Back to Journals » Infection and Drug Resistance » Volume 14
Surgical Antibiotic Prophylaxis in an Era of Antibiotic Resistance: Common Resistant Bacteria and Wider Considerations for Practice
Authors Menz BD , Charani E, Gordon DL , Leather AJM, Moonesinghe SR, Phillips CJ
Received 27 September 2021
Accepted for publication 19 November 2021
Published 7 December 2021 Volume 2021:14 Pages 5235—5252
DOI https://doi.org/10.2147/IDR.S319780
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Bradley D Menz,1 Esmita Charani,2,3 David L Gordon,4,5 Andrew JM Leather,6 S Ramani Moonesinghe,7,8 Cameron J Phillips1,4,9
1SA Pharmacy, Flinders Medical Centre, Southern Adelaide Local Health Network, Adelaide, South Australia, Australia; 2Division of Infectious Diseases and HIV Medicine, Department of Medicine, Groote Schuur Hospital, University of Cape Town, Cape Town, South Africa; 3National Institute for Health Research, Health Protection Research Unit in Healthcare Associated Infections and Antimicrobial Resistance, Department of Medicine, Imperial College London, London, UK; 4Flinders Health & Medical Research Institute, College of Medicine and Public Health, Flinders University, Adelaide, South Australia, Australia; 5Division of Medicine, Flinders Medical Centre, Southern Adelaide Local Health Network, Adelaide, South Australia, Australia; 6Centre for Global Health and Health Partnerships, School of Population Health and Environmental Science, Kings College London, London, UK; 7Centre for Perioperative Medicine, UCL Division of Surgery and Interventional Science, London, UK; 8UCL Hospitals NIHR Biomedical Research Centre, London, UK; 9Clinical and Health Sciences, University of South Australia, Adelaide, South Australia, Australia
Correspondence: Cameron J Phillips
College of Medicine and Public Health, Flinders University, Adelaide, South Australia, 5001, Australia
Tel +61 8 8204 4400
Email [email protected]
Abstract: The increasing incidence of antimicrobial resistance (AMR) presents a global crisis to healthcare, with longstanding antimicrobial agents becoming less effective at treating and preventing infection. In the surgical setting, antibiotic prophylaxis has long been established as routine standard of care to prevent surgical site infection (SSI), which remains one of the most common hospital-acquired infections. The growing incidence of AMR increases the risk of SSI complicated with resistant bacteria, resulting in poorer surgical outcomes (prolonged hospitalisation, extended durations of antibiotic therapy, higher rates of surgical revision and mortality). Despite these increasing challenges, more data are required on approaches at the institutional and patient level to optimise surgical antibiotic prophylaxis in the era of antibiotic resistance (AR). This review provides an overview of the common resistant bacteria encountered in the surgical setting and covers wider considerations for practice to optimise surgical antibiotic prophylaxis in the perioperative setting.
Keywords: antibiotic resistance, perioperative care, surgical antibiotic prophylaxis, surgical site infection, antimicrobial stewardship
Introduction
Antimicrobial resistance (AMR) presents a modern global crisis. The incidence of AMR is rapidly increasing and infection-related mortality is expected to exceed 10 million cases per year by 2050.1 Over the past decade, the emergence of AMR across fungal, viral, bacterial, and protozoal infections is increasingly reported.2 Resistance has been highlighted in recent times to various microbes, including fungal infection with Candida auris, which is showing growing resistance to many first-line anti-fungal treatments.3 AMR is set to exceed current annual cancer-related deaths world-wide by 2050.1 While the emergence of AMR occurs across the microbiome, bacterial resistance poses the greatest threat with longstanding antibiotics becoming less effective at treating and preventing bacterial infection, resulting in poorer patient and surgical outcomes.4,5
Globally, multiple factors contribute to increasing prevalence of antibiotic-resistant organisms, including social determinants, economic factors, provision and governance of healthcare, and environmental factors, all of which impact on both humans and animals.6,7 Additionally, inappropriate antibiotic use is becoming an increasing concern as a major contributor to the emergence of AMR.8,9 The US Centers for Disease Control and Prevention (CDC-US) reports a concerning increase in the prevalence of AMR, with at least 2.8 million people suffering from a drug-resistant infection each year in the US alone, and 35,000 of these subsequently dying.10 Similarly, throughout Europe, it is estimated that approximately 33,000 patients die each year due to drug-resistant infections, of which more than half are healthcare-acquired.11 The actual world-wide prevalence of people affected with drug-resistant bacteria is likely to be much larger, as data drawn from many low- and middle-income countries are scarce.12
In the surgical setting, antibiotic resistance (AR) leads to prolonged hospitalisation, extended durations of antibiotic therapy, higher rates of mortality, increased need for surgical revision, and requirement for novel antibiotics which may have increased toxicity.13 Across the United States, Europe and Australia, expenditure on AMR and its sequelae is expected to cost approximately $USD 3.5 billion annually.14 Longer term, the cumulative global expenditure between now and 2050 on AMR is expected to exceed USD$100 trillion.1 Despite our advances and increasing knowledge on this topic, AR remains a critical consideration for the clinician and an important barrier for successfully preventing surgical site infection (SSI).9 SSIs are one of the most prevalent hospital-acquired infections, and AR poses significant risks in surgery due to commonplace antibiotics having limited or no effect against some resistant organisms.15
This review aims to provide insight on common resistant bacteria, which pose a threat in surgery and any ensuring SSI that may develop. We discuss common methods to optimise current surgical antibiotic prophylaxis (SAP) and highlight broader considerations for mitigating the risk of SSI in the setting of AR.
The Role of Surgical Antibiotic Prophylaxis
SSI is defined as occurring in the superficial, deep or organ space around the surgical site within 30 days of the surgical procedure, or within 1 year of implants in-situ.16 In attempts to classify risks of developing SSI, factors including patient age, sex, surgery type and duration, American Society of Anesthesiologists (ASA) grade, and assessment of wound contamination score are routinely assessed.17 Wound contamination assessment is undertaken prior to and during surgery. Wounds are classified by the CDC-US as clean, clean-contaminated, contaminated and dirty/infected.18 The inclusion of the ASA grade acknowledges the potential impact of patient functional capacity (fitness) and long-term conditions as contributing factors to infection risk.17 While providing insight to infection risk, guidance is lacking on these classifications regarding how to assess, prevent and manage patients with an increased risk of carrying or acquiring antibiotic-resistant organisms and ultimately SSI.
The rationale of SAP is to deliver adequate antibiotic exposure throughout the surgical site to cover the organisms commonly associated with SSI for a procedure (Table 1).19 The pooled rate of SSI across all surgeries when stratified by wound class is approximately 1.3–2.9% for clean, 2.4–7.7% for clean-contaminated, 6.4–15.2% for contaminated and 7.1–40% for dirty.20 In many clean, clean-contaminated, and contaminated procedures, SAP is employed as a routine standard of care demonstrating good safety and efficacy. Dirty procedures are considered as established infections and thus, a targeted treatment approach is generally indicated. An international multicentre study of SSIs post gastrointestinal surgery provides some confronting results when stratified by country income status using the United Nations Human Development Index (HDI). In this prospective study, cohort analysis of 610 patients that had both an SSI and microbiology culture result, 132 (21.6%) acquired an infection with an organism resistant to the SAP administered. Low-HDI countries reported resistant infections in 46/128 (36%) of patients compared to 37/187 (20%) in middle-HDI countries and 49/295 (16.6%) in high-HDI countries.21
 |
Table 1 Examples of Bacteria Implicated in Surgical Site Infection Categorised by Surgical Procedure |
AR is a threat across both primary healthcare (general/family practice, residential aged care facilities), secondary and tertiary care (hospitalisations). However, AR remains especially problematic in areas with high usage of antibiotics (intensive and critical care, and perioperative care),22–24 and in lower HDI country contexts.21 Three common organisms associated with resistance include Staphylococcus aureus in approximately 50% of the cases, followed by Escherichia coli (17%), and Pseudomonas aeruginosa (10%). Infection with resistant Klebsiella pneumoniae, Proteus mirabilis, Enterobacter aerogenes, and non-fermenting Gram-negative bacterial infections also regularly occur.23
Antibiotic selection for SAP is dependent on local resistance patterns and institutional protocols, though most SAP regimens are selected by their spectrum of antibiotic activity in relation to the procedure performed. This assumes that local resistance patterns are known, and institutional protocols are in place and adhered to.
Narrower spectrum first-generation cephalosporins (ie, cefazolin) are common antibiotics utilised for SAP. Cephalosporins are widely used due to their spectrum of activity (on commensal skin flora, and some Gram-negative bacteria), favourable safety profile, and extensive experience in SAP.19 Additional coverage for Gram-negative bacteria and anaerobes is occasionally utilised, often with aminoglycosides (gentamicin) for Gram-negative bacteria and nitroimidazole agents (eg metronidazole or tinidazole) for anaerobes.8 Standard SAP is often hindered by the presence of β-lactam allergy, whereby immediate hypersensitivity to penicillins or cephalosporins necessitates use of alternate classes of antibiotics such as lincosamides (clindamycin, lincomycin) or glycopeptides (vancomycin, teicoplanin) for Gram-positive coverage.25 Moreover, the routine use of vancomycin in place of cefazolin is discouraged in the absence of methicillin-resistant S. aureus (MRSA) and/or cephalosporin allergy due to reduced efficacy and the potential to drive AR.26 Currently, there is limited guidance on selecting appropriate SAP in patients colonised with antibiotic-resistant bacteria, mostly due to an absence of established clinical trials and published data. A targeted approach using alternative antibiotics based on spectrum of activity in discussion with infectious diseases/medical microbiology may be considered if the resistant bacteria pose a significant risk of SSI (Table 2). Clinician discretion is imperative as unnecessary use of broader spectrum agents may exacerbate local resistance profiles.
 |
Table 2 Common Agents in Surgical Antibiotic Prophylaxis and Possible Alternatives for Consideration |
Patient-Specific Risk Factors for SSI
Patient-specific risk factors such as older age, obesity, active smoking status, medical comorbidities, ASA-grade and use of immunosuppressive drugs can influence the risk of SSI and postoperative mortality.27,28 In low-income and middle-income countries, risk of SSI and postoperative mortality is greater than high-income countries.29
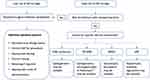
In the perioperative period, factors influencing SSI risk include availability, selection of antibiotic, timing and dose of antibiotic agent, and appropriate antiseptic preparation of the skin.25,28,30 During surgery, physiological factors affecting the risk of SSI include the presence of hyperglycaemia, trauma, shock, requirement for blood transfusion, hypoxia and hypothermia.28 In addition, some patients may be known or at risk of carriage of multidrug-resistant organisms (MROs), which preclude the use of first-line SAP agents. Risk factors to consider include recent antibiotic usage, travel or residence in jurisdictions with high rates of AR, known or prior colonisation with MROs, or exposure to health facilities (intensive care units, hospitals/care facilities) with high rates of MROs.31 Collectively, there are many steps in the patient’s surgical journey that can influence SSI risk (Figure 1). Structural measures, for example, the use of laminar airflow in orthopaedic operating theatres have been implemented in some high-income settings to support reduction in SSI, although their effectiveness has been questioned.32
Gram-Positive Antibiotic Resistance of Concern in Surgical Antibiotic Prophylaxis
MRSA and Vancomycin-Resistant S. aureus (VRSA)
S. aureus is an important organism in the human microbiome, remaining the most common organism associated with cutaneous infection.33 It has widespread presence as a commensal organism on the skin, skin-folds, and external nares of the nose.34 In the perioperative setting, S. aureus remains the most common organism associated with SSI, with a majority of S. aureus isolates sensitive to first-line narrow spectrum penicillins and cephalosporins (flucloxacillin, nafcillin, cefazolin).16,35 While the longstanding use of first-line penicillins and cephalosporins is routinely effective in preventing these infections, the emergence of resistance to β-lactams is challenging for SAP selection.
