Back to Journals » Infection and Drug Resistance » Volume 13
Synergistic Effect of Pseudolaric Acid B with Fluconazole Against Resistant Isolates and Biofilm of Candida tropicalis
Authors Li Z , Yin H, Chen W , Jiang C, Hu J, Xue Y, Yao D, Peng Y, Hu X
Received 12 May 2020
Accepted for publication 9 July 2020
Published 5 August 2020 Volume 2020:13 Pages 2733—2743
DOI https://doi.org/10.2147/IDR.S261299
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Zhen Li,1,* Hongmei Yin,1,* Weiqin Chen,1 Cen Jiang,2 Jun Hu,1 Yingjun Xue,1 Dongting Yao,1 Yibing Peng,2 Xiaobo Hu1
1Department of Laboratory Medicine, Longhua Hospital, Shanghai University of Traditional Chinese Medicine, Shanghai, People’s Republic of China; 2Department of Laboratory Medicine, Ruijin Hospital, Shanghai Jiaotong University School of Medicine, Shanghai, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Xiaobo Hu
Department of Laboratory Medicine, Longhua Hospital, Shanghai University of Traditional Chinese Medicine, No. 725 South Wanping Road, Shanghai 200032, People’s Republic of China
Tel +86 1 376 121 6435
Email [email protected]
Yibing Peng
Department of Laboratory Medicine, Ruijin Hospital, Shanghai Jiaotong University School of Medicine, No. 197 Ruijin ER Road, Shanghai 200025, People’s Republic of China
Email [email protected]
Purpose: Candida tropicalis (C. tropicalis) has emerged as an important fungal pathogen due to its increasing resistance to conventional antifungal agents, especially fluconazole (FLC). Pseudolaric acid B (PAB), a herbal-originated diterpene acid from Pseudolarix kaempferi Gordon, has been reported to possess inhibitory activity against fungus. The present study aims to investigate the antifungal effect of PAB alone and in combination with FLC on planktonic and biofilm cells of C. tropicalis.
Methods: The antifungal activity of PAB against planktonic isolates was evaluated alone and in combination with FLC using the chequerboard microdilution method and growth curve assay. The anti-biofilm effects were quantified by tetrazolium (XTT) reduction assay, which were further confirmed by scanning electron microscopy (SEM) and fluorescent microscope to observe morphological changes of biofilm treated with PAB and FLC.
Results: It was revealed that PAB alone exhibited similar inhibitory activity against FLC-resistant and FLC-susceptible strains with median MIC ranging from 8 to 16 μg/mL. When administered in combination, synergism was observed in all (13/13) FLC-resistant and (2/9) FLC-susceptible strains with FICI ranging from 0.070 to 0.375. Moreover, the concomitant use of PAB and FLC exhibited a strong dose-dependent synergistic inhibitory effect on the early and mature biofilm, eliminating more than 80% biofilm formation. SEM found that PAB, different from azoles, could significantly inhibit spore germination and destroy the cell integrity causing cell deformation, swelling, collapse and outer membrane perforation.
Conclusion: PAB was highly active against FLC-resistant isolates and biofilm of C. tropicalis, particularly when combined with FLC. These findings suggest that PAB may have potential as a novel antifungal agent with different targets from azole drugs.
Keywords: C. tropicalis, pseudolaric acid B, fluconazole, biofilm, antifungal susceptibility
Introduction
C. tropicalis has received widespread attention in recent years due to increased prevalence and high mortality. Although C. albicans remains the predominant pathogenic fungus causing invasive candidiasis (IC), C. tropicalis has emerged as the most or second frequently isolated non-albicans Candida (NCAC) species.1–3 Moreover, C. tropicalis exhibit higher levels of resistance and cross-resistance to azole drugs compared with C. albicans isolates, which led to the failure of therapeutic strategies.1,4
It was revealed that biofilm formation might be more influential than invasiveness on the development of persistent candidemia.5 The National Institutes of Health estimates that biofilms are responsible for approximately 80% of microbial infections in humans.6 With the surrounding of extracellular matrix, C. tropicalis biofilm is a closely packed microbial community constituted of yeast and hyphae, which can establish on implanted medical devices, such as intravascular catheters, dentures and pacemakers. Such growth characteristics of biofilm confer C. tropicalis cells some properties that are distinct from their planktonic counterparts, for example, they exhibit greater resistance to antifungal agent and host immune defenses, serving as reservoirs for persistent infections.5,7,8 It has been reported that biofilm of candida may be up to 1000 times more resistant to antifungal agents when compared with their planktonic cell.9 Therefore, efforts are needed to explore promising antifungal drugs that are effective against azole-resistant isolates and biofilms of C. tropicalis.
