by chemotactic signals released at the injury site (Figure 2).42,43 Consequently, one of the key benefits of MSC-based therapies is their ability to preferentially target damaged tissues. In the case of inflammatory injuries, MSCs tend to become inflamed and colonize damaged organs.44 Several studies have validated a direct role in binding to adhesion molecules and chemokines secreted by damaged tissues through self-expressed adhesion molecules and chemokine receptors after MSCs are transplanted into blood vessels, thereby homing to the site of injury.45 The expression of stromal cell-derived factor 1 (SDF-1) is upregulated during local inflammation, and SDF-1/CXCR4 will lay the foundation for future studies on MSCs implantation. SDF-1 expression increases after the injury site undergoes inflammation. Under hypoxic conditions, the receptor CXCR4 of MSCs expressing SDF-1 experiences ischemic injury, which significantly increases the expression of SDF-1, thereby increasing the colonization of MSCs at the injured site.46 Overexpression of CXCR4 on the surface of MSCs promoted the secretion of bone morphogenetic protein 7, IL-10, and hepatocyte growth factor (HGF), which enhanced the effect of para-secretion, ensuring that the repair effect was improved. Scientists are currently conducting further research on the homing of MSCs to improve their homing rates. Si’s team found that the importance and originality of their study lies in the idea that addition of TGFβ1 to plasma can significantly raise the expression of CXCR4 on the surface of MSCs and the homing rate of MSCs.47 Hypoxic pretreatment of MSCs in vitro also upregulates the expression of CXCR4, which significantly increases the homing rate of MSCs and enhances the repair effect.48
Differentiation
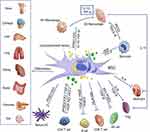
Multi-lineage differentiation of MSCs has been extensively studied in vitro and in vivo since their discovery. The multipotent stem cell components of MSCs isolates can differentiate into derivatives of different lineages, including osteocytes, myocytes, adipocytes, and chondrocytes (Figure 3).49,50 Mushroom growth in molecular biology and transplantation techniques has benefitted MSCs applications in regenerative medicine. The ideal cell source for tissue regeneration is MSCs, which possess powerful proliferation, differentiation, chemotactic, and immune-modulatory properties required for promoting tissue repair and recovery from damage caused by inflammation.51 Therefore, differentiation is also a pivotal mechanism for MSCs to treat inflammatory diseases; however, MSCs differentiate into different cells in diverse diseases, and the specific differentiation mechanism will be explained in related diseases.
Exosomes
The enormous role of MSC-Exos in inflammation progression has drawn widespread attention in recent decades. Accumulating evidence has linked miRNAs in MSC-Exos to the inhibition of inflammation development. MSC-Exos can mimic the function of parental MSCs by transferring their components – such as DNA, proteins/peptides, mRNA, microRNA (miRNA), lipids, and organelles – to recipient cells.52 It has been reported that MSC-Exos are derived from porcine adipose tissue, and that miRNA sequence analysis annotated 413 miRNAs.53 In another study, miRNA profiling of MSC-Exos revealed 171 miRNAs. Among them, the top 23 miRNAs accounted for 79.1% of all miRNAs, which targeted 5481 high-intensity genes through prediction, whereas the remaining 148 miRNAs represented a very small percentage of the total reads.54 At the system level, targeted genes participate in angiogenesis. The targeted pathways included pro-fibrotic signaling, Wnt signaling, cell proliferation, and apoptosis. The prominence of MSC-Exos miRNAs in diverse diseases will be described in detail in the following sections.
Kidney Injury
Acute kidney injury (AKI) is a heterogeneous group of diseases characterized by a sudden decrease in glomerular filtration rate (GFR) followed by an increase in serum creatinine concentration (SCC) or oliguria. Its pathogenesis includes pre-renal azotemia, acute tubular necrosis, acute glomerulonephritis, acute interstitial nephritis, and acute renal vascular obstructive nephropathy. AKI can be divided into pre-renal, intra-renal, and post-renal injuries according to severity and causes affecting prognosis and treatment, of which pre-renal and intra-renal injuries are the most common types, accounting for 60–70% of AKI cases.55 Pre-renal AKI is caused by decreased renal perfusion, with ischemia and obstructions leading to intrarenal and post-renal AKI, respectively. Nevertheless, there are no overwhelming treatments in the literature that can improve the clinical outcomes of AKI, and the care of such patients is limited to supportive treatment, including renal replacement therapy. The mortality rate of patients undergoing renal replacement therapy is as high as 50%.56 Consequently, scientists have turned their attention to MSCs with strong proliferation and differentiation capabilities, advantages of autologous transplantation, low immune rejection, no tumorigenicity, and ethical restrictions. Herein, the potential mechanism underlying the protective effect of MSCs on kidney injury is outlined.
Differentiation in Kidney Injury
Under physiological conditions, MSCs can differentiate into renal tubular epithelial cells and glomerular endothelial cells.57 Renal tubular epithelial cells function in the reabsorption, secretion, and excretion of renal tubules, while the repair of the kidneys also depend on the regeneration and reconstruction of renal tubular epithelial cells.58 If the renal tubular epithelial and endothelial damage exceeds the regeneration potential, the damaged kidney may develop interstitial fibrosis or chronic kidney disease. Therefore, promoting epithelial/endothelial cell repair may be the best way to target AKI and to maintain renal function. Researchers have found that resveratrol-modified human umbilical cord MSCs (Res-hucMSCs) activate the ERK pathway of renal tubular cells through paracrine PDGF-DD, promoting endothelial cell angiogenesis and improving kidney protection. More effective repair and stronger homing abilities were observed compared to normal hucMSCs.59 Tubular epithelial cells (TECs) differentiated from MSCs and induced by collecting tube cell-conditioned medium (CDC-CM) were injected into the rat renal cortex. Compared with untreated cells, using a small amount of gel complex to pre-process cells for three-dimensional culture before implantation can induce more renin-like structures. The insights gained from this study may assist in the repair of dysfunctional kidney tissue.60
Restore Mitochondrial Function
The kidney, with its physiological function of blood purification, is rich in mitochondria and consumes approximately 7% of ATP in the body every day. Mitochondrial pathology is a common feature of different forms of human AKI, and proximal tubules are more susceptible to mitochondrial dysfunction.61 During the occurrence and development of AKI, changes in the ultrastructure, metabolism, and bioenergy centered on the mitochondria often occur. To maintain stable function, it is necessary to maintain the balance of the mitochondrial environment.62 Consequently, many studies have been conducted to improve mitochondrial function in the recovery of renal function.
In the cisplatin-induced AKI mouse model, human umbilical cord mesenchymal stem cells (UC-MSCs) protect the quality and function of mitochondria by regulating microtubule-rich protrusions (the role of these protrusions is to maintain the mitochondria between adjacent renal tubular cells). At the same time, it also stimulates the mitochondrial biogenesis, antioxidant defense and energy production of the damaged proximal tubules through the SIRT3-dependent mechanism, which is constructive to the repair of the kidney.63 Researchers injected MSCs into rats with cisplatin-induced AKI and found that cytochrome C released by mitochondria in the cytoplasm and malondialdehyde levels were reduced, indicating that MSCs can protect renal cells from mitochondrial dysfunction and oxidative damage, and alleviate acute kidney injury induced by cisplatin-induced AKI by inhibiting the activation of the mitochondrial signaling pathway.64
Exosomes
RNase III Drosha and Dicer are key enzymes that regulate intracellular miRNA maturation. RNase III Drosha acts on the nucleus to cut inactivated pri-miRNA into pre-miRNA, which is exported from the nucleus to the cytoplasm through Dicer division, and finally produces a functionally mature miRNA.65 After the injection of Drosha-knockout MSC-Exos, normal MSCs, and normal MSC-Exos intravenously in the AKI model, it was found that only Drosha-knockout MSC-Exos did not restore the morphology and function of AKI. RNA sequence analysis also demonstrated that drosha-knockout MSC-Exos could not restore the expression of kidney genes after injury. It can be concluded that the absence of miRNA significantly reduces the inherent regenerative potential of MSC-Exos in AKI, suggesting that the recovery of miRNA after AKI is of great significance.66
In the early stages of renal ischemia-reperfusion (I/R) injury, MSC-Exos overexpressing miR-199a-5p can be transferred to renal tubular epithelial cells and markedly bind to immunoglobulin protein (BIP) by inhibiting I/R-induced endoplasmic reticulum stress.67 MSC-exos can not only transmit miR-199a-3p to the receptor renal tubular epithelial cells, but also inhibit semaphorin 3A (Sema3A) by activating protein kinase B (Akt) and extracellular signal-regulated kinase (ERK) pathways to achieve the purpose of inhibiting apoptosis.68 The miRNA of MSC-Exos was analyzed by high-throughput miRNA sequencing (miRNA-seq), indicating that MSC-Exos inhibited the expression of p53 protein in TECs by miR-125b-5p and regulated Bcl-2 and Bax to inhibit TECs apoptosis by upregulating CDK1 and cyclin B1 and reversing the G2 M block. In conclusion, these results suggest that targeted cell cycle arrest and apoptosis improve ischemic AKI and promote the repair of renal tubules.69
Application of MSCs and MSC-Exos in Kidney Injury
Inflammation, ischemia, anoikis, and imbalance between reactive oxygen species and antioxidants can lead to a low survival rate of transplanted MSCs.70 Therefore, MSCs have been pretreated to protect them from damage via the following methods: (1) the use of cytokines or compounds to improve the survival of MSCs in vivo and accelerate the recovery of renal function. MSCs pretreated with melatonin showed increased expression of P-Erk1/2, superoxide dismutase-1 (SOD-1), P-Akt, and heme oxygenase-1 (HO-1), which have anti-inflammatory and antioxidant properties. The proliferation, pro-survival, paracrine, and migration capabilities of MSCs were significantly improved after transplantation. Melatonin can be used as a dietary supplement with safety advantages.71 MSCs pretreated with atorvastatin were administered through the carotid artery to an animal model of I/R injury, and it was discovered that the toll-like receptor 4 (TLR4) signaling pathway was inhibited. Serum creatinine and blood urea nitrogen levels and severity of renal damage were reduced 24 h after I/R injury. Furthermore, pretreatment with atorvastatin not only improves oxidative stress and inhibits inflammation, but also promotes the vitality of implanted MSCs.72 The combination of noninvasive image-guided pulsed focused ultrasound (pFUS) and MSCs can enhance the homing ability of MSCs, increase the secretion of IL-10, reduce the level of TNF-α, and improve the survival rate of cisplatin-induced AKI mouse models.73 (2) Improvement of the cell culture environment: The use of three-dimensional (3D) cell culture MSCs can promote the interaction between cells and ECM, produce higher levels of ECM proteins, and show stronger anti-apoptotic and antioxidant capabilities. In vivo and in vitro experiments confirmed that 3D culture enhanced the paracrine effect of MSCs and increased the secretion of antioxidant factors, anti-inflammatory proteins, anti-apoptotic factors (epidermal growth factor and HGF), vascular endothelial growth factor (VEGF), basic fibroblast growth factor (bFGF)), and insulin-like growth factors.74 (3) Combination of MSCs with hydrogel scaffolds: The mixed β-sheet polypeptide hydrogel composed of the IGF-1C domain and D-form polypeptide has the properties of a biomimetic ECM and cell delivery carrier. MSCs can be delivered to promote angiogenesis, leading to the recovery of renal function.75 The thermosensitive chitosan chloride (CSCl) hydrogel used as a scaffold to deliver MSCs to I/R-induced AKI rats can improve the retention and survival of transplanted kidney MSCs and enhance the proliferation of host kidney cells.76 (4) Through genetic modification, MSCs overexpress cytokine or antiapoptotic genes: There are two methods of viral transduction and non-viral gene transfer, among which viral transduction is more commonly used.77 In vivo and in vitro experiments have demonstrated that MSCs overexpressing CXCR4 can activate PI3K/Akt and MAPK/ERK transduction pathways to enhance the ability of MSCs to migrate to the kidneys and improve renal function.78 Transplantation of Nrf2 overexpressing MSCs increases the expression of repair genes, such as AQP-1 and CK-18, while downregulating the expression of damage markers can also increase the resistance of MSCs to stress-related environments in vivo. Renal regeneration was promoted by reducing creatinine and blood urea nitrogen within 14 days. The expression of repair markers was significantly upregulated, and the expression of injury markers was significantly downregulated after transplantation with Nrf2 overexpression 14 days later.79 (5) In addition to genetics, physical and chemical methods have been used to modify MSCs: The physical methods include microinjection, electroporation, and sonication. Chemical methods can deliver nucleic acid materials containing siRNA, mRNA, and DNA into MSCs via cationic lipids, polymers, and inorganic nanoparticles.80 However, these two methods have limited clinical application because they may lead to obvious cell damage, transient expression of transgenes, and relatively low efficiency of gene transfer.81
Paracrine action is the main mechanism by which MSCs repair damaged tissue; therefore, there are approaches to enhance paracrine action similar to pretreatment.82,83 After hypoxia treatment, VEGF, bFGF, and other angiogenic factors in MSCs were markedly enhanced, causing an increase in the secretion of anti-oxidant cytokines and anti-apoptotic cytokine activated Akt (p-Akt), and reduced the level of pro-apoptotic factor caspase 3.84 As mentioned in the in vivo literature, vascular remodeling of kidneys treated with hypoxia is the most conspicuous. Tregs are indispensable in immunomodulation and protect against AKI (IRI-AKI) caused by ischemia-reperfusion injury in animal models. IL-17A pretreatment increased PGE2 levels in the supernatant of MSCs, and PGE2 induced Treg Foxp3 gene expression in vivo. Therefore, studies evaluating the growth of MSCs revealed that IL-17A can ameliorate MSC-mediated increase in Tregs by promoting the production of PGE2. In the meantime, the efficacy of MSCs in IRI-AKI treatment can be upgraded by boosting the serum anti-inflammatory cytokine IL-10 and abating the serum pro-inflammatory cytokines IL-6, TNF-α and IFN-g.85 Genetic modification can more accurately reinforce the paracrine effect of MSCs than other pretreatment strategies. Lcn2 is an antioxidant, antiapoptotic, and anti-inflammatory protein. When the kidney is damaged, the expression of Lcn2 is immediately induced to re-establish homeostasis. MSCs overexpressing Lcn2 can stimulate the secretion of several key growth factors such as HGF, IGF-1, FGF, and VEGF, reduce the expression of injury markers KIM-1 and cystatin C, and increase the expression of proximal tubular epithelial markers AQP-1 and CK-18.86
In addition, the half-life and curative effects of MSC-Exos were extended when used in combination with hydrogels. There is a smart design for arginine-glycine-aspartate (RGD) hydrogels for MSC-Exos as carriers binding to integrins. The stability and retention time of MSC-Exos added to the RGD hydrogel in vivo were determined using bioluminescence imaging technology and compared with ordinary MSC-Exos. As a result, the RGD hydrogel can promote cell adhesion and uptake of MSC-Exos by renal tubular epithelial cells to protect against AKI. In addition, let-7a-5p contained in MSC-Exos can activate autophagy by downregulating the amino acid-sensing pathway and reducing the death of tubular epithelial cells. The RGD hydrogel can amplify autophagy activation and improve the bioavailability and therapeutic efficacy of MSC-Exos.87
In an early clinical trial (NCT00733876), all study participants were closely followed up for the first six months and the following three years after undergoing pump heart surgery, confirming the safety and efficacy of MSCs in high-risk AKI patients.88 However, in a multicenter, randomized, controlled Phase 2 clinical trial (NCT01602328), patients with early AKI after cardiac surgery were treated with allogeneic MSCs. There was no significant difference in the time to recovery of renal functions between the placebo and the MSCs groups. These conflicting results hinder the clinical application of MSCs for the treatment of AKI.89 Although MSCs research has made great breakthroughs, there are still many crucial questions to be answered regarding the clinical efficacy of MSCs in the treatment of AKI.