The emergence of MRSA dates back to the mid-20th century where isolates of S. aureus showed alterations to penicillin binding protein 2a (PBP2a).36 Vancomycin overcomes this resistance mechanism by targeting an alternate pathway via inhibition of S. aureus peptidoglycan synthesis.37 Nowadays, the use of vancomycin is integrated into SAP practice for patients previously colonised with MRSA, at high-risk of MRSA carriage or exposure to institutions with high prevalence of MRSA.26
The incidence of S. aureus resistance is increasing throughout the world, with the World Health Organization (WHO) Global Report on AMR and Surveillance identifying 20% of S. aureus isolates worldwide as MRSA, with some regions reporting rates ≥80%.38 In the early 2000s, there was the emergence of vancomycin-resistant S. aureus (VRSA), which is recognised as a concern and high priority by the CDC-US and WHO.39–41 While VRSA has been documented across the globe, the absolute rates of infection remain low and there are minimal reports of transmissions across patients, particularly in the healthcare setting.39,42,43 Despite small incidences, reports of infection since 2010 have doubled, with the highest increases seen in the United States and throughout Asia.44 Surgical recommendations, management and data remain isolated to a series of case reports of VRSA requiring antibiotic treatment with daptomycin, linezolid, ceftaroline or tigecycline.42,45
Vancomycin-Resistant Enterococcus (VRE)
Many of the approximately 12 Enterococcus spp. can harbor vancomycin resistance. Patients can be colonised with one or multiple species, including E. faecalis, E. faecium, E. gallinarum, E. casseliflavus, E. avium, and E. mundtii.46 Clinically, vancomycin resistance is most important in E. faecium and E. faecalis. A prevalence study in the US consisting of over 24,179 patients with hospital-acquired bacteraemia, approximately 9% were Enterococcus spp. with two-thirds of E. faecium isolates displaying resistance to vancomycin.47 While there are many phenotypes of resistance within the Enterococcus spp., VRE often occurs in E. faecalis and E. faecium as phenotypes VanA or VanB, resulting in target binding-site alteration.48 Both are inducible on administration of glycopeptides, with VanA typically conferring resistance to vancomycin and teicoplanin, and VanB conferring resistance to vancomycin alone.46,48
The CDC-US reported down-trending VRE infections in the US between 2012 and 2017 (84,000 down to 54,500 cases) as a testament to improved infection control practices and antibiotic stewardship.49 Alarmingly, in some other countries such as Germany, an increasing prevalence of SSI with VRE is observed with reports showing VRE infection increasing from 1% to 5% of all SSI between 2007 and 2016.50 Globally, it is estimated that carriage of VRE is increasing, though VRE colonisation does not always manifest to infection with VRE. Enterococcus spp. show intrinsic resistance to cephalosporins, clindamycin, penicillins and trimethoprim/sulfamethoxazole. Patients with VRE colonisation may require SAP with agents such as linezolid or daptomycin to reduce the risk of SSI, particularly in the context of abdominal surgery.
Gram-Negative Antibiotic Resistance of Concern in Surgical Antibiotic Prophylaxis
ß-lactamase and Carbapenemase-Producing Gram-Negative Bacteria
ß-lactamase production can render cephalosporin, penicillin and carbapenem type ß-lactam antibiotics ineffective.51 Carriage with resistant Enterobacterales is increasing, with common examples of ß-lactamase-producing organisms including E. coli, Klebsiella spp, and Enterobacter spp.51 In addition to Enterobacterales, P. aeruginosa is another common organism-producing ß-lactamase and is considered to be an increasing concern among SSI.52 ß-lactamases exist in molecular subclasses A, B, C and D, determined by amino acid sequence, of which each resistance profile can be grouped by phenotype and bacterial isolate.53 Three groups, defined by Bush et al, include the following: group 1 (cephalosporinases), group 2 (serine ß-lactamases) and group 3 (metallo-ß-lactamases).54 Group 1 and 2 ß-lactamases hydrolyse the ß-lactam antibiotic via serine mediated catabolism of the ß-lactamase enzyme active site, whereas group 3 metallo-ß-lactamases require catalysation by a cation – most commonly zinc 2+ at the active site.55,56 Two common terms used frequently throughout the literature to illustrate carbapenem resistance are carbapenem-resistant Enterobacterales (CRE) and a subset of this in which the mechanism of resistance is mediated by carbapenemase production (carbapenemase-producing Enterobacterales (CPE). CPE remains the most concerning mechanism driving carbapenem resistance.57
Group 1 cephalosporinases comprise molecular subclass C and a common representative enzyme (AmpC).58 AmpC-mediated resistance occurs by hydrolysis of narrow spectrum cephalosporins (cefazolin, cefalexin), though increased and repeated exposure to cephalosporins can induce AmpC production – leading to resistance to broader-spectrum cephalosporins ceftriaxone, ceftazidime and cefotaxime.58 This is concerning as common ß-lactamase inhibitors such as clavulanic acid and tazobactam are generally ineffective at overcoming such resistance mechanisms and the presence of other resistance mechanisms such as extended-spectrum ß-lactamases (ESBL) can co-exist.59,60 Recent advances have seen the advent of the ß-lactamase inhibitor avibactam, which is used in combination with broad spectrum cephalosporin agent ceftazidime in attempts to provide efficacy for AmpC-mediated resistance.61
Group 2 serine ß-lactamases consist of molecular subclasses A and D, with common representative enzymes and their many respective enzymes variants (Class A: PC, TEM, SHV, CepA, KPC; and, Class D: OXA).54 Following their characterisation was the development of β-lactamase inhibitors clavulanic acid and tazobactam, though resistance has also emerged to these agents.62 ß-lactamases within subgroup 2be and group 2d are ESBL and are associated with the greatest emergence of resistance with an approximately 8-fold increase in identified variants over a 20-year period.54 In the surgical setting, ESBL-producing Enterobacterales have shown a prevalence of approximately 15%, which is expected to grow sharply over the coming years.63 ESBLs show increased resistance to broader spectrum penicillins and cephalosporins. The reader is referred to Bush et al for a comprehensive overview on the many enzyme subclasses and complexities of ESBL-producing bacteria.54
Carbapenemase produced in K. pneumoniae (KPC) is an important class A enzyme, first identified in the US in 2001; shortly after multiple variants (KPC-2, to KPC-11) were discovered worldwide.64 KPC has emerged as one of the most common carbapenem resistance mechanisms across the US, China, South America and Greece.65–67 While KPC is most common in K. pneumoniae, other organisms harbouring KPC include both Enterobacterales and non-Enterobacterales (P. aeruginosa, Acinetobacter spp.).65 The presence of KPC-producing bacteria hinders the efficacy of antibiotics used for SAP, as routine cephalosporins are precluded by class-A ß-lactamase hydrolysis.68 SAP in such instances may require sensitivity-guided/-targeted prophylaxis or combinations of aminoglycosides, polymyxins, tigecycline, fosfomycin or ceftazidime/avibactam.69
Group 3, Class B metallo-ß-lactamases (MBL), differ functionally and structurally to their serine counterparts by the presence of zinc 2+ at the enzyme active site.70 Common representative enzymes and variants (IMP, VIM, NDM) confer resistance to carbapenems, oxyimino-β-lactams, ß-lactamase inhibitors and monobactams.71 MBL-mediated carbapenem resistance is the commonest mechanism across Europe, Scandinavia and India.66
Fluroquinolone-Resistant Gram-Negative Bacteria (FR-GNB)
Fluroquinolones (quinolones) are effective first-line SAP agents in urological procedures such as transrectal prostatic biopsy or for patients with confirmed β-lactam allergy (IgE mediated) to first-line agents.72,73 Quinolones (eg, oral ciprofloxacin) are also effective against P. aeruginosa.19
Traditionally, quinolones are used predominantly for the prevention and treatment of Gram-negative organisms, via their effect on inhibition of DNA gyrase. Additionally, quinolones also target topoisomerase IV, inhibiting the relaxation of coiled DNA.74 Resistance occurs predominantly due to mutations of DNA gyrase subunits GyrA/GyrB and/or topoisomerase IV subunit ParC/ParE.75 Further, co-existence of resistance to quinolones can occur by reduced membrane permeability and/or overexpression of drug efflux pumps.76
Recent exposure to quinolones (surgical prophylaxis or treatment) is a known risk factor for acquired quinolone resistance.77 A meta-analysis by Zhu et al highlights the significant increase of resistance of E. coli in those with recent fluoroquinolone use (OR 7.67; 95% CI 4.79–12.26).78 In addition to acquired quinolone resistance, transfer of plasmids containing resistance genes qnrA, qnrB, qnrS are of growing concern within the hospital environment.79 QnrA, qnrB and qnrS prevent quinolone effects at DNA gyrase/topoisomerase IV and are hypothesised to contribute and potentiate quinolone resistance, though their effect is not entirely understood.79,80 Resistance to quinolones is increasing globally, threatening many elements of modern medicine including SAP.81
