Back to Journals » International Journal of Nanomedicine » Volume 14
Synergistic antibacterial effect of ultrasound microbubbles combined with chitosan-modified polymyxin B-loaded liposomes on biofilm-producing Acinetobacter baumannii
Authors Fu Y, Zhang L, Yang Y, Liu C, He Y, Li P , Yu X
Received 5 September 2018
Accepted for publication 3 February 2019
Published 8 March 2019 Volume 2019:14 Pages 1805—1815
DOI https://doi.org/10.2147/IJN.S186571
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Lei Yang
Yu-Ying Fu,1 Liang Zhang,2 Yi Yang,1 Cheng-Wei Liu,3 Ying-Na He,4 Pan Li,2 Xian Yu1
1Phase I Clinical Trial Center, The Second Affiliated Hospital of Chongqing Medical University, Chongqing, China; 2Chongqing Key Laboratory of Ultrasound Molecular Imaging, Institute of Ultrasound Imaging, Second Affiliated Hospital, Chongqing Medical University, Chongqing, China; 3State Key Laboratory of Infectious Diseases and Parasites, First Affiliated Hospital, Chongqing Medical University, Chongqing, China; 4Department of Pharmacy, Hebei University of Chinese Medicine, Shijiazhuang, China
Purpose: Resistant strains of Acinetobacter baumannii (AB) that can form biofilms are resistant to polymyxin. Therefore, effective and safe polymyxin preparations against biofilm-producing AB are urgently needed. This study aims to prepare chitosan-modified polymyxin B-loaded liposomes (CLPs) and ultrasound microbubbles (USMBs) and then explore the synergistic antibacterial effects of USMBs combined with CLPs in vitro.
Methods: CLPs were prepared using a modified injection method, and microbubbles were prepared using a simple mechanical vibration method. Minimal biofilm inhibitory concentration (MBIC) of CLPs against resistant biofilm-producing AB was determined. Antibacterial activities of CLPs with or without USMBs were analyzed by crystal violet staining and resazurin assays to evaluate biofilm mass and viable counts, respectively. Then, the anti-biofilm effects of CLPs with or without USMBs on biofilm-producing AB were confirmed via scanning electron microscopy (SEM) analysis.
Results: We prepared CLPs that were 225.17±17.85 nm in size and carried positive charges of 12.64±1.44 mV. These CLPs, with higher encapsulation efficiency and drug loading, could exhibit a sustained release effect. We prepared microbubbles that were 2.391±0.052 µm in size and carried negative charges of -4.32±0.43 mV. The MBICs of the CLPs on the biofilm-producing AB was 8±2 µg/mL, while that of polymyxin B was 32±2 µg/mL. USMBs in combination with 2 µg/mL of polymyxin B could completely eliminate the biofilm-producing AB and achieve the maximum antimicrobial effects (P>0.05 vs sterile blank control). SEM imaging revealed some scattered bacteria without a biofilm structure in the USMB combined with the CLP group, confirming that this combination has the greatest anti-biofilm effects.
Conclusion: In this research, we successfully prepared USMBs and CLPs that have a more significant antibacterial effect on biofilm-forming AB than polymyxin B alone. Experiments in vitro indicate that the synergistic antibacterial effect of combining USMBs with CLPs containing as little as 2 µg/mL of polymyxin B is sufficient to almost eliminate drug-resistant biofilm-producing AB.
Keywords: liposome, chitosan, polymyxin B, ultrasound microbubbles, Acinetobacter baumannii, biofilms
Introduction
Acinetobacter baumannii (AB) is one of the most serious opportunistic pathogens in nosocomial infections. It can persist and form biofilms on various abiotic materials in a hospital environment, thereby coming into contact with susceptible patients and causing outbreaks of ventilator-associated pneumonia, meningitis, septicemia, urinary tract infections, and skin and soft tissue infections (SSTIs).1 A biofilm is an aggregate of microbial cells embedded in a self-produced matrix on living or non-living surfaces.2 It can be viewed as a protected mode of microbial growth that can provide protection from hostile environments, eg, in cases involving Acinetobacter, biofilm-forming isolates can survive longer than their non-biofilm-forming counterparts.3 Biofilms have significantly higher antibiotic resistance than their planktonic counterparts and thus have serious consequences for the treatment of biofilm-associated infections.4
Reports from various parts of the world have indicated a growing concern regarding multi-, extensive-, and pan-drug-resistant (MDR, XDR, and PDR) strains of AB, some of which are resistant to even polymyxin.5–7 Polymyxin comprises a class of cyclic polypeptide antibiotics that include polymyxins A–E, of which only polymyxins B and E are used in the clinic. Polymyxin B or E is applied to treat severe infections caused by Gram-negative bacteria. However, due to their severe renal toxicity, neurotoxicity, and narrow therapeutic window, their clinical applications are limited to use as a last resort for treating MDR-AB or other MDR Gram-negative bacterial infections.8 Therefore, effective and safe polymyxin B or E preparations against biofilm-producing AB are urgently needed.
