Back to Journals » Clinical Interventions in Aging » Volume 13
Superior articular process cross-sectional area is a new sensitive parameter for the diagnosis of lumbar central canal spinal stenosis
Authors An SJ ![]() , Mun JU, Kang KN, Kim YU
, Mun JU, Kang KN, Kim YU ![]()
Received 26 April 2018
Accepted for publication 25 July 2018
Published 17 September 2018 Volume 2018:13 Pages 1763—1767
DOI https://doi.org/10.2147/CIA.S172355
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Walker
Sang Joon An,1 Jong-Uk Mun,2 Keum Nae Kang,3 Young Uk Kim4
1Department of Neurology, Catholic Kwandong University of Korea College of Medicine, International St Mary’s Hospital, Incheon, Korea; 2Department of Orthopaedic Surgery, Changwon Gyeongsang National University Hospital, Changwon, Korea; 3Department of Anesthesiology and Pain Medicine, National Police Hospital, Seoul, Korea; 4Department of Anesthesiology and Pain Medicine, Catholic Kwandong University of Korea College of Medicine, International St Mary’s Hospital, Incheon, Korea
Purpose: Previous studies reported that hypertrophied superior articular process (SAP) was associated with an increased risk of lumbar foraminal stenosis. However, no study investigated the effect of SAP hypertrophy in lumbar central canal spinal stenosis (LCCSS). We hypothesized that the SAP cross-sectional area (SAPCSA) is the main morphologic feature in the diagnosis of LCCSS.
Patients and methods: Data regarding the SAPCSA were collected from 109 patients with LCCSS. All patients were enrolled after the LCCSS diagnosis was confirmed by an experienced, board-certified neuroradiologist. All patients had clinical manifestations compatible with LCCSS. A total of 120 subjects in the control group underwent lumbar spine MRI as part of non-symptomatic medical examination. T2-weighted axial images were obtained from the 2 groups. Using a picture archiving and communications system, we analyzed the CSA of the bone margin of SAP at the level of L4–L5 facet joint on MRI.
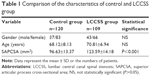
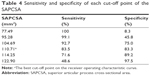
Results: The average SAPCSA was 96.63±13.37 mm2 in the control group, and 123.59±14.18 mm2 in the LCCSS. The LCCSS group showed significantly higher levels of the SAPCSA (P<0.001) compared with the control one. Receiver operator characteristic (ROC) curve analysis was performed to determine the validity of the SAPCSA as a predictor of LCCSS. In the LCCSS group, the optimal cut-off-point was 110.71 mm2, with 83.5% sensitivity, 83.3% specificity, and area under the curve of 0.92 (95% CI: 0.88–0.95).
Conclusion: Higher SAPCSA values were associated with a higher possibility of LCCSS. These results are important in the evaluation of patients with LCCSS.
Keywords: superior articular process, lumbar central canal spinal stenosis, cross-sectional area
Introduction
Lumbar spinal stenosis results from progressive degenerative disease of the spinal canal, and is the most common spinal disorder in patients aged ≥60 years.1–3 It typically presents with buttock or low back pain, sensory and motor disturbances in the lower leg, and neurogenic intermittent claudication.4 Lumbar spinal stenosis is characterized by narrowing of the lumbar spinal canal, which is caused by facet joint hypertrophy, hypertrophy of the ligamentum flavum, mechanical compression of the lumbar spinal nerve roots, and disc herniation combined with osteophytes.5,6 Anatomically, degenerative lumbar spinal stenosis may involve the lumbar central canal spinal stenosis (LCCSS), the foramina, the extraforaminal zone, or a combination of these locations. LCCSS is a narrowing of the central spinal canal causing spinal nerve compression, which can result in painful and chronic symptoms.7–9 Previous studies have indicated that morphological parameters, including the spinal canal area, dural sac area, and ligamentum flavum thickness are associated with disc degeneration, aging, and lumbar spinal stenosis.10–12 Superior articular process (SAP) hypertrophy is associated with a higher possibility of lumbar foraminal stenosis.13 However, no studies have determined the effect of SAP hypertrophy on LCCSS until now. The cross-sectional area (CSA) of the SAP is an important morphological parameter in the identification of facet joint hypertrophy.13,14 Therefore, to evaluate the connection between LCCSS and hypertrophy of the SAP, we analyzed the superior articular process cross-sectional area (SAPCSA). We hypothesized that the SAPCSA is an important morphological parameter in the diagnosis of LCCSS. We used MRI to compare the SAPCSA between LCCSS patients and normal controls.