Optimising Current Practice Across the Patient Pathway
Revisiting Patient Allergy Status
Patient allergy status is an important consideration when it comes to selecting a suitable agent for SAP. Inappropriate or spurious patient labelling of antibiotic allergies is problematic. A recent multicentre UK study of hospitalised patients with common infections found that two-thirds of those patients with documented penicillin allergy had no details recorded on their purported allergy.82 Similarly, in the perioperative setting, a UK study of over 21,000 elective surgical patients found that 27% of those with an allergy label were likely suitable for a direct provocation test.83 A US study by Blumenthal et al of 8385 patients undergoing arthroplasty, gastrointestinal surgery, cardiovascular surgery, and hysterectomy found those listed with a penicillin allergy to have a 50% increased SSI risk which was largely due to patients receiving second-line agents for SAP.84 In addition, the labelling of penicillin allergy can lead to increased length of hospital stay, admission into intensive care units, increased hospital readmission rates and the promotion of antibiotic resistance.83
A US study with over 50,000 patients demonstrated that patients labelled as penicillin allergic received significantly more prophylaxis or treatment with fluoroquinolones, clindamycin and vancomycin compared with control subjects.85 Further, those labelled with penicillin allergy had 23.4% more cases of C. difficile infection, 14.1% more MRSA infection and 30.1% more VRE infection.85 Surgical specific examples are illustrated in arthroplasty and, head and neck surgery. In arthroplasty, the use of vancomycin as a sole agent in a cohort of patients (where 54% of the patients had a penicillin allergy) was associated with more SSI than prophylaxis with cefazolin in those without penicillin allergy.86 Similarly, when patients labelled penicillin allergic undergoing head and neck surgery received clindamycin as an alternative to cephalosporins, SSIs increased four-fold.87
While penicillin allergy labelling is associated with increased SSI, penicillin and sulphonamide antibiotics remain the most reported causes of drug-induced hypersensitivity and anaphylaxis; surgeons and anaesthetists should thus remain vigilant in ensuring true penicillin allergy is managed appropriately.88
Steps to appropriately manage patients with suspected antibiotic allergy stem from institutional protocols development in accordance with appropriate antimicrobial stewardship and hospital resistance patterns. Consideration should be given where possible to implementation of prescreening “antibiotic allergy testing” patients in surgical preadmission clinics.83 Development of a protocol for taking an accurate patient penicillin/cephalosporin allergy history was undertaken by Blumenthal et al, whereby patients deemed low risk of type 1 hypersensitivity reaction were administered a test dose of the antibiotic in question under medical supervision. This approach narrowed the spectrum of SAP used (reducing the use of vancomycin, aztreonam, fluoroquinolones, and aminoglycosides), with no reported increase in adverse drug reactions (type and/or severity).89 In a US study, preoperative penicillin allergy skin testing was performed in patients self-reporting penicillin allergy (1204 of 11,819 screened patients). In those patients with reported penicillin allergy, only 4% demonstrated a true reaction to penicillin, resulting in a significant reduction of empirical vancomycin use from 30% to 16%.90
Pharmacokinetic Alteration – Obesity
Patient risk factors for altered pharmacokinetics are frequently seen in surgical patients and can lead to variable requirements for SAP dosing. Obesity is increasing worldwide and the number of hospitalised patients with obesity is expected to grow in coming years.91 A cross-sectional study in the UK revealed approximately one in five inpatients were obese and those receiving antibiotics typically required more complicated dosing than non-obese patients.92 Obesity influences the pharmacokinetic disposition of common antibiotics used in SAP by alterations in volume of distribution, hepatic metabolism, renal clearance and protein binding, resulting in less predictable antibiotic exposure.93,94 In attempts to ensure adequate SAP dosage that provides sufficient tissue concentrations, minimise toxicity and prevent the development of antibiotic resistance, clinicians may need to tailor dosing to ideal body weight, lean body weight, or adjusted body weight recommendations.93 Examples of antibiotics which may require dose alteration in obesity include cefazolin, ceftriaxone, clindamycin, gentamicin, aztreonam, daptomycin and vancomycin.95 However, it is important to note there is a lack of well-established and robust data on dosing antibiotics in obesity, as many studies have small sample sizes, are subject to heterogenous data and often provide conflicting results.96 Appropriate dose alteration should be balanced, comparing the risk of over-exposure (possible adverse events) with that of under dosing (risk of SSI and promotion of AR).
Surgical Antibiotic Prophylaxis Redosing
Timing of antibiotic dosing is an important consideration during the perioperative phase, although extended re-dosing of an antibiotic following completion of surgery is not recommended due to increased risks of toxicity (ie, acute kidney injury and C. difficile infection), with no overall improvements in SSI prevention.97,98 The Australian National Antibiotic Prescribing Survey (NAPS) has shown that extended antibiotic exposure beyond 24 hours is one of the most common divergences from SAP guidelines, with approximately 30% of all SAP employed exceeding 24 hours.99 Intraoperative antibiotic re-dosing is sometimes employed in surgeries exceeding 4 hours. This is done to ensure serum and tissue concentrations exceed the microbial minimum inhibitory concentration (MIC) throughout the duration of surgery.100,101 Common examples include procedure durations exceeding two half-lives of the SAP utilised or in procedures where there is excessive blood loss, exceeding 1500mL. Key considerations for the clinician include factors that may affect antibiotic exposure time, such as extensive burns, concomitant medicines with drug–drug interactions or renal impairment.19
Cefazolin pharmacokinetics suggest that additional doses could be administered in surgical procedures exceeding 3 hours in patients with normal renal function.98,102 SSI rates with such procedures are reportedly reduced (6.1% to 1.3%; P = <0.01) by re-dosing cefazolin or substituting another cephalosporin with a longer half-life, such as cefotetan.98 Similarly, intraoperative re-dosing of cefazolin has been adopted in prolonged cardiac surgery, where a 16% reduction in SSI has been demonstrated.103 An important study conducted in Brazil between 2011 and 2012 revealed the average length of surgery and total time spent in the operating theatre for 8337 procedures across various surgical sub-specialities.104 Considerations and planning for possible antibiotic redosing could be achieved by reviewing usual surgery duration for longer procedures.
While not routinely re-dosed perioperatively, gentamicin dosing more than 30 minutes prior to surgical incision is generally recommended to allow for peak concentration-dependant activity.102 Gentamicin concentration upon surgery completion remains a predictable risk factor for SSI, where concentrations <0.5 mg/L were associated with up to an 80% infection rate in a very small study of 10 patients.100 Difficulties in SAP with gentamicin are highlighted during intraoperative blood loss and fluid replacement, where 2mg/kg doses of gentamicin for colorectal surgery did not achieve concentrations above the MIC of the organism in serum and/or tissue samples.105
Optimising Current Practice Across Institutional Processes
The increasing frequency of community transmission and patient colonisation with ESBL producers, CRE, VRE as well as MRSA threatens the efficacy of routine SAP and perioperative patient pathways. This calls for a major overhaul of established perioperative processes, SAP guidelines and considerations of individual patient factors. Screening patients for colonisation with MROs will have an increasing role.106,107 Antimicrobial stewardship (AMS) programs, infection prevention and control initiatives, and quality use of medicines principles (right antibiotic(s), right dose and timing of administration) will have increasing importance (Figure 2).19 AMS programs are also effective in improving SAP in low- and middle-income countries (LMICs).108 The CDC-US provides guidance on the prevention of SSI, stipulating that SAP should only be used when appropriate and should follow established guidelines.109 Despite these influential recommendations, obstacles and challenges remain at the institutional level.