Lung Injury
Acute respiratory distress syndrome (ARDS) is a respiratory failure syndrome characterized by hypoxemia and respiratory mechanical changes, which is associated with high incidence rate, mortality and high economic costs.90 The recent global outbreak of pneumonia caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and terminal acute lung injury (ALI) can lead to ARDS. Coronavirus disease 2019 (COVID-19) caused by SARS-CoV-2 is characterized by high fever, fatigue, loss of taste and smell, respiratory symptoms, decreased oxygen saturation, and shortness of breath. It can cause neurological diseases such as encephalopathy, encephalitis, loss of sense of smell, deafness, and Guillain–Barré syndrome.91 High-level activation of immune cells and excessive production of a large number of inflammatory cytokines and chemical mediators will lead to a cytokine storm related to high levels of circulating cytokines, thrombosis, a large amount of mononuclear cell infiltration in multiple organs, and severe lymphopenia. Cytokine storm is considered the key cause of disease and even death in COVID-19 patients.92 ALI – a common clinical disease with high mortality – is caused by direct or indirect causes of lung epithelial cell and capillary endothelial cell damage, enhanced pulmonary capillary permeability (leading to acute hypoxic respiratory failure), and an imbalance between anti-inflammatory and pro-inflammatory factors in the lungs.93 There is a lack of recognized evaluation standards and treatment schemes, but the current clinical treatment plan can still achieve satisfactory results.94 These findings contribute in several ways to our understanding of inflammation, which has an important impact on the development of ALI, and provide a basis for our study. As MSCs can secrete anti-inflammatory, anti-fibrotic, and anti-apoptotic cytokines, the following section will explain the possible mechanisms of MSC-based treatment of COVID-19 and ALI.
Differentiation in Lung Injury
Damage to alveolar endothelial cells and epithelial cells, as well as a decrease in alveolar clearance (AFC), are crucial to the pathological changes associated with lung injury. MSCs can secrete or promote the production of a series of cytokines and growth factors including keratinocyte growth factor (KGF), VEGF, HGF, TGF-β, PGE-2, IL-6, macrophage colony-stimulating factor (M-CSF), and angiopoietin-1 (Ang-1).95 Among them, HGF, KGF, and Ang-1 promote angiogenesis, anti-inflammatory activity, and proliferation. MSCs can reduce the apoptosis of alveolar epithelial and endothelial cells by secreting these three growth factors. HGF induces the proliferation of vascular endothelial cells, promotes angiogenesis, inhibits Rho GTPase (improving the stability and integrity of lung endothelial cells), and inhibits cell apoptosis. HGF improves the protein permeability of pulmonary microvascular endothelial cells (HMVEc), repairs tight junctions between cells and junctions of endothelial cells, and restores the cytoskeleton.96 As a powerful epithelial tissue-specific mitogen and differentiation factor, KGF enhances the mitotic activity of type II alveolar epithelial cells; stimulates cell proliferation, differentiation, and DNA repair; increases the secretion of alveolar epithelial lipids, proteins, and alveolar surfactants; increases the transcription and translation of sodium and chloride transporters; and inhibits damaged alveolar epithelium and endothelial cell apoptosis. It is worth noting that KGF is only effective before or at the same time as when the lung tissue is injured.97 Ang-1 can make a difference to maintain homeostasis in the pulmonary vascular endothelium and may indirectly participate in the repair of alveolar epithelial cells by reducing the protein level in bronchoalveolar lavage fluid (BALF).98
Exosomes
MSC-Exos have anti-inflammatory and tissue repair effects similar to those of MSCs, which have an important impact in the treatment of lung injury. MSC-Exos can also regulate lung injury through miRNAs and reduce the infiltration of neutrophils and macrophages into the lungs.99 Serum amyloid A3 (SAA3) is the main component of acute inflammation and is highly expressed in ALI. Yi et al injected MSC-Exos overexpressing mir-30b-3p into LPS-induced ALI mice. It was found that miR-30b-3p can reduce the expression of SAA3 in receptor alveolar epithelial cells (AECs) and ultimately promote the proliferation of AECs and inhibit the apoptosis of AECs, thereby protecting against ALI.100 Similarly, MSC-Exos overexpressing MiR-21-5P inhibits endogenous and exogenous apoptosis pathways in mouse lung endothelial cells by targeting pro-apoptotic genes, such as PTEN and PDCD4, and reduce lung cell apoptosis and lung injury.101 TLR4 signalling plays a pivotal role in inflammation. A typical case of this is that MSC-Exos overexpressing miR-451 inhibited the expression of TLR4 and its downstream target proteins – NF-κB/P65 and p-P65 – in vivo, increased the levels of malondialdehyde, MPO, and SOD in lung tissues, and reduced the levels of TNF-α, IL-1β, and IL-6. Conversely, when these miRNAs are inhibited, the situation is reversed.102 The above studies showed that MSC-Exos carrying different types of miRNAs can affect lung injury by regulating inflammation, promoting alveolar epithelial regeneration, and enhancing endothelial cell proliferation, which could provide a a theoretical basis for MSC-Exos to target lung injury.
Application of MSCs and MSC-Exos in Lung Injury
Most clinical trials have evaluated the safety and effectiveness of MSCs in patients.103,104 In 2015, Wilson et al published the results of MSCs treatment for ARDS trials (NCT01775774). In nine of the patients, there were no pre-specified infusion-related events or treatment-related adverse events. Three patients were found to have serious adverse events several weeks after the injection; however, based on the MRI results, they were not considered to be related to MSCs. Nine patients with moderate-to-severe acute respiratory distress syndrome received a single intravenous infusion of allogeneic MSCs. Therefore, the author believes that more patients are needed to thoroughly study the response of ARDS patients to MSCs; the second-phase trial is, thus, still in progress, mainly focusing on safety and secondary results, including respiratory, systemic, and biological endpoints.105 Researchers conducted a prospective, double-blind, multicenter, randomized phase 2a trial (NCT02097641) to evaluate the effect of a single intravenous injection of MSCs and placebo in the treatment of moderate-to-severe ARDS. Experiments showed that for patients with moderate to severe ARDS, there was no difference in the 28-day mortality between the MSCs and placebo groups, and a single intravenous injection of MSCs was safe.106 MiR-142a-5p mediates autophagy in pulmonary endothelial cells by increasing Beclin-1 protein expression. MSCs may reduce autophagy in pulmonary artery endothelial cells (PECs) by downregulating miR-142a-5p and promoting the survival of PECs to alleviate LPS-induced ALI.107
Relatively speaking, more clinical trials are using MSCs for the treatment of COVID-19. The first was a single-center, open-label pilot study in which MSCs were used to treat seven acute ARDS patients with COVID-19 of varying severity. The symptoms of all patients treated with MSCs improved clinically after 2 days. Three patients (one severe and two moderate) were discharged after 10 days. CD4 T cells, CD8 T cells, and NK cells were depleted in all critically ill patients who received MSCs treatment. Compared with the placebo group, the levels of C-reactive protein and TNF-α were reduced in patients treated with MSCs, whereas the level of IL-10 was increased. It was further shown that the patient changed from a pro-inflammatory to an anti-inflammatory state. Taken together, these results indicate that intravenous transplantation of MSCs is safe and effective for treating COVID-19 pneumonia, especially in critically ill patients.108 Additionally, in a single-case study of a 65-year-old Chinese woman with ARDS caused by COVID-19, the patient’s inflammatory symptoms were not significantly ameliorated after conventional treatment for 12 days, whereas symptoms improved significantly after the second dose of allogeneic UC-MSCs treatment, pneumonia on chest CT scan decreased, neutrophil and lymphocyte counts returned to normal, and no adverse reactions were found.109
Although current research shows that MSCs are expected to provide a means to inhibit the aggressive inflammatory response of patients with severe COVID-19 infections, more powerful experimental data are required to make more conclusive judgments. Moreover, the optimal dose of MSCs for the treatment of other lung diseases remains unclear. Therefore, the MSCs treatment and placebo groups must be considered when evaluating the treatment efficiency in comparison with the dose, route of administration, time interval, and disease severity of the two groups.
MSC-Exos have been used in clinical trials owing to their anti-inflammatory, anti-apoptotic, and immune-rebuilding capabilities, and have shown good efficacy in the treatment of some diseases. The results of a published prospective clinical study showed that 24 critically ill COVID-19 patients received a single 15 mL intravenous injection of MSC-Exos, 17 patients (71%) recovered, and 3 patients (13%) were still critically ill, but stable After a series of treatments, the absolute value of neutrophils in this group of patients was significantly reduced, the absolute values of CD4+ T lymphocytes and CD8+ T lymphocytes were significantly increased, and no adverse events occurred.110 Owing to their intrinsic properties, such as biodegradability, biocompatibility, prolonged circulation time, low immunogenicity, and ability to pass the blood barrier, MSC-Exos can be ideal carriers for therapeutic drugs.111 For example, remdesivir, an antiviral drug, can be used for the treatment of COVID-19. Thus, MSC-Exos can be used as drug carriers for the treatment of COVID-19. Considerably more work is needed on the combined use of exosomal immunomodulatory products and remdesivir to determine whether it is a new intervention tool for the treatment of COVID-19.112 In summary, MSC-Exos may be a powerful tool for the treatment of lung injury caused by inflammation; however, its safety and specific mechanisms require further study.
Osteoarthritis and Rheumatoid Arthritis
OA is a chronic degenerative joint disease characterized by subchondral bone sclerosis, articular cartilage degeneration, synovial inflammation, and osteophyte formation.113 The main clinical symptoms are joint deformities, joint swelling, and joint pain. The main pathological change is the destruction of articular cartilage.114 On the one hand, joint pain is caused by dysfunction, and on the other hand, it also affects the patient’s sleep and quality of life.115 RA is a chronic, systemic inflammatory disease that can cause cartilage and bone damage and disability, manifesting as symmetrical pain and swelling of the hands, wrists, feet, and knees.116 RA includes several subgroups of diseases characterized by immune disorders. If left untreated, this may lead to chronic inflammation and irreversible joint or organ damage. Therefore, progressive disability and shortened lifespan are the most common symptoms in patients with RA.117 Although the cause of RA is not fully understood, the long-term prognosis of RA has improved through early diagnosis in recent years.118 Currently, the most widely used methods for the treatment of cartilage injury are drug therapy, surgical therapy, and autologous chondrocyte transplantation.119,120 In contrast, MSCs can regulate the local microenvironment of damaged cartilage through their anti-inflammatory and immunomodulatory capabilities and secrete a variety of biologically active factors to protect the cartilage from further damage. Thus, MSCs have been widely studied as ideal seed cells for the treatment of cartilage regeneration in OA.