Liposomes, which are a type of a drug delivery system (DDS), are spherical vesicles consisting of one or more phospholipid bilayers surrounding a drug and thus affect pharmacokinetics, pharmacodynamics, toxicity, immunogenicity, and biological identification.9 They can protect antimicrobial agents from binding to matrix material and from enzymatic inactivation, thus making chemical treatments more effective, reducing the toxicity of antimicrobials, and increasing the safety of chemical treatments.10 However, liposomes still have some shortcomings, such as the chemical instability due to the hydrolysis of ester bonds in structures, the oxidation of unsaturated acyl chains in lipids, and the physical instability caused by the leakage of encapsulated drugs.11 Chitosan as a polycationic heteropolysaccharide has attracted the attention of researchers due to its low toxicity, bacteriostasis, biocompatibility, and moisture-retention properties. Using chitosan to modify liposomes can improve the stability of preparations.12,13 Ultrasound microbubbles (USMBs) are a new type of DDS for the treatment of bacterial infection.14–17 A number of publications have indicated that ultrasound with cavitation can enhance the inhibitory effects of antimicrobial agents on bacterial biofilms, which can be amplified by microbubbles.18,19 As a result, USMBs can promote the bacterial uptake of antimicrobials and improve the antibacterial efficacy of drugs.20–22 This study aims to explore the synergistic antibacterial effects of combining USMBs with chitosan-modified polymyxin B-loaded liposomes (CLPs) in vitro to assess the feasibility of employing this combined DDS in systemic or topical antibacterial treatment of biofilm-producing AB infections.
Materials and methods
Bacterial strains and culture conditions
In this study, the bacterial strain AB W1340, the strain of AB that had been clinically isolated from the sputum of a pneumonia patient in the First Affiliated Hospital of Chongqing Medical University as part of a routine hospital laboratory procedure, was used. The minimum inhibitory concentration (MIC) values of the antibacterial agent against this clinical isolate had been determined, and the results showed that this strain is resistant to multiple antibiotics, including polymyxin B (Table S1). The strain forms dense biofilms on polystyrene and glass surfaces, and the mature biofilms are resistant to polymyxin B. The strain was inoculated onto blood agar plates (Jiangmen Caring Trading Company, Jiangmen, China) and cultivated for 18 hours at 37°C. Ninety-six-well polystyrene microtiter plates were used for the cultivation of AB biofilms. Briefly, the organisms were grown in Luria-Bertani broth (LB; Qingdao Haibo Biochemistry Instrument, Qingdao, China) overnight at 37°C with agitation, and bacterial suspensions were adjusted to contain an equivalence of the McFarland standard of 0.5. Then, 200 μL of bacterial suspensions was added to 96-well plates and incubated at 37°C for 4 days, and the LB was replaced every 24 hours. Biofilms were cultivated in 24-well polystyrene microtiter plates in a similar manner. In short, sterilized 12 mm coverslip disks were placed into the bottoms of 24-well plates. Then, 200 μL of bacterial suspensions with a McFarland standard of 0.5 and 1.5 mL of LB were added to each well. The 24-well plates were incubated at 37°C for 4 days, and the LB was replaced every 24 hours.