Antimicrobial stewardship programs, clinical practice guidelines and protocols
A international survey of surgeons yielded 588 responses, with all considering antibiotic resistance to be an important concern and that local guidelines were of greater use than national guidelines.110 Key considerations for local guideline development are theatre workflow, organisms of concern, antibiotic selection, timing of dosage in relation commencement of surgery (knife-to-skin time), route of administration, duration of surgery and unforeseen delays in surgery time.19,111 Despite these considerations, many studies have demonstrated that even when such protocols and guidelines exist, they are often poorly adhered to.112–120 Studies investigating the implementation of AMS programs in the surgical setting have shown improved guideline adherence in several studies.121–123 Segala et al found that prior to the implementation of stewardship programs, antimicrobial prophylaxis was either not required, or appropriate but not given, occured 41% of the time and that 29% of the cases were prescribed for longer than indicated. Post-implementation data from this study showed improvement in overall antibiotic appropriateness, duration and guideline adherence (36.6% vs 57.9%; p = <0.001).124
Cohen et al performed a large study across a cohort of 22,138 patients investigating the use of appropriate prophylactic antibiotics in various low-risk procedures and concluded there was no risk of increased antibiotic resistance in their cohort.125 Such data are promising for the use of protocols and their role in preventing the emergence of resistance within the surgical setting. The successful implementation of stewardship principles and protocols is demonstrated in previous studies involving a multidisciplinary team approach consisting of medical, pharmacy and nursing staff.121,123,126 Despite successful implementation of these programs, clinicians prescribing SAP are often unaware of existing protocols and therefore adherence is adversely impacted. Strategies to overcome these issues are likely to be improved by multidisciplinary engagement in the development and implementation of protocols.127
The WHO recommend using a 19-step Surgical Safety Checklist (SSC) to capture critical tasks across three phases of the patients’ surgical journey: before induction of anaesthesia, before skin incision and prior to the patient leaving the operating room.128 A study across 8 countries following implementation of the SSC assessed complications and mortality occurring 30 days after non-cardiac surgery. SSC implementation decreased surgical complications (11% to 7%; p = <0.001), and mortality (1.5% to 0.8%; p = 0.003).129 The current WHO SSC (2009) aims to achieve SAP within 60 minutes prior to incision and assessment of patient allergies, both of which optimise appropriate antibiotic use.128 A retrospective cohort study of 772 patients undergoing various surgeries in the Netherlands investigated the impact of SSC implementation and optimisation on the timing of SAP (previously 0–60 minutes) prior to incision.130 Significantly fewer patients received SAP post-incision (decreasing from 12% to 7.1%,) while SAP use within the pre-specified SAP time-frame (30–60 minutes) prior to incision improved.130 The issue of how well the WHO SSC is implemented into practice is a major consideration in both high- and low- to middle-income countries. While the evidence for SSC is strong, the evidence to supporting implementation has been lacking, particularly in LMICs. A recent systematic review found that the SSC is used with high fidelity in LMICs and strategies covering domains of training, adapting to context, provision of interactive assistance, stakeholder engagement and clinician support may offer solutions to augment implementation.131 Encouragingly, several studies in LMICs have shown that use of the WHO SSC is possible and can be sustained.132–134 Furthermore, when scale-up of the WHO SSC underwent economic evaluation in three African countries (Benin, Cameroon and Madagascar) it was found to be very cost-effective with every USD$1 spent producing a potential return on investment of USD$9–62.135
Prescreening Patients and Decolonisation
In attempts to curb institutional transmission of antibiotic resistance within the perioperative setting, focus has shifted towards prescreening patients prior to surgery and/or hospital admission. This strategy was seen after the emergence of MRSA, whereby prescreening for MRSA by the use of nasal swabs and subsequently patient isolation, decolonisation and tailored SAP has been adopted in some institutions.136 A multi-site study conducted across a number of European countries showed that combinations of prescreening, patient isolation and decolonisation 5-days prior to clean surgery (using chlorhexidine body wash and nasal mupirocin) decreased MRSA infection by 17%.137 Further, in a study of patients undergoing cardiac surgery, targeted prophylaxis with vancomycin (± standard of care, cefazolin) in patients colonised with MRSA reduced SSI (OR 0.58; 95% CI 0.40–0.86).138
There are challenges with ß-lactamase-producing Enterobacterales, where bacterial carriage in the gastrointestinal tract limits the ability of decolonisation and is recognised in the WHO Surgical Site Infection Prevention Guidelines.139 Dubinsky-Pertzov et al screened surgical patients given routine SAP for ESBL colonisation prior to gastrointestinal surgery, and found SSI rates in those colonised more than double that of non-ESBL carriers (OR 2.36; 95% CI 1.50–3.71).140
The increasing prevalence of fluoroquinolone resistance and the longstanding use for prophylaxis in transrectal prostate biopsy has received critique by the European Association of Urology, suggesting prescreening of these patients for fluoroquinolone resistance or the use of prophylactic Fosfomycin.141 Recommendations for fosfomycin in transrectal prostate biopsy are supported by a meta-analysis, which shows improved patient outcomes compared with ciprofloxacin standard of care, although data in this meta-analysis came from some countries with high rates of ciprofloxacin resistance.142 Additionally, increasing fluoroquinolone resistance has been seen in patients with P. aeruginosa SSI post endoscopic sinus surgery.143
Timing of Surgical Antibiotic Prophylaxis
The premise of appropriate timing of SAP stems from the pharmacokinetic and pharmacodynamic properties of the antibiotic. The most commonly employed SAP agents are cephalosporins (ie cefazolin) which are traditionally administered up to 60 minutes prior to surgical incision (knife-to-skin time).144 This allows appropriate time for peak concentration, distribution and penetration into the peripheral tissue and time above the MIC of the anticipated organism.144 Evidence for administration of cefazolin <120 minutes prior to incision originated from a study in 1992 consisting of 2847 patients.145 A subsequent meta-analysis of over 54,552 patients (from 14 studies) concurred with this finding.146 The timing of vancomycin and fluoroquinolone administration for SAP differs to cephalosporin due to the increased infusion time required with these agents.19 Typically, commencement of vancomycin and fluoroquinolone infusions commences 1-hour prior to surgical incision, as shortened infusions can cause adverse effects, including “red-man syndrome” with vancomycin and venous irritation with fluoroquinolones.147 While standard infusion times may vary at an institutional level, vancomycin is typically administered at a rate of 1g/hour. Similarly, ciprofloxacin is infused over 60 minutes and levofloxacin over 60 or 90 minutes for doses of ≤500mg and >500mg, respectively.148
Timing of antibiotic administration remains a challenging component of SAP and clinical practice can be divergent with key guidelines.149 Studies have shown improved timing of antibiotic administration following antibiotic stewardship initiatives in high-income countries,126,150 though mixed results have been observed from similar studies in LMICs. A study performed in Egypt found that after AMS initiatives, appropriate timing of SAP first dose improved from 6.7% to 38.7% (p = <0.01).108 A study conducted in Ethiopia found that the recorded timing of SAP administration remained poor post-implementation of an institutional SAP protocol 69.3% to 63.9% (p = 0.26).30
While various studies have investigated the impact of SAP timing, there are many nuances within the surgical environment that can hinder appropriate timing of antibiotic administration. Tan et al interviewed surgeons, anaesthetists and perioperative administrators across two large hospitals in the US and highlighted key obstacles affecting SAP: 1) timing as lower priority to the clinician, 2) inconvenience in patient management, 3) potential to disrupt workflow, 4) limited communication between the treating team and differences in role perception and clinician hierarchy.111
Team Communications, and Cultures of Practice
Research has highlighted the power dynamics that exist in surgical teams in relation to antibiotic decision-making, including an individualistic and hierarchical culture, which may limit integrated care in surgical pathways.151,152 Historically, AMS initiatives have overlooked surgical specialties, with the notion that surgical teams are more difficult to engage with.153 Where they do exist, the focus has been on SSI prevention and SAP.154 Antibiotic prescribing in hospital settings is ubiquitous and doctors of different specialties must be able to diagnose and treat infections. The issue of ownership of the antibiotic prescription or order is often not clear, particularly in surgical pathways where multiple healthcare professionals have a role to play.155 The issue of ownership is also unclear when it comes to SAP, with surgeons and anaesthetists both identified as bearing the responsibility for the selection and administration of initial and any follow-up doses.156 The issue of clinician autonomy taking precedence over policy and guidelines is an important factor, which can influence the selection of antibiotics and the duration of SAP.156,157 Studies have demonstrated that SAP is often prolonged beyond a single dose and is used as a safety net to allay the surgeons fears of SSI and post-operative complications.158 A systematic review of the effect of post-operative continuation of SAP has demonstrated no benefits over single-dose therapy.159 Therefore, we need to communicate the risks associated with prolonged antibiotic use more effectively with surgical teams, to facilitate their decision-making and their ability to understand infection-related risks associated with their surgical intervention versus the risks associated with prolonged use of antibiotics. Team communication both peri- and post-operatively also impacts the infection-related care that patients receive, including SAP, and SSI-related care.160 Research is needed to identify effective mechanisms for interdisciplinary work across the surgical pathway including within the theatre between surgeons and anaesthetists. The roles of anaesthetists in SAP need to be more clearly defined to enable a more sustainable path to optimising SAP.
Alternative Agents and Means to Approach Antibiotic Resistance
Machine learning, while still in early stages, is an evolving field for the prediction and management of patients colonised with MROs. The use of machine learning aims to predict the presence of resistance by using genomic data, assisting the clinician with early diagnosis, prevention and provide guidance on treatment options.161 Early-stage trials within ICU departments in Greece have used machine learning to review patients with antibiotic-resistant bacteria and recommend appropriate empirical antimicrobials.162 Other promising options of machine learning are the ability to utilise health registry data to identify resistance patterns and promote stewardship programs within the hospital environment.161 While not currently trialled for SAP, such systems may be appropriate to assist clinicians in the growing climate of antibiotic resistance.
The slow rate of new antimicrobial development and at times diminishing efficacy of existing antibiotics has led to the investigation of alternative options, such as bacteriophage therapy and faecal microbiota transplant (FMT). While still in their discovery phase in medicine, bacteriophages are viruses with restricted bacterial activity often resulting in bacterial cell lysis.163 To date, application and evidence of bacteriophage therapy is limited to small case studies and experimental trials. A series of patients (n=8) in Finland with multidrug-resistant bacterial infection post-cardiothoracic surgery received conventional targeted antibiotics and bacteriophages, resulting in successfully treated infections in 7/8 (87%) patients.164 Hypothesised applications of bacteriophages in the surgical setting with topical application of phage solutions to a surgical wound before closure are proposed by Brives and Pourraz.165 The growing concerns of mupirocin resistant MRSA and difficulty in eradicating mucosal Staphylococcal carriage has sparked interest in the use of bacteriophage for nasal decolonisation of MRSA; studies are currently limited to animal models but show favourable efficacy.166 The role of FMT is an emerging treatment for multidrug-resistant organisms of the gastrointestinal tract, with use increasingly common for the treatment of refractory C. difficile infection. Treatment outcomes in these patients are showing response rates in approximately 90% of the cases.167 Currently, there is limited evidence supporting gastrointestinal MRO decolonisation with antibiotic therapy, where use may potentiate resistance due to gastrointestinal rebound of resistant bacteria.168 FMT is a novel method, with growing interest in its role in gastrointestinal decolonisation of multidrug-resistant bacteria.168 A recent systematic review showed approximately 50% of the patients to have successful gastrointestinal decolonisation when treated with FMT for multidrug-resistant bacteria. Higher rates of decolonisation were noted for P. aeruginosa than K. pneumoniae (ESBL or NDM-1); however, it is important to note the studies were of low-quality evidence (high risk of bias), small sample size (total 52 patients) and decolonisation was temporary.169
Conclusion
Overcoming challenges in SAP in the modern era of antibiotic resistance is essential to optimise patient care and surgical outcomes. The increasing and complex issue of antibiotic-resistant bacteria and strategies required to facilitate appropriate SAP are becoming increasingly challenging for those working in the surgical setting. Optimising current practice and implementing appropriate antibiotic stewardship initiatives through collaborative multidisciplinary teams is likely to remain fundamental to mitigate AR and limit SSI with resistant organisms.170 Future research is urgently required to establish effective and appropriate alternative antibiotics for surgical antibiotic prophylaxis in those colonised with antibiotic-resistant bacteria. Currently, a targeted approach to using alternative antibiotics based on pre-colonisation data, antibiotic sensitives and/or spectrum of antibiotic activity could be considered.
Key Considerations
- The incidence of antibiotic-resistant bacteria is increasing across the world: threatening the efficacy of surgical antibiotic prophylaxis and increasing the risk of surgical site infection.
- Drivers of antibiotic resistance are multifactorial and consist of misuse/inappropriate use across many areas, including primary and tertiary care.
- Awareness of common and problematic resistant bacteria and appropriate prophylaxis is imperative to optimise surgical outcomes, these include methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Staphylococcus aureus (VRSA), vancomycin-resistant Enterococcus (VRE), carbapenem-resistant Enterobacterales (CRE), fluoroquinolone-resistant Gram-negative bacteria, and extended-spectrum β-lactamase (ESBL)-producing bacteria.
- There is a lack of well-established data to support the use of broader spectrum or targeted alternative agents in those colonised with antibiotic-resistant bacteria, future research is essential on this topic.