Differentiation in OA and RA
The articular cartilage is a weight-bearing tissue that provides a low-friction surface for effective shock absorption, weight loss, and efficient movement. In the absence of vascularization, innervation, and low cell density in cartilage tissue characteristics, the ability to self-repair articular cartilage is very limited.121 When MSCs migrate from the subchondral bone to the damaged site, they can differentiate into chondrocytes and osteoblasts and integrate into new and surrounding tissues while repairing cartilage and subchondral bone tissue. After being co-cultured with chondrocytes, it can substantially increase the mRNA expression levels of chondrocyte marker genes aggrecan, col-2 and sox-9, promote chondrocyte proliferation, and inhibit chondrocyte apoptosis.122 The main factors of chondrogenic differentiation are cell proliferation and migration of MSCs promoted by vitamin D. TGF-β1 participates in the function of vitamin D by regulating the ERK/JNK signaling pathway.123 Some studies have interfered with the chondrogenic differentiation potential of MSCs using growth factors, scaffolds, and co-culture technology. A notable example is that nerve growth factor (NGF), which can significantly upregulate the expression of cartilage-specific markers.124 Nanofiber-based polyethersulfone (PES) scaffolds can enhance the potential of MSCs to differentiate into cartilage while maintaining their proliferation and differentiation capabilities.125 These studies suggest that MSCs have the potential to differentiate into chondroblasts, which can then be used to promote cartilage damage and repair.
Exosomes
Specific protein 1 (Sp1) is a transcription factor that inhibits Sp1 and promotes chondrocyte proliferation. MSC-Exos-derived miR-135b can down-regulate the expression of Sp1 in chondrocytes. There are a large volume of published studies describing the enhanced expression of miR-135b in MSC-Exos after TGF-β1 stimulation. Rat chondrocyte line (C5.18 cell) cells were co-cultured with TGF-β1-MSC-Exos, and it was found that Sp1 expression in C5.18 cells was reduced, cell viability was enhanced, and chondrocyte proliferation was promoted.126 Angiogenesis is a sign of RA, with long-lasting angiogenesis leading to chronic changes in the RA synovium. MMP14 plays a role in vascular remodeling by promoting the secretion of vasoactive cytokines, such as VEGF, from the matrix, while VEGF is upregulated in RA, which can also promote the development of RA. The research results show that MSC-Exos overexpressing miR-150-5p can inhibit the migration and invasion of RA FLS, inhibit angiogenesis, and relieve the symptoms of RA by down-regulating MMP14 and VEGF.127
Application of MSCs and MSC-Exos in OA and RA
The vast majority of tissue engineering studies involve combining MSCs with scaffold materials and adding factors to induce MSCs to differentiate into chondrocytes and transplanting them into cartilage defects to promote cartilage regeneration and repair. Cases reported by Zhou et al incorporated human MSCs and graphene oxide (GO) to adsorb the growth factor TGFβ3 into a 3D scaffold. TGFβ3 adsorbed by GO enhances chondrogenic differentiation and cartilage tissue synthesis of MSCs in the entire scaffold without the need to repeat the exogenous supply of TGFβ3.128 In addition, cartilage-specific ECM deposition and chondrogenic gene expression were improved. MSCs have also been used in clinical trials for the treatment of OA and RA. In 2010, researchers from the Korea Stem Cell Research Center used MSCs in the first clinical trial of RA. A total of ten patients with different autoimmune diseases were included in this study, four of whom had RA. Different numbers of MSCs were intravenously injected at single, double, or four doses. All the patients were monitored for 13 months and showed good clinical efficacy and safety.129 A study conducted in the PLA Air Force Hospital demonstrated for the first time the long-term beneficial effect of MSC combined with low-dose disease-modifying anti-rheumatic drugs (DMARDs) in the treatment of RA patients (NCT01547091) and was materially better than the control group treated with DMARDS alone. However, multicenter experiments are required to confirm the reliability of the research results.130 The first clinical trial reported the use of allogeneic MSCs and hyaluronic acid hydrogel complexes for the regeneration of articular cartilage defects in patients with OA. The 1-year histological evaluation revealed high hyaline-like cartilage. Delayed gadolinium-enhanced MRI of the cartilage three years after transplantation showed continuous regeneration of cartilage and high glycosaminoglycan (GAG) content. During the 7-year follow-up, improvement in pain and function did not show palpable deterioration. This suggests that the new treatment product appears to be safe and effective, and further studies are needed with a larger number of patients.131 Additionally, there is a newly registered clinical trial to inject MSC-Exos into joints to treat OA (NCT05060107), which is expected to be completed by 2023. Ten patients were recruited in the Phase 1 trial and followed up for up to 12 months to verify the safety of MSC-Exos in the treatment of OA.
Whether it is an intravenous injection, subcutaneous injection, or other method of administration, MSC-Exos will be excreted from the body in a short time. Therefore, the insights gained from this study on how to prolong the half-life of exosomes may have a positive impact on the effect of local treatment with MSC-Exos.132 Hydrogels can target drugs to specific sites and control drug release, which can effectively solve deficiencies in the application of MSC-Exos. In vitro studies have shown that hydrogels are non-toxic, biodegradable, retain the chondrocyte phenotype, and can secrete cartilage-specific ECM.133
Zhang’s research group found that chitosan hydrogels as carriers loaded with MSC-Exos could improve the stability of proteins and miRNAs in MSC-Exos and prolong their retention time in vivo. The researchers also used MSC-Exos of hydrogels in a hind limb ischemia model to increase angiogenesis and endothelial cell survival, thereby accelerating the recovery of ischemic hind limbs, which were administered in a mouse hind limb ischemia model to increase angiogenesis and endothelial cell survival, accelerating the recovery of ischemic hind limbs.134 Liu et al encapsulated MSC-Exos into photoinduced imine crosslinking (PIC) hydrogel glue and prepared MSC-Exos composite hydrogel tissue patch EHG, which can seamlessly combine with natural cartilage, effectively prolong the activity of MSC-Exos, and exhibit positive cell regulation in vitro and in vivo to promote cartilage repair and regeneration, thus providing a more effective scaffold for promoting the repair and regeneration of articular cartilage defects.135 MSC-Exos can also be used as carriers to encapsulate curcumin, an anti-inflammatory drug with poor bioavailability. Therefore, curcumin was loaded into MSC-Exos as a nanolevel curcumin drug delivery system. Curcumin-encapsulated MSC-Exos can reduce the phosphorylation of Erk1/2, PI3K/Akt and p-38MAPK induced by il-1β, thereby reducing the activity of pathways involving these kinases. On this basis, the curcumin-encapsulated MSC-Exos can also induce the up-regulation of miR-126-3p expression in target cells, and reduce the degradation of OA chondrocytes induced by il-1β.136 Although the clinical application of MSC-Exos has shown good therapeutic effects, the mechanism and safety of MSC-Exos need to be further studied before clinical application.
Inflammatory Bowel Disease
IBD is a chronic and debilitating disease characterized by idiopathic mucosal inflammation of the gastrointestinal tract and is classified as Crohn’s disease (CD) and ulcerative colitis (UC).137,138 UC generally only affects the mucosa of the large intestine, whereas CD may cause harmful inflammation in any part of the gastrointestinal tract, including all layers of the intestinal wall.139 It is unclear what causes inflammatory bowel disease, but a variety of factors – including genetic susceptibility, changes in the gut microbiome, defects in the innate and adaptive immune system, and various environmental exposures – may cause the disease.140,141 Although significant progress has been made in the treatment of IBD – mainly using biological agents such as anti-TNF-α antibodies – there are still some patients who face difficulties in being cured via traditional methods.142,143
Differentiation in IBD
The intestinal epithelium is composed of closely connected columnar epithelial cells. Most previous studies defined intestinal epithelial cells as an important barrier against the translocation of microorganisms, toxins, and foreign antigens in the cavity.144 Intestinal epithelial cell death is a common pathological feature of inflammatory bowel disease (IBD). It not only destroys the integrity of the barrier but also causes inflammation and increases the levels of inflammatory cytokines, such as TNF-α.145 As mentioned in the literature review, MSCs have been demonstrated to enter the injured site of the intestinal tract of mice with experimental colitis, and differentiate directly into intestinal epithelial cells or endothelial cells, or by combining with intestinal epithelial stem cells and secreting cytokines to exert their protective effects on the intestinal mucosa.146 MSCs can also induce neovascularization and promote intestinal mucosal repair by differentiating into vascular endothelial cells, promoting angiogenesis, improving the local blood supply, and playing a noteworthy role in tissue repair.147,148
Exosomes
miRNAs are loaded into MSC-Exos during exosome biogenesis. The miRNAs contained in exosomes are pre-miRNAs, which are inactive before being converted into mature miRNAs.149 MiR-146a as a negative feedback regulator of innate immune response, has a significant anti-inflammatory effect. An initial objective of the project conducted by Wu’s team was to identify whether or not MSC-Exos can target inflammatory genes TNF receptor-associated factor 6 (TRAF6) and IRAK1 through miR-146a so as to inhibit gene expression and the activation of the NF-κB pathway. In this way, 2,4,6‑trinitrobenzenesulfonic acid (TNBS)-induced inflammation of the colon in rats was controlled.150 Epithelial cells lose their phenotype and functional characteristics and acquire mesenchymal properties, resulting in intestinal fibrosis. MSC-Exos can also prevent colonic fibrosis by inhibiting the transformation of epithelium to mesenchyme through miR-200b, but whether this therapy can reverse fibrosis still requires greater and more in-depth research.151 Inflammasomes would make a valuable contribution to many immune and inflammation-related diseases by activating caspase-1 to produce IL-18 and IL-1β for the initiation of the inflammatory process. NLRP3 inflammasome is one of the most studied subtypes, and its increase and abnormality can aggravate the incidence and development of inflammatory diseases.152 MSC-Exos can reduce the activation of NLRP3 inflammasomes in macrophages, directly bind to NLRP3 mRNA through miR-410, and inhibit the NLRP3 pathway.153
Application of MSCs and MSC-Exos in IBD
Areas where significant differences have been found in theMSCs administration methods used for IBD include systemic and topical treatments. Systemic administration is mainly achieved by intravenous infusion, whereas local administration methods involve intraperitoneal injection and fistula injection. Intravenous injection of MSCs is simple and minimally invasive, but the injected MSCs are mainly retained in the lungs; however, few MSCs reach the intestinal inflammation site, curbing their ability to play a role, thus affecting their efficacy to a certain extent.154,155 Local injection of MSCs is superior to intravenous injection, which has proven to be an effective way to treat CD via an anal fistula.156 However, no studies have shown a correlation between the therapeutic effect and dose of MSCs.
In summary, MSCs are promising candidates for the treatment of IBD through mechanisms such as immune regulation and promotion of intestinal repair. However, the current related trials are all small-sample studies, there is no sufficient exploration of the mechanism of MSCs, and there is still no clear consensus on patient inclusion criteria, transplantation methods, treatment dose selection, treatment frequency, and treatment of transplant complications. For this reason, more studies are needed to confirm the safety and effectiveness of MSCs in the treatment of IBD and to explore additional methods so as to improve the therapeutic effect of MSCs.