Patients and methods
Patients
This study was registered at the Eulji University College of Medicine, Republic of Korea. The Institutional Review Board (IRB) approved and reviewed the research protocol (EMC IRB number 15–94). All participants provided written informed consent to review their radiological images. We retrospectively reviewed the patients who had visited the Catholic Kwandong Spine Center and Pain Clinic between March 2014 and December 2017 and were diagnosed with LCCSS. Patients aged >60 years were included if they had clinical manifestations compatible with LCCSS (such as low back pain and/or neurogenic intermittent claudication), maximum stenosis occurring at L4–L5, and had MRI within 1 year of the diagnosis that was available for chart review. Patients were excluded if they had a past history of spinal injury or lumbar surgery, congenital spine defect, lumbar spondylolisthesis, complex regional pain syndrome, or history of spinal interventions, such as kyphoplasty or vertebroplasty.
A total of 109 patients were enrolled after the LCCSS diagnosis was confirmed by an experienced, board-certified neuroradiologist. The LCCSS group included 43 (39.4%) men and 66 (60.6%) women with a mean age of 70.81±6.94 years (range, 60–88 years) (Table 1). The SAPCSA between patients with and without LCCSS was compared with a group of control subjects who had undergone lumbar spine MRI as part of non-symptomatic medical examination. Patients in the control group had no LCCSS-related symptoms. The control group consisted of 120 patients (37 men and 83 women) with a mean age of 68.12±8.13 years (range, 60–87 years) (Table 1). The SAPCSA in the control group was similarly examined at the L4–L5 facet joint level.
Imaging parameters
Lumbar spine MRI was performed with 3T Avanto (Siemens Healthcare, Erlangen, Germany) with 3T scanners (Achieva; Philips Healthcare, Amsterdam, the Netherlands). The lumbar MRI examination was conducted using axial T2-weighted images obtained with a slice thickness <3 mm, 0.9 mm intersection gap, 6,507-ms/120-ms repetition time/echo time, 150×150 field of view, and 256×241 matrix. All MRI data were transferred from the MRI unit to an INFINITT system (INFINITT Healthcare Co., Seoul, Korea).
Image analysis
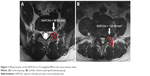
The measurement analysis and data collection were performed in a double-blind fashion. The axial T2-weighted MR images in individual patients were acquired at the level of facet joint. A picture archiving and communications system was used to measure the SAPCSA at the L4–L5 facet joint level on MRI. The SAPCSA was measured as the CSA by outlining the SAP at the L4–L5 level (Figure 1). To find age-related changes of SAPCSA, we also investigated age-related subgroup analysis.
Statistical analysis
The data were expressed as means ± SD. Unpaired t-tests were used to compare the SAPCSA between the control and LCCSS groups. P-values<0.05 were considered statistically significant. The correlation between the SAPCSA and age-related changes were analyzed using a one-way ANOVA. The validity of the SAPCSA for diagnosis was estimated using Receiver Operator Characteristic (ROC) curves, optimal cut-off values, area under the curve (AUC), sensitivity, and specificity with 95% CIs. SPSS version 22 for Windows (IBM SPSS, IBM Corp, Armonk, NY, USA) was used for the statistical analysis.