Preparation of CLPs and chitosan-modified liposomes
CLPs were prepared using a modified injection method.23 Briefly, the components dipalmitoylphosphatidylcholine (DPPC), distearoylphosphatidylcholine (DSPE), and cholesterol (CHOL) (Corden Pharma Switzerland LLC, Liestal, Switzerland) (at a weight ratio of 3:1:1) were completely dissolved in 10 mL of chloroform as the lipid phase. Polymyxin B (Sangon Biotech, Shanghai, China) was dissolved in 2 mL of PBS (pH 7.4) at 50°C as the aqueous phase. Then, the lipid phase was dropped into either the aqueous phase or PBS (pH 7.4) under gentle magnetic stirring, followed by evaporation via rotary evaporator to remove the organic solvent (Shanghai Yarong Biochemistry Instrument, Shanghai, China). Next, the liposomes and 0.1% chitosan (Sangon Biotech) solution (chitosan was dissolved in a 0.1 M acetic acid glacial solution) were mixed at equal volumes, followed by 10 minutes of sonication in an ice bath using an ultrasonic processor (175 W; Sonics & Material, Newtown, CT, USA). Then, CLPs and chitosan-modified liposomes were prepared. Finally, ultrafiltration was used to remove the unencapsulated polymyxin B using the liposome solution in a 15 mL ultrafiltration tube and centrifuging at 5,000 rpm for 20 minutes at 4°C. Then, the CLPs were collected. All CLP dispersions were stored at 4°C for further analysis. The particle size and potential of the final liposomes were analyzed using a laser particle size analyzer system (Zetasizer 3000 HS; Malvern Instruments, Malvern, UK). For morphological analysis, the liposomes were stained with 0.1% phosphotungstic acid for 10 minutes, washed with ultrapure water, and subjected to transmission electron microscopy (TEM, TECNAI-10; Philips, Eindhoven, the Netherlands).
Preparation of microbubbles
Microbubbles were prepared using a simple mechanical vibration method.16 Briefly, 5 mg of DPPC, 2 mg of DSPE, and 0.5 mg of CHOL were dissolved in 500 μL of 10% glycerol solution in a vial. After heating in a water bath at 50°C for 30 minutes, the air in the vial was exchanged with perfluoropropane (C3F8; Research Institute of Physical and Chemical Engineering of Nuclear Industry, Tianjin, China), followed by vigorous shaking for 45 seconds via dental amalgamator (YJT; Shanghai Medical Apparatus and Instruments, Shanghai, China). Finally, the microbubbles were washed with PBS twice to obtain supernatant microbubbles via centrifugation at 500 rpm, and the concentration was then adjusted to 108 per mL. The particle size and potential of the microbubbles were analyzed using a laser particle size analyzer system (Zetasizer 3000 HS; Malvern Instruments). The morphological characteristics of microbubbles were analyzed using an inverted optical microscope (IX71; Olympus, Tokyo, Japan).
Encapsulation and drug loading efficiency of CLPs
Free polymyxin B was separated by ultrafiltration–centrifugation technique using 10-kDa MWCO Amicon centrifugal filters (EMD Millipore, Billerica, CA, USA) at 5,000 rpm for 20 minutes at 4°C. Then, the free polymyxin B content in the filtrate was determined by measuring the absorbance at 215 nm using ultraviolet spectrophotometry (UV 2600; Shimadzu, Kyoto, Japan). Blank liposomes were used as controls.
In vitro drug release study of CLPs
The release kinetics of CLPs was monitored as previously described.24 In short, a dialysis bag containing 2 mL of CLPs was placed in an opaque bottle filled with 100 mL of PBS as the release medium. The medium was then incubated at 37°C under mild continuous agitation. At 1, 2, 3, 6, 9, 12, 24, and 48 hours, 2 mL of the release medium was withdrawn and replaced with 2 mL of fresh PBS, and the amount of polymyxin B released was determined by measuring absorbance at 215 nm. Blank liposomes were used as controls.
Minimal biofilm inhibitory concentration (MBIC) determinations
First, 96-well plates were used to cultivate AB W1340 biofilms. The biofilms were gently washed three times using sterile water to remove planktonic bacteria and then incubated at 37°C in 200 μL of LB containing twofold serial dilutions of polymyxin B or CLPs. After 24 hours, the plates were gently washed and supplemented with 200 μL of fresh LB. The lowest concentration of polymyxin B that prevented the appearance of visible growth within the inoculation area after 24 hours at 37°C was defined as the MBIC.
Ultrasound experiments
Ultrasound was applied using a gene ultrasonic transfer machine (UGT 1025; CQMU, Chongqing, China). The frequency of this unfocused ultrasonic transducer was 1.0 MHz, and the acoustic intensity was set as a continuous ultrasonic intensity of 3 W/cm2. The duration of the intervention was 5 minutes. During the experiment, we placed an ultrasonic probe underneath the bottom of a well using coupling gel.