Pseudolaric acid B (PAB), a diterpene acid, is the major antifungal constituent of a traditional Chinese herb known as “Tu-Jin-Pi”, which has been prescribed as a treatment for fungal infections of skin since the 17th century.10,11 The antifungal activity of PAB against planktonic cells of FLC-resistant C. albicans has been demonstrated, displaying a significant synergistic effect when it was combined with FLC.12,13 However, it has not been described on the effectiveness of PAB alone or in combination with azoles against planktonic cells and biofilms of C. tropicalis, whose resistance to clinical commonly used antifungal agents is more serious.
The combination therapy is a common approach to improve the efficacy of antifungal agents and reduce the adverse reactions of these drugs. The present study aimed to elucidate the antifungal activity of PAB alone and in combination with FLC on planktonic and biofilm cells of C. tropicalis. It is hoped that our research will contribute to overcome the drug resistance of C. tropicalis.
Materials and Methods
Fungal Strains and Materials
Twenty-one clinical isolates of C. tropicalis involved in our study were kindly provided by the Clinical laboratory of Ruijin Hospital affiliated to Shanghai Jiaotong University School of Medicine. In addition, C. parapsilosis ATCC 22019 and C. tropicalis ATCC 750 were used as controls. Pseudolaric acid B (PAB), a diterpene acid from Pseudolarix kaempferi Gordon (Pinaceae), was purchased from the Tauto Biotech Co., Ltd (Shanghai, China). And Fluconazole (FLC) was obtained from Sigma-Aldrich. Stock solutions of PAB and FLC were both prepared in dimethyl sulfoxide. The final concentration of DMSO was not higher than 0.14%. What’s more, RPMI 1640 (Thermo Fisher Scientific), menadione (Sangon Biotech, Shanghai) and 2,3-bis(2-methoxy-4-nitro-5-sulfophenyl)-2H-tetrazolium-5-carboxanilide, XTT, (Sigma-Aldrich) were used in this study.
Antifungal Susceptibility Testing
The minimum inhibitory concentrations (MICs) of antifungal agents (PAB and FLC) against the C. tropicalis strains were determined according to the broth microdilution assay in 96-well flat-bottomed microtitration plates as described by the method M27-A3 of the Clinical and Laboratory Standards Institute (CLSI, formerly NCCLS).14–16 Twofold serial dilutions in RPMI 1640 medium were performed to obtain the final concentrations ranged from 0.125 to 64 μg/mL for PAB and from 0.125 to 512 µg/mL for FLC. To ensure the quality of susceptibility tests, reference strains C. parapsilosis ATCC 22019 and C. tropicalis ATCC 750 were included in each batch of test. All plates were incubated at 37°C for 24h, after which the MICs were determined by both visual reading and optical density (OD) determination as previously reported.11 Three independent experiments were performed.
Microdilution Chequerboard Assay
The synergistic effects of PAB with FLC against planktonic C. tropicalis isolates were analyzed using the method of microdilution chequerboard as previously described.12 Drug dilutions were prepared in RPMI 1640 medium to obtain four times of the final concentration. Then, 50 μL of medium containing PAB with different concentrations were added to rows A to H of the 96-well microtitration plates, and 50 μL of FLC medium was added to columns 1 to 11. Finally, 100 μL of Candida suspensions (103 CFU/mL) was added to each well. Wells in column 12 served as the positive (only containing Candida suspensions) and negative (only containing medium) growth control. The final concentrations ranged from 0.125 to 16 µg/mL for PAB, 0.03125 to 32 µg/mL for FLC. After incubation at 37°C for 24h, the MICs were determined by visual analysis, which represent the lowest concentration of 100% growth inhibition.
The Fractional inhibitory concentration index (FICI) was used to evaluate the synergistic effects of drugs.12 FICI = FICA + FICB = C Acomb ⁄ MIC A alone + C B comb ⁄MIC B alone. MIC A alone and MICB alone are the MIC values of drug A and B when acting alone. C Acomb and C B comb are concentrations of drug A and B in combination. The FICI value of ≤0.5 represented synergy, FICI value between 0.5 and 4 represented indifference and FICI value >4 represented antagonism.