Results
Demographic characteristics were not significantly different between the 2 groups (Table 1). The average SAPCSA was 96.63±13.37 mm2 in the control group, and 123.59±14.18 mm2 in the LCCSS one. LCCSS patients had significantly greater SAPCSA (P<0.001) than control subjects (Table 1). The mean SAPCSA of control group was 96.88±11.60 mm2 in subjects aged 60–69 years, 96.43±15.56 mm2 in subjects aged 70–79 years, and 95.85±17.37 mm2 in subjects in the 80–88 years age group (Table 2). In the control group, we found no statistically significant relationships between the SAPCSA and age-related changes in the one-way ANOVA (F=0.43; df =2; and P=0.958). The mean SAPCSA of LCCSS group measured 123.97±14.28 mm2 in patients aged 60–69 years, 123.68±14.58 mm2 in patients in the 70–79 years age group, and 120.37±11.51 mm2 in patients in the 80–88 years age category (Table 3). In the LCCSS group, we also found no statistically significant relationships between SAPCSA and age-related changes (F=0.195; df =2; and P=0.823). Regarding the validity of the SAPCSA as predictors of LCCSS, the ROC curve analysis showed that the optimal cut-off point of the SAPCSA was 110.71 mm2, with 83.5% sensitivity, 83.3% specificity (Table 4), and AUC of 0.92 (95% CI: 0.88–0.95) (Figure 2).
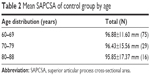  | Table 2 Mean SAPCSA of control group by age |
  | Table 3 Mean SAPCSA of LCCSS group by age |
Discussion
LCCSS is the most common spinal disease in elderly individuals that causes low back or buttock pain and intermittent neurogenic claudication.15 LCCSS results from a combination of pathogenic factors, including a decrease in the area of dural sac, hypertrophy of ligamentum flavum, and loss of intervertebral disk height.1–3 Facet joint hypertrophy also is a major step in the development of LCCSS.14 Previous studies investigated the association between ligamentum flavum, dural sac area, and spinal canal area on MRI and the signs and symptoms of LCCSS. Abbas et al reported that the thickness of ligamentum flavum is gender-independent and age-dependent.10 Altinkaya et al found that the ligamentum flavum thickness was correlated with body mass index, spinal level, aging, disc degeneration, and disc herniation.16 Ogikubo et al found a significant relationship between shorter walking distances and a smaller dural sac area.12 Kim et al reported that a larger dural sac area is associated with a longer subjective walking distance before the onset of claudication.11 Lim et al demonstrated that hypertrophy of SAP is a sensitive parameter for the assessment of lumbar foraminal stenosis.13 However, there are no previous reports correlating LCCSS and hypertrophy of SAPCSA as a morphologic parameter on MRI. Panjabi et al described the CSA of the superior articular facet.17 These CSAs were analyzed in autopsy specimens. Barry et al measured the CSA of the SAP using CT.14 In this study, we analyzed the SAPCSA from MRI images, especially visualizing the SAP in the axial T2-weighted MRI. The lumbar MRI provides an enhanced view of the SAP and SAP hypertrophic changes.13 Our results demonstrate the association between the SAPCSA and LCCSS. The positive correlation between the SAPCSA and the LCCSS is explained by the increase in SAPCSA associated with the increase in LCCSS. In our study, the best cut-off point for SAPCSA was 110.71 mm2, with 83.5% sensitivity, 83.3% specificity, and AUC of 0.92 (95% CI: 0.88–0.95). Our interpretation of these correlations is based on the understanding that the process of SAP hypertrophy begins with mechanical continuous stress during lumbar extension, flexion, and rotation. These stressors exert pressure on the facet joints, which leads to a high degree of abrasion,18,19 and alter the morphological features of SAP. Bajek et al demonstrated that osteophyte formation in the lumbar spinal canal to stabilize an unstable segment, may result in SAP hypertrophy.20 Disc degeneration and herniation may also increase the stress on the morphologic changes in SAP.21
Therefore, our findings suggest that SAPCSA is an objective and accurate morphological parameter for LCCSS prediction. Our study only included individuals aged >60 years because only minimal changes in cartilage associated with SAP occurred before the age of 45 years, as reported by Lim et al, and that osteoarthritic hypertrophy advances with age.13
The present study had a number of weaknesses. First, we could not compare our SAPCSA with patients’ clinical outcomes and symptoms. Second, there may be measurement errors associated with analyzing the SAPCSA on MRI. We measured the SAPCSA in the axial T2 images at the level of the L4–L5 facet joint. However, these axial images may be inhomogeneous due to differences in the cutting angle of the MRI resulting from individual anatomic variations and technical difficulties. A 3.0-mm slice of axial T2-weighted MR image is also thicker than an ideal slice. Third, several methods are known to effectively discriminate LCCSS, such as analysis of cauda equina, morphological grading, and sedimentation signs.22–30 However, this study only used SAPCSA measurement. Therefore, our results may be limited regarding measurement of the epidural pressure or morphologic changes. The ultimate weakness of the study is that it was retrospective in nature.