- In the absence of high-quality data, a targeted approach using antibiotics in those colonised with antibiotic-resistant bacteria and those with a high risk of SSI may be an option. Inappropriate use of broad-spectrum antibiotics in the absence of drug-resistant bacteria and low risk of SSI is likely to potentiate antibiotic resistance, provide no benefit in patient care and may be potentially harmful.
- In addition to the consideration of targeted prophylaxis, optimisation of current practice is essential to slow the emergence of antibiotic resistance. Such steps occur at the patient and institutional levels. Steps of optimisation at a patient level include rationalisation of appropriate antibiotic use in those with a documented penicillin allergy and tailored dosing in those with concurrent obesity. Steps of optimisation at the institutional level include developing and implementation of antibiotic stewardship programs, which involve staff in the perioperative setting and ensuring appropriate timing of administration.
Acknowledgments
EC, AL and RM acknowledge funding from the Economic and Social Research Council (ESRC) and the National Institute for Health Research ASPIRES project (Antibiotic use across Surgical Pathways: Investigating, Redesigning and Evaluating Systems) (https://www.imperial.ac.uk/arc/aspires/). ASPIRES aims to address antimicrobial resistance and improve clinical outcomes optimising antibiotic usage along surgical pathways. EC acknowledges the National Institute for Health Research, UK Department of Health [HPRU-2012-10047] in partnership with Public Health England. The funders had no role in the design and conduct of this review; collection, management, analysis, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Disclosure
Dr Esmita Charani reports personal fees from Pfizer, outside the submitted work. Dr Andrew JM Leather reports grants from Economic and Social Research Council (ESRC). The authors report no other conflicts of interest in this work.
References
1. O’Neill J. Tackling drug-resistant infections globally: final report and recommendations. Government of the United Kingdom; 2016.
2. World Health Organization. World action plan on antimicrobial resistance; 2015.
3. Ademe M, Girma F. Candida auris: from multidrug resistance to pan-resistant strains. Infect Drug Resist. 2020;13:1287–1294. doi:10.2147/IDR.S249864
4. Cosgrove SE. The relationship between antimicrobial resistance and patient outcomes: mortality, length of hospital stay, and health care costs. Clin Infect Dis. 2006;42(Supp2):S82–S89. doi:10.1086/499406
5. Prestinaci F, Pezzotti P, Pantosti A. Antimicrobial resistance: a global multifaceted phenomenon. Pathog Glob Health. 2015;109(7):309–318. doi:10.1179/2047773215Y.0000000030
6. Vikesland P, Garner E, Gupta S, Kang S, Maile-Moskowitz A, Zhu N. Differential drivers of antimicrobial resistance across the world. Acc Chem Res. 2019;52(4):916–924. doi:10.1021/acs.accounts.8b00643
7. Birgand G, Castro-Sánchez E, Hansen S, et al. Comparison of governance approaches for the control of antimicrobial resistance: analysis of three European countries. Antimicrob Resist Infect Control. 2018;7(1):28. doi:10.1186/s13756-018-0321-5
8. Munckhof W. Antibiotics for surgical prophylaxis. Aust Prescr. 2005;28:38–40. doi:10.18773/austprescr.2005.030
9. Raymond DP, Kuehnert MJ, Sawyer RG. Preventing antimicrobial-resistant bacterial infections in surgical patients. Surg Infect. 2002;3(4):375–385. doi:10.1089/109629602762539599
10. Strathdee SA, Davies SC, Marcelin JR. Confronting antimicrobial resistance beyond the COVID-19 pandemic and the 2020 US election. Lancet. 2020;396(10257):1050–1053. doi:10.1016/S0140-6736(20)32063-8
11. Cassini A, Högberg LD, Plachouras D, et al. Attributable deaths and disability-adjusted life-years caused by infections with antibiotic-resistant bacteria in the EU and the European Economic Area in 2015: a population-level modelling analysis. Lancet Infect Dis. 2019;19(1):56–66. doi:10.1016/S1473-3099(18)30605-4
12. Oldenkamp R, Schultsz C, Mancini E, Cappuccio A. Filling the gaps in the global prevalence map of clinical antimicrobial resistance. Proc Natl Acad Sci USA. 2021;118(1):e2013515118. doi:10.1073/pnas.2013515118
13. Sipahi OR. Economics of antibiotic resistance. Expert Rev Anti Infect Ther. 2008;6(4):523–539. doi:10.1586/14787210.6.4.523
14. Hofer U. The cost of antimicrobial resistance. Nat Rev Microbiol. 2019;17(1):3. doi:10.1038/s41579-018-0125-x
15. Magill SS, Edwards JR, Bamberg W, et al. Multistate point-prevalence survey of health care–associated infections. N Engl J Med. 2014;370(13):1198–1208. doi:10.1056/NEJMoa1306801
16. Owens CD, Stoessel K. Surgical site infections: epidemiology, microbiology and prevention. J Hosp Infect. 2008;70(Suppl 2):3–10. doi:10.1016/S0195-6701(08)60017-1
17. World Health Organization. Protocol for surgical site infection surveillance with a focus on settings with limited resources; 2018.
18. Centers for Disease Control and Prevention. Surgical site infection event. National Healthcare Safety Network; 2021:1–39.
19. Bratzler DW, Dellinger EP, Olsen KM, et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health-Syst Pharm. 2013;70(3):195–283. doi:10.2146/ajhp120568
20. Anderson DJ, Sexton DJ. Antimicrobial prophylaxis for prevention of surgical site infection in adults. Waltham, MA: UptoDate; 2021.
21. Bhangu A, Ademuyiwa AO, Aguilera ML, et al. Surgical site infection after gastrointestinal surgery in high-income, middle-income, and low-income countries: a prospective, international, multicentre cohort study. Lancet Infect Dis. 2018;18(5):516–525.
22. Vincent J-L, Rello J, Marshall J, et al. International study of the prevalence and outcomes of infection in intensive care units. JAMA. 2009;302(21):2323–2329. doi:10.1001/jama.2009.1754
23. Călina D, Docea AO, Rosu L, et al. Antimicrobial resistance development following surgical site infections. Mol Med Rep. 2017;15(2):681–688. doi:10.3892/mmr.2016.6034
24. Ben-Ami R, Rodríguez-Baño J, Arslan H, et al. A multinational survey of risk factors for infection with extended-spectrum beta-lactamase-producing Enterobacteriaceae in nonhospitalized patients. Clin Infect Dis. 2009;49(5):682–690. doi:10.1086/604713
25. Crader MF, Varacallo M. Preoperative antibiotic prophylaxis. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021.
26. Crawford T, Rodvold KA, Solomkin JS. Vancomycin for surgical prophylaxis? Clin Infect Dis. 2012;54(10):1474–1479. doi:10.1093/cid/cis027
27. Korol E, Johnston K, Waser N, et al. A systematic review of risk factors associated with surgical site infections among surgical patients. PLoS One. 2013;8(12):e83743. doi:10.1371/journal.pone.0083743
28. Cheadle WG. Risk factors for surgical site infection. Surg Infect. 2006;7(Suppl 1):S7–S11. doi:10.1089/sur.2006.7.s1-7
29. Biccard BM, Madiba TE, Kluyts HL, et al. Perioperative patient outcomes in the African surgical outcomes study: a 7-day prospective observational cohort study. Lancet. 2018;391(10130):1589–1598. doi:10.1016/S0140-6736(18)30001-1
30. Forrester JA, Koritsanszky LA, Amenu D, et al. Developing process maps as a tool for a surgical infection prevention quality improvement Initiative in resource-constrained settings. J Am Coll Surg. 2018;226(6):1103–1116.e1103. doi:10.1016/j.jamcollsurg.2018.03.020
31. Holubar S. Antimicrobial stewardship in hospital settings. In: Hooper DC, editor. UpToDate. Waltham, MA: UpToDate; 2021.
32. Bischoff P, Kubilay NZ, Allegranzi B, Egger M, Gastmeier P. Effect of laminar airflow ventilation on surgical site infections: a systematic review and meta-analysis. Lancet Infect Dis. 2017;17(5):553–561. doi:10.1016/S1473-3099(17)30059-2
33. Wenzel RP. Minimizing surgical-site infections. N Engl J Med. 2010;362(1):75–77. doi:10.1056/NEJMe0908753
34. Lowy FD. Staphylococcus aureus Infections. N Engl J Med. 1998;339(8):520–532. doi:10.1056/NEJM199808203390806
35. Gern BH, Greninger AL, Weissman SJ, Stapp JR, Tao Y, Qin X. Continued in vitro cefazolin susceptibility in methicillin-susceptible Staphylococcus aureus. Ann Clin Microbiol Antimicrob. 2018;17(1):5. doi:10.1186/s12941-018-0257-x
36. Stapleton PD, Taylor PW. Methicillin resistance in Staphylococcus aureus: mechanisms and modulation. Sci Prog. 2002;85(Pt 1):57–72. doi:10.3184/003685002783238870
37. Malabarba A, Nicas TI, Thompson RC. Structural modifications of glycopeptide antibiotics. Med Res Rev. 1997;17(1):69–137. doi:10.1002/(SICI)1098-1128(199701)17:1<69::AID-MED3>3.0.CO;2-R
38. World Health Organization. Antimicrobial resistance: global report on surveillance. Geneva: World Health Organization; 2014.
39. Centers for Disease Control and Prevention. Staphylococcus aureus resistant to vancomycin–United States, 2002. MMWR Morb Mortal Wkly Rep. 2002;51(26):565–567.
40. Centers for Disease Control and Prevention. Vancomycin-resistant Staphylococcus aureus–New York, 2004. MMWR Morb Mortal Wkly Rep. 2004;53(15):322–323.
41. World Health Organization. Global priority list of antibiotic-resistant bacteria to guide research, discovery, and development of new antibiotics, Geneva; 2017.
42. Cong Y, Yang S, Rao X. Vancomycin resistant Staphylococcus aureus infections: a review of case updating and clinical features. J Adv Res. 2019;21:169–176. doi:10.1016/j.jare.2019.10.005
43. McGuinness WA, Malachowa N, DeLeo FR. Vancomycin resistance in Staphylococcus aureus. Yale J Biol Med. 2017;90(2):269–281.