In vitro antimicrobial activity
Biofilms were cultivated on 24- or 96-well polystyrene microtiter plates, and planktonic bacteria were removed by washing the plates with sterile water. Then, the biofilms were randomly divided into eight treatment groups and treated as follows: biofilm control (BF control), ultrasound (US), chitosan-modified liposome (CL), USMB, polymyxin B (PMB), CLP, USMB and polymyxin B (USMB + PMB), and USMB and CLP (USMB + CLP); a sterile blank control group (SB control) was used as the background control. The biofilms of each group were treated with LB supplemented with various preparation solutions or the mixture of various preparation solutions and microbubbles (1:1 vol) with various concentrations of polymyxin B (2 μg/mL, 4 μg/mL, 8 μg/mL, and 16 μg/mL). The final concentration of microbubbles was 4% (v/v). After ultrasound treatment, the plates were incubated at 37°C for 24 hours. Then, crystal violet staining assays were applied to evaluate the biofilm mass, and resazurin assays were used to estimate viable counts, as described in the following section.
Crystal violet staining assay
The crystal violet staining assay used in this study was performed according to a previously described method with some modifications.25 After biofilms in 24-well plates were gently washed to remove planktonic bacteria, plates were air-dried at room temperature, and each well was stained with 1 mL of 1% crystal violet solution for 10 minutes. Then, the plates were washed with sterile distilled water for five times. The whole process was performed without light. One milliliter of 95% ethanol was added to destain the wells and 200 μL of destaining solution from each well was transferred to a 96-well plate. The destaining solution was measured by the absorbance at 570 nm using a Varioskan Flash Microplate Reader (Thermo Fisher Scientific, Waltham, MA, USA).
Resazurin assay
The resazurin assay was performed as previously described.16 After biofilms in 96-well plates were gently washed to remove planktonic bacteria, 100 μL of LB was added to each well. The LB was then mixed with 10 μL of resazurin solution (Alamar blue, Yeasen, Shanghai, China), followed by shaking and incubation at 37°C for 1 hour. The percent reduction in Alamar blue was calculated by measuring absorbances at 570 and 600 nm.
Scanning electron microscopy (SEM) analysis
AB biofilms were cultivated using 24-well plates and treated according to the different groups. Then, the AB biofilms were fixed with 2.5% glutaraldehyde for 2 hours and rinsed twice with PBS. They were dehydrated with 30%, 50%, 70%, 90%, and 100% concentrations of ethanol in series, and then replaced with 50%, 70%, 90%, and 100% concentrations of tertiary butyl alcohol in series. Finally, each sample was coated with gold by a sputter coater and imaged via SEM (S-3000N; Hitachi, Tokyo, Japan).
Statistical analysis
Statistical analysis was conducted using SPSS 19.0. One-way analysis of variance (ANOVA) was used for multiple comparisons, and the least-significant difference (LSD) test was used for comparisons between two groups. Statistical significance was determined at P<0.05.
Results
Characterization of CLPs and microbubbles
First, we prepared CLPs composed of DPPC, DSPE, and CHOL at a mass ratio of 3:1:1. The sizes of the CLPs were 225.17±17.85 nm (Figure 1A) and CLPs had positive charges of 12.64±1.44 mV, as their outer layer was coated with chitosan. The morphology of the CLPs revealed that the liposomes were regularly spherical, and the outer layer was composed of chitosan and phospholipid (Figure 1B). A modified injection method was used to prepare the CLPs, yielding an encapsulation efficiency as high as 90.31%±2.84% and a drug loading of 15.62%±1.97%. Next, we prepared microbubbles containing DPPC, DSPE, and CHOL at a mass ratio of 10:4:1. The microbubbles had an average diameter of 2.391±0.052 μm (Figure 1C) and showed negative charges of −4.32±0.43, as the surfaces were composed of phospholipid shells and the cores were filled with C3F8. As shown in Figure 1D, phospholipid-coated microbubbles were annular and uniformly dispersed without aggregation.
In vitro drug release of CLPs
The dialysis method was adopted to determine the accumulated release percentages of polymyxin B and CLPs in vitro over time (Figure 2). Polymyxin B showed sudden release and was completely released from the dialysis medium within 12 hours, while polymyxin B in CLPs was released more slowly, with nearly all of the drug released in 24 hours. These data suggest that the polymyxin B preparation of CLPs prolonged the release time of polymyxin B.
MBIC of CLPs
MBIC is defined as the lowest concentration that inhibits visible biofilm cell growth. The MBIC of CLPs on the biofilm-producing AB was 8±2 μg/mL, while that of polymyxin B alone was 32±2 μg/mL (Figure 3A). Compared with polymyxin B alone, CLPs induced a significant antibacterial effect on the biofilm-producing AB (P<0.01).