Despite these limitations, this is the first study to document the association of SAPCSA with LCCSS. In our results, the AUC is 0.92 and its accuracy is in the excellent range (0.90–1). Therefore these results are sufficient for SAPCSA to be a senstive parameter to evaluate LCCSS.
Conclusion
SAPCSA is a sensitive parameter for the diagnosis of LCCSS with an optimal cut-off-point of 110.71 mm2, sensitivity of 83.5%, specificity of 83.3%, and AUC of 0.92. We believe that this measurement tool will facilitate the evaluation of patients with LCCSS.
Acknowledgment
This research did not receive any specific grant from funding agencies in the public.
Author contributions
All authors contributed toward data analysis, drafting, and critically revising the paper, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
Patel J, Osburn I, Wanaselja A, Nobles R. Optimal treatment for lumbar spinal stenosis: an update. Curr Opin Anaesthesiol. 2017;30(5):598–603. | ||
Bin Y, de Cheng W, Wei WZ, Hui L. Muscle gap approach under a minimally invasive channel technique for treating long segmental lumbar spinal stenosis: A retrospective study. Medicine. 2017;96(32):e7779. | ||
Lubelski D, Thompson NR, Agrawal B, et al. Prediction of quality of life improvements in patients with lumbar stenosis following use of membrane stabilizing agents. Clin Neurol Neurosurg. 2015;139:234–240. | ||
Hong JH, Lee MY, Jung SW, Lee SY. Does spinal stenosis correlate with MRI findings and pain, psychologic factor and quality of life? Korean J Anesthesiol. 2015;68(5):481–487. | ||
Hughes A, Makirov SK, Osadchiy V. Measuring spinal canal size in lumbar spinal stenosis: description of method and preliminary results. Int J Spine Surg. 2015;9:3. | ||
Lim YS, Mun JU, Seo MS, et al. Dural sac area is a more sensitive parameter for evaluating lumbar spinal stenosis than spinal canal area: A retrospective study. Medicine. 2017;96(49):e9087. | ||
Mannion AF, Fekete TF, Pacifico D, et al. Dural sac cross-sectional area and morphological grade show significant associations with patient-rated outcome of surgery for lumbar central spinal stenosis. Eur Spine J. 2017;26(10):2552–2564. | ||
Manchikanti L, Pampati V, Benyamin RM, Hirsch JA. Cost Utility Analysis of Lumbar Interlaminar Epidural Injections in the Treatment of Lumbar Disc Herniation, Central Spinal Stenosis, and Axial or Discogenic Low Back Pain. Pain Physician. 2017;20(4):219–228. | ||
Lee GY, Guen YL, Lee JW, Choi HS, et al. A new grading system of lumbar central canal stenosis on MRI: an easy and reliable method. Skeletal Radiol. 2011;40(8):1127–1039. | ||
Abbas J, Hamoud K, Masharawi YM, et al. Ligamentum flavum thickness in normal and stenotic lumbar spines. Spine. 2010;35(12):1225–1230. | ||
Kim YU, Kong YG, Lee J, et al. Clinical symptoms of lumbar spinal stenosis associated with morphological parameters on magnetic resonance images. Eur Spine J. 2015;24(10):2236–2243. | ||
Ogikubo O, Forsberg L, Hansson T. The relationship between the cross-sectional area of the cauda equina and the preoperative symptoms in central lumbar spinal stenosis. Spine. 2007;32(13):1423–1428. | ||