44. Shariati A, Dadashi M, Moghadam MT, van Belkum A, Yaslianifard S, Darban-Sarokhalil D. Global prevalence and distribution of vancomycin resistant, vancomycin intermediate and heterogeneously vancomycin intermediate Staphylococcus aureus clinical isolates: a systematic review and meta-analysis. Sci Rep. 2020;10(1):12689. doi:10.1038/s41598-020-69058-z
45. Saravolatz LD, Pawlak J, Johnson LB. In vitro susceptibilities and molecular analysis of vancomycin-intermediate and vancomycin-resistant Staphylococcus aureus isolates. Clin Infect Dis. 2012;55(4):582–586. doi:10.1093/cid/cis492
46. Cetinkaya Y, Falk P, Mayhall CG. Vancomycin-resistant enterococci. Clin Microbiol Rev. 2000;13(4):686–707. doi:10.1128/CMR.13.4.686
47. Wisplinghoff H, Bischoff T, Tallent SM, Seifert H, Wenzel RP, Edmond MB. Nosocomial bloodstream infections in US hospitals: analysis of 24,179 cases from a prospective nationwide surveillance study. Clin Infect Dis. 2004;39(3):309–317. doi:10.1086/421946
48. Eliopoulos GM, Gold HS. Vancomycin-resistant Enterococci: mechanisms and clinical observations. Clin Infect Dis. 2001;33(2):210–219. doi:10.1086/321815
49. Centers for Disease Prevention and Control. Antibiotic resistance threats in the United States; 2013.
50. Remschmidt C, Schröder C, Behnke M, Gastmeier P, Geffers C, Kramer TS. Continuous increase of vancomycin resistance in enterococci causing nosocomial infections in Germany - 10 years of surveillance. Antimicrob Resist Infect Control. 2018;7:54. doi:10.1186/s13756-018-0353-x
51. Bush K, Bradford PA. Interplay between β-lactamases and new β-lactamase inhibitors. Nat Rev Microbiol. 2019;17(5):295–306. doi:10.1038/s41579-019-0159-8
52. Bassetti M, Vena A, Croxatto A, Righi E, Guery B. How to manage Pseudomonas aeruginosa infections. Drugs Context. 2018;7:212527. doi:10.7573/dic.212527
53. Bush K. The ABCD’s of β-lactamase nomenclature. J Infect Chemother. 2013;19(4):549–559. doi:10.1007/s10156-013-0640-7
54. Bush K, Jacoby GA. Updated functional classification of β-lactamases. Antimicrob Agents Chemother. 2010;54(3):969–976. doi:10.1128/AAC.01009-09
55. Page MI, Badarau A. The mechanisms of catalysis by metallo beta-lactamases. Bioinorg Chem Appl. 2008;2008:576297. doi:10.1155/2008/576297
56. Brem J, Cain R, Cahill S, et al. Structural basis of metallo-β-lactamase, serine-β-lactamase and penicillin-binding protein inhibition by cyclic boronates. Nat Commun. 2016;7:12406. doi:10.1038/ncomms12406
57. Iovleva A, Doi Y. Carbapenem-resistant Enterobacteriaceae. Clin Lab Med. 2017;37(2):303–315. doi:10.1016/j.cll.2017.01.005
58. Jacoby GA. AmpC β-lactamases. Clin Microbiol Rev. 2009;22(1):161–182. doi:10.1128/CMR.00036-08
59. Grover N, Sahni AK, Bhattacharya S. Therapeutic challenges of ESBLS and AmpC beta-lactamase producers in a tertiary care center. Med J Armed Forces India. 2013;69(1):4–10. doi:10.1016/j.mjafi.2012.02.001
60. Paterson DL, Bonomo RA. Extended-spectrum β-lactamases: a clinical update. Clin Microbiol Rev. 2005;18(4):657–686. doi:10.1128/CMR.18.4.657-686.2005
61. Lahiri SD, Johnstone MR, Ross PL, McLaughlin RE, Olivier NB, Alm RA. Avibactam and class C β-lactamases: mechanism of inhibition, conservation of the binding pocket, and implications for resistance. Antimicrob Agents Chemother. 2014;58(10):5704–5713. doi:10.1128/AAC.03057-14
62. Drawz SM, Bonomo RA. Three decades of beta-lactamase inhibitors. Clin Microbiol Rev. 2010;23(1):160–201. doi:10.1128/CMR.00037-09
63. Pilmis B, Cattoir V, Lecointe D, et al. Carriage of ESBL-producing Enterobacteriaceae in French hospitals: the PORTABLSE study. J Hosp Infect. 2018;98(3):247–252. doi:10.1016/j.jhin.2017.11.022
64. Chen LF, Anderson DJ, Paterson DL. Overview of the epidemiology and the threat of Klebsiella pneumoniae carbapenemases (KPC) resistance. Infect Drug Resist. 2012;5:133–141. doi:10.2147/IDR.S26613
65. Arnold RS, Thom KA, Sharma S, Phillips M, Kristie Johnson J, Morgan DJ. Emergence of Klebsiella pneumoniae carbapenemase-producing bacteria. South Med J. 2011;104(1):40–45. doi:10.1097/SMJ.0b013e3181fd7d5a
66. van Duin D, Doi Y. The global epidemiology of carbapenemase-producing Enterobacteriaceae. Virulence. 2017;8(4):460–469. doi:10.1080/21505594.2016.1222343
67. Pesesky MW, Hussain T, Wallace M, et al. KPC and NDM-1 genes in related Enterobacteriaceae strains and plasmids from Pakistan and the United States. Emerg Infect Dis. 2015;21(6):1034–1037. doi:10.3201/eid2106.141504
68. Palzkill T. Structural and mechanistic basis for extended-spectrum drug-resistance mutations in altering the specificity of TEM, CTX-M, and KPC β-lactamases. Front Mol Biosci. 2018;5(16). doi:10.3389/fmolb.2018.00016
69. Bassetti M, Giacobbe DR, Giamarellou H, et al. Management of KPC-producing Klebsiella pneumoniae infections. Clin Microbiol Infect. 2018;24(2):133–144. doi:10.1016/j.cmi.2017.08.030
70. Palzkill T. Metallo-β-lactamase structure and function. Ann N Y Acad Sci. 2013;1277:91–104. doi:10.1111/j.1749-6632.2012.06796.x
71. Mojica MF, Bonomo RA, Fast W. B1-metallo-β-lactamases: where do we stand? Curr Drug Targets. 2016;17(9):1029–1050. doi:10.2174/1389450116666151001105622
72. Oliphant CM, Green GM. Quinolones: a comprehensive review. Am Fam Physician. 2002;65(3):455–464.
73. Shandera KC, Thibault GP, Deshon GE. Efficacy of one dose fluoroquinolone before prostate biopsy. Urology. 1998;52(4):641–643. doi:10.1016/S0090-4295(98)00311-2
74. Ruiz J. Mechanisms of resistance to quinolones: target alterations, decreased accumulation and DNA gyrase protection. J Antimicrob Chemother. 2003;51(5):1109–1117. doi:10.1093/jac/dkg222
75. Hooper DC, Jacoby GA. Mechanisms of drug resistance: quinolone resistance. Ann N Y Acad Sci. 2015;1354(1):12–31. doi:10.1111/nyas.12830
76. Aldred KJ, Kerns RJ, Osheroff N. Mechanism of quinolone action and resistance. Biochemistry. 2014;53(10):1565–1574. doi:10.1021/bi5000564
77. Lautenbach E, Fishman NO, Bilker WB, et al. Risk factors for fluoroquinolone resistance in nosocomial Escherichia coli and Klebsiella pneumoniae infections. Arch Intern Med. 2002;162(21):2469–2477. doi:10.1001/archinte.162.21.2469
78. Zhu D-M, Li Q-H, Shen Y, Zhang Q. Risk factors for quinolone-resistant Escherichia coli infection: a systematic review and meta-analysis. Antimicrob Resist Infec Control. 2020;9(1):11. doi:10.1186/s13756-019-0675-3
79. Rezazadeh M, Baghchesaraei H, Peymani A. Plasmid-mediated quinolone-resistance (qnr) genes in clinical isolates of Escherichia coli collected from several hospitals of Qazvin and Zanjan Provinces, Iran. Osong Public Health Res Perspect. 2016;7(5):307–312. doi:10.1016/j.phrp.2016.08.003
80. Robicsek A, Jacoby GA, Hooper DC. The worldwide emergence of plasmid-mediated quinolone resistance. Lancet Infect Dis. 2006;6(10):629–640. doi:10.1016/S1473-3099(06)70599-0
81. Kim ES, Hooper DC. Clinical importance and epidemiology of quinolone resistance. Infect Chemother. 2014;46(4):226–238. doi:10.3947/ic.2014.46.4.226
82. Phillips CJ, Gilchrist M, Cooke FJ, et al. Adherence to antibiotic guidelines and reported penicillin allergy: pooled cohort data on prescribing and allergy documentation from two English National Health Service (NHS) trusts. BMJ Open. 2019;9(2):e026624. doi:10.1136/bmjopen-2018-026624
83. Savic LC, Khan DA, Kopac P, et al. Management of a surgical patient with a label of penicillin allergy: narrative review and consensus recommendations. Br J Anaesth. 2019;123(1):e82–e94. doi:10.1016/j.bja.2019.01.026
84. Blumenthal KG, Ryan EE, Li Y, Lee H, Kuhlen JL, Shenoy ES. The impact of a reported penicillin allergy on surgical site infection risk. Clin Infect Dis. 2018;66(3):329–336. doi:10.1093/cid/cix794
85. Macy E, Contreras R. Health care use and serious infection prevalence associated with penicillin “allergy” in hospitalized patients: a cohort study. J Allergy Clin Immunol. 2014;133(3):790–796. doi:10.1016/j.jaci.2013.09.021
86. Ponce B, Raines BT, Reed RD, Vick C, Richman J, Hawn M. Surgical site infection after arthroplasty: comparative effectiveness of prophylactic antibiotics: do surgical care improvement project guidelines need to be updated? J Bone Joint Surg Am. 2014;96(12):970–977. doi:10.2106/JBJS.M.00663
87. Pool C, Kass J, Spivack J, et al. Increased surgical site infection rates following clindamycin use in head and neck free tissue transfer. Otolaryngol Head Neck Surg. 2016;154(2):272–278. doi:10.1177/0194599815617129
88. Dhopeshwarkar N, Sheikh A, Doan R, et al. Drug-induced anaphylaxis documented in electronic health records. J Allergy Clin Immunol Pract. 2019;7(1):103–111. doi:10.1016/j.jaip.2018.06.010
89. Blumenthal KG, Shenoy ES, Varughese CA, Hurwitz S, Hooper DC, Banerji A. Impact of a clinical guideline for prescribing antibiotics to inpatients reporting penicillin or cephalosporin allergy. Ann Allergy, Asthma Immunol. 2015;115(4):294–300.e292. doi:10.1016/j.anai.2015.05.011
90. Park M, Markus P, Matesic D, Li JT. Safety and effectiveness of a preoperative allergy clinic in decreasing vancomycin use in patients with a history of penicillin allergy. Ann Allergy Asthma Immunol. 2006;97(5):681–687. doi:10.1016/S1081-1206(10)61100-3
91. Hruby A, Hu FB. The epidemiology of obesity: a big picture. Pharmacoeconomics. 2015;33(7):673–689. doi:10.1007/s40273-014-0243-x
92. Charani E, Gharbi M, Frost G, Drumright L, Holmes A. Antimicrobial therapy in obesity: a multicentre cross-sectional study. J Antimicrob Chemother. 2015;70(10):2906–2912. doi:10.1093/jac/dkv189
93. Polso AK, Lassiter JL, Nagel JL. Impact of hospital guideline for weight-based antimicrobial dosing in morbidly obese adults and comprehensive literature review. J Clin Pharm Therap. 2014;39(6):584–608. doi:10.1111/jcpt.12200
94. Janson B, Thursky K. Dosing of antibiotics in obesity. Curr Opin Infect Dis. 2012;25(6):634–649. doi:10.1097/QCO.0b013e328359a4c1
95. Chopra T, Zhao JJ, Alangaden G, Wood MH, Kaye KS. Preventing surgical site infections after bariatric surgery: value of perioperative antibiotic regimens. Expert Rev Pharmacoecon Outcomes Res. 2010;10(3):317–328. doi:10.1586/erp.10.26
96. Meng L, Mui E, Holubar MK, Deresinski SC. Comprehensive guidance for antibiotic dosing in obese adults. Pharmacother. 2017;37(11):1415–1431. doi:10.1002/phar.2023
97. Branch-Elliman W, O’Brien W, Strymish J, Itani K, Wyatt C, Gupta K. Association of duration and type of surgical prophylaxis with antimicrobial-associated adverse events. JAMA Surg. 2019;154(7):590–598. doi:10.1001/jamasurg.2019.0569
