Back to Journals » OncoTargets and Therapy » Volume 13
Successful Management of a ROS1-Rearranged Pulmonary Pleomorphic Carcinoma Using Serial Tyrosine Kinase Inhibitors
Authors Wu CW , Yang CY, Chang YL, Shih JY
Received 13 May 2020
Accepted for publication 16 August 2020
Published 9 October 2020 Volume 2020:13 Pages 10123—10127
DOI https://doi.org/10.2147/OTT.S262653
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Leo Jen-Liang Su
Chang-Wei Wu,1 Ching-Yao Yang,1 Yih-Leong Chang,2 Jin-Yuan Shih1
1Division of Pulmonary and Critical Care Medicine, Department of Internal Medicine, National Taiwan University Hospital and College of Medicine, National Taiwan University, Taipei, Taiwan; 2Department of Pathology, National Taiwan University Cancer Center, National Taiwan University Hospital and National Taiwan University College of Medicine, Taipei, Taiwan
Correspondence: Jin-Yuan Shih
Division of Pulmonary and Critical Care Medicine, Department of Internal Medicine, National Taiwan University Hospital and College of Medicine, National Taiwan University, No. 7, Chung-Shan South Road, Taipei 100, Taiwan
Tel +886-2-23562905
Fax +886-2-23582867
Email [email protected]
Abstract: Pulmonary pleomorphic carcinoma (PPC) generally lacks actionable driver mutations such as epidermal growth factor receptor mutations or anaplastic lymphoma kinase or c-ros oncogene 1 (ROS1) rearrangements. The response to crizotinib, ceritinib, brigatinib, and lorlatinib in ROS1-positive advanced non-small cell lung carcinoma is well established; however, there is little mention of their successful administration in pulmonary pleomorphic carcinoma cases. We report a case of a stage II PPC with recurrence after surgical resection and developed multiple distant metastasis. The tumor was refractory to chemotherapy and immunotherapy with progressive disease. EZR-ROS1 fusion was detected by next-generation sequencing and showed a good response to serial ROS1 inhibitors combined with surgery and radiotherapy. Now under lorlatinib, all her lesions responded well during the follow-up with sustained partial remission for more than 18 months. A sustainable treatment effect can be achieved in pulmonary pleomorphic carcinoma with driver mutations with tyrosine kinase inhibitor treatment. Driver mutations should be regularly tested in pulmonary pleomorphic carcinomas.
Keywords: pulmonary pleomorphic carcinoma, lung cancer, ROS1-rearranged, tyrosine kinase inhibitor, lorlatinib
Introduction
Pulmonary pleomorphic carcinoma (PPC) is a rare malignant tumor of the lung, accounting for around 0.1–0.3% of all lung cancers.1 According to the revised 2015 World Health Organization classification, PPC is one of the five subtypes of pulmonary sarcomatoid carcinoma (PSC) and contains at least 10% spindle and/or giant cells. The clinical course and response to systemic therapy are generally poor in PPC, which has a lower incidence of druggable driver mutations than lung adenocarcinoma. Kirsten rat sarcoma virus oncogene homolog (KRAS), MET exon 14 skipping, and epidermal growth factor receptor (EGFR) mutations are the most common driver oncogenes in PSC, whereas anaplastic lymphoma kinase (ALK) and c-ros oncogene 1 (ROS1) fusions are seldom identified.2 In recent years, several newer generation tyrosine kinase inhibitors (TKIs) have emerged and shown good activity in treatment-naïve or heavily treated patients, including alectinib, ceritinib, brigatinib, and lorlatinib. However, in the real-world practice of lung adenocarcinoma harboring druggable driver mutations, not all patients respond well to each type of TKI, making the switch between TKIs a mandatory and reasonable approach to maximize the benefit of targeted therapy. However, the data regarding TKIs other than crizotinib in PPC with fusion genes are lacking.
Herein, we describe a heavily treated ROS1-rearranged PPC patient successfully managed by serial ROS1 TKIs. Appropriate written informed consent was obtained for publication of this case report and accompanying images.