Growth Curve Assay
Growth curve assay was used to dynamically monitor the synergistic effect of PAB with FLC against planktonic C. tropicalis isolates.17 The fungal supernatant (103 CFU/mL) containing agents (1 µg/mL PAB, 2 µg/mL FLC, and 1µg/mL PAB + 2µg/mL FLC) was grown with constant shaking at 37◦C. The control was free of FLC and PAB. At designated time points (0, 12, 24, 48 h), the cell broths were pipetted out to determine the OD with microplate reader at 495 nm. What’s more, 20 μL of cell broths was filled into the fast counting plate to observe the growth state of strains with microscope at 48h.
Effects of PAB and FLC Against Biofilm Formation
The effects of PAB and FLC against C. tropicalis biofilm were quantified by tetrazolium (XTT) reduction assays.18,19 Biofilms were produced on commercially available polystyrene, flat-bottomed 96-well microtiter plates. At first, 100 μL of a standardized cell suspension (106 CFU/mL) in RPMI 1640 medium was transferred into selected wells for biofilm formation, and were incubated at 37°C. At the early phase of biofilm formation (0, 2h), 100 μL of RPMI 1640 medium containing PAB alone (or FLC) were added to each well. To detect the developmental and mature biofilms, drugs were, respectively, added at 6h and 24h, before which non-adhered cells were removed by sterile PBS. After incubation at 37°C for 48h, biofilm growth was analyzed with XTT assay as described.19 As for the anti-biofilm effect of combination, 50 μL of RPMI 1640 medium containing PAB or FLC was added to each well at designated time points (0, 2, 6, 24h) according to the method of microdilution chequerboard mentioned above.
Each experiment was repeated for three times.
Fluorescein Diacetate Assay
The fluorescence microscope assay was performed according to the method as reported.20 The fungal supernatant of ATCC750 (106 CFU/mL) containing agents (4µg/mL PAB, 16 µg/mL FLC, and 4 µg/mL PAB + 16 µg/mL FLC) was incubated to form biofilms on 6-well plate at 37◦C. The control was free of PAB and FLC. After 24 h culture, the plate was rinsed by sterile PBS to remove planktonic cell. Subsequently, 1.5mL fresh RPMI 1640 medium and 1.5mL diluent of 0.2 mg/mL fluorescein diacetate (FDA, Sigma, Shanghai, China) were added into each well. The plate was incubated at 37◦C for 30 min in the dark. The morphology of biofilms was visualized by a fluorescent microscope (Oumeng,EUROStar III plus) at the excitation wavelength of 494 nm and the emission wavelength of 518nm.
Scanning Electron Microscope
Firstly, biofilms (ATCC750) were formed on 6-well plate with different concentrations of drugs (4µg/mL PAB, 16µg/mL FLC, and 4µg/mL PAB + 16 µg/mL FLC). The control had only fungal suspension (106 CFU/mL) without drug. After incubated at 37◦C for 24h, the wells were cut into square pieces of 1x1cm, which were fixed in 2.5% glutaraldehyde overnight at 4◦C. The pieces were washed three times with PBS, and post-fixed with 1% osmium acid. Thereafter, they were dehydrated sequentially by 30, 50, 70, 80, 95 and 100% (twice) ethanol for each step 15 min. After critical point drying, the biofilms on pieces were sputter coated with gold in a vacuum evaporator and observed by scanning electron microscope (FEI, Quanta-250).
Statistical Analysis
The MICs were presented as Median (Range). The inhibition rates were expressed as mean values with corresponding standard deviations. Statistical analysis was performed with SPSS 23.0. Differences between groups were determined using analysis of variance (ANOVA). A P<0.05 was considered statistically significant.