98. Scher KS. Studies on the duration of antibiotic administration for surgical prophylaxis. Am Surg. 1997;63(1):59–62.
99. Australian Commission on Safety and Quality in Health Care. Antimicrobial prescribing practice in Australian Hospitals: results of the 2019 Hospital National Antimicrobial Prescribing survey; 2021:1–45.
100. Zelenitsky SA, Ariano RE, Harding GK, Silverman RE. Antibiotic pharmacodynamics in surgical prophylaxis: an association between intraoperative antibiotic concentrations and efficacy. Antimicrob Agents Chemother. 2002;46(9):3026–3030. doi:10.1128/AAC.46.9.3026-3030.2002
101. Naik BI, Roger C, Ikeda K, et al. Comparative total and unbound pharmacokinetics of cefazolin administered by bolus versus continuous infusion in patients undergoing major surgery: a randomized controlled trial. Br J Anaesth. 2017;118(6):876–882. doi:10.1093/bja/aex026
102. Swoboda SM, Merz C, Kostuik J, Trentler B, Lipsett PA. Does intraoperative blood loss affect antibiotic serum and tissue concentrations? Arch Surg. 1996;131(11):1165–1171. doi:10.1001/archsurg.1996.01430230047009
103. Zanetti G, Giardina R, Platt R. Intraoperative redosing of cefazolin and risk for surgical site infection in cardiac surgery. Emerg Infect Dis. 2001;7(5):828–831. doi:10.3201/eid0705.017509
104. Costa AD. Assessment of operative times of multiple surgical specialties in a public university hospital. Einstein. 2017;15(2):200–205. doi:10.1590/s1679-45082017gs3902
105. Markantonis SL, Kostopanagiotou G, Panidis D, Smirniotis V, Voros D. Effects of blood loss and fluid volume replacement on serum and tissue gentamicin concentrations during colorectal surgery. Clin Ther. 2004;26(2):271–281. doi:10.1016/S0149-2918(04)90025-2
106. Ben-Ami R, Schwaber MJ, Navon-Venezia S, et al. Influx of extended-spectrum β-lactamase—producing Enterobacteriaceae into the hospital. Clin Infect Dis. 2006;42(7):925–934. doi:10.1086/500936
107. Bhattacharya S. Is screening patients for antibiotic-resistant bacteria justified in the Indian context? Indian J Med Microbiol. 2011;29(3):213–217. doi:10.4103/0255-0857.83902
108. Saied T, Hafez SF, Kandeel A, et al. Antimicrobial stewardship to optimize the use of antimicrobials for surgical prophylaxis in Egypt: a multicenter pilot intervention study. Am J Infect Control. 2015;43(11):e67–e71. doi:10.1016/j.ajic.2015.07.004
109. Berríos-Torres SI, Umscheid CA, Bratzler DW, et al. Centers for disease control and prevention guideline for the prevention of surgical site infection, 2017. JAMA Surg. 2017;152(8):784–791. doi:10.1001/jamasurg.2017.0904
110. Labricciosa FM, Sartelli M, Correia S, et al. Emergency surgeons’ perceptions and attitudes towards antibiotic prescribing and resistance: a worldwide cross-sectional survey. World J Emerg Surg. 2018;13(1):27. doi:10.1186/s13017-018-0190-5
111. Tan JA, Naik VN, Lingard L. Exploring obstacles to proper timing of prophylactic antibiotics for surgical site infections. Qual Saf Health Care. 2006;15(1):32–38. doi:10.1136/qshc.2004.012534
112. Abdel-Aziz A, El-Menyar A, Al-Thani H, et al. Adherence of surgeons to antimicrobial prophylaxis guidelines in a tertiary general hospital in a rapidly developing country. Adv Pharmaco Sci. 2013;2013:842593.
113. Bedouch P, Labarère J, Chirpaz E, et al. Compliance with guidelines on antibiotic prophylaxis in total hip replacement surgery: results of a retrospective study of 416 patients in a teaching hospital. Infect Control Hosp Epidemiol. 2004;25(4):302–307. doi:10.1086/502396
114. Bull AL, Russo PL, Friedman ND, Bennett NJ, Boardman CJ, Richards MJ. Compliance with surgical antibiotic prophylaxis – reporting from a statewide surveillance programme in Victoria, Australia. J Hosp Infect. 2006;63(2):140–147. doi:10.1016/j.jhin.2006.01.018
115. Graham H, Vasireddy A, Nehra D. A national audit of antibiotic prophylaxis in elective laparoscopic cholecystectomy. Ann R Coll Surg Engl. 2014;96(5):377–380. doi:10.1308/003588414X13946184900688
116. Miliani K, L’Hériteau F, Astagneau P. Group obotINS. Non-compliance with recommendations for the practice of antibiotic prophylaxis and risk of surgical site infection: results of a multilevel analysis from the INCISO Surveillance Network. J Antimicrob Chemother. 2009;64(6):1307–1315. doi:10.1093/jac/dkp367
117. Mohamed Rizvi Z, Palasanthiran P, Wu C, Mostaghim M, McMullan B. Adherence to surgical antibiotic prophylaxis guidelines in children: a cohort study. J Paed Child Health. 2020;56(1):34–40. doi:10.1111/jpc.14484
118. Wright JD, Hassan K, Ananth CV, et al. Use of guideline-based antibiotic prophylaxis in women undergoing gynecologic surgery. Obstet Gynecol. 2013;122(6):1145–1153. doi:10.1097/AOG.0b013e3182a8a36a
119. Romero Viamonte K, Salvent Tames A, Sepúlveda Correa R, Rojo Manteca MV, Martín-Suárez A. Compliance with antibiotic prophylaxis guidelines in caesarean delivery: a retrospective, drug utilization study (indication-prescription type) at an Ecuadorian hospital. Antimicrob Resist Infect Control. 2021;10(1):12. doi:10.1186/s13756-020-00843-1
120. Hawkins RB, Levy SM, Senter CE, et al. Beyond surgical care improvement program compliance: antibiotic prophylaxis implementation gaps. Am J Surg. 2013;206(4):451–456. doi:10.1016/j.amjsurg.2013.02.009
121. Haney V, Maman S, Prozesky J, Bezinover D, Karamchandani K. Improving intraoperative administration of surgical antimicrobial prophylaxis: a quality improvement report. BMJ Open Quality. 2020;9(3):e001042. doi:10.1136/bmjoq-2020-001042
122. Mahmoudi L, Ghouchani M, Mahi-Birjand M, Bananzadeh A, Akbari A. Optimizing compliance with surgical antimicrobial prophylaxis guidelines in patients undergoing gastrointestinal surgery at a referral teaching hospital in southern Iran: clinical and economic impact. Infect Drug Resist. 2019;12:2437–2444. doi:10.2147/IDR.S212728
123. Telfah S, Nazer L, Dirani M, Daoud F. Improvement in adherence to surgical antimicrobial prophylaxis guidelines after implementation of a multidisciplinary quality improvement project. Sultan Qaboos Univ Med J. 2015;15(4):e523–e527. doi:10.18295/squmj.2015.15.04.014
124. Segala FV, Murri R, Taddei E, et al. Antibiotic appropriateness and adherence to local guidelines in perioperative prophylaxis: results from an antimicrobial stewardship intervention. Antimicrob Resist Infect Control. 2020;9(1):164. doi:10.1186/s13756-020-00814-6
125. Cohen ME, Salmasian H, Li J, et al. Surgical antibiotic prophylaxis and risk for postoperative antibiotic-resistant infections. J Am Coll Surg. 2017;225(5):631–638.e633. doi:10.1016/j.jamcollsurg.2017.08.010
126. Ribed A, Monje B, García-González X, et al. Improving surgical antibiotic prophylaxis adherence and reducing hospital readmissions: a bundle of interventions including health information technologies. Eur J Hosp Pharm. 2020;27(4):237–242. doi:10.1136/ejhpharm-2018-001666
127. Pons-Busom M, Aguas-Compaired M, Delás J, Eguileor-Partearroyo B. Compliance with local guidelines for antibiotic prophylaxis in surgery. Infect Control Hosp Epidemiol. 2004;25(4):308–312. doi:10.1086/502397
128. World Health Organization. World Health Alliance for Patient Safety. Implementation Manual: WHO Surgical Safety Checklist.