Liver Injury
Liver toxicity factors – such as viruses, drugs, lipid deposits, and autoimmune reactions – can induce acute or chronic liver injury and liver failure, among which acute liver injury is characterized by abnormal liver function in a short period of time.157 The main pathological changes are extensive necrosis and apoptosis of liver cells, which prevent the liver from performing normal synthetic and metabolic functions.158,159 Liver failure can cause complications including encephalopathy, cerebral edema, sepsis, renal failure, gastrointestinal bleeding, and respiratory failure.160 Chronic liver inflammation can also lead to fibrosis and cirrhosis, which are the twelfth leading causes of death in the United States.161 Autologous liver transplantation remains the most effective method for patients with severe liver injury or liver failure. However, liver donor risks still exist, such as lack of liver donors, high surgical costs, post-transplant complications, and immune rejection, which result in the limited use of liver transplantation. MSCs show great potential and advantages in the treatment of acute and chronic liver diseases, owing to their unique characteristics.162
Differentiation in Liver Injury
MSCs from bone marrow (BM-MSCs), menstrual blood (Men-SCs), UC-MSCs, and adipose tissue (AT-MSCs) can differentiate into hepatocyte-like cells (HLCs) under selective growth conditions in vitro.163 Multiple liver transcription factors and cytoplasmic proteins are selectively expressed during MSCs differentiation. In the early stage, early markers – such as hepatocyte nuclear factor (HNF) 3β, transthyretin (TTR), and alpha-fetoprotein (AFP) – are expressed, followed by the expression of late-stage markers such as albumin (ALB), cytokeratin (CK), HNF1α, and HNF4α. In the later stage, markers and proteins of MSCs, similar to mature hepatocytes – such as antitrypsin (AAT), CCAAT-enhanced binding protein (C/EBP) α, tryptophan 2.3-dioxygenase (TO), and tyrosine aminotransferase (TAT) – are gradually lost after MSCs expression.164 Inducing MSCs to differentiate into mature hepatocytes in vitro requires sufficient stimulation to maintain cellular function. Many cytokines and growth factors – including oncostatin M (OSM), insulin-like growth factor (IGF), HGF, epidermal growth factor (EGF), TGF-β, and bFGF – affect the growth and differentiation of hepatocytes in vitro. Among them, HGF, bFGF, and OSM are particularly important for liver differentiation of MSCs. In addition, dexamethasone (Dex), tretinoin, sodium butyrate, nicotinamide (NTA), norepinephrine, dimethyl sulfoxide, and other compounds may have significant effects on the promotion of liver differentiation.165,166
Exosomes
Nucleoside binding and the NLRP3 inflammasome are newly discovered pattern recognition receptors that are involved in the pathogenesis of many liver diseases due to their high expression in the liver. After activation of NLRP3, it induces the death of new inflammatory cells and pyroptosis, and simultaneously leads to the maturation and secretion of IL-1β. Many studies have suggested that abnormal NLRP3 activation is involved in the progression of autoimmune hepatitis (AIH). MSC-Exos overexpressing miRNA-233 can inhibit the activation of NLRP3 by binding to the NLRP3ʹ3-UTR, leading to degradation of NLRP3 mRNA and downregulation of protein levels, thereby inhibiting liver inflammation and cell death.167 In addition, the absence of thioredoxin-interacting protein (TXNIP) affects NLRP3 inflammasomes and the secretion of IL-1β. Liver expression of TXNIP and the interaction between TXNIP and NLRP3 were upregulated in the ALF mouse model, indicating that TXNIP-mediated activation of NLRP3 is far-reaching for ALF. After miRNA-17 is expressed by MSC-Exos, the activation of NLRP3 inflammatory bodies is reduced, thereby protecting ALF.168
Application of MSCs and MSC-Exos in Liver Injury
At present, MSCs and MSC-Exos have been used in various clinical trials for liver injury and liver failure with good curative effects.169–171 An open-label, unblinded, randomized controlled study served as a control between 24 patients treated with MSCs and 19 patients treated with normal saline. These patients were administered MSCs three times every four weeks, and liver function, adverse events, and survival rates were evaluated during a 48-week or 72-week follow-up period. Experiments showed that there were no obvious side effects after MSCs infusion, and serum-total bilirubin and alanine aminotransferase levels were significantly reduced.172 Another study enrolled 110 patients with acute-on-chronic liver failure (ACLF – NCT01322906). The experimental group was followed up with for 24 weeks after MSC injection. Although the symptoms of fever appeared in the experimental group throughout the 5–24 week span, they were all relieved after appropriate treatment, and the incidence and mortality of severe infections in the experimental group were significantly lower than those in the control group. These results suggest that MSCs can not only increase liver function but also significantly enhance 24-week survival by reducing severe infection. The study was conducted at a single center, which limits the generalizability of the results. Therefore, the results of this study need to be further confirmed in a multicenter, with a more diverse patient population and with a longer follow-up times.169 Despite this, the long-term effects of MSCs transplantation on immune function and tumorigenesis risk are due to the suboptimal frequency of transplantation and differentiation.173 Large-scale clinical trials are needed to confirm the therapeutic potential of MSCs, which are still in their infancy for the treatment of liver disease.
Pancreatitis
Pancreatitis can be divided into acute and chronic forms. Acute pancreatitis (AP) is a rapid inflammation of the pancreas that is the most common cause of hospitalization for gastrointestinal diseases worldwide.174 Although the exact pathogenesis of AP is currently unknown, premature activation of pancreatic enzymes leading to self-digestion of the pancreas is considered to be a consensus mechanism initiated by AP which, in turn, triggers local inflammation as well as a systemic inflammatory response affecting remote organs, such as the kidneys and lungs.175 Due to the lack of effective therapies for AP and the high mortality rate associated with severe acute pancreatitis (SAP), a new therapeutic approach is urgently needed. MSCs are included in the treatment plan for AP because of their immunomodulatory effects, such as secretion of anti-inflammatory cytokines, inhibition of pro-inflammatory cytokines, and regulation of immune cell activation.176
Organ failure is a decisive factor in the prognosis of AP, and secondary myocardial injury is the main cause of death from SAP. miR-29a-3p secreted by MSC-Exos inhibits cardiomyocyte apoptosis by reducing collagen production in myocardial fibroblasts. The high mobility group Box 1 protein (HMGB1), which is actively released in response to inflammatory stimuli, may be a hypothetical target gene of Mir-29a-3p. HMGB1 expression is upregulated in pancreatitis; however, overexpression of HMGB1 can lead to serious consequences of myocardial injury. The TLR4/Akt pathway also promotes the progression of inflammation-induced cell damage. MiR-29a-3p participating in the regulation of SAP to reduce myocardial injury and can inhibit the inflammatory response during SAP by reducing the expression of HMGB1 to inhibit oxidative stress and blocking the Akt signaling pathway. However, existing studies only used a single dose of MSC-Exos, and the optimal dose and injection time need to be further explored.177
Application of MSCs and MSC-Exos in AP
Although large and small animal models have been used to study AP, there are few reports on the use of MSCs in large animals. However, the transition to clinical research is usually carried out from small animal models to large animal models, which is a necessary step before the first human clinical trials are considered.178 Moreover, even in small animal models, the optimal dose of MSCs for the most effective treatment of AP has not yet been determined, and there are many drawbacks of intravenous infusion (according to previous studies).179 Therefore, the best delivery path to the pancreas may be through intra-arterial (IA) injection to the organ’s main blood supply, which has not been reported in the literature. However, because most studies are conducted in small animals, size limitations will make this method infeasible. If MSCs can be combined in AP studies and IA infusion is feasible in large animals, they could certainly be used in human clinical trials.180
Neurodegenerative Disease
Neurodegenerative disease refers to a pathological state in which neurological function progressively declines owing to brain atrophy. Due to its lethality, it imposes a heavy economic burden on society. Although a great deal of research has been conducted on the treatment of neurodegenerative diseases, there has been little success.181 Recent studies have established that inflammatory processes are closely related to multiple neurodegenerative pathways, such as Alzheimer’s disease (AD), multiple system atrophy (MSA), Parkinson’s disease (PD), narcolepsy, and autism. The immunomodulatory effects of MSCs have been shown to have therapeutic effects on dynamic inflammation in neurodegenerative diseases.182 Microglia are key cells involved in brain inflammation and inflammatory neurodegenerative diseases. As the first indication of neuritis, activated microglia undergo morphological and functional transitions, release various oxidants, activate various proteins and genes, and produce neurotoxic compounds that may lead to neuronal dysfunction and cell death. It remains activated for longer periods, leading to long-term neurodegeneration. TLRs, which are abundantly expressed in microglia, are the detection and initiation factors for immune responses. TLRs can induce the production of pro-inflammatory cytokines and chemokines, which amplify inflammation.183 Often, multiple T cell populations can also infiltrate central nervous system tissues in response to specific injury signals, exacerbating injury while being pro-inflammatory. MSCs play several roles in the treatment process. MSCs have the potential to transform into neuron-like cells, but few studies have demonstrated the differentiation of transplanted MSCs into neurons or microglia-like cells, and the therapeutic effect is largely attributed to immunomodulation mediated by the interaction between MSCs and various immune cells.184 MSCs can switch resident microglia from a pro-inflammatory phenotype to an anti-inflammatory or tissue-healing phenotype by activating the CX3CL1/CX3CR1 signaling pathway.185 In addition, many studies have shown that important factors such as IDO, PGE2, TSG6, NO, and indoleamine 2 expressed by MSCs have strong immunosuppressive effects on T cell activation in vivo and in vitro.186 MSC-EXOs are beneficial for neuronal preservation and have neuroprotective and regenerative effects on neurons, synapses, and myelin sheaths, all of which have been demonstrated in models of neuroinflammation, Alzheimer’s disease, stroke, and spinal cord injury.187
One study used MSCs to treat MSA patients for the first time. During the subsequent 12-month follow-up, the disease course of patients treated with MSCs was slower than that of the control group.188 Another open-label study showed that patients with PD showed improvement in the Unified PD Rating Scale and other measures after treatment with MSCs.189 None of the aforementioned studies reported any significant adverse reactions. In addition, several clinical trials using MSC-EXOs are currently underway. If existing clinical studies yield positive results, further studies should be conducted in patients with neurodegenerative diseases, especially PD.
Ocular Diseases
The eye, a unique visual sensory organ, is considered an immune-exempt site for preventing immunogenic inflammation. Nevertheless, there are still several inflammatory diseases and immune-mediated diseases involving the eye, which can cause severe visual disturbances, including corneal allograft rejection, dry eye syndrome, age-related macular degeneration, and uveitis wait.190,191 These eye diseases have brought about significant hindrances to people’s daily lives, so it is necessary to explore new treatment methods in which MSCs are one of the candidate methods.192 However, the safety of MSCs transplantations remains controversial. A notable example of this is that three women with macular degeneration experienced vision loss, retinal detachment, and bleeding due to the undesirable differentiation of transplanted stem cells, which eventually led to complete blindness. As a result, MSC-Exos with similar functions and migration potential as MSCs began to emerge.193,194 However, the frequency and dosage of injections that can maintain the lasting effect of MSC-Exos need to be further studied, and the most suitable source of MSC-Exos for each disease also requires careful exploration. To be successfully transformed into a clinical treatment, the mechanism of action of MSCs and MSC-Exos in ocular diseases needs to be studied more thoroughly.
Moreover, the author organized the clinical trials mentioned in this review into a table for easy reference (Table 1).
 |
Table 1 Summary of Some Clinical Trials Based MSCs in Inflammatory Diseases |
Conclusion and Future Perspectives
Inflammation is a complex series of interactions between soluble factors and cells that can occur in any tissue and lead to persistent tissue damage to leukocytes, lymphocytes, or collagen, leading to various diseases. Therefore, in order to achieve the main purpose of maximizing their therapeutic efficiency and minimizing their side effects, drugs for the treatment of inflammatory diseases have undergone a major surge over the last few decades. The rapid development of MSC-based therapies has a strong potential to promote cell proliferation and angiogenesis, differentiation into injured cells, homing to inflammatory sites, inhibiting cell apoptosis, and anti-inflammatory and immunomodulatory functions, all of which are critical points in inflammatory disease treatment. Nevertheless, after intravenous infusion, MSCs can easily locate in the lungs, causing pulmonary embolism, and be cleared by macrophages around the blood vessels in the tissues, which greatly limits their persistence. Research on the dosage and time of administration is particularly important. Currently, most studies use simple incremental methods to explore the relationship between efficacy, dose, and time. If the specific distribution, survival time, and metabolism of MSCs in the body can be clarified through pharmacokinetic studies, it would be more reasonable to determine the optimal dosage and time. Therefore, it is also expected to improve the clinical efficacy of MSCs and reduce their toxic side effects.195
Most previous studies have focused on the therapeutic effects of MSC-Exos on miRNAs. Nevertheless, recent studies have shown that exosomes contain more total protein than total RNA, and are rich in extracellular proteins.196 Generally, all exosomes share a group of proteins, but they also contain specific proteins that reflect the origin and pathophysiological state of the cell source.197 The MSC-Exos proteome participates in many key biological processes and plays important roles in inflammation, cell structure, cell communication, exosome biogenesis, development, tissue repair and regeneration, and metabolism. Consequently, more attention should be paid to the proteins present in exosomes.198 In addition, the yield and purity of MSC-Exos depended on the extraction method used. Unfortunately, there is currently no standardized method for isolating exosomes. Therefore, the separation and purification of MSC-Exos require further research. Other studies have shown that exosomes are unstable, tend to aggregate, have a short half-life, and may have only short-term beneficial effects. Ultrasound is commonly used to disperse aggregated exosomes.199 These findings contribute to solving the problem of the half-life of MSC-Exos. In recent years, attention has been paid to the use of hybrid cell membrane camouflage and synthesis of nanoparticles, which is the direction of our group’s research.200 The camouflage of synthetic nanoparticles (NPs) with natural cell membranes not only retains the physical and chemical characteristics of synthetic carriers, but also inherits the inherent functions of cell membranes, providing significant features such as immune evasion, long-term circulation, and homotypic targeting. Compared with single-cell membranes, hybrid cell membranes can produce synthetic nanoparticles with a variety of biological functions from the original cell.201 The cell membrane can also be combined with liposomes, and the cell membrane can be doped with synthetic lipid molecules of different types and different degrees of saturation to enhance the specificity and biocompatibility of the target.202
Beyond their application to inflammation, MSCs and MSC-Exos can also treat diseases such as cancer. These studies combined the targeting profile and biocompatibility of well-designed MSCs and MSC-Exos with chemotherapy/gene/photothermal therapy.203,204 The surface of MSC-Exos was wrapped with magnetic nanoparticles combined with molecular beacons that can target miR-21 for responsive molecular imaging. Under the guidance of an external magnetic field, MSC-Exos are enriched at the tumor site and near-infrared radiation (NIR) is used to induce local hyperthermia, which triggers the release of the cargo loaded in MSC-Exos.205 This research paves the way for the design of MSC-Exos with multiple functions to achieve precise cancer diagnosis and treatment. These findings have many important implications for future practice, and greater efforts are needed to ensure that this approach can be applied to the treatment of inflammation. The development of targeted drug delivery is remarkable in the field of biomedical research.195 A detailed study of cell-based drug carrier design, different cellular functionalization pathways, and cell-assisted drug delivery vehicles can enable targeted drug delivery to designated locations with minimal or no immune response or toxicity.206 However, technical issues, such as route of administration, injection rate, cell transplantation dose, transplantation frequency, and selection of cell transplantation time window, still need to be solved, and the best clinical effect and safety cannot be guaranteed. Therefore, more large-scale, multi-sample, multi-center, long-term follow-up studies are needed to verify the efficacy and safety of MSCs and MSC-Exos for the treatment of various clinical diseases. In addition, heterogeneity also has a great impact on the clinical application of MSCs; therefore, it is imperative to establish a stem cell bank and strengthen the development and quality control of stem cell preparations.204 These methods provide new ideas for the future treatment of inflammation-related diseases using MSCs and MSC-Exos.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (81973265) and the Fundamental Research Funds for the Central Universities, Southwest Minzu University (2021107).