Results
Antifungal Activities Against Planktonic Isolates
The antifungal activities of PAB and FLC alone were determined by the broth microdilution assay. Among the 22 isolates of C. tropicalis tested, 13 isolates were resistant to FLC with MIC values ranging from 8 to 256µg/mL, and 9 isolates were sensitive to FLC with MICs ranging from 1 to 4µg/mL. The median MICs of PAB were in a range of 8–16 µg/mL against all the C. tropicalis isolates, indicating that PAB exhibited similar inhibitory activity against FLC-resistant and FLC-susceptible strains when it was used alone (Table 1).
 |
Table 1 The Interaction Between Pseudolaric Acid B and Fluconazole Against Isolates of C. tropicalis in vitro |
The synergistic effects of PAB with FLC against planktonic C. tropicalis isolates were analyzed using the method of microdilution chequerboard. As is presented in Table 1, a significant decrease in MICs of FLC was observed when planktonic cells were tested in the presence of PAB. For instance, MIC of isolate 1782 to FLC in combination with 0.5 µg/mL PAB was found to be 2μg/mL, which was 128 times less than that of fluconazole alone (256μg/mL). What’s more, the FICI values ranged from 0.070 to 0.375 for FLC-resistant isolates, showing a good synergistic effect against FLC-resistant C. tropicalis s isolates when PAB was combined with FLC. While for 9 of the FLC-S strains, including ATCC 750, the PAB/FLC combination utilization displayed synergy (2/9) or indifference (7/9) with FICI values ranging from 0.281 to 1.008. No antagonism interactions between PAB and FLC were observed in either FLC-resistant or FLC-susceptible C. tropicalis isolates.
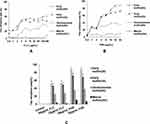
In order to further evaluate the synergism of FLC and PAB against resistant C. tropicalis, we used the Growth curve assay. As shown in Figure 1, little differences in growth were seen among the 4 groups in the first 12h. At 48h, the OD value was reduced more than 2-fold in the combination group (FLC+PAB) than in the FLC-alone group, indicating the synergistic effect of 2µg/mL of FLC and 1µg/mL of PAB against C. tropicalis isolate (Figure 1). The microscope results showed reduction of spores and hyphae, which also confirmed the synergistic antifungal activity of PAB/FLC combination utilization (Figure 2).
Effects of PAB/FLC Alone on Biofilm Formation
The effects of PAB/FLC on biofilm formation at different growth stages were analyzed using a tetrazolium salt XTT reduction assay by calculating the percent reduction in biofilm growth compared to untreated control (Figure 3). During the early phase (0h) of biofilm formation, drug intervention was added at the beginning of biofilm formation. PAB alone repressed the formation of biofilm in a dose-dependent manner, while no significant correlation was found between the FLC doses and its inhibitory effects on biofilm formation. When PAB was used alone, the percent reduction in biofilm growth elevated with the increasing of drug concentration, and treatment with 32 µg/mL of PAB inhibited more than 80% biofilm formation of C. tropicalis isolates. However, the inhibition of biofilm in 256 µg/mL FLC was about 60% for ATCC750, although about 20% biofilms could be repressed at a lower concentration of 0.25 µg/mL at the early phase. At the developmental phase (6h), C. tropicalis cells grew for 6 hours to form metaphase biofilm, and then FLC+PAB was added. Both PAB and FLC decreased about 30% biofilm formation. Mature biofilms (24h) were completely resistant to PAB and FLC, when they were used alone. At the early stage (0h), the inhibition rate of 32 µg/mL PAB on biofilm formation was more than 80%, while at the same concentration, the inhibition rate of mature biofilm (24h) was less than 10% (Figure 3).
Effects of PAB and FLC Combination on Biofilm Formation
The invitro activities of combinations against C. tropicalis biofilms were investigated with checkerboard assays. It was found that the combination utilization of PAB and FLC displayed strong inhibitory effects on both early and mature biofilms (Figure 4). And the percent reduction in biofilm formation increases with the increase of FLC concentration in the presence of PAB. During the early phase, the combination of 16 µg/mL of FLC and 2 µg/mL of PAB on ATCC750 could inhibit 80.36% biofilm formation, while the inhibition of biofilm in 256 µg/mL FLC alone was about 60.99%. FLC alone was ineffective against mature biofilms, but when combined with 64 µg/mL of PAB, could inhibit about 50%, 70%, 80% biofilms at concentrations of 2,32,256 µg/mL, respectively. Treated with 4 µg/mL of PAB+16 µg/mL of FLC inhibited the biofilm formation significantly (P<0.05) compared with the separate use of PAB or FLC.