129. Haynes AB, Weiser TG, Berry WR, et al. A surgical safety checklist to reduce morbidity and mortality in a global population. N Engl J Med. 2009;360(5):491–499. doi:10.1056/NEJMsa0810119
130. de Vries EN, Dijkstra L, Smorenburg SM, Meijer RP, Boermeester MA. The SURgical PAtient Safety System (SURPASS) checklist optimizes timing of antibiotic prophylaxis. Patient Saf Surg. 2010;4(1):6. doi:10.1186/1754-9493-4-6
131. White MC, Peven K, Clancy O, et al. Implementation strategies and the uptake of the World Health Organization surgical safety checklist in low and middle income countries: a systematic review and meta-analysis. Ann Surg. 2021;273:6. doi:10.1097/SLA.0000000000003944
132. White MC, Randall K, Ravelojaona VA, et al. Sustainability of using the WHO surgical safety checklist: a mixed-methods longitudinal evaluation following a nationwide blended educational implementation strategy in Madagascar. BMJ Glob Health. 2018;3(6):e001104. doi:10.1136/bmjgh-2018-001104
133. White MC, Daya L, Karel FKB, et al. Using the knowledge to action framework to describe a nationwide implementation of the WHO surgical safety checklist in cameroon. Anesth Analg. 2020;130(5):1425–1434. doi:10.1213/ANE.0000000000004586
134. White MC, Randall K, Capo-Chichi NFE, et al. Implementation and evaluation of nationwide scale-up of the surgical safety checklist. Br J Surg. 2019;106(2):e91–e102. doi:10.1002/bjs.11034
135. White MC, Leather AJM, Sevdalis N, Healey A. Economic case for scale-up of the WHO surgical safety checklist at the national level in Sub-Saharan Africa. Ann Surg. 2020. doi:10.1097/SLA.0000000000004498
136. Kavanagh KT, Calderon LE, Saman DM, Abusalem SK. The use of surveillance and preventative measures for methicillin-resistant Staphylococcus aureus infections in surgical patients. Antimicrob Resist Infect Control. 2014;3(1):18. doi:10.1186/2047-2994-3-18
137. Lee AS, Cooper BS, Malhotra-Kumar S, et al. Comparison of strategies to reduce meticillin-resistant Staphylococcus aureus rates in surgical patients: a controlled multicentre intervention trial. BMJ Open. 2013;3(9):e003126. doi:10.1136/bmjopen-2013-003126
138. Saraswat MK, Magruder JT, Crawford TC, et al. Preoperative Staphylococcus aureus screening and targeted decolonization in cardiac surgery. Ann Thorac Surg. 2017;104(4):1349–1356. doi:10.1016/j.athoracsur.2017.03.018
139. World Health Organization. WHO surgical site infection prevention guidelines web appendix 4. Summary of a systematic review on screening for extended spectrum beta-lactamase and the impact on surgical antibiotic prophylaxis. Geneva; 2018.
140. Dubinsky-Pertzov B, Temkin E, Harbarth S, et al. Carriage of extended-spectrum beta-lactamase-producing Enterobacteriaceae and the risk of surgical site infection after colorectal surgery: a prospective cohort study. Clin Infect Dis. 2019;68(10):1699–1704. doi:10.1093/cid/ciy768
141. Bonkat G, Pilatz A, Wagenlehner F. Time to adapt our practice? The European commission has restricted the use of fluoroquinolones since March 2019. Eur Urol. 2019;76(3):273–275. doi:10.1016/j.eururo.2019.06.011
142. Freitas DM, Moreira DM. Fosfomycin trometamol vs ciprofloxacin for antibiotic prophylaxis before transrectal ultrasonography-guided prostate biopsy: a meta-analysis of clinical studies. Arab J Urol. 2019;17(2):114–119. doi:10.1080/2090598X.2019.1592636
143. Guss J, Abuzeid WM, Doghramji L, Edelstein PH, Chiu AG. Fluoroquinolone-resistant Pseudomonas aeruginosa in chronic rhinosinusitis. Orl J Otorhinolaryngol Relat Spec. 2009;71(5):263–267. doi:10.1159/000242428
144. Bratzler DW, Houck PM. Antimicrobial prophylaxis for surgery: an advisory statement from the National Surgical Infection Prevention Project. Am J Surg. 2005;189(4):395–404. doi:10.1016/j.amjsurg.2005.01.015
145. Classen DC, Evans RS, Pestotnik SL, Horn SD, Menlove RL, Burke JP. The timing of prophylactic administration of antibiotics and the risk of surgical-wound infection. N Engl J Med. 1992;326(5):281–286. doi:10.1056/NEJM199201303260501
146. de Jonge SW, Gans SL, Atema JJ, Solomkin JS, Dellinger PE, Boermeester MA. Timing of preoperative antibiotic prophylaxis in 54,552 patients and the risk of surgical site infection: a systematic review and meta-analysis. Medicine. 2017;96(29):e6903. doi:10.1097/MD.0000000000006903
147. Spencer S, Ipema H, Hartke P, et al. Intravenous push administration of antibiotics: literature and considerations. Hosp Pharm. 2018;53(3):157–169. doi:10.1177/0018578718760257
148. Lexicomp. Adult Drug Information Handbook. Waltham, MA; 2020.
149. Knox MC, Edye M. Adherence to surgical antibiotic prophylaxis guidelines in New South Wales, Australia: identifying deficiencies and regression analysis of contributing factors. Surg Infect (Larchmt). 2016;17(2):203–209. doi:10.1089/sur.2015.195
150. Zvonar RK, Bush P, Roth V. Practice changes to improve delivery of surgical antibiotic prophylaxis. Healthc Q. 2008;11(3):141–144. doi:10.12927/hcq.2008.19664
151. Charani E, Ahmad R, Rawson TM, Castro-Sanchèz E, Tarrant C, Holmes AH. The differences in antibiotic decision-making between acute surgical and acute medical teams: an ethnographic study of culture and team dynamics. Clin Infect Dis. 2019;69(1):12–20. doi:10.1093/cid/ciy844
152. Charani E, Tarrant C, Moorthy K, Sevdalis N, Brennan L, Holmes AH. Understanding antibiotic decision making in surgery-a qualitative analysis. Clin Microbiol Infect. 2017;23(10):752–760. doi:10.1016/j.cmi.2017.03.013
153. Charani E, Smith I, Skodvin B, et al. Investigating the cultural and contextual determinants of antimicrobial stewardship programmes across low-, middle- and high-income countries-a qualitative study. PLoS One. 2019;14(1):e0209847. doi:10.1371/journal.pone.0209847
154. Davey P, Marwick CA, Scott CL, et al. Interventions to improve antibiotic prescribing practices for hospital inpatients. Cochrane Database Syst Rev. 2017;2:CD003543.
155. Singh S, Mendelson M, Surendran S, et al. Investigating infection management and antimicrobial stewardship in surgery: a qualitative study from India and South Africa. Clin Microbiol Infect. 2021;27(10):1455–1464. doi:10.1016/j.cmi.2020.12.013
156. Ierano C, Thursky K, Peel T, Rajkhowa A, Marshall C, Ayton D. Influences on surgical antimicrobial prophylaxis decision making by surgical craft groups, anaesthetists, pharmacists and nurses in public and private hospitals. PLoS One. 2019;14(11):e0225011. doi:10.1371/journal.pone.0225011
157. Charani E, Castro-Sanchez E, Sevdalis N, et al. Understanding the determinants of antimicrobial prescribing within hospitals: the role of “prescribing etiquette”. Clin Infect Dis. 2013;57(2):188–196. doi:10.1093/cid/cit212
158. Charani E, de Barra E, Rawson TM, et al. Antibiotic prescribing in general medical and surgical specialties: a prospective cohort study. Antimicrob Resist Infect Control. 2019;8:151. doi:10.1186/s13756-019-0603-6
159. de Jonge SW, Boldingh QJJ, Solomkin JS, et al. Effect of postoperative continuation of antibiotic prophylaxis on the incidence of surgical site infection: a systematic review and meta-analysis. Lancet Infect Dis. 2020;20(10):1182–1192. doi:10.1016/S1473-3099(20)30084-0
160. Bonaconsa C, Mbamalu O, Mendelson M, et al. Visual mapping of team dynamics and communication patterns on surgical ward rounds: an ethnographic study. BMJ Qual Saf. 2021;30:812–824. doi:10.1136/bmjqs-2020-012372
161. Anahtar MN, Yang JH, Kanjilal S, McAdam AJ. Applications of machine learning to the problem of antimicrobial resistance: an emerging model for translational research. J Clin Microbiol. 2021;59(7):e01260–01220. doi:10.1128/JCM.01260-20
162. Feretzakis G, Loupelis E, Sakagianni A, et al. Using machine learning techniques to aid empirical antibiotic therapy decisions in the intensive care unit of a general hospital in Greece. Antibiotics. 2020;9(2):50. doi:10.3390/antibiotics9020050
163. Luong T, Salabarria AC, Roach DR. Phage therapy in the resistance era: where do we Stand and where are we going? Clin Ther. 2020;42(9):1659–1680. doi:10.1016/j.clinthera.2020.07.014
164. Rubalskii E, Ruemke S, Salmoukas C, et al. Bacteriophage therapy for critical infections related to cardiothoracic surgery. Antibiotics. 2020;9(5):232. doi:10.3390/antibiotics9050232
165. Brives C, Pourraz J. Phage therapy as a potential solution in the fight against AMR: obstacles and possible futures. Palgrave Commun. 2020;6(1):100. doi:10.1057/s41599-020-0478-4
166. Narasimhaiah MH, Asrani JY, Palaniswamy SM, et al. Therapeutic potential of Staphylococcal bacteriophages for nasal decolonization of Staphylococcus aureus in Mice. Adv Microbiol. 2013;03(01):9. doi:10.4236/aim.2013.31008
167. Rohlke F, Stollman N. Fecal microbiota transplantation in relapsing Clostridium difficile infection. Therap Adv Gastroenterol. 2012;5(6):403–420. doi:10.1177/1756283X12453637
168. Gargiullo L, Del Chierico F, D’Argenio P, Putignani L. Gut Microbiota modulation for multidrug-resistant organism decolonization: present and future perspectives. Front Microbiol. 2019;10:1704. doi:10.3389/fmicb.2019.01704
169. Tavoukjian V. Faecal microbiota transplantation for the decolonization of antibiotic-resistant bacteria in the gut: a systematic review and meta-analysis. J Hosp Infect. 2019;102(2):174–188. doi:10.1016/j.jhin.2019.03.010
170. Paskovaty A, Pflomm JM, Myke N, Seo SK. A multidisciplinary approach to antimicrobial stewardship: evolution into the 21st century. Int J Antimicrob Agents. 2005;25(1):1–10. doi:10.1016/j.ijantimicag.2004.09.001
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.