Synergistic antibacterial effects of USMBs and CLPs against biofilm-producing AB
We further explored the synergistic antibacterial effects of USMBs combined with different preparations of polymyxin B. The crystal violet staining assay was applied to evaluate biofilm mass, and the resazurin assay was used to estimate viable counts. As shown in Figure 3B and C, compared with the biofilm control, USMBs induced an inhibitory effect on both biofilm mass and viable bacteria against biofilm-producing AB (P<0.05), while ultrasound alone and chitosan-modified liposomes exerted no antibacterial effects (P>0.05). When the polymyxin B concentration of each preparation was 2 μg/mL, which is the susceptible breakpoint for polymyxin B in the Clinical and Laboratory Standards Institute (CLSI),26 the inhibitory effects of CLPs or polymyxin B alone were enhanced compared with those of USMBs (P<0.01). Furthermore, CLPs inhibited biofilm mass and viable bacteria more obviously than polymyxin B alone (P<0.01). Compared with polymyxin B or CLPs alone, USMBs combined with polymyxin B or CLPs induced significant inhibitory effects on the biofilm mass and viable bacteria (P<0.01), suggesting that USMBs enhanced the antibacterial effects of polymyxin B preparations on biofilm-producing AB. Among these treatments, USMBs combined with CLPs induced significantly greater inhibition of the biofilm-producing AB than that of USMBs combined with polymyxin B (P<0.01). There was no significant difference in the biofilm mass and viable counts of USMBs combined with CLPs and the sterile blank control (P>0.05). These results indicate that the combination of USMBs and CLPs yielded the most significant synergistic antibacterial effect, which could almost completely eliminate biofilm-producing AB.
Dose–effect relationship of antibacterial effects against biofilm-producing AB
In this section, we further illuminated the dose–effect relationship of antibacterial effects of polymyxin B or CLPs with or without USMBs. As shown in the biofilm mass (Figure 4A) and the viable count (Figure 4B) results, with increasing polymyxin B concentrations (2, 4, 8, and 16 μg/mL), the scavenging effect of each treatment on bacterial biofilms was gradually enhanced within a certain dose range. Though the antibacterial activity of polymyxin B alone showed a dose–effect relationship within the range from 2 to 16 μg/mL, there were significant differences in biofilm mass and viable counts between 16 μg/mL polymyxin B and the sterile blank control (P<0.05), suggesting that 16 μg/mL polymyxin B, which was the maximum dose in this study, could not eliminate the biofilm-producing AB. The antibacterial activity of CLPs also showed a dose–effect relationship between 2 and 16 μg/mL concentrations. Furthermore, between CLPs with 8 μg/mL polymyxin B and the sterile blank control, there was no significant difference in the biofilm mass (P>0.05), while between CLPs with 16 μg/mL polymyxin B and the sterile blank control, there was a significant difference in the viable counts (P<0.05), suggesting that CLPs even with 16 μg/mL polymyxin B could not eliminate the biofilm-producing AB. The antibacterial activity of USMBs combined with polymyxin B showed a dose–effect relationship within the range from 2 μg/mL to 4 μg/mL polymyxin B, and USMBs combined with 4 μg/mL polymyxin B could effectively remove the biofilm-producing AB (P>0.05 vs sterile blank control). However, USMBs in combination with 2 μg/mL polymyxin B could completely eliminate the biofilm-producing AB and achieved the maximum antimicrobial effects (P>0.05 vs sterile blank control).
Morphological evaluation of the biofilm
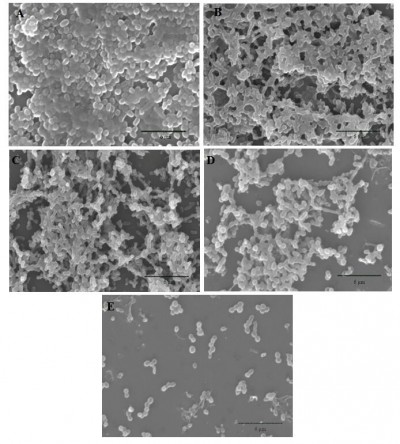
The anti-biofilm effects of different treatments against biofilm-producing AB were then confirmed by SEM analysis. The results of the biofilm control (Figure 5A) showed that AB adhered to the coverslip surface to form dense biofilms, which were rod shaped with a cluster distribution, closely linked with the extracellular matrix. USMBs (Figure 5B) had sparser biofilms than the biofilm control. Although the density was sparser than that of USMBs, most of the biofilms were still present in the polymyxin B group (Figure 5C). Compared with those of the 2 μg/mL polymyxin B group, the biofilms of the CLPs with 2 μg/mL polymyxin B group were obviously sparser, and some planktonic bacteria were present (Figure 5D). However, USMBs combined with CLPs showed the greatest anti-biofilm effects. A few scattered bacteria without biofilm structure were observed, as shown in Figure 5E.