Case Report
Chest computed tomography revealed a 3.5 cm tumor in the right middle lobe in April 2017 in a 45-year-old non-smoking woman. She was diagnosed with stage II pleomorphic carcinoma (pT2aN1M0 according to the American Joint Committee on Cancer handbook 7th edition) and underwent right middle lobe lobectomy in May 2017, followed by four cycles of adjuvant chemotherapy (cisplatin and pemetrexed every 3 weeks, which ended in August 2017). However, tumor recurrence occurred 3 months later, with a new left lower lung tumor, bone (left clavicle and right femur) and brain (left cerebral frontal lobe) metastases (Figure 1). The tests for driver oncogenes, including EGFR, ALK, and ROS1 were negative, using the MassARRAY® system (Agena, CA, USA), VENTANA ALK (D5F3) CDx Assay, and ROS1 (D4D6) rabbit monoclonal antibody immunohistochemical (IHC) assay, respectively.
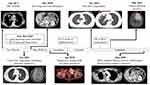 |
Figure 1 Summary of the treatment course in this case report. Arrows indicate the target lesions in the imaging studies. |
Because of the chemoresistant nature of PPC, the patient received an aggressive multidisciplinary approach including lung tumor resection (S6 segmentectomy) in February 2018, radiation to the bone metastases (35 Gy in five fractions to the left clavicle and right femur) in February 2018, and brain tumor resection in March 2018 followed by radiation to the brain tumor bed (18 Gy in a single fraction) (Figure 1). The pathologic findings of the lung (Figure 2A) and brain tumors showed pleomorphic carcinoma (Figure 2B) arising in an adenocarcinoma, which was positive for thyroid transcription factor 1 (Figure 2C) and vimentin (Figure 2D) and negative for CK7. PD-L1 (Dako PD-L1 IHC 22C3 pharmDx assay) was expressed in 80% and 45% of tumor cells in the lung and brain tumors, respectively. Unfortunately, new lung, and renal metastases developed during treatment; therefore, a combination of docetaxel (35 mg/m2 on days 1 and 8 every 3 weeks) and pembrolizumab (100 mg every 3 weeks) was administered as salvage treatment for the new metastases. However, progressive disease with new bone metastasis (left rib, right ischium, right femur) was identified by positron emission tomography (PET) after two cycles of chemotherapy-immune checkpoint inhibitor combination therapy.
Owing to the refractory nature of her disease, we performed next-generation sequencing (NGS) using the previous surgical specimens (Foundation One assay, hybrid-capture-based NGS to detect 324 genes, Foundation Medicine, Inc.) to find possible driver mutations in tumor of young non-smoker female patient. Unexpectedly, an EZR-ROS1 fusion was identified. Using a more sensitive ROS-1 antibody (SP384), we disclosed both resected lung tumor and brain tumor had ROS-1 protein expression (Figure 2F). Therefore, crizotinib treatment was started in April 2018, and she achieved a partial response 2 months later (according to the Response Evaluation Criteria in Solid Tumors, version 1.1), with shrinkage of the lung and renal tumors. Despite the remarkable efficacy of TKIs, grade 4 treatment-related hepatoxicity was observed during crizotinib therapy. The peak levels of aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were 865 U/L and 2451 U/L, respectively; total bilirubin and alkaline phosphatase were within the normal limits. Her liver enzymes returned to normal levels at day 39 after crizotinib discontinuation (AST 19 U/L, ALT 20 U/L). We changed the treatment to ceritinib in July 2018, and the partial response to TKIs persisted until November 2018, although new lung tumors emerged in the right upper and lower lobes (Figure 1). Therefore, we changed the treatment to the third-generation ROS1 TKI lorlatinib (100 mg daily) in combination with radiation therapy to the lung tumors (5250 cGy in 15 fractions). The lung tumor, bone, and renal metastases responded well during follow-up (last image study in June 2020 as showed in Figure 1), except a pseudoprogression of the left frontal lobe brain tumor occurring in March 2019, which was confirmed as radiation necrosis after surgical resection (Figure 3). Recurrence of radiation necrosis was noted 3 months after surgery, and it later spontaneously resolved. The patient is still under lorlatinib therapy since November 2018 with a sustained systemic partial remission. The total treatment course is summarized in Figure 1.