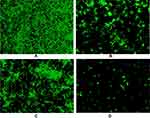
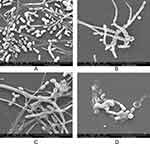
To further confirm the synergistic inhibitory effects of PAB/FLC on biofilm formation, we also employed fluorescent microscope (Figure 5) and SEM (Figure 6) to observe the morphological changes of C. tropicalis ATCC750 after the treatment of PAB and FLC. As shown in Figure 6A, biofilms of the drug-free control were mainly composed of blast conidia and elongated hyphae. When it was treated with 4 µg/mL PAB, both hyphal and blast conidia cells were reduced, especially blast conidia cells, indicating that PAB has strong ability to inhibit spore germination (Figure 6C). Moreover, PAB could cause alterations in hyphal morphology of C. tropicalis, such as distortion, swelling, collapse and perforated outer membrane. However, biofilm formed in the presence of 16 µg/mL FLC exhibited less hyphae and blast conidia, suggesting that fluconazole mainly postpone spore proliferation and mycelium formation (Figure 6B). It is noteworthy that 16 µg/mL FLC did not destroy cell structure, which was consistent with the results of XTT assay. Because the biofilm activity in the presence of 16 µg/mL FLC was about 50% of the control group, although the number of cells has been significantly reduced. When PAB and FLC were used in combination, the inhibitory effects of the two drugs were superimposed. Only a few scattered yeast cells could be found, showing broken and deformed morphology (Figure 6D).
Discussion
Due to the extensive use of conventional antifungal agents in past decades, azole-resistant C. tropicalis has emerged worldwide, particularly in the Asia Pacific region. In China, the fluconazole and voriconazole resistant rates of C. tropicalis substantially increased from <8% in 2009–2010 to over 22% in 2013–2014.21 In the US, C. tropicalis (4–9%) also has a higher incidence of fluconazole resistance than C. albicans(0.5–2%).22 It is reported that C. tropicalis appears to develop fluconazole resistance much more rapidly than other Candida species under in vitro selection.23 Thus, novel anticandidal agents are urgently needed to relieve the pressure caused by C. tropicalis.
Pseudolaric acid B (PAB) is considered as the major antifungal component of Cortex pseudolaricis (tujingpi). Previous studies have shown that the extract of “tujingpi” is effective against C. albicans, Trichophyton mentagrophytes, Microsporum gypseum and Torulopsis petrophilum at micromolar levels.10 However, the anticandidal effect of PAB against C. tropicalis has rarely been reported. The present study evaluated the inhibitory activity of PAB against planktonic cell and biofilm of C. tropicalis in vitro.
As for planktonic cell, PAB exhibited the similar antifungal activity against clinically isolated FLC-resistant and FLC-susceptible C. tropicalis strains with MIC ranging from 8 to 16 µg/mL (Table 1), when it was used alone. This potent nonselective effect of PAB on susceptible and resistant C. tropicalis suggests that it may have potential as a novel antifungal agent with different mechanism from azole drugs. What’s more, when PAB was combined with FLC, they showed better synergistic effects on FLC-resistant C. tropicalis isolates, rather than their FLC-susceptible counterparts. Such similar synergistic phenomenon has been reported on palmatine, berberine, and calcium channel blockers.17,24,25
It is necessary to find effective methods to inhibit the growth of biofilm, because the biofilm formed on living and non-living surfaces is better protected from immune defense and antimicrobial agents than their free-living cells, which are associated with the pathogenesis resistance of Candida.8,26 As it is known, Candida biofilm formation comprises several specific stages including the early phase (adherence of round yeast cells to a solid surface), the developmental phase (cell proliferation and early-stage filamentation), and the biofilm maturation stage.27 Mature biofilm is more difficult to be eradicated by antifungal agent, owing to the complex network of polymorphic cells encased in an extracellular matrix.
In the present study, our results found that the concomitant use of PAB and FLC was applicable in the removal of developmental and mature biofilms. The morphological changes showed by SEM further confirmed the synergistic inhibitory effect of PAB and FLC on biofilm. PAB and FLC may have different antifungal targets, which contributes to their synergistic action. Unlike azoles, which target the ergosterol biosynthesis in fungal cell membranes, PAB not only inhibited spore germination and mycelium formation, but also destroyed the cell integrity, leading to cell deformation, swelling, collapse and outer membrane perforation. The morphological changes induced by PAB in C. gloeosporioides are similar to those caused by microtubule inhibitors.28 Recent studies have suggested that the antitumor activity of PAB could be associated with its ability to inhibit cell proliferation and induce cell apoptosis related to microtubule depolymerization.29 Microtubules, composed of α - and β-tubulin heterodimers, are responsible for a variety of biological functions including sustained shapes, the intracellular transport and the cell division.30 Disruption of microtubule dynamics leads to cell cycle arrest and cell death.30 These findings warrant further investigation to find the potential antifungal targets and molecular mechanism of PAB against resistance candida.