Disclosure
The authors declare no conflicts of interest for this work.
References
1. Zhong J, Shi G. Editorial: regulation of inflammation in chronic disease. Front Immunol. 2019;10:737. doi:10.3389/fimmu.2019.00737
2. Alessandri AL, Sousa LP, Lucas CD, et al. Resolution of inflammation: mechanisms and opportunity for drug development. Pharmacol Ther. 2013;139(2):189–212. doi:10.1016/j.pharmthera.2013.04.006
3. Sugimoto MA, Sousa LP, Pinho V, et al. Resolution of inflammation: what controls its onset?. Front Immunol. 2016;7:160. doi:10.3389/fimmu.2016.00160
4. Germolec DR, Shipkowski KA, Frawley RP, Evans E, et al. Markers of inflammation. Methods Mol Biol. 2018;1803:57–79. doi:10.1007/978-1-4939-8549-4_5
5. Dhingra AK, Chopra B, Dass R, et al. An update on anti-inflammatory compounds: a review. Antiinflamm Antiallergy Agents Med Chem. 2015;14(2):81–97. doi:10.2174/1871523014666150514102027
6. Patil KR, Mahajan UB, Unger BS, et al. Animal models of inflammation for screening of anti-inflammatory drugs: implications for the discovery and development of phytopharmaceuticals. Int J Mol Sci. 2019;20(18):4367. doi:10.3390/ijms20184367
7. Menshikov M, Zubkova E, Stafeev I, et al. Autophagy, mesenchymal stem cell differentiation, and secretion. Biomedicines. 2021;9(9):1178. doi:10.3390/biomedicines9091178
8. Pires AO, Mendes-Pinheiro B, Teixeira FG, et al. Unveiling the differences of secretome of human bone marrow mesenchymal stem cells, adipose tissue-derived stem cells, and human umbilical cord perivascular cells: a proteomic analysis. Stem Cells Dev. 2016;25(14):1073–1083. doi:10.1089/scd.2016.0048
9. Guadix JA, Zugaza JL, Gálvez-Martín P, et al. Characteristics, applications and prospects of mesenchymal stem cells in cell therapy. Med Clin (Barc). 2017;148(9):408–414. doi:10.1016/j.medcli.2016.11.033
10. Wei X, Yang X, Han Z-P, et al. Mesenchymal stem cells: a new trend for cell therapy. Acta Pharmacol Sin. 2013;34(6):747–754. doi:10.1038/aps.2013.50
11. Shi Y, Wang Y, Li Q, et al. Immunoregulatory mechanisms of mesenchymal stem and stromal cells in inflammatory diseases. Nat Rev Nephrol. 2018;14(8):493–507. doi:10.1038/s41581-018-0023-5
12. Phinney DG, Pittenger MF. Concise review: MSC-derived exosomes for cell-free therapy. Stem Cells. 2017;35(4):851–858. doi:10.1002/stem.2575
13. Zhang W, Wang Y, Kong Y, et al. Exosomes derived from mesenchymal stem cells modulate miR-126 to ameliorate hyperglycemia-induced retinal inflammation via targeting HMGB1. Invest Ophthalmol Vis Sci. 2019;60(1):294–303. doi:10.1167/iovs.18-25617
14. Wu P, Zhang B, Shi H, et al. MSC-exosome: a novel cell-free therapy for cutaneous regeneration. Cytotherapy. 2018;20(3):291–301. doi:10.1016/j.jcyt.2017.11.002
15. Stanko P, Kaiserova K, Altanerova V, et al. Comparison of human mesenchymal stem cells derived from dental pulp, bone marrow, adipose tissue, and umbilical cord tissue by gene expression. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2014;158(3):373–377. doi:10.5507/bp.2013.078
16. Dominici M, Le Blanc K, Mueller I, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315–317. doi:10.1080/14653240600855905
17. Heo JS, Choi Y, Kim H-S, et al. Comparison of molecular profiles of human mesenchymal stem cells derived from bone marrow, umbilical cord blood, placenta and adipose tissue. Int J Mol Med. 2016;37(1):115–125. doi:10.3892/ijmm.2015.2413
18. Almalki SG, Agrawal DK. Key transcription factors in the differentiation of mesenchymal stem cells. Differentiation. 2016;92(1–2):41–51. doi:10.1016/j.diff.2016.02.005
19. Kobolak J, Dinnyes A, Memic A, et al. Mesenchymal stem cells: identification, phenotypic characterization, biological properties and potential for regenerative medicine through biomaterial micro-engineering of their niche. Methods. 2016;99:62–68. doi:10.1016/j.ymeth.2015.09.016
20. Brown C, McKee C, Bakshi S, et al. Mesenchymal stem cells: cell therapy and regeneration potential. J Tissue Eng Regen Med. 2019;13(9):1738–1755. doi:10.1002/term.2914
21. Uder C, Brückner S, Winkler S, et al. Mammalian MSC from selected species: features and applications. Cytometry A. 2018;93(1):32–49. doi:10.1002/cyto.a.23239
22. Kalluri R, Lebleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367(6478). doi:10.1126/science.aau6977
23. Yu X, Odenthal M, Fries JW, et al. Exosomes as miRNA carriers: formation-function-future. Int J Mol Sci. 2016;17(12):2028. doi:10.3390/ijms17122028
24. Sun Z, Shi K, Yang S, et al. Effect of exosomal miRNA on cancer biology and clinical applications. Mol Cancer. 2018;17(1):147. doi:10.1186/s12943-018-0897-7
25. Liu H, Chen Y, Yin G, et al. Therapeutic prospects of MicroRNAs carried by mesenchymal stem cells-derived extracellular vesicles in autoimmune diseases. Life Sci. 2021;277:119458. doi:10.1016/j.lfs.2021.119458
26. Hamidzadeh K, Christensen SM, Dalby E, et al. Macrophages and the recovery from acute and chronic inflammation. Annu Rev Physiol. 2017;79(1):567–592. doi:10.1146/annurev-physiol-022516-034348
27. Song W-J, Li Q, Ryu M-O, et al. TSG-6 released from intraperitoneally injected canine adipose tissue-derived mesenchymal stem cells ameliorate inflammatory bowel disease by inducing M2 macrophage switch in mice. Stem Cell Res Ther. 2018;9(1):91. doi:10.1186/s13287-018-0841-1
28. Geng Y, Zhang L, Fu B, et al. Mesenchymal stem cells ameliorate rhabdomyolysis-induced acute kidney injury via the activation of M2 macrophages. Stem Cell Res Ther. 2014;5(3):80. doi:10.1186/scrt469
29. Bacskai I, Mázló A, Kis-Tóth K, et al. Mesenchymal stromal cell-like cells set the balance of stimulatory and inhibitory signals in monocyte-derived dendritic cells. Stem Cells Dev. 2015;24(15):1805–1816. doi:10.1089/scd.2014.0509
30. Lu Z, Chang W, Meng S, et al. Mesenchymal stem cells induce dendritic cell immune tolerance via paracrine hepatocyte growth factor to alleviate acute lung injury. Stem Cell Res Ther. 2019;10(1):372. doi:10.1186/s13287-019-1488-2
31. Ðokić JM, Z. Tomic S, J. Colic M, et al. Cross-talk between mesenchymal stem/stromal cells and dendritic cells. Curr Stem Cell Res Ther. 2016;11(1):51–65. doi:10.2174/1574888X10666150904114035
32. Cui R, Rekasi H, Hepner-Schefczyk M, et al. Human mesenchymal stromal/stem cells acquire immunostimulatory capacity upon cross-talk with natural killer cells and might improve the NK cell function of immunocompromised patients. Stem Cell Res Ther. 2016;7(1):88. doi:10.1186/s13287-016-0353-9
33. Markov A, Thangavelu L, Aravindhan S, et al. Mesenchymal stem/stromal cells as a valuable source for the treatment of immune-mediated disorders. Stem Cell Res Ther. 2021;12(1):192. doi:10.1186/s13287-021-02265-1
34. Song J-Y, Kang HJ, Ju HM, et al. Umbilical cord-derived mesenchymal stem cell extracts ameliorate atopic dermatitis in mice by reducing the T cell responses. Sci Rep. 2019;9(1):6623. doi:10.1038/s41598-019-42964-7
35. Ma Y, Wang Z, Zhang A, et al. Human placenta-derived mesenchymal stem cells ameliorate GVHD by modulating Th17/Tr1 balance via expression of PD-L2. Life Sci. 2018;214:98–105. doi:10.1016/j.lfs.2018.10.061
36. Euler M, Hoffmann MH. The double-edged role of neutrophil extracellular traps in inflammation. Biochem Soc Trans. 2019;47(6):1921–1930. doi:10.1042/bst20190629
37. Liu S, Su X, Pan P, et al. Neutrophil extracellular traps are indirectly triggered by lipopolysaccharide and contribute to acute lung injury. Sci Rep. 2016;6(1):37252. doi:10.1038/srep37252
38. Mehta P, McAuley DF, Brown M, et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395(10229):1033–1034. doi:10.1016/s0140-6736(20)30628-0
39. Ye Q, Wang B, Mao J, et al. The pathogenesis and treatment of the “Cytokine Storm” in COVID-19. J Infect. 2020;80(6):607–613. doi:10.1016/j.jinf.2020.03.037
40. Chen J, Shao Y, Xu G, et al. Bone marrow-derived mesenchymal stem cells attenuate phosgene-induced acute lung injury in rats. Inhal Toxicol. 2015;27(5):254–261. doi:10.3109/08958378.2015.1037029
41. Nitzsche F, Müller C, Lukomska B, et al. Concise review: MSC adhesion cascade-insights into homing and transendothelial migration. Stem Cells. 2017;35(6):1446–1460. doi:10.1002/stem.2614
42. Ullah M, Liu DD, Thakor AS, et al. Mesenchymal stromal cell homing: mechanisms and strategies for improvement. iScience. 2019;15:421–438. doi:10.1016/j.isci.2019.05.004
43. Liesveld JL, Sharma N, Aljitawi OS, et al. Stem cell homing: from physiology to therapeutics. Stem Cells. 2020;38(10):1241–1253. doi:10.1002/stem.3242
44. Leibacher J, Henschler R. Biodistribution, migration and homing of systemically applied mesenchymal stem/stromal cells. Stem Cell Res Ther. 2016;7:7. doi:10.1186/s13287-015-0271-2
45. Szydlak R. Biological, chemical and mechanical factors regulating migration and homing of mesenchymal stem cells. World J Stem Cells. 2021;13(6):619–631. doi:10.4252/wjsc.v13.i6.619
46. Jiang Q, Huang K, Lu F, et al. Modifying strategies for SDF-1/CXCR4 interaction during mesenchymal stem cell transplantation. Gen Thorac Cardiovasc Surg. 2021;70:1–10. doi:10.1007/s11748-021-01696-0
47. Si XY, Li -J-J, Yao T, et al. Transforming growth factor-β1 in the microenvironment of ischemia reperfusion-injured kidney enhances the chemotaxis of mesenchymal stem cells to stromal cell-derived factor-1 through upregulation of surface chemokine (C-X-C motif) receptor 4. Mol Med Rep. 2014;9(5):1794–1798. doi:10.3892/mmr.2014.1989
48. Yu X, Lu C, Liu H, et al. Hypoxic preconditioning with cobalt of bone marrow mesenchymal stem cells improves cell migration and enhances therapy for treatment of ischemic acute kidney injury. PLoS One. 2013;8(5):e62703. doi:10.1371/journal.pone.0062703
49. Li Q, Gao Z, Chen Y, et al. The role of mitochondria in osteogenic, adipogenic and chondrogenic differentiation of mesenchymal stem cells. Protein Cell. 2017;8(6):439–445. doi:10.1007/s13238-017-0385-7
50. Atashi F, Modarressi A, Pepper MS, et al. The role of reactive oxygen species in mesenchymal stem cell adipogenic and osteogenic differentiation: a review. Stem Cells Dev. 2015;24(10):1150–1163. doi:10.1089/scd.2014.0484
51. Han Y, Li X, Zhang Y, et al. Mesenchymal stem cells for regenerative medicine. Cells. 2019;8(8):886. doi:10.3390/cells8080886
52. Baglio SR, Rooijers K, Koppers-Lalic D, et al. Human bone marrow- and adipose-mesenchymal stem cells secrete exosomes enriched in distinctive miRNA and tRNA species. Stem Cell Res Ther. 2015;6(1):127. doi:10.1186/s13287-015-0116-z
53. Meng Y, Eirin A, Zhu X-Y, et al. The metabolic syndrome alters the miRNA signature of porcine adipose tissue-derived mesenchymal stem cells. Cytometry A. 2018;93(1):93–103. doi:10.1002/cyto.a.23165
54. Li Y, Meng Y, Zhu X, et al. Metabolic syndrome is associated with altered mRNA and miRNA content in human circulating extracellular vesicles. Front Endocrinol (Lausanne). 2021;12:687586. doi:10.3389/fendo.2021.687586
55. Levey AS, James MT. Acute kidney injury. Ann Intern Med. 2017;167(9):ITC66–ITC80. doi:10.7326/AITC201711070
56. Schneider AG, Bagshaw SM. Effects of renal replacement therapy on renal recovery after acute kidney injury. Nephron Clin Pract. 2014;127(1–4):35–41. doi:10.1159/000363671
57. De Almeida DC, Donizetti-Oliveira C, Barbosa-Costa P, et al. In search of mechanisms associated with mesenchymal stem cell-based therapies for acute kidney injury. Clin Biochem Rev. 2013;34(3):131–144.