Discussion
AB is one of the most serious opportunistic pathogens in clinical settings, which can largely be attributed to the following characteristics. First, AB has such a strong adhesion capacity that it can easily adhere to living or non-living surfaces and form biofilms both in vitro and in vivo.27,28 The formation of biofilms makes it difficult for antibacterial drugs to act directly on bacteria. Furthermore, biofilm bacteria have slower growth rates than planktonic bacteria, resulting in slower responses to antibiotics and increased drug resistance, as there is a lack of oxygen and nutrients in biofilms.4 Next, AB induces a poor inflammatory response in human cells.29 The effective treatment of MDR, XDR, and PDR AB remains a great medical challenge. In this study, we prepared CLPs and microbubbles and then explored the synergistic antibacterial effects of the combination of USMBs and CLPs against biofilm-producing AB.
Compared with liposomes, besides improving the stability of preparations, chitosan-modified liposomes might have the following advantages. First, the surface charge of liposomes is generally neutral or slightly negative, which is unfavorable for the electrostatic interaction with membranes.9 However, the positive charge of chitosan could facilitate electrostatic cross-linking with bacterial membranes and drug delivery into cells, thus enhancing antibacterial activity.23 Second, the hydrophilicity of chitosan may prolong the cycle time of drugs encapsulated in liposomes and enhance the drugs’ interactions with bacteria. Third, chitosan has the pharmacological capacity to promote wound healing and prevent scarring.30 To increase the antibacterial effectiveness of polymyxin B and reduce its toxicity, we prepared CLPs that were 225.17±17.85 nm in size and carried a positive charge of 12.64±1.44 mV (Figure 1A and B). These CLPs, which had higher encapsulation efficiency and drug loading, could have a sustained release effect, as shown in Figure 2. Next, we discovered that the MBIC of CLPs on the biofilm-producing AB was 8±2 μg/mL (Figure 3A), but the CLSI susceptible breakpoint for polymyxin B was ≤2 μg/mL, suggesting that CLPs could still not effectively eliminate the polymyxin B-resistant biofilm-producing AB, though it had four times more anti-biofilm efficacy than polymyxin B. As the study assumed, CLPs could significantly enhance the anti-biofilm effects of polymyxin B (Figure 3B and C). These results were reconfirmed by morphological assays (Figure 5C and D).
In this study, ultrasound alone could not inhibit biofilm bacteria (Figure 3B and C). However, USMBs can obviously inhibit biofilm-producing AB (Figure 3B and C). The morphological analysis via SEM confirmed the inhibitory effect of USMBs on biofilms (Figure 5B). Previous reports indicated that high-intensity ultrasound could induce cavitation to cause tiny air bubbles in liquid culture medium to expand and break, and thus opening temporary channels in biofilms. However, when no antibiotics were present, these temporary channels only promoted oxygen and nutrient penetration into biofilms and bacterial metabolite discharge.31 However, in our study, microbubbles filled with C3F8 were large, stable, and present in high concentrations, and microbubbles were affected by high-intensity ultrasound at 3 W/cm2. All of these parameters were enough to make the microbubbles instantly cavitate and release energy, causing the formation of temporary channels in the biofilms. As a result, the planktonic bacteria were freed from biofilms, and the continuous ultrasound disintegrated these planktonic bacteria.22,32,33 However, USMBs had the less anti-biofilm effectivity than 2 μg/mL of polymyxin B (Figure 3B and C). Moreover, as the study assumed, CLPs could significantly enhance the anti-biofilm effects of polymyxin B (Figure 3B and C). These results were reconfirmed via morphological assay (Figure 5C and D).