Conclusion
In conclusion, the present study confirmed the antifungal effects of PAB alone and in combination with FLC on planktonic and biofilm cells of C. tropicalis. PAB alone showed similar antifungal activity against FLC-resistant and FLC-susceptible planktonic cells, and exhibited better synergistic effects on FLC-resistant isolates and mature biofilms of C. tropicalis when combined with FLC. PAB not only inhibited spore germination and mycelium formation, but also destroyed the cell integrity. These findings suggest that PAB may have potential as a novel antifungal agent with different mechanism from azole drugs.
Acknowledgments
This study was supported by the national clinical research base of traditional Chinese medicine of Longhua Hospital (Grant number: Seedling program LYTD66).
Author Contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Wang H, Xu YC, Hsueh PR. Epidemiology of candidemia and antifungal susceptibility in invasive Candida species in the Asia-Pacific region. Future Microbiol. 2016;11(11):1461–1477. doi:10.2217/fmb-2016-0099
2. Orasch C, Marchetti O, Garbino J, et al. Candida species distribution and antifungal susceptibility testing according to EUCAST and new vs old CLSI clinical breakpoints: a six-year prospective candidemia survey from the Fungal Infection Network of Switzerland. Clin Microbiol Infect. 2014;20(7):698–705. doi:10.1111/1469-0691.12440
3. Trong LN, Viet HD, Quoc Doan TT, et al. Biological activities of essential oils from leaves of paramignya trimera (Oliv.) Guillaum and Limnocitrus littoralis (Miq.) Swingle. Antibiotics (Basel). 2020;9(4):E207. doi:10.3390/antibiotics9040207
4. Xiao M, Sun ZY, Kang M, et al. Five-year national surveillance of invasive candidiasis: species distribution and azole susceptibility from the china hospital invasive fungal surveillance net (CHIF-NET) study. J Clin Microbiol. 2018;56(7):00577–618. doi:10.1128/JCM.00577-18
5. Li WS, Chen YC, Kuo SF, Chen FJ, Lee CH. The impact of biofilm formation on the persistence of candidemia. Front Microbiol. 2018;9:1196. doi:10.3389/fmicb.2018.01196
6. Lohse MB, Gulati M, Johnson AD, Nobile CJ. Development and regulation of single- and multi-species Candida albicans biofilms. Nat Rev Microbiol. 2018;16(1):19–31. doi:10.1038/nrmicro.2017.107
7. Cavalheiro M, Teixeira MC. Candida biofilms: threats, challenges, and promising strategies. Front Med (Lausanne). 2018;5:28. doi:10.3389/fmed.2018.00028
8. Marioni J, Bresoli-Obach R, Agut M, et al. On the mechanism of Candida tropicalis biofilm reduction by the combined action of naturally-occurring anthraquinones and blue light. PLoS One. 2017;12(7):e0181517. doi:10.1371/journal.pone.0181517
9. Hacioglu M, Birteksoz Tan AS, Dosler S, Inan N, Otuk G. In vitro activities of antifungals alone and in combination with tigecycline against Candida albicans biofilms. Peer J. 2018;6:e5263. doi:10.7717/peerj.5263
10. Chiu P, Leung LT, Ko BC. Pseudolaric acids: isolation, bioactivity and synthetic studies. Nat Prod Rep. 2010;27(7):1066–1083. doi:10.1039/b906520m
11. Wen C, Chen J, Zhang D, et al. Pseudolaric acid B induces mitotic arrest and apoptosis in both 5-fluorouracil-sensitive and -resistant colorectal cancer cells. Cancer Lett. 2016;383(2):295–308. doi:10.1016/j.canlet.2016.09.007
12. Guo N, Ling G, Liang X, et al. In vitro synergy of pseudolaric acid B and fluconazole against clinical isolates of Candida albicans. Mycoses. 2011;54(5):e400–406. doi:10.1111/j.1439-0507.2010.01935.x
13. Liu S, Hou Y, Chen X, Gao Y, Li H, Sun S. Combination of fluconazole with non-antifungal agents: a promising approach to cope with resistant Candida albicans infections and insight into new antifungal agent discovery. Int J Antimicrob Agents. 2014;43(5):395–402. doi:10.1016/j.ijantimicag.2013.12.009
14. National Committee for Clinical and Laboratory Standards. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts. Approved Standard. M27-A. Wayne, PA: National Committee for Clinical and Laboratory Standards; 1997.