58. Yu SM-W, Bonventre JV. Acute kidney injury and maladaptive tubular repair leading to renal fibrosis. Curr Opin Nephrol Hypertens. 2020;29(3):310–318. doi:10.1097/mnh.0000000000000605
59. Zhang R, Yin L, Zhang B, et al. Resveratrol improves human umbilical cord-derived mesenchymal stem cells repair for cisplatin-induced acute kidney injury. Cell Death Dis. 2018;9(10):965. doi:10.1038/s41419-018-0959-1
60. Machiguchi T, Nakamura T. Nephron generation in kidney cortices through injection of pretreated mesenchymal stem cell-differentiated tubular epithelial cells. Biochem Biophys Res Commun. 2019;518(1):141–147. doi:10.1016/j.bbrc.2019.08.022
61. Zhao L, Hu C, Zhang P, et al. Mesenchymal stem cell therapy targeting mitochondrial dysfunction in acute kidney injury. J Transl Med. 2019;17(1):142. doi:10.1186/s12967-019-1893-4
62. Bhargava P, Schnellmann RG. Mitochondrial energetics in the kidney. Nat Rev Nephrol. 2017;13(10):629–646. doi:10.1038/nrneph.2017.107
63. Perico L, Morigi M, Rota C, et al. Human mesenchymal stromal cells transplanted into mice stimulate renal tubular cells and enhance mitochondrial function. Nat Commun. 2017;8(1):983. doi:10.1038/s41467-017-00937-2
64. Peng X, Xu H, Zhou Y, et al. Human umbilical cord mesenchymal stem cells attenuate cisplatin-induced acute and chronic renal injury. Exp Biol Med (Maywood). 2013;238(8):960–970. doi:10.1177/1535370213497176
65. Treiber T, Treiber N, Meister G, et al. Regulation of microRNA biogenesis and its crosstalk with other cellular pathways. Nat Rev Mol Cell Biol. 2019;20(1):5–20. doi:10.1038/s41580-018-0059-1
66. Collino F, Bruno S, Incarnato D, et al. AKI recovery induced by mesenchymal stromal cell-derived extracellular vesicles carrying MicroRNAs. J Am Soc Nephrol. 2015;26(10):2349–2360. doi:10.1681/asn.2014070710
67. Wang C, Zhu G, He W, et al. BMSCs protect against renal ischemia-reperfusion injury by secreting exosomes loaded with miR-199a-5p that target BIP to inhibit endoplasmic reticulum stress at the very early reperfusion stages. FASEB j. 2019;33(4):5440–5456. doi:10.1096/fj.201801821R
68. Zhu G, Pei L, Lin F, et al. Exosomes from human-bone-marrow-derived mesenchymal stem cells protect against renal ischemia/reperfusion injury via transferring miR-199a-3p. J Cell Physiol. 2019;234(12):23736–23749. doi:10.1002/jcp.28941
69. Cao J-Y, Wang B, Tang -T-T, et al. Exosomal miR-125b-5p deriving from mesenchymal stem cells promotes tubular repair by suppression of p53 in ischemic acute kidney injury. Theranostics. 2021;11(11):5248–5266. doi:10.7150/thno.54550
70. Galipeau J, Sensébé L. Mesenchymal stromal cells: clinical challenges and therapeutic opportunities. Cell Stem Cell. 2018;22(6):824–833. doi:10.1016/j.stem.2018.05.004
71. Zhao L, Hu C, Zhang P, et al. Melatonin preconditioning is an effective strategy for mesenchymal stem cell-based therapy for kidney disease. J Cell Mol Med. 2020;24(1):25–33. doi:10.1111/jcmm.14769
72. Cai J, Yu X, Zhang B, et al. Atorvastatin improves survival of implanted stem cells in a rat model of renal ischemia-reperfusion injury. Am J Nephrol. 2014;39(6):466–475. doi:10.1159/000362623
73. Burks SR, Nguyen BA, Tebebi PA, et al. Pulsed focused ultrasound pretreatment improves mesenchymal stromal cell efficacy in preventing and rescuing established acute kidney injury in mice. Stem Cells. 2015;33(4):1241–1253. doi:10.1002/stem.1965
74. Xu Y, Shi T, Xu A, et al. 3D spheroid culture enhances survival and therapeutic capacities of MSC s injected into ischemic kidney. J Cell Mol Med. 2016;20(7):1203–1213. doi:10.1111/jcmm.12651
75. Wang H, Shang Y, Chen X, et al. Delivery of MSCs with a Hybrid β-sheet peptide hydrogel consisting IGF-1C domain and D-form peptide for acute kidney injury therapy. Int J Nanomedicine. 2020;15:4311–4324. doi:10.2147/ijn.S254635
76. Gao J, Liu R, Wu J, et al. The use of chitosan based hydrogel for enhancing the therapeutic benefits of adipose-derived MSCs for acute kidney injury. Biomaterials. 2012;33(14):3673–3681. doi:10.1016/j.biomaterials.2012.01.061
77. Liu Y, Fang J, Mareschi K. Mesenchymal stem cells as therapeutic agents and novel carriers for the delivery of candidate genes in acute kidney injury. Stem Cells Int. 2020;2020:8875554. doi:10.1155/2020/8875554
78. Liu N, Tian J, Cheng J, et al. Migration of CXCR4 gene-modified bone marrow-derived mesenchymal stem cells to the acute injured kidney. J Cell Biochem. 2013;114(12):2677–2689. doi:10.1002/jcb.24615
79. Zhaleh F, Amiri F, Mohammadzadeh-Vardin M, et al. Nuclear factor erythroid-2 related factor 2 overexpressed mesenchymal stem cells transplantation, improves renal function, decreases injuries markers and increases repair markers in glycerol-induced Acute kidney injury rats.. Iran J Basic Med Sci. 2016;19(3):323–329.
80. Mashel TV, Tarakanchikova YV, Muslimov AR, et al. Overcoming the delivery problem for therapeutic genome editing: current status and perspective of non-viral methods. Biomaterials. 2020;258:120282. doi:10.1016/j.biomaterials.2020.120282
81. Wang W, Xu X, Li Z, et al. Genetic engineering of mesenchymal stem cells by non-viral gene delivery. Clin Hemorheol Microcirc. 2014;58(1):19–48. doi:10.3233/ch-141883
82. Cheng K, Rai P, Plagov A, et al. Transplantation of bone marrow-derived MSCs improves cisplatinum-induced renal injury through paracrine mechanisms. Exp Mol Pathol. 2013;94(3):466–473. doi:10.1016/j.yexmp.2013.03.002
83. Zhao L, Hu C, Zhang P, et al. Preconditioning strategies for improving the survival rate and paracrine ability of mesenchymal stem cells in acute kidney injury. J Cell Mol Med. 2019;23(2):720–730. doi:10.1111/jcmm.14035
84. Zhang W, Liu L, Huo Y, et al. Hypoxia-pretreated human MSCs attenuate acute kidney injury through enhanced angiogenic and antioxidative capacities. Biomed Res Int. 2014;2014:462472. doi:10.1155/2014/462472
85. Bai M, Zhang L, Fu B, et al. IL-17A improves the efficacy of mesenchymal stem cells in ischemic-reperfusion renal injury by increasing Treg percentages by the COX-2/PGE2 pathway. Kidney Int. 2018;93(4):814–825. doi:10.1016/j.kint.2017.08.030
86. Roudkenar MH, Halabian R, Tehrani HA, et al. Lipocalin 2 enhances mesenchymal stem cell-based cell therapy in acute kidney injury rat model. Cytotechnology. 2018;70(1):103–117. doi:10.1007/s10616-017-0107-2
87. Zhang C, Shang Y, Chen X, et al. Supramolecular nanofibers containing arginine-glycine-aspartate (RGD) peptides boost therapeutic efficacy of extracellular vesicles in kidney repair. ACS Nano. 2020;14(9):12133–12147. doi:10.1021/acsnano.0c05681
88. Tögel FE, Westenfelder C. Kidney protection and regeneration following acute injury: progress through stem cell therapy. Am J Kidney Dis. 2012;60(6):1012–1022. doi:10.1053/j.ajkd.2012.08.034
89. Swaminathan M, Stafford-Smith M, Chertow GM, et al. Allogeneic mesenchymal stem cells for treatment of AKI after cardiac surgery. J Am Soc Nephrol. 2018;29(1):260–267. doi:10.1681/asn.2016101150
90. Peck TJ, Hibbert KA. Recent advances in the understanding and management of ARDS. F1000Res. 2019;8:1959. doi:10.12688/f1000research.20411.1
91. Salian VS, Wright JA, Vedell PT, et al. COVID-19 transmission, current treatment, and future therapeutic strategies. Mol Pharm. 2021;18(3):754–771. doi:10.1021/acs.molpharmaceut.0c00608
92. Hu B, Huang S, Yin L, et al. The cytokine storm and COVID-19. J Med Virol. 2021;93(1):250–256. doi:10.1002/jmv.26232
93. Butt Y, Kurdowska A, Allen TC, et al. Acute lung injury: a clinical and molecular review. Arch Pathol Lab Med. 2016;140(4):345–350. doi:10.5858/arpa.2015-0519-RA
94. Mokra D, Mikolka P, Kosutova P, Mokry J, et al. Corticosteroids in acute lung injury: the dilemma continues. Int J Mol Sci. 2019;20(19):4765. doi:10.3390/ijms20194765
95. Shi X, Mao J, Liu Y, et al. Pulp stem cells derived from human permanent and deciduous teeth: biological characteristics and therapeutic applications. Stem Cells Transl Med. 2020;9(4):445–464. doi:10.1002/sctm.19-0398
96. Higginbotham K, Tian Y, Gawlak G, et al. Hepatocyte growth factor triggers distinct mechanisms of Asef and Tiam1 activation to induce endothelial barrier enhancement. Cell Signal. 2014;26(11):2306–2316. doi:10.1016/j.cellsig.2014.07.032
97. Chen J, Li C, Gao X, et al. Keratinocyte growth factor gene delivery via mesenchymal stem cells protects against lipopolysaccharide-induced acute lung injury in mice. PLoS One. 2013;8(12):e83303. doi:10.1371/journal.pone.0083303
98. Li JW, Wu X. Mesenchymal stem cells ameliorate LPS-induced acute lung injury through KGF promoting alveolar fluid clearance of alveolar type II cells. Eur Rev Med Pharmacol Sci. 2015;19(13):2368–2378.