Lim TH, Choi SI, Cho HR, et al. Optimal Cut-Off Value of the Superior Articular Process Area as a Morphological Parameter to Predict Lumbar Foraminal Stenosis. Pain Res Manag. 2017;2017:7914836. | ||
Barry M, Livesley P. Facet joint hypertrophy: the cross-sectional area of the superior articular process of L4 and L5. Eur Spine J. 1997;6(2):121–124. | ||
Genevay S, Atlas SJ, Stenosis Lspinal. Lumbar spinal stenosis. Best Pract Res Clin Rheumatol. 2010;24(2):253–265. | ||
Altinkaya N, Yildirim T, Demir S, Alkan O, Sarica FB. Factors associated with the thickness of the ligamentum flavum: is ligamentum flavum thickening due to hypertrophy or buckling? Spine. 2011;36(16):E1093–E1097. | ||
Panjabi MM, Oxland T, Takata K, Goel V, Duranceau J, Krag M. Articular facets of the human spine. Quantitative three-dimensional anatomy. Spine. 1993;18(10):1298–1310. | ||
Dunlop RB, Adams MA, Hutton WC. Disc space narrowing and the lumbar facet joints. J Bone Joint Surg Br. 1984;66(5):706–710. | ||
Wang J, Yang X. Age-related changes in the orientation of lumbar facet joints. Spine. 2009;34(17):E596–E598. | ||
Bajek G, Bajek S, Cvek SZ, Bobinac D, Splavski B, Grahovac DS. Histomorphological analysis of the osteophytic appositions in patients with lumbar lateral recess syndrome. Coll Antropol. 2010;34(Suppl 2):79–84. | ||
Chaput CD, Allred JJ, Pandorf JJ, Song J, Rahm MD. The significance of facet joint cross-sectional area on magnetic resonance imaging in relationship to cervical degenerative spondylolisthesis. Spine J. 2013;13(8):856–861. | ||
Ko S. Correlations between sedimentation sign, dural sac cross-sectional area, and clinical symptoms of degenerative lumbar spinal stenosis. Eur Spine J. 2018;27(7):1623–1628. | ||
Zhang L, Chen R, Liu B, Zhang W, Zhu Y, Rong L. The nerve root sedimentation sign for differential diagnosis of lumbar spinal stenosis: a retrospective, consecutive cohort study. Eur Spine J. 2017;26(10):2512–2519. | ||
Barz T, Staub LP, Melloh M, et al. Clinical validity of the nerve root sedimentation sign in patients with suspected lumbar spinal stenosis. Spine J. 2014;14(4):667–674. | ||
Park SH, Lee PB, Choe GY, Moon JY, Nahm FS, Kim YC. Therapeutic effect of epidurally administered lipo-prostaglandin e1 agonist in a rat spinal stenosis model. Korean J Pain. 2014;27(3):219–228. | ||
An JW, Koh JC, Sun JM, et al. Clinical Identification of the Vertebral Level at Which the Lumbar Sympathetic Ganglia Aggregate. Korean J Pain. 2016;29(2):103–109. | ||
Manchikanti L, Pampati V, Kaye AD, Hirsch JA. Therapeutic lumbar facet joint nerve blocks in the treatment of chronic low back pain: cost utility analysis based on a randomized controlled trial. Korean J Pain. 2018;31(1):27–38. | ||
Sanie MS, Ghobadifar MA. Risks and Pitfalls of Epidural Injections during Management of Lumbar Disc Herniation: Few Comments. Korean J Pain. 2015;28(3):217–218. | ||
Byun JM, Park HS, Woo JH, Kim J. The effects of a forceful transforaminal epidural steroid injection on radicular pain: a preliminary study. Korean J Pain. 2014;27(4):334–338. | ||
Kim SY. Radiculopathy caused by discal cyst. Korean J Pain. 2014;27(1):86–89. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.