15. Le N T, Ho D V, Quoc Doan T, et al. In vitro antimicrobial activity of essential oil extracted from leaves of leoheo domatiophorus Chaowasku, D.T. Ngo and H.T. Le in Vietnam. Plants (Basel). 2020;9(4):E453.
16. Donadu MG, Usai D, Marchetti M, et al. Antifungal activity of oils macerates of North Sardinia plants against Candida species isolated from clinical patients with candidiasis. Nat Prod Res. 2019;24:1–5. doi:10.1080/14786419.2018.1557175
17. Xu Y, Quan H, Wang Y, et al. Requirement for ergosterol in berberine tolerance underlies synergism of fluconazole and berberine against fluconazole-resistant Candida albicans Isolates. Front Cell Infect Microbiol. 2017;7:491. doi:10.3389/fcimb.2017.00491
18. Sun L, Liao K, Wang D. Effects of magnolol and honokiol on adhesion, yeast-hyphal transition, and formation of biofilm by Candida albicans. PLoS One. 2015;10(2):e0117695. doi:10.1371/journal.pone.0117695
19. Dovigo LN, Pavarina AC, Carmello JC, Machado AL, Brunetti IL, Bagnato VS. Susceptibility of clinical isolates of Candida to photodynamic effects of curcumin. Lasers Surg Med. 2011;43(9):927–934. doi:10.1002/lsm.21110
20. Shao J, Shi G, Wang T, Wu D, Wang C. Antiproliferation of berberine in combination with fluconazole from the perspectives of reactive oxygen species, ergosterol and drug efflux in a fluconazole-resistant candida tropicalis isolate. Front Microbiol. 2016;7:1516. doi:10.3389/fmicb.2016.01516
21. Guo LN, Xiao M, Cao B, et al. Epidemiology and antifungal susceptibilities of yeast isolates causing invasive infections across urban Beijing, China. Future Microbiol. 2017;12:1075–1086. doi:10.2217/fmb-2017-0036
22. Berkow EL, Lockhart SR. Fluconazole resistance in Candida species: a current perspective. Infect Drug Resist. 2017;10:237–245. doi:10.2147/IDR.S118892
23. Jin L, Cao Z, Wang Q, et al. MDR1 overexpression combined with ERG11 mutations induce high-level fluconazole resistance in Candida tropicalis clinical isolates. BMC Infect Dis. 2018;18(1):162. doi:10.1186/s12879-018-3082-0
24. Wang T, Shao J, Da W, et al. Strong synergism of palmatine and fluconazole/itraconazole against planktonic and biofilm cells of candida species and efflux-associated antifungal mechanism. Front Microbiol. 2018;9:2892. doi:10.3389/fmicb.2018.02892
25. Liu S, Yue L, Gu W, Li X, Zhang L, Sun S. Synergistic effect of fluconazole and calcium channel blockers against resistant candida albicans. PLoS One. 2016;11(3):e0150859. doi:10.1371/journal.pone.0150859
26. Nobile CJ, Johnson AD. Candida albicans biofilms and human disease. Annu Rev Microbiol. 2015;69:71–92. doi:10.1146/annurev-micro-091014-104330
27. Gulati M, Nobile CJ. Candida albicans biofilms: development, regulation, and molecular mechanisms. Microbes Infect. 2016;18(5):310–321. doi:10.1016/j.micinf.2016.01.002
28. Zhang J, Yan LT, Yuan EL, et al. Antifungal activity of compounds extracted from Cortex Pseudolaricis against Colletotrichum gloeosporioides. J Agric Food Chem. 2014;62(21):4905–4910. doi:10.1021/jf500968b
29. Liu ML, Sun D, Li T, Chen H. A systematic review of the immune-regulating and anticancer activities of Pseudolaric Acid B. Front Pharmacol. 2017;8:394. doi:10.3389/fphar.2017.00394
30. Ding Y, Li Y, Li Z, et al. Alteramide B is a microtubule antagonist of inhibiting Candida albicans. Biochim Biophys Acta. 2016;1860(10):2097–2106. doi:10.1016/j.bbagen.2016.06.025
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.