99. Monsel A, Zhu Y-G, Gennai S, et al. Therapeutic effects of human mesenchymal stem cell-derived microvesicles in severe pneumonia in mice. Am J Respir Crit Care Med. 2015;192(3):324–336. doi:10.1164/rccm.201410-1765OC
100. Yi X, Wei X, Lv H, et al. Exosomes derived from microRNA-30b-3p-overexpressing mesenchymal stem cells protect against lipopolysaccharide-induced acute lung injury by inhibiting SAA3. Exp Cell Res. 2019;383(2):111454. doi:10.1016/j.yexcr.2019.05.035
101. Li JW, Wei L, Han Z, Chen Z, et al. Mesenchymal stromal cells-derived exosomes alleviate ischemia/reperfusion injury in mouse lung by transporting anti-apoptotic miR-21-5p. Eur J Pharmacol. 2019;852:68–76. doi:10.1016/j.ejphar.2019.01.022
102. Liu JS, Du J, Cheng X, et al. Exosomal miR-451 from human umbilical cord mesenchymal stem cells attenuates burn-induced acute lung injury. J Chin Med Assoc. 2019;82(12):895–901. doi:10.1097/jcma.0000000000000189
103. Zhu YG, Hao Q, Monsel A, et al. Adult stem cells for acute lung injury: remaining questions and concerns. Respirology. 2013;18(5):744–756. doi:10.1111/resp.12093
104. Zheng G, Huang L, Tong H, et al. Treatment of acute respiratory distress syndrome with allogeneic adipose-derived mesenchymal stem cells: a randomized, placebo-controlled pilot study. Respir Res. 2014;15(1):39. doi:10.1186/1465-9921-15-39
105. Wilson JG, Liu KD, Zhuo H, et al. Mesenchymal stem (stromal) cells for treatment of ARDS: a phase 1 clinical trial. Lancet Respir Med. 2015;3(1):24–32. doi:10.1016/s2213-2600(14)70291-7
106. Matthay MA, Calfee CS, Zhuo H, et al. Treatment with allogeneic mesenchymal stromal cells for moderate to severe acute respiratory distress syndrome (START study): a randomised phase 2a safety trial. Lancet Respir Med. 2019;7(2):154–162. doi:10.1016/s2213-2600(18)30418-1
107. Zhou Z, You Z. Mesenchymal stem cells alleviate LPS-induced acute lung injury in mice by MiR-142a-5p-controlled pulmonary endothelial cell autophagy. Cell Physiol Biochem. 2016;38(1):258–266. doi:10.1159/000438627
108. Leng Z, Zhu R, Hou W, et al. Transplantation of ACE2- mesenchymal stem cells improves the outcome of patients with COVID-19 Pneumonia. Aging Dis. 2020;11(2):216–228. doi:10.14336/ad.2020.0228
109. Liang B, Chen J, Li T, et al. Clinical remission of a critically ill COVID-19 patient treated by human umbilical cord mesenchymal stem cells: a case report. Medicine. 2020;99(31):e21429. doi:10.1097/md.0000000000021429
110. Sengupta V, Sengupta S, Lazo A, et al. Exosomes derived from bone marrow mesenchymal stem cells as treatment for severe COVID-19. Stem Cells Dev. 2020;29(12):747–754. doi:10.1089/scd.2020.0080
111. Yousefi Dehbidi M, Goodarzi N, Azhdari MH, et al. Mesenchymal stem cells and their derived exosomes to combat Covid–19. Rev Med Virol. 2022;32(2):e2281. doi:10.1002/rmv.2281
112. Grein J, Ohmagari N, Shin D, et al. Compassionate use of remdesivir for patients with severe Covid-19. N Engl J Med. 2020;382(24):2327–2336. doi:10.1056/NEJMoa2007016
113. Arden NK, Leyland KM. Osteoarthritis year 2013 in review: clinical. Osteoarthritis Cartilage. 2013;21(10):1409–1413. doi:10.1016/j.joca.2013.06.021
114. Kim SJ, Shetty AA, Kurian NM, et al. Articular cartilage repair using autologous collagen-induced chondrogenesis (ACIC): a pragmatic and cost-effective enhancement of a traditional technique. Knee Surg Sports Traumatol Arthrosc. 2020;28(8):2598–2603. doi:10.1007/s00167-020-05884-y
115. Kong L, Zheng L-Z, Qin L, et al. Role of mesenchymal stem cells in osteoarthritis treatment. J Orthop Translat. 2017;9:89–103. doi:10.1016/j.jot.2017.03.006
116. Cush JJ. Rheumatoid arthritis: early diagnosis and treatment. Med Clin North Am. 2021;105(2):355–365. doi:10.1016/j.mcna.2020.10.006
117. Scherer HU, Häupl T, Burmester GR, et al. The etiology of rheumatoid arthritis. J Autoimmun. 2020;110:102400. doi:10.1016/j.jaut.2019.102400
118. Lin YJ, Anzaghe M, Schülke S, et al. Update on the pathomechanism, diagnosis, and treatment options for rheumatoid arthritis. Cells. 2020;9(4). doi:10.3390/cells9040880
119. Singh JA, Saag KG, Bridges SL, et al. 2015 American College of Rheumatology Guideline for the treatment of rheumatoid arthritis. Arthritis Care Res. 2016;68(1):1–25. doi:10.1002/acr.22783
120. Beasley J. Osteoarthritis and rheumatoid arthritis: conservative therapeutic management. J Hand Ther. 2012;25(2):
121. Guo W-Z, Di H, Chu G-H, et al. Comparative analysis of conventional and biological treatment in healing of bone disease. Saudi J Biol Sci. 2018;25(1):162–166. doi:10.1016/j.sjbs.2017.02.003
122. Ter Huurne M, Schelbergen R, Blattes R, et al. Antiinflammatory and chondroprotective effects of intraarticular injection of adipose-derived stem cells in experimental osteoarthritis. Arthritis Rheum. 2012;64(11):3604–3613. doi:10.1002/art.34626
123. Jiang X, Huang B, Yang H, et al. TGF-β1 is involved in Vitamin D-induced chondrogenic differentiation of bone marrow-derived mesenchymal stem cells by regulating the ERK/JNK pathway. Cell Physiol Biochem. 2017;42(6):2230–2241. doi:10.1159/000479997
124. Lu Z, Lei D, Jiang T, et al. Nerve growth factor from Chinese cobra venom stimulates chondrogenic differentiation of mesenchymal stem cells. Cell Death Dis. 2017;8(5):e2801. doi:10.1038/cddis.2017.208
125. Mahboudi H, Kazemi B, Soleimani M, et al. Enhanced chondrogenesis of human bone marrow mesenchymal Stem Cell (BMSC) on nanofiber-based polyethersulfone (PES) scaffold. Gene. 2018;643:98–106. doi:10.1016/j.gene.2017.11.073
126. Wang R, Xu B, Xu H, et al. TGF-β1 promoted chondrocyte proliferation by regulating Sp1 through MSC-exosomes derived miR-135b. Cell Cycle. 2018;17(24):2756–2765. doi:10.1080/15384101.2018.1556063
127. Chen Z, Wang H, Xia Y, et al. Therapeutic potential of mesenchymal cell-derived miRNA-150-5p-expressing exosomes in rheumatoid arthritis mediated by the modulation of MMP14 and VEGF. J Immunol. 2018;201(8):2472–2482. doi:10.4049/jimmunol.1800304
128. Zhou M, Lozano N, Wychowaniec JK, et al. Graphene oxide: a growth factor delivery carrier to enhance chondrogenic differentiation of human mesenchymal stem cells in 3D hydrogels. Acta Biomater. 2019;96:271–280. doi:10.1016/j.actbio.2019.07.027
129. Ra JC, Kang SK, Shin IS, et al. Stem cell treatment for patients with autoimmune disease by systemic infusion of culture-expanded autologous adipose tissue derived mesenchymal stem cells. J Transl Med. 2011;9(1):181. doi:10.1186/1479-5876-9-181
130. Wang L, Huang S, Li S, et al. Efficacy and safety of umbilical cord mesenchymal stem cell therapy for rheumatoid arthritis patients: a prospective Phase I/II study. Drug Des Devel Ther. 2019;13:4331–4340. doi:10.2147/dddt.S225613
131. Park YB, Ha C-W, Lee C-H, et al. Cartilage regeneration in osteoarthritic patients by a composite of allogeneic umbilical cord blood-derived mesenchymal stem cells and hyaluronate hydrogel: results from a clinical trial for safety and proof-of-concept with 7 years of extended follow-up. Stem Cells Transl Med. 2017;6(2):613–621. doi:10.5966/sctm.2016-0157
132. Charoenviriyakul C, Takahashi Y, Morishita M, et al. Cell type-specific and common characteristics of exosomes derived from mouse cell lines: yield, physicochemical properties, and pharmacokinetics. Eur J Pharm Sci. 2017;96:316–322. doi:10.1016/j.ejps.2016.10.009
133. Mohan N, Mohanan PV, Sabareeswaran A, Nair P, et al. Chitosan-hyaluronic acid hydrogel for cartilage repair. Int J Biol Macromol. 2017;104(Pt B):1936–1945. doi:10.1016/j.ijbiomac.2017.03.142
134. Zhang K, Zhao X, Chen X, et al. Enhanced therapeutic effects of mesenchymal stem cell-derived exosomes with an injectable hydrogel for hindlimb ischemia treatment. ACS Appl Mater Interfaces. 2018;10(36):30081–30091. doi:10.1021/acsami.8b08449
135. Liu X, Yang Y, Li Y, et al. Integration of stem cell-derived exosomes with in situ hydrogel glue as a promising tissue patch for articular cartilage regeneration. Nanoscale. 2017;9(13):4430–4438. doi:10.1039/c7nr00352h
136. Li S, Stöckl S, Lukas C, et al. Curcumin-primed human BMSC-derived extracellular vesicles reverse IL-1β-induced catabolic responses of OA chondrocytes by upregulating miR-126-3p. Stem Cell Res Ther. 2021;12(1):252. doi:10.1186/s13287-021-02317-6
137. Malik TA. Inflammatory bowel disease: historical perspective, epidemiology, and risk factors. Surg Clin North Am. 2015;95(6):1105–1122. doi:10.1016/j.suc.2015.07.006
138. Crowley E, Muise A. Inflammatory bowel disease: what very early onset disease teaches us. Gastroenterol Clin North Am. 2018;47(4):755–772. doi:10.1016/j.gtc.2018.07.004
139. Shapiro JM, Subedi S, LeLeiko NS, et al. Inflammatory bowel disease. Pediatr Rev. 2016;37(8):337–347. doi:10.1542/pir.2015-0110
140. Zhang YZ, Li YY. Inflammatory bowel disease: pathogenesis. World J Gastroenterol. 2014;20(1):91–99. doi:10.3748/wjg.v20.i1.91
141. Flynn S, Eisenstein S. Inflammatory bowel disease presentation and diagnosis. Surg Clin North Am. 2019;99(6):1051–1062. doi:10.1016/j.suc.2019.08.001
142. Sairenji T, Collins KL, Evans DV, et al. An update on inflammatory bowel disease. Prim Care. 2017;44(4):673–692. doi:10.1016/j.pop.2017.07.010
143. Lightner AL. Stem cell therapies for inflammatory bowel disease. Curr Gastroenterol Rep. 2019;21(4):16. doi:10.1007/s11894-019-0672-y
144. Subramanian S, Geng H, Tan X-D, et al. Cell death of intestinal epithelial cells in intestinal diseases. Sheng Li Xue Bao. 2020;72(3):308–324.