Though USMBs combined with 2 μg/mL of polymyxin B had significant synergistic inhibitory effects on biofilm-producing AB, this combination could still not completely inhibit biofilm-producing AB (Figures 3B and C and 4A and B), while USMBs combined with CLPs containing 2 μg/mL of polymyxin B could almost eliminate AB (Figures 3B and C and 4A and B), as confirmed by the morphological assay of USMBs combined with CLPs (Figure 5E). The possible mechanisms of these significant synergistic anti-biofilm effects might be the formation of temporary channels caused by cavitation, which induced CLPs penetration into biofilms and the disintegration of planktonic bacteria dissociated from biofilms. In addition, this study revealed that polymyxin B or CLPs alone and in combination with USMBs had obvious dose–effect relationships, suggesting that each treatment specifically inhibited biofilm-producing AB (Figure 4A and B).
Conclusion
In this research, we successfully prepared USMBs and CLPs, which had a more significant antibacterial effect on biofilm-forming AB than polymyxin B alone. Experiments in vitro indicate that the synergistic antibacterial effect of USMBs in combination with CLPs containing as little as 2 μg/mL of polymyxin B is sufficient to almost eliminate drug-resistant biofilm-producing AB. In subsequent studies, we will further verify the biological toxicity of CLPs, and develop AB biofilm lung infection animal models or SSTI animal models to further evaluate whether USMBs combined with CLPs can enhance the antibacterial effect of polymyxin B and reduce its toxicity for treating AB infection in systemic or topical antibacterial treatment conditions.
Acknowledgment
This work was supported by a grant from the Health Commission of Chongqing, China (2016ZDXM010).
Disclosure
The authors report no conflicts of interest in this work.
References
Krzyściak P, Chmielarczyk A, Pobiega M, Romaniszyn D, Wójkowska-Mach J. Acinetobacter baumannii isolated from hospital-acquired infection: biofilm production and drug susceptibility. APMIS. 2017;125(11):1017–1026. | ||
Flemming H-C, Wingender J. The biofilm matrix. Nat Rev Microbiol. 2010;8(9):623–633. | ||
Greene C, Vadlamudi G, Newton D, Foxman B, Xi C. The influence of biofilm formation and multidrug resistance on environmental survival of clinical and environmental isolates of Acinetobacter baumannii. Am J Infect Control. 2016;44(5):e65–e71. | ||
Sun F, Qu F, Ling Y, et al. Biofilm-associated infections: Antibiotic resistance and novel therapeutic strategies. Future Microbiol. 2013;8(7):877–886. | ||
Moradi J, Hashemi FB, Bahador A. Antibiotic resistance of Acinetobacter baumannii in Iran: a systemic review of the published literature. Osong Public Health Res Perspect. 2015;6(2):79–86. | ||
Inchai J, Liwsrisakun C, Theerakittikul T, et al. Risk factors of multidrug-resistant, extensively drug-resistant and pandrug-resistant Acinetobacter baumannii ventilator-associated pneumonia in a medical intensive care unit of university hospital in Thailand. J Infect Chemother. 2015;21(8):570–574. | ||
Gupta M, Lakhina K, Kamath A, et al. Colistin-resistant Acinetobacter baumannii ventilator-associated pneumonia in a tertiary care hospital: an evolving threat. J Hosp Infect. 2016;94(1):72–73. | ||
Nation RL, Li J, Cars O, et al. Framework for optimisation of the clinical use of colistin and polymyxin B: the Prato polymyxin consensus. Lancet Infect Dis. 2015;15(2):225–234. | ||
Drulis-Kawa Z, Dorotkiewicz-Jach A. Liposomes as delivery systems for antibiotics. Int J Pharm. 2010;387(1–2):187–198. | ||
Forier K, Raemdonck K, De Smedt SC, et al. Lipid and polymer nanoparticles for drug delivery to bacterial biofilms. J Control Release. 2014;190:607–623. | ||
Sharma A, Sharma US. Liposomes in drug delivery: progress and limitations. Int J Pharm. 1997;154(2):123–140. | ||
Yang Z, Liu J, Gao J, Chen S, Huang G. Chitosan coated vancomycin hydrochloride liposomes: characterizations and evaluation. Int J Pharm. 2015;495(1):508–515. | ||