Discussion
Herein, we describe a patient with heavily treated ROS1-rearranged PPC successfully managed by serial ROS1 TKIs. The patient developed severe liver toxicity after a partial response to crizotinib and was successfully switched to ceritinib and lorlatinib uneventfully. The disease was well-controlled for years. Our report demonstrates the efficacy of newer generation ROS1 TKIs even in PPC, a tumor type with a lower incidence of driver mutations.
PPC is known for its resistance to chemotherapy and low responsiveness to radiotherapy. Therefore, different treatment strategies have been assessed in recent years. Surgery is the treatment of choice in early-stage PPC with a high recurrence rate; these patients may also benefit from perioperative chemotherapy.3 In advanced PPC, response to first-line chemotherapy is poor, with an overall survival of 5 months.4 Multimodality treatment with surgery, radiotherapy, and immunotherapy has been attempted but rarely achieves good response. A high frequency of PD-L1 expression has been observed in PPC patients, but variable response to immune checkpoint inhibitors is reported.5 Our patient had a poor response to pembrolizumab combined with docetaxel despite a high expression of PD-L1 in the primary and metastatic tumors.
Druggable driver mutation genes are detected by comprehensive genomic profiling in 30% of PSC patients, with MET exon 14 skipping being the most common mutation, adding an additional option for treatment.2 ROS1 rearrangement is rare in PPC and the specificity of ROS1 in IHC was low hence ROS1 status should be confirmed with fluorescence in situ hybridization (FISH) or NGS. The reason for the discrepancy of results of ROS1 IHC stains in our case might be caused by different sensitivity of antibodies. As previous studies reported, SP384 is more sensitive than D4D6 in FISH detected ROS1 gene arrangement lung cancer.6,7 The discordancy between SP384 and D4D6, though rare, was also shown in previous studies.6,7
Crizotinib and ceritinib are ROS1 inhibitors with overall response rates of 72% and 62% in NSCLC patients, respectively, and median progression-free survivals of 19.2 and 19.3 months, respectively.8,9 Lorlatinib also showed promising results in a Phase 1–2 trial.10 However, only one PSC case with ROS1 alteration has been reported; the patient showed a sustained response to crizotinib for 46 months.11 Our case describes the first successful treatment with serial ROS1 inhibitors. In our patient, crizotinib showed a good effect; however, hepatoxicity occurred, and the treatment was shifted to ceritinib. New lesions developed after 4 months. The treatment was changed to lorlatinib in combination with local radiation treatment. The resistance to ceritinib may be related to kinase domain mutation as opposed to activation of other pathways because the patient had a sustained response to lorlatinib.
Radiation necrosis presenting as pseudoprogression during alectinib treatment of previously radiated brain metastases has been reported by Ou et al.12 No other similar presentation of ALK inhibitors has been reported. Our patient developed pseudoprogression 11 months after Cyberknife surgery and about 3 months after starting lorlatinib, which is similar to what was observed in two previously reported cases.12 However, the causal relationship and pathophysiology remain unclear.
Conclusion
To our knowledge, this is the first report of ROS1-rearranged lung PPC successfully treated by serial TKIs. Our case highlights the importance of molecular testing even in PPC, which has a low prevalence of driver mutations, sequential use of TKIs, the delicate management of treatment-related adverse events, multidisciplinary treatment, and the careful differential diagnosis of radiation necrosis rather than true progression, which may contribute to extended patient survival.
Abbreviations
ROS1, c-ros oncogene 1; PPC, pulmonary pleomorphic carcinoma; PSC, pulmonary sarcomatoid carcinoma; KRAS, Kirsten rat sarcoma virus oncogene homolog; EGFR, epidermal growth factor receptor; ALK, anaplastic lymphoma kinase; TKI, tyrosine kinase inhibitor; PD-L1, programmed death-ligand 1; NGS, next-generation sequencing; AST, aspartate aminotransferase; ALT, alanine aminotransferase.