145. Blander JM. Death in the intestinal epithelium-basic biology and implications for inflammatory bowel disease. Febs j. 2016;283(14):2720–2730. doi:10.1111/febs.13771
146. Barker N. Adult intestinal stem cells: critical drivers of epithelial homeostasis and regeneration. Nat Rev Mol Cell Biol. 2014;15(1):19–33. doi:10.1038/nrm3721
147. Wang C, Li Y, Yang M, et al. Efficient differentiation of bone marrow mesenchymal stem cells into endothelial cells in vitro. Eur J Vasc Endovasc Surg. 2018;55(2):257–265. doi:10.1016/j.ejvs.2017.10.012
148. Deng X, Szabo S, Chen L, et al. New cell therapy using bone marrow-derived stem cells/endothelial progenitor cells to accelerate neovascularization in healing of experimental ulcerative colitis. Curr Pharm Des. 2011;17(16):1643–1651. doi:10.2174/138161211796197007
149. Deng H, Sun C, Sun Y, et al. Lipid, protein, and MicroRNA composition within mesenchymal stem cell-derived exosomes. Cell Reprogram. 2018;20(3):178–186. doi:10.1089/cell.2017.0047
150. Wu H, Fan H, Shou Z, et al. Extracellular vesicles containing miR-146a attenuate experimental colitis by targeting TRAF6 and IRAK1. Int Immunopharmacol. 2019;68:204–212. doi:10.1016/j.intimp.2018.12.043
151. Yang J, Zhou C-Z, Zhu R, et al. miR-200b-containing microvesicles attenuate experimental colitis associated intestinal fibrosis by inhibiting epithelial-mesenchymal transition. J Gastroenterol Hepatol. 2017;32(12):1966–1974. doi:10.1111/jgh.13797
152. Zhen Y, Zhang H. NLRP3 inflammasome and inflammatory bowel disease. Front Immunol. 2019;10:276. doi:10.3389/fimmu.2019.00276
153. Cai X, Zhang Z-Y, Yuan J-T, et al. hucMSC-derived exosomes attenuate colitis by regulating macrophage pyroptosis via the miR-378a-5p/NLRP3 axis. Stem Cell Res Ther. 2021;12(1):416. doi:10.1186/s13287-021-02492-6
154. He XW, He X-S, Lian L, et al. Systemic infusion of bone marrow-derived mesenchymal stem cells for treatment of experimental colitis in mice. Dig Dis Sci. 2012;57(12):3136–3144. doi:10.1007/s10620-012-2290-5
155. Grégoire C, Lechanteur C, Briquet A, et al. Review article: mesenchymal stromal cell therapy for inflammatory bowel diseases. Aliment Pharmacol Ther. 2017;45(2):205–221. doi:10.1111/apt.13864
156. Ciccocioppo R, Bernardo ME, Sgarella A, et al. Autologous bone marrow-derived mesenchymal stromal cells in the treatment of fistulising Crohn’s disease. Gut. 2011;60(6):788–798. doi:10.1136/gut.2010.214841
157. Liou IW. Management of end-stage liver disease. Med Clin North Am. 2014;98(1):119–152. doi:10.1016/j.mcna.2013.09.006
158. Marcellin P, Kutala BK. Liver diseases: a major, neglected global public health problem requiring urgent actions and large-scale screening. Liver Int. 2018;38(Suppl 1):2–6. doi:10.1111/liv.13682
159. Hu C, Li L. In vitro culture of isolated primary hepatocytes and stem cell-derived hepatocyte-like cells for liver regeneration. Protein Cell. 2015;6(8):562–574. doi:10.1007/s13238-015-0180-2
160. Fyfe B, Zaldana F, Liu C, et al. The pathology of acute liver failure. Clin Liver Dis. 2018;22(2):257–268. doi:10.1016/j.cld.2018.01.003
161. Koyama Y, Brenner DA. Liver inflammation and fibrosis. J Clin Invest. 2017;127(1):55–64. doi:10.1172/jci88881
162. Lee CA, Sinha S, Fitzpatrick E, et al. Hepatocyte transplantation and advancements in alternative cell sources for liver-based regenerative medicine. J Mol Med (Berl). 2018;96(6):469–481. doi:10.1007/s00109-018-1638-5
163. Liu WH, Song F-Q, Ren L-N, et al. The multiple functional roles of mesenchymal stem cells in participating in treating liver diseases. J Cell Mol Med. 2015;19(3):511–520. doi:10.1111/jcmm.12482
164. Afshari A, Shamdani S, Uzan G, et al. Different approaches for transformation of mesenchymal stem cells into hepatocyte-like cells. Stem Cell Res Ther. 2020;11(1):54. doi:10.1186/s13287-020-1555-8
165. Yang Y, Zhao Y, Zhang L, et al. The application of mesenchymal stem cells in the treatment of liver diseases: mechanism, efficacy, and safety issues. Front Med. 2021;8:655268. doi:10.3389/fmed.2021.655268
166. Wu XB, Tao R. Hepatocyte differentiation of mesenchymal stem cells. Hepatobiliary Pancreat Dis Int. 2012;11(4):360–371. doi:10.1016/s1499-3872(12)60193-3
167. Chen L, Lu F-B, Chen D-Z, et al. BMSCs-derived miR-223-containing exosomes contribute to liver protection in experimental autoimmune hepatitis. Mol Immunol. 2018;93:38–46. doi:10.1016/j.molimm.2017.11.008
168. Liu Y, Lou G, Li A, et al. AMSC-derived exosomes alleviate lipopolysaccharide/d-galactosamine-induced acute liver failure by miR-17-mediated reduction of TXNIP/NLRP3 inflammasome activation in macrophages. EBioMedicine. 2018;36:140–150. doi:10.1016/j.ebiom.2018.08.054
169. Lin B-L, Chen J-F, Qiu W-H, et al. Allogeneic bone marrow-derived mesenchymal stromal cells for hepatitis B virus-related acute-on-chronic liver failure: a randomized controlled trial. Hepatology. 2017;66(1):209–219. doi:10.1002/hep.29189
170. Li Y-H, Xu Y, Wu H-M, et al. Umbilical cord-derived mesenchymal stem cell transplantation in Hepatitis B virus related acute-on-chronic liver failure treated with plasma exchange and entecavir: a 24-month prospective study. Stem Cell Rev Rep. 2016;12(6):645–653. doi:10.1007/s12015-016-9683-3
171. Schacher FC, Martins Pezzi da Silva A, Silla LMDR, et al. Bone marrow mesenchymal stem cells in acute-on-chronic liver failure Grades 2 and 3: a Phase I-II Randomized Clinical Trial. Can J Gastroenterol Hepatol. 2021;2021:3662776. doi:10.1155/2021/3662776
172. Shi M, Zhang Z, Xu R, et al. Human mesenchymal stem cell transfusion is safe and improves liver function in acute-on-chronic liver failure patients. Stem Cells Transl Med. 2012;1(10):725–731. doi:10.5966/sctm.2012-0034
173. Owen A, Newsome PN. Mesenchymal stromal cell therapy in liver disease: opportunities and lessons to be learnt?. Am J Physiol Gastrointest Liver Physiol. 2015;309(10):G791–800. doi:10.1152/ajpgi.00036.2015
174. Kawakubo K, Ohnishi S, Kuwatani M, Sakamoto N, et al. Mesenchymal stem cell therapy for acute and chronic pancreatitis. J Gastroenterol. 2018;53(1):1–5. doi:10.1007/s00535-017-1363-9
175. Goodman RR, Jong MK, Davies JE, et al. Concise review: the challenges and opportunities of employing mesenchymal stromal cells in the treatment of acute pancreatitis. Biotechnol Adv. 2020;42:107338. doi:10.1016/j.biotechadv.2019.01.005
176. Ahmed SM, Morsi M, Ghoneim NI, et al. Mesenchymal stromal cell therapy for pancreatitis: a systematic review. Oxid Med Cell Longev. 2018;2018:3250864. doi:10.1155/2018/3250864
177. Ren S, Pan L, Yang L, et al. miR-29a-3p transferred by mesenchymal stem cells-derived extracellular vesicles protects against myocardial injury after severe acute pancreatitis. Life Sci. 2021;272:119189. doi:10.1016/j.lfs.2021.119189
178. Yin G, Hu G, Wan R, et al. Role of microvesicles from bone marrow mesenchymal stem cells in acute pancreatitis. Pancreas. 2016;45(9):1282–1293. doi:10.1097/mpa.0000000000000694
179. Kawakubo K, Ohnishi S, Fujita H, et al. Effect of fetal membrane-derived mesenchymal stem cell transplantation in rats with acute and chronic pancreatitis. Pancreas. 2016;45(5):707–713. doi:10.1097/mpa.0000000000000541
180. Song G, Ma Z, Liu D, et al. Bone marrow-derived mesenchymal stem cells ameliorate severe acute pancreatitis by inhibiting necroptosis in rats. Mol Cell Biochem. 2019;459(1–2):7–19. doi:10.1007/s11010-019-03546-3
181. Vaquer-Alicea J, Diamond MI. Propagation of protein aggregation in neurodegenerative diseases. Annu Rev Biochem. 2019;88:785–810. doi:10.1146/annurev-biochem-061516-045049
182. Chen WW, Zhang X, Huang W-J, et al. Role of neuroinflammation in neurodegenerative diseases (Review). Mol Med Rep. 2016;13(4):3391–3396. doi:10.3892/mmr.2016.4948
183. Bonafede R, Mariotti R. ALS pathogenesis and therapeutic approaches: the role of mesenchymal stem cells and extracellular vesicles. Front Cell Neurosci. 2017;11:80. doi:10.3389/fncel.2017.00080
184. Zriek F, Di Battista JA, Alaaeddine N, et al. Mesenchymal stromal cell secretome: immunomodulation, tissue repair and effects on neurodegenerative conditions. Curr Stem Cell Res Ther. 2021;16(6):656–669. doi:10.2174/1574888x16666210202145639
185. Joo HS, Suh JH, Lee HJ, Bang ES, Lee JM, et al. Current knowledge and future perspectives on mesenchymal stem cell-derived exosomes as a new therapeutic agent. Int J Mol Sci. 2020;21(3):727. doi:10.3390/ijms21030727
186. Chen X, Wang S, Cao W, et al. Mesenchymal stem cell-mediated immunomodulation in cell therapy of neurodegenerative diseases. Cell Immunol. 2018;326:8–14. doi:10.1016/j.cellimm.2017.06.006
187. Staff NP, Jones DT, Singer W, et al. Mesenchymal stromal cell therapies for neurodegenerative diseases. Mayo Clin Proc. 2019;94(5):892–905. doi:10.1016/j.mayocp.2019.01.001
188. Lee PH, Kim JW, Bang OY, et al. Autologous mesenchymal stem cell therapy delays the progression of neurological deficits in patients with multiple system atrophy. Clin Pharmacol Ther. 2008;83(5):723–730. doi:10.1038/sj.clpt.6100386
189. Venkataramana NK, Pal R, Rao SAV, et al. Bilateral transplantation of allogenic adult human bone marrow-derived mesenchymal stem cells into the subventricular zone of Parkinson’s disease: a pilot clinical study. Stem Cells Int. 2012;2012:931902. doi:10.1155/2012/931902
190. Li JJ, Yi S, Wei L, et al. Ocular microbiota and intraocular inflammation. Front Immunol. 2020;11:609765. doi:10.3389/fimmu.2020.609765
191. Albini TA, Davis JL. Ocular immunity and inflammation. Dev Ophthalmol. 2016;55:38–45. doi:10.1159/000431131
192. Lee HJ, Ko JH, Jeong HJ, et al. Mesenchymal stem/stromal cells protect against autoimmunity via CCL2-dependent recruitment of myeloid-derived suppressor cells. J Immunol. 2015;194(8):3634–3645. doi:10.4049/jimmunol.1402139
193. Harrell CR, Simovic Markovic B, Fellabaum C, et al. Therapeutic potential of mesenchymal stem cell-derived exosomes in the treatment of eye diseases. Adv Exp Med Biol. 2018;1089:47–57. doi:10.1007/5584_2018_219
194. Rajool Dezfuly A, Safaee A, Salehi H, et al. Therapeutic effects of mesenchymal stem cells-derived extracellular vesicles’ miRNAs on retinal regeneration: a review. Stem Cell Res Ther. 2021;12(1):530. doi:10.1186/s13287-021-02588-z
195. Timin AS, Peltek OO, Zyuzin MV, et al. Safe and effective delivery of antitumor drug using mesenchymal stem cells impregnated with submicron carriers. ACS Appl Mater Interfaces. 2019;11(14):13091–13104. doi:10.1021/acsami.8b22685
196. Elahi FM, Farwell DG, Nolta JA, et al. Preclinical translation of exosomes derived from mesenchymal stem/stromal cells. Stem Cells. 2020;38(1):15–21. doi:10.1002/stem.3061
197. Samanta S, Rajasingh S, Drosos N, et al. Exosomes: new molecular targets of diseases. Acta Pharmacol Sin. 2018;39(4):501–513. doi:10.1038/aps.2017.162
198. Toh WS, Lai RC, Zhang B, et al. MSC exosome works through a protein-based mechanism of action. Biochem Soc Trans. 2018;46(4):843–853. doi:10.1042/bst20180079
199. Campbell CR, Berman AE, Weintraub NL, et al. Electrical stimulation to optimize cardioprotective exosomes from cardiac stem cells. Med Hypotheses. 2016;88:6–9. doi:10.1016/j.mehy.2015.12.022
200. Liu H, Deng S, Han L, et al. Mesenchymal stem cells, exosomes and exosome-mimics as smart drug carriers for targeted cancer therapy. Colloids Surf B Biointerfaces. 2021;209(Pt 1):112163. doi:10.1016/j.colsurfb.2021.112163
201. Chen HY, Deng J, Wang Y, et al. Hybrid cell membrane-coated nanoparticles: a multifunctional biomimetic platform for cancer diagnosis and therapy. Acta Biomater. 2020;112:1–13. doi:10.1016/j.actbio.2020.05.028
202. Himbert S, Blacker MJ, Kihm A, et al. Hybrid erythrocyte liposomes: functionalized red blood cell membranes for molecule encapsulation. Adv Biosyst. 2020;4(3):e1900185. doi:10.1002/adbi.201900185
203. Patil SM, Sawant SS, Kunda NK, et al. Exosomes as drug delivery systems: a brief overview and progress update. Eur J Pharm Biopharm. 2020;154:259–269. doi:10.1016/j.ejpb.2020.07.026
204. Muslimov AR, Timin AS, Bichaykina VR, et al. Biomimetic drug delivery platforms based on mesenchymal stem cells impregnated with light-responsive submicron sized carriers. Biomater Sci. 2020;8(4):1137–1147. doi:10.1039/c9bm00926d
205. Wang J, Chen P, Dong Y, et al. Designer exosomes enabling tumor targeted efficient chemo/gene/photothermal therapy. Biomaterials. 2021;276:121056. doi:10.1016/j.biomaterials.2021.121056
206. Timin AS, Litvak MM, Gorin DA, et al. Cell-based drug delivery and use of nano-and microcarriers for cell functionalization. Adv Healthc Mater. 2018;7(3). doi:10.1002/adhm.201700818
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.