Manconi M, Manca ML, Valenti D, et al. Chitosan and hyaluronan coated liposomes for pulmonary administration of curcumin. Int J Pharm. 2017;525(1):203–210. | ||
Zhu C, He N, Cheng T, et al. Ultrasound-targeted microbubble destruction enhances human β-defensin 3 activity against antibiotic-resistant Staphylococcus biofilms. Inflammation. 2013;36(5):983–996. | ||
Liao A-H, Hung C-R, Chen H-K, Chiang C-P. Ultrasound-Mediated EGF-Coated-Microbubble cavitation in dressings for wound-healing applications. Sci Rep. 2018;8(1):8327. | ||
Guo H, Wang Z, du Q, et al. Stimulated phase-shift acoustic nanodroplets enhance vancomycin efficacy against methicillin-resistant Staphylococcus aureus biofilms. Int J Nanomedicine. 2017;12:4679–4690. | ||
Li S, Zhu C, Fang S, et al. Ultrasound microbubbles enhance human β-defensin 3 against biofilms. J Surg Res. 2015;199(2):458–469. | ||
Hu J, Zhang N, Li L, et al. The synergistic bactericidal effect of vancomycin on UTMD treated biofilm involves damage to bacterial cells and enhancement of metabolic activities. Sci Rep. 2018;8(1):192. | ||
Ohl CD, Arora M, Ikink R, et al. Sonoporation from jetting cavitation bubbles. Biophysical Journal. 2006;91(11):4285–4295. | ||
Dong Y, Chen S, Wang Z, Peng N, Yu J. Synergy of ultrasound microbubbles and vancomycin against Staphylococcus epidermidis biofilm. J Antimicrob Chemother. 2013;68(4):816–826. | ||
Runyan CM, Carmen JC, Beckstead BL, et al. Low-frequency ultrasound increases outer membrane permeability of Pseudomonas aeruginosa. J Gen Appl Microbiol. 2006;52(5):295–301. | ||
He N, Hu J, Liu H, et al. Enhancement of vancomycin activity against biofilms by using ultrasound-targeted microbubble destruction. Antimicrob Agents Chemother. 2011;55(11):5331–5337. | ||
Wang M, Zhao T, Liu Y, et al. Ursolic acid liposomes with chitosan modification: promising antitumor drug delivery and efficacy. Mat Sci Eng C. 2017;71:1231–1240. | ||
Alipour M, Halwani M, Omri A, Suntres ZE. Antimicrobial effectiveness of liposomal polymyxin B against resistant gram-negative bacterial strains. Int J Pharm. 2008;355(1–2):293–298. | ||
Djordjevic D, Wiedmann M, Mclandsborough LA. Microtiter plate assay for assessment of Listeria monocytogenes biofilm formation. Appl Environ Microbiol. 2002;68(6):2950–2958. | ||
Ellington MJ, Ekelund O, Aarestrup FM, et al. The role of whole genome sequencing in antimicrobial susceptibility testing of Bacteria: report from the EUCAST Subcommittee. Clin Microbiol Infect. 2017;23(1):2–22. | ||
Sivaranjani M, Srinivasan R, Aravindraja C, Karutha Pandian S, Veera Ravi A. Inhibitory effect of α-mangostin on Acinetobacter baumannii biofilms-an in vitro study. Biofouling. 2018;34(5):579–593. | ||
Pakharukova N, Tuittila M, Paavilainen S, et al. Structural basis for Acinetobacter baumannii biofilm formation. Proc Natl Acad Sci U S A. 2018;115(21):5558–5563. | ||
de Breij A, Dijkshoorn L, Lagendijk E, et al. Do biofilm formation and interactions with human cells explain the clinical success of Acinetobacter baumannii? PLoS One. 2010;5(5):e10732. | ||
Thakur K, Sharma G, Singh B, et al. Chitosan-tailored lipidic nanoconstructs of fusidic acid as promising vehicle for wound infections: an explorative study. Int J Biol Macromol. 2018;115:1012–1025. | ||
Walters MC, Roe F, Bugnicourt A, Franklin MJ, Stewart PS. Contributions of antibiotic penetration, oxygen limitation, and low metabolic activity to tolerance of Pseudomonas aeruginosa biofilms to ciprofloxacin and tobramycin. Antimicrob Agents Chemother. 2003;47(1):317–323. | ||
Dong Y, Xu Y, Li P, et al. Antibiofilm effect of ultrasound combined with microbubbles against Staphylococcus epidermidis biofilm. Int J Med Microbiol. 2017;307(6):321–328. | ||
Hernot S, Klibanov AL. Microbubbles in ultrasound-triggered drug and gene delivery. Adv Drug Deliv Rev. 2008;60(10):1153–1166. |
Supplementary material
  | Table S1 MIC values of antibacterial agents against AB W1340 |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.