Consent for Publication
The patient signed the written informed consent to use and publish the patient’s data, case details, and images for study. No institutional approval was required to publish the case details.
Acknowledgments
We would like to thank Editage for English language editing. There was no funding source to acknowledge.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
Disclosure
Chang-Wei Wu, Ching-Yao Yang, and Yih‐Leong Chang report no conflicts of interest for this work.
Jin-Yuan Shih has served as an advisory board member for and has received speaker honoraria from AbbVie, AstraZeneca, Boehringer Ingelheim, Bristol‐Myers Squibb, Chugai, Eli Lilly, Merck Sharp & Dohme, Novartis, Ono Pharmaceutical, and Roche, and has received allowances for expenses for travel and accommodation from Boehringer Ingelheim, Bristol‐Myers Squibb, Merck Sharp & Dohme, Pfizer, and Roche, and reports no other potential conflicts of interest in this work.
References
1. Chang Y-L, Lee Y-C, Shih J-Y, Wu C-T. Pulmonary pleomorphic (spindle) cell carcinoma: peculiar clinicopathologic manifestations different from ordinary non-small cell carcinoma. Lung Cancer. 2001;34(1):91–97.
2. Schrock AB, Li SD, Frampton GM, et al. Pulmonary sarcomatoid carcinomas commonly harbor either potentially targetable genomic alterations or high tumor mutational burden as observed by comprehensive genomic profiling. J Thorac Oncol. 2017;12(6):932–942.
3. Chaft JE, Sima CS, Ginsberg MS, et al. Clinical outcomes with perioperative chemotherapy in sarcomatoid carcinomas of the lung. J Thorac Oncol. 2012;7(9):1400–1405.
4. Bae HM, Min HS, Lee SH, et al. Palliative chemotherapy for pulmonary pleomorphic carcinoma. Lung Cancer. 2007;58(1):112–115.
5. Chang Y-L, Yang C-Y, Lin M-W, Wu C-T, Yang P-C. High co-expression of PD-L1 and HIF-1α correlates with tumour necrosis in pulmonary pleomorphic carcinoma. Eur J Cancer. 2016;60:125–135.
6. Hofman V, Rouquette I, Long-Mira E, et al. Multicenter evaluation of a novel ROS1 immunohistochemistry assay (SP384) for detection of ROS1 rearrangements in a large cohort of lung adenocarcinoma patients. J Thorac Oncol. 2019;14(7):1204–1212.
7. Conde E, Hernandez S, Martinez R, et al. Assessment of a new ROS1 immunohistochemistry clone (SP384) for the identification of ROS1 rearrangements in patients with non–small cell lung carcinoma: the ROSING study. J Thorac Oncol. 2019;14(12):2120–2132.
8. Shaw AT, Ou S-HI, Bang Y-J, et al. Crizotinib in ROS1-rearranged non–small-cell lung cancer. N Engl J Med. 2014;371(21):1963–1971.
9. Lim SM, Kim HR, Lee J-S, et al. Open-label, multicenter, Phase II study of ceritinib in patients with non–small-cell lung cancer harboring ROS1 rearrangement. J Clin Oncol. 2017;35(23):2613–2618.
10. Shaw AT, Solomon BJ, Chiari R, et al. Lorlatinib in advanced ROS1-positive non-small-cell lung cancer: a multicentre, open-label, single-arm, phase 1–2 trial. Lancet Oncol. 2019;20(12):1691–1701.
11. Mizuno T, Fujiwara Y, Yoshida K, Kohno T, Ohe Y. Next-generation sequencer analysis of pulmonary pleomorphic carcinoma with a CD74-ROS1 fusion successfully treated with crizotinib. J Thorac Oncol. 2019;14(5):e106–e108.
12. Ou SH, Klempner SJ, Azada MC, Rausei-Mills V, Duma C. Radiation necrosis presenting as pseudoprogression (PsP) during alectinib treatment of previously radiated brain metastases in ALK-positive NSCLC: implications for disease assessment and management. Lung Cancer. 2015;88(3):355–